Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Cohorts
2.2. Kawasaki Disease Risk
2.3. Blood Cadmium, Lead, Manganese, Mercury, Selenium
2.4. Statistical Analysis
3. Results
3.1. KID Cohort
| Ethnicity | KID (1997–2006) 1 | NHANES (2011–2012) 2 | U.S. Census (2012) 3 |
|---|---|---|---|
| African | n = 1540 (19.2%) | n = 238 (33.4%) | n = 10,235,449 (13.9%) |
| Asian | n = 823 (10.2%) | n = 60 (8.4%) | n = 3,501,690 (4.8%) |
| Caucasian | n = 3751 (46.7%) | n = 119 (16.7%) | n = 38,915,681 (52.8%) |
| Hispanic | n = 1614 (20.1%) | n = 256 (35.9%) | n = 17,569,191 (23.8%) |
| Other | n = 307 (3.8%) | n = 40 (5.6%) | n = 3,486,168 (4.7%) |
| TOTAL | n = 8035 (73.9%) | n = 713 (100%) | n = 73,708,179 |
3.2. NHANES Cohort
3.3. Kawasaki Disease Hospitalizations
| Ethnicity | Averaged (OR) | 1997 (OR) | 2000 (OR) | 2006 (OR) |
|---|---|---|---|---|
| African | 18.0 (0.97) | 16.9 (0.96) | 19.7 (1.15) | 17.5 (0.84) |
| Asian | 33.9 (1.83) | 32.5 (1.85) | 39.0 (2.28) | 30.3 (1.46) |
| Caucasian | 10.8 (0.58) | 9.1 (0.52) | 11.4 (0.67) | 12.0 (0.58) |
| Hispanic | 13.5 (0.73) | 11.1 (0.63) | 13.6 (0.80) | 15.7 (0.75) |
| Other 1 | n/a | n/a | n/a | n/a |
| Weighted | 18.5 | 17.6 (0.95) | 17.1 (0.92) | 20.8 (1.12) |
3.4. Mean Blood Determinations
| Ethnicity | Cd µg/L 2 (OR) | Hg µg/L 2 (OR) | Mn µg/L 3 (OR) | Pb µg/dL 4 (OR) | Se µg/L 5 (OR) |
|---|---|---|---|---|---|
| [min–max] | [min–max] | [min–max] | [min–max] | [min–max] | |
| African | 0.13 (1.00) | 0.50 (1.39) | 9.23 (0.84) ** | 1.63 (1.15) ** | 165.25 (0.99) |
| [0.11–0.43] | [0.11–18.89] | [4.83–17.49] | [0.29–18.37] | [110.24–217.25] | |
| Asian | 0.17 (1.31) ** | 0.92 (2.56) ** | 12.69 (1.15) ** | 0.92 (0.65) ** | 165.00 (0.98) |
| [0.11–0.39] | [0.11–5.18] | [5.94–25.42] | [0.29–3.26] | [102.34–204.90] | |
| Caucasian | 0.12 (0.92) ** | 0.26 (0.72) ** | 10.86 (0.98) | 1.59 (1.12) | 169.22 (1.01) |
| [0.11–0.28] | [0.11–1.05] | [6.28–24.49] | [0.18–27.88] | [126.37–241.38] | |
| Hispanic | 0.13 (1.00) | 0.38 (1.06) * | 12.04 (1.09) ** | 1.07 (0.75) ** | 165.82 (0.99) |
| [0.11–0.27] | [0.11–1.98] | [3.97–25.54] | [0.26–5.24] | [96.24–221.36] | |
| Other 1 | 0.13 (1.00) | 0.44 (1.22) | 11.58 (1.05) | 1.11 (0.78) | 166.73 (1.00) |
| [0.11–0.22] | [0.11–4.57] | [4.84–21.94] | [0.44–3.14] | [126.95–202.08] | |
| TOTAL 6 | 0.13 (1.00) ** | 0.45 (1.25) ** | 10.93 (0.99) ** | 1.33 (0.94) ** | 166.16 (0.99) |
| [0.11–0.43] | [0.11–18.89] | [3.97–25.54] | [0.18–27.88] | [96.24–241.38] | |
| Weighted | 0.13 | 0.36 | 11.04 | 1.42 | 167.54 |
3.5. Detectable Blood Determinations
| Ethnicity | Cd % (OR) 2 | Hg % (OR) 2 | Mn % 3 | Pb % (OR) 4 | Se % 5 |
|---|---|---|---|---|---|
| African | 20.2% (1.22) | 86.1% (1.12) ** | 100% | 100% (1.01) | 100% |
| Asian | 46.7% (2.81) ** | 96.7% (1.26) ** | 100% | 100% (1.01) | 100% |
| Caucasian | 10.9% (0.66) ** | 73.1% (0.95) * | 100% | 98.3% (0.99) | 100% |
| Hispanic | 19.9% (1.20) | 77.0% (1.00) | 100% | 100% (1.01) | 100% |
| Other 1 | 22.5% (1.36) | 72.5% (0.94) | 100% | 100% (1.01) | 100% |
| TOTAL 6 | 20.9% (1.26) ** | 80.8% (1.05) ** | 100% | 99.7% (1.01) * | 100% |
| Weighted | 16.6% | 76.9% | 100% | 99.1% | 100% |
3.6. Linear Dose-Responsive Analysis
| Elements | Average µg/L | Detection Rate | Bottom Percentile | Top Percentile |
|---|---|---|---|---|
| Cd | 96.8% * | 96.7% * | n/a 2,3 | 99.6% **,4 |
| Hg | 99.5% ** | 93.6% * | 91.7% *,3 | 97.2% *,4 |
| Mn | 22.5% | n/a 1 | 0.1% 3 | 21.0% 4 |
| Pb | 40.4% | 28.3% | 0.8% 3 | 43.0% 5 |
| Se | 42.4% | n/a 1 | 12.6% 3 | 55.8% 4 |
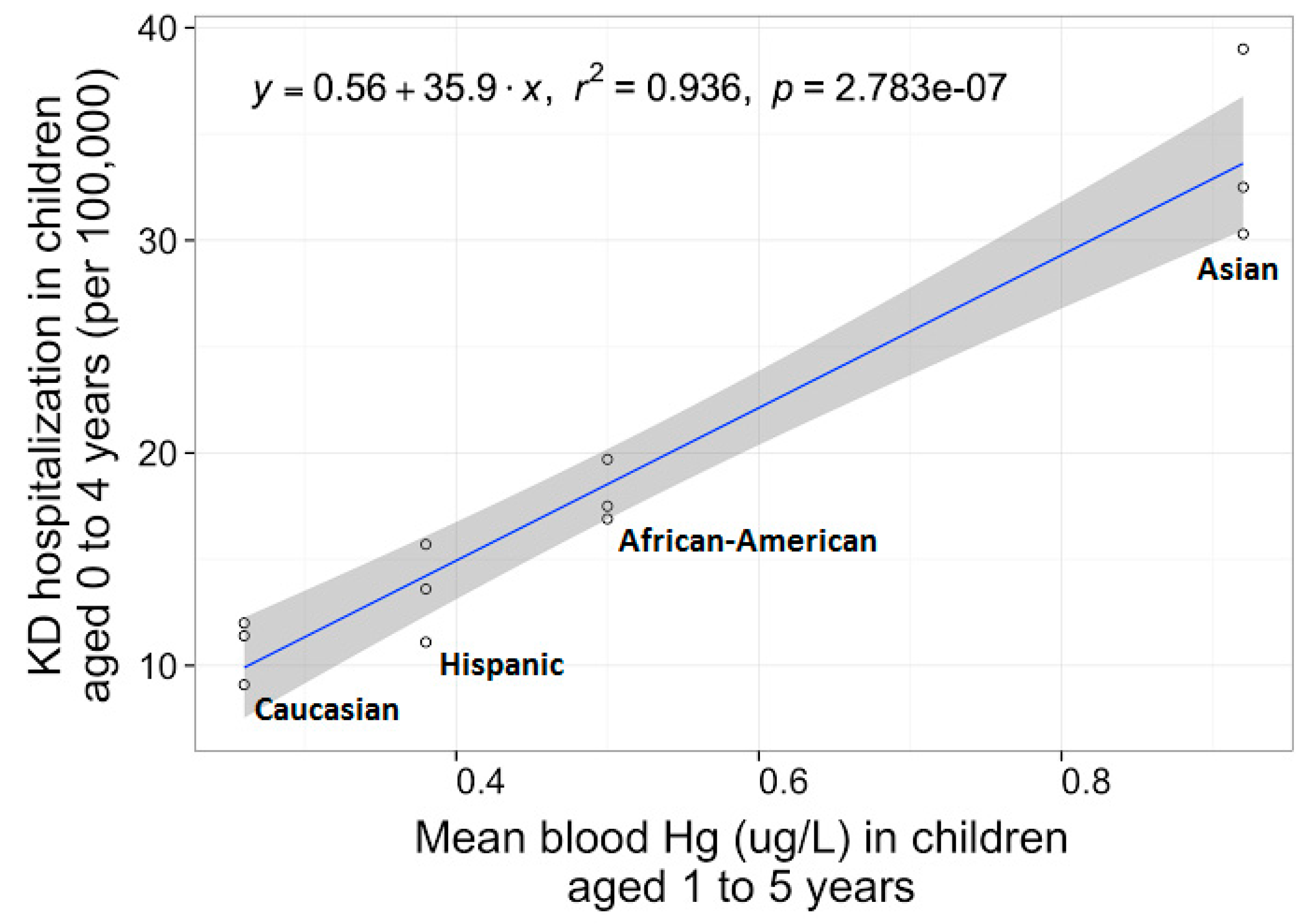
4. Discussion
5. Conclusions
Supplemental Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guo, M.M.; Tseng, W.N.; Ko, C.H.; Pan, H.M.; Hsieh, K.S.; Kuo, H.C. Th17- and Treg-related cytokine and mRNA expression are associated with acute and resolving Kawasaki disease. Allergy 2015, 70, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C.; Hsu, Y.W.; Wu, M.S.; Chien, S.C.; Liu, S.F.; Chang, W.C. Intravenous immunoglobulin, pharmacogenomics, and Kawasaki disease. J. Microbiol. Immunol. Infect. 2014. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Y.; Ye, B.; Zhao, X. Clinical characteristics of Kawasaki syndrome and the risk factors for coronary artery lesions in China. Pediatr. Infect. Dis. J. 2013, 32, e397–e402. [Google Scholar] [CrossRef] [PubMed]
- Newburger, J.W.; Takahashi, M.; Gerber, M.A.; Gewitz, M.H.; Tani, L.Y.; Burns, J.C.; Shulman, S.T.; Bolger, A.F.; Ferrieri, P.; Baltimore, R.S.; et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2004, 110, 2747–2771. [Google Scholar] [CrossRef] [PubMed]
- Mason, W.H.; Takahashi, M. Kawasaki syndrome. Clin. Infect. Dis. 1999, 28, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Makino, N.; Nakamura, Y.; Yashiro, M.; Ae, R.; Tsuboi, S.; Aoyama, Y.; Kojo, T.; Uehara, R.; Kotani, K.; Yanagawa, H. Descriptive epidemiology of kawasaki disease in Japan, 2011–2012: From the results of the 22nd nationwide survey. J. Epidemiol. 2015, 25, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.B.; Han, J.W.; Park, Y.W.; Song, M.S.; Hong, Y.M.; Cha, S.H.; Kim, D.S.; Park, S. Epidemiologic features of Kawasaki disease in South Korea: Data from nationwide survey, 2009–2011. Pediatr. Infect. Dis. J. 2014, 33, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.K.; Lin, M.T.; Chen, H.C.; Huang, S.C.; Wu, M.H. Epidemiology of Kawasaki disease: Prevalence from national database and future trends projection by system dynamics modeling. J. Pediatr. 2013, 163, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Uehara, R.; Belay, E.D. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J. Epidemiol. 2012, 22, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.C.; Christensen, K.Y.; Belay, E.D.; Steiner, C.A.; Effler, P.V.; Miyamura, J.; Forbes, S.; Schonberger, L.B.; Melish, M. Racial/ethnic differences in the incidence of Kawasaki syndrome among children in Hawaii. Hawaii Med. J. 2010, 69, 194–197. [Google Scholar] [PubMed]
- Duan, J.; Lou, J.; Zhang, Q.; Ke, J.; Qi, Y.; Shen, N.; Zhu, B.; Zhong, R.; Wang, Z.; Liu, L.; et al. A genetic variant rs1801274 in FCGR2A as a potential risk marker for Kawasaki disease: A case-control study and meta-analysis. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Kuo, H.C.; Hsu, Y.W.; Lo, M.H.; Huang, Y.H.; Chien, S.C.; Chang, W.C. Single-nucleotide polymorphism rs7251246 in ITPKC is associated with susceptibility and coronary artery lesions in Kawasaki disease. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Xing, Y.; Wang, H.; Liu, X.; Yu, X.; Chen, R.; Wang, C.; Sun, L. Meta-analysis of the relationship between single nucleotide polymorphism rs72689236 of caspase-3 and Kawasaki disease. Mol. Biol. Rep. 2014, 41, 6377–6381. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.J.; Kuo, H.C.; Chang, J.S.; Lee, J.K.; Tsai, F.J.; Khor, C.C.; Chang, L.C.; Chen, S.P.; Ko, T.M.; Liu, Y.M.; et al. Replication and meta-analysis of GWAS identified susceptibility loci in Kawasaki disease confirm the importance of B lymphoid tyrosine kinase (BLK) in disease susceptibility. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Chen, C.H.; Wu, Q.; Yang, Y. Meta-analyses of the associations of genome-wide association study-linked gene loci with Kawasaki disease. Chin. J. Pediatr. 2013, 51, 571–577. [Google Scholar]
- Lou, J.; Xu, S.; Zou, L.; Zhong, R.; Zhang, T.; Sun, Y.; Lu, X.; Liu, L.; Li, C.; Wang, L.; et al. A functional polymorphism, rs28493229, in ITPKC and risk of Kawasaki disease: An integrated meta-analysis. Mol. Biol. Rep. 2012, 39, 11137–11144. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Kuo, H.C.; Chang, J.S.; Chang, L.Y.; Huang, L.M.; Chen, M.R.; Liang, C.D.; Chi, H.; Huang, F.Y.; Lee, M.L.; et al. Two new susceptibility loci for Kawasaki disease identified through genome-wide association analysis. Nat. Genet. 2012, 44, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Khor, C.C.; Davila, S.; Breunis, W.B.; Lee, Y.C.; Shimizu, C.; Wright, V.J.; Yeung, R.S.; Tan, D.E.; Sim, K.S.; Wang, J.J.; et al. Genome-wide association study identifies FCGR2A as a susceptibility locus for Kawasaki disease. Nat. Genet. 2011, 43, 1241–1246. [Google Scholar] [CrossRef] [PubMed]
- Onouchi, Y. Genetics of Kawasaki disease: What we know and don’t know. Circ. J. 2012, 76, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Yeter, D.; Deth, R. ITPKC susceptibility in Kawasaki syndrome as a sensitizing factor for autoimmunity and coronary arterial wall relaxation induced by thimerosal’s effects on calcium signaling via IP3. Autoimmun. Rev. 2012, 11, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.Y.; Lu, C.Y.; Shao, P.L.; Lee, P.I.; Lin, M.T.; Fan, T.Y.; Cheng, A.L.; Lee, W.L.; Hu, J.J.; Yeh, S.J.; et al. Viral infections associated with Kawasaki disease. J. Formos. Med. Assoc. 2014, 113, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Turnier, J.L.; Anderson, M.S.; Heizer, H.R.; Jone, P.N.; Glode, M.P.; Dominguez, S.R. Concurrent respiratory viruses and Kawasaki disease. Pediatrics 2015, 136, e609–e614. [Google Scholar] [CrossRef] [PubMed]
- Jaggi, P.; Kajon, A.E.; Mejias, A.; Ramilo, O.; Leber, A. Human adenovirus infection in Kawasaki disease: A confounding bystander? Clin. Infect. Dis. 2013, 56, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Jordan-Villegas, A.; Chang, M.L.; Ramilo, O.; Mejias, A. Concomitant respiratory viral infections in children with Kawasaki disease. Pediatr. Infect. Dis. J. 2010, 29, 770–772. [Google Scholar] [CrossRef] [PubMed]
- Cheek, D.B. Letter: Comment on mucocutaneous lymph node syndrome: Could it be a heavy metal poisoning? Pediatrics 1975, 56, 335–336. [Google Scholar] [PubMed]
- Nishida, G. Letter: Mercury poisoning and MLNS. Pediatrics 1976, 58. [Google Scholar] [CrossRef]
- Adler, R.; Boxstein, D.; Schaff, P.; Kelly, D. Metallic mercury vapor poisoning simulating mucocutaneous lymph node syndrome. J. Pediatr. 1982, 101, 967–968. [Google Scholar] [CrossRef]
- Orlowski, J.P.; Mercer, R.D. Urine mercury levels in Kawasaki disease. Pediatrics 1980, 66, 633–636. [Google Scholar] [PubMed]
- Kawasaki, T. Letter To The Editor. Pediatrics 1975, 56, 336–337. [Google Scholar]
- Aschner, M.; Aschner, J.L. Mucocutaneous lymph node syndrome: Is there a relationship to mercury exposure? Am. J. Dis. Child. 1989, 143, 1133–1134. [Google Scholar] [CrossRef] [PubMed]
- Warkany, J.; Hubbard, D.M. Adverse mercurial reactions in the form of acrodynia and related conditions, AMA. Am. J. Dis. Child. 1951, 81, 335–373. [Google Scholar]
- Warkany, J.; Hubbard, D.M. Acrodynia and mercury. J. Pediatr. 1953, 42, 365–386. [Google Scholar] [CrossRef]
- The Kids’ Inpatient Database (KID). Available online: http://hcup-us.ahrq.gov/kidoverview.jsp (accessed on 15 February 2015).
- The National Health and Nutrition Examination Survey. Available online: http://www.cdc.gov/nchs/nhanes.htm (accessed on 15 February 2015).
- Holman, R.C.; Curns, A.T.; Belay, E.D.; Steiner, C.A.; Schonberger, L.B. Kawasaki syndrome hospitalizations in the United States, 1997 and 2000. Pediatrics 2003, 112, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.C.; Belay, E.D.; Christensen, K.Y.; Folkema, A.M.; Steiner, C.A.; Schonberger, L.B. Hospitalizations for Kawasaki syndrome among children in the United States, 1997–2007. Pediatr. Infect. Dis. J. 2010, 29, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Woon, P.Y.; Chang, W.C.; Liang, C.C.; Hsu, C.H.; Klahan, S.; Huang, Y.H.; Chang, W.P.; Kuo, H.C. Increased risk of atopic dermatitis in preschool children with Kawasaki disease: A population-based study in Taiwan. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.C.; Lin, C.L.; Kao, C.H.; Liao, Y.H.; Shen, T.C.; Tsai, J.D.; Chang, Y.J.; Li, T.C. Increased risk of Kawasaki disease in children with common allergic diseases. Ann. Epidemiol. 2014, 24, 340–343. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.J.; Lin, C.H.; Fu, L.S.; Fu, Y.C.; Lin, M.C.; Jan, S.L. The association between Kawasaki disease and allergic diseases, from infancy to school age. Allergy Asthma Proc. 2013, 34, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.Y.; Leem, J.H.; Kim, H.C.; Kim, J.H.; Hwang, S.S.; Lee, J.Y.; Kim, B.J.; Hong, Y.C.; Hong, S.J.; Kwon, H.J. Effect of Traffic-Related Air Pollution on Allergic Disease: Results of the Children’s Health and Environmental Research. Allergy Asthma Immunol. Res. 2015, 7, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Lee, C.S.; Jeon, J.M.; Yu, S.D.; Lee, C.W.; Park, J.H.; Shin, D.C.; Lim, Y.W. Analysis of the association between air pollution and allergic diseases exposure from nearby sources of ambient air pollution within elementary school zones in four Korean cities. Environ. Sci. Pollut. Res. Int. 2013, 20, 4831–4846. [Google Scholar] [CrossRef] [PubMed]
- Feleszko, W.; Ruszczynski, M.; Jaworska, J.; Strzelak, A.; Zalewski, B.M.; Kulus, M. Environmental tobacco smoke exposure and risk of allergic sensitisation in children: A systematic review and meta-analysis. Arch. Dis. Child. 2014, 99, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Thacher, J.D.; Gruzieva, O.; Pershagen, G.; Neuman, A.; Wickman, M.; Kull, I.; Melen, E.; Bergstrom, A. Pre- and postnatal exposure to parental smoking and allergic disease through adolescence. Pediatrics 2014, 134, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Awaya, A.; Nishimura, C. A combination of cross correlation and trend analyses reveals that Kawasaki disease is a pollen-induced delayed-type hyper-sensitivity disease. Int. J. Environ. Res. Public Health 2014, 11, 2628–2641. [Google Scholar] [CrossRef] [PubMed]
- Rodo, X.; Ballester, J.; Cayan, D.; Melish, M.E.; Nakamura, Y.; Uehara, R.; Burns, J.C. Association of Kawasaki disease with tropospheric wind patterns. Sci. Rep. 2011, 1. [Google Scholar] [CrossRef] [PubMed]
- Rodo, X.; Curcoll, R.; Robinson, M.; Ballester, J.; Burns, J.C.; Cayan, D.R.; Lipkin, W.I.; Williams, B.L.; Couto-Rodriguez, M.; Nakamura, Y.; et al. Tropospheric winds from northeastern China carry the etiologic agent of Kawasaki disease from its source to Japan. Proc. Natl. Acad. Sci. USA 2014, 111, 7952–7957. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Teng, Y.; Lu, S.; Wang, Y.; Wang, J. Contamination features and health risk of soil heavy metals in China. Sci. Total Environ. 2015, 512–513, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Baba, H.; Tsuneyama, K.; Kumada, T.; Aoshima, K.; Imura, J. Histopathological analysis for osteomalacia and tubulopathy in itai-itai disease. J. Toxicol. Sci. 2014, 39, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Satoh, H.; Murata, K.; Eto, K. Adverse effects of methylmercury: Environmental health research implications. Environ. Health Perspect. 2010, 118, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, N.; Shibuya, K.; Kato, H.; Yanagisawa, M. Kawasaki disease before kawasaki at Tokyo university hospital. Pediatrics 2002, 110. [Google Scholar] [CrossRef]
- Tashyrev, O.B.; Ie, P.P.; Tashyreva, G.O.; Bielikova, O. Resistance of microbial communities from ecuador ecosystems to representative toxic metals–CrO4(2−), Co2+, Ni2+, Cu2+, Hg2+. Mikrobiol. Z. 2015, 77, 44–61. [Google Scholar] [PubMed]
- Jiang, C.B.; Hsi, H.C.; Fan, C.H.; Chien, L.C. Fetal exposure to environmental neurotoxins in Taiwan. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Diez, S.; Delgado, S.; Aguilera, I.; Astray, J.; Perez-Gomez, B.; Torrent, M.; Sunyer, J.; Bayona, J.M. Prenatal and early childhood exposure to mercury and methylmercury in Spain, a high-fish-consumer country. Arch. Environ. Contam. Toxicol. 2009, 56, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Ralston, N.V.; Ralston, C.R.; Blackwell, J.L., 3rd; Raymond, L.J. Dietary and tissue selenium in relation to methylmercury toxicity. Neurotoxicology 2008, 29, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.L.; Budtz-Jorgensen, E.; Jorgensen, P.J.; Steuerwald, U.; Debes, F.; Weihe, P.; Grandjean, P. Selenium as a potential protective factor against mercury developmental neurotoxicity. Environ. Res. 2008, 107, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Pichichero, M.E.; Gentile, A.; Giglio, N.; Umido, V.; Clarkson, T.; Cernichiari, E.; Zareba, G.; Gotelli, C.; Gotelli, M.; Yan, L.; et al. Mercury levels in newborns and infants after receipt of thimerosal-containing vaccines. Pediatrics 2008, 121, e208–e214. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Zartarian, V.G.; Liu, S.V.; Geller, A.M. Methyl mercury exposure from fish consumption in vulnerable racial/ethnic populations: Probabilistic SHEDS-Dietary model analyses using 1999–2006 NHANES and 1990–2002 TDS data. Sci. Total Environ. 2012, 414, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Lue, H.C.; Chen, L.R.; Lin, M.T.; Chang, L.Y.; Wang, J.K.; Lee, C.Y.; Wu, M.H. Epidemiological features of Kawasaki disease in Taiwan, 1976–2007: Results of five nationwide questionnaire hospital surveys. Pediatr. Neonatol. 2014, 55, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.J.; Aoki, Y.; Kit, B.K.; Ogden, C.L. More than half of US youth consume seafood and most have blood mercury concentrations below the EPA reference level, 2009–2012. J. Nutr. 2015, 145, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, K.L.; Mortensen, M.E.; Jones, R.L.; Caudill, S.P.; Osterloh, J.D. Total blood mercury concentrations in the U.S. population: 1999–2006. Int. J. Hyg. Environ. Health 2009, 212, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Portman, M.A. Kawasaki disease and soy: Potential role for isoflavone interaction with Fcgamma receptors. Pediatr. Res. 2013, 73, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Wiener, H.; Shendre, A.; Kaslow, R.A.; Wu, J.; Olson, A.; Bowles, N.E.; Patel, H.; Edberg, J.C.; Portman, M.A. Role of activating FcgammaR gene polymorphisms in Kawasaki disease susceptibility and intravenous immunoglobulin response. Circ. Cardiovasc. Genet. 2012, 5, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Myers, R.; Wei, T.; Bind, E.; Kassim, P.; Wang, G.; Ji, Y.; Hong, X.; Caruso, D.; Bartell, T.; et al. Placental transfer and concentrations of cadmium, mercury, lead, and selenium in mothers, newborns, and young children. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Arefieva, A.S.; Kamaeva, A.G.; Krasilshchikova, M.S. Low doses of mercuric chloride cause the main features of antinucleolar autoimmune process in female outbred CFW mice. Toxicol. Ind. Health 2015. [Google Scholar] [CrossRef] [PubMed]
- Havarinasab, S.; Bjorn, E.; Ekstrand, J.; Hultman, P. Dose and Hg species determine the T-helper cell activation in murine autoimmunity. Toxicology 2007, 229, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Gallucci, S.; Gaughan, J.P.; Gross, J.A.; Monestier, M. A role for B cell-activating factor of the TNF family in chemically induced autoimmunity. J. Immunol. 2005, 175, 6163–6168. [Google Scholar] [CrossRef] [PubMed]
- Toomey, C.B.; Cauvi, D.M.; Hamel, J.C.; Ramirez, A.E.; Pollard, K.M. Cathepsin B regulates the appearance and severity of mercury-induced inflammation and autoimmunity. Toxicol. Sci. 2014, 142, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Häggqvist, B.; Havarinasab, S.; Björn, E.; Hultman, P. The immunosuppressive effect of methylmercury does not preclude development of autoimmunity in genetically susceptible mice. Toxicology 2005, 208, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Havarinasab, S.; Lambertsson, L.; Qvarnstrom, J.; Hultman, P. Dose-response study of thimerosal-induced murine systemic autoimmunity. Toxicol. Appl. Pharmacol. 2004, 194, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Sapin, C.; Hirsch, F.; Delaporte, J.P.; Bazin, H.; Druet, P. Polyclonal IgE increase after HgCl2 injections in BN and LEW rats: A genetic analysis. Immunogenetics 1984, 20, 227–236. [Google Scholar] [CrossRef] [PubMed]
- De Castro, N.S.; Mde, O.L. Biomarkers of mercury exposure in the Amazon. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Motts, J.A.; Shirley, D.L.; Silbergeld, E.K.; Nyland, J.F. Novel biomarkers of mercury-induced autoimmune dysfunction: A cross-sectional study in Amazonian Brazil. Environ. Res. 2014, 132, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.M.; Nyland, J.F.; Silva, I.A.; Ventura, A.M.; de Souza, J.M.; Silbergeld, E.K. Mercury exposure, serum antinuclear/antinucleolar antibodies, and serum cytokine levels in mining populations in Amazonian Brazil: A cross-sectional study. Environ. Res. 2010, 110, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Silva, I.A.; Nyland, J.F.; Gorman, A.; Perisse, A.; Ventura, A.M.; Santos, E.C.; Souza, J.M.; Burek, C.L.; Rose, N.R.; Silbergeld, E.K. Mercury exposure, malaria, and serum antinuclear/antinucleolar antibodies in Amazon populations in Brazil: A cross-sectional study. Environ. Health 2004, 3. [Google Scholar] [CrossRef] [PubMed]
- Gattineni, J.; Weiser, S.; Becker, A.M.; Baum, M. Mercury intoxication: Lack of correlation between symptoms and levels. Clin. Pediatr. 2007, 46, 844–846. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeter, D.; Portman, M.A.; Aschner, M.; Farina, M.; Chan, W.-C.; Hsieh, K.-S.; Kuo, H.-C. Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children. Int. J. Environ. Res. Public Health 2016, 13, 101. https://doi.org/10.3390/ijerph13010101
Yeter D, Portman MA, Aschner M, Farina M, Chan W-C, Hsieh K-S, Kuo H-C. Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children. International Journal of Environmental Research and Public Health. 2016; 13(1):101. https://doi.org/10.3390/ijerph13010101
Chicago/Turabian StyleYeter, Deniz, Michael A. Portman, Michael Aschner, Marcelo Farina, Wen-Ching Chan, Kai-Sheng Hsieh, and Ho-Chang Kuo. 2016. "Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children" International Journal of Environmental Research and Public Health 13, no. 1: 101. https://doi.org/10.3390/ijerph13010101
APA StyleYeter, D., Portman, M. A., Aschner, M., Farina, M., Chan, W.-C., Hsieh, K.-S., & Kuo, H.-C. (2016). Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children. International Journal of Environmental Research and Public Health, 13(1), 101. https://doi.org/10.3390/ijerph13010101









