Low-Dose Cadmium Upregulates VEGF Expression in Lung Adenocarcinoma Cells
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Culture
2.2. Cell Proliferation Assay
2.3. Wound-Healing Assay
2.4. Annexin V-FITC/PI Analyses
2.5. Quantitative Real-Time PCR (qRT-PCR)
| Gene | Sequence | Size (bp) | Tm (ºC) |
|---|---|---|---|
| VEGFA | |||
| Sense | AAAGGGAAAGGGGCAAAAACGAA | 110 | 60.5 |
| Anti-sense | AGGAACATTTACACGTCTGCGG | ||
| b-FGF | |||
| Sense | AGCGACCCTCACATCAAG | 106 | 61 |
| Anti-sense | ATCTTCCATCTTCCTTCATAGC | ||
| GAPDH | |||
| Sense | TGATGACATCAAGAAGGTGGTGAAG | 240 | 60 |
| Anti-sense | TCCTTGGAGGCCATGTGGGCCAT |
2.6. Western Blotting
2.7. ELISA
2.8. Preparation of Tumor-Conditioned Medium (CM)
2.9. Electric Cell-Substrate Impedance Sensing (ECIS) Analysis
2.10. Statistical Analysis
3. Results
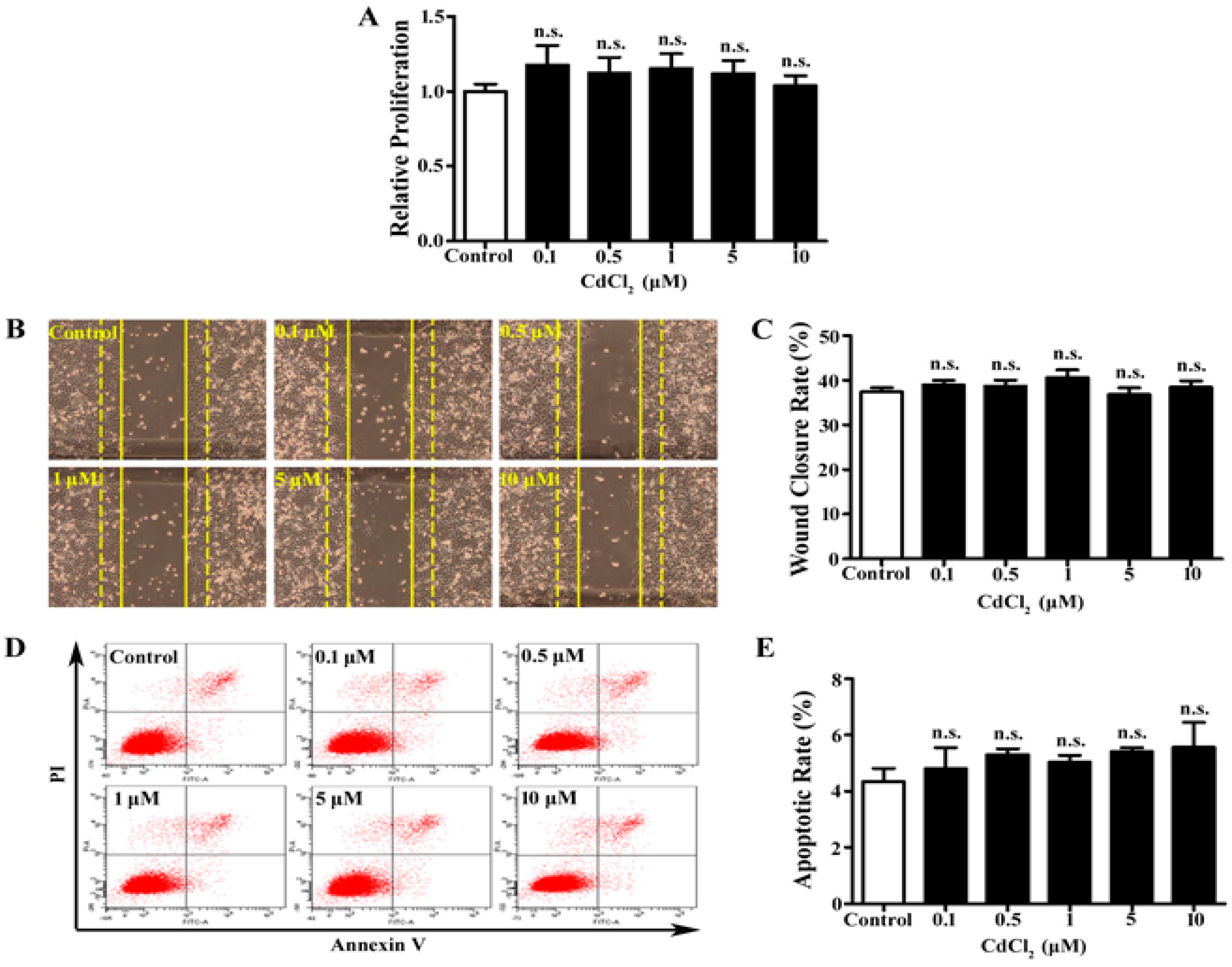
3.1. Low Dose of Cd Has No Significant Effect on Proliferation, Migration, and Apoptosis of A549 Cells
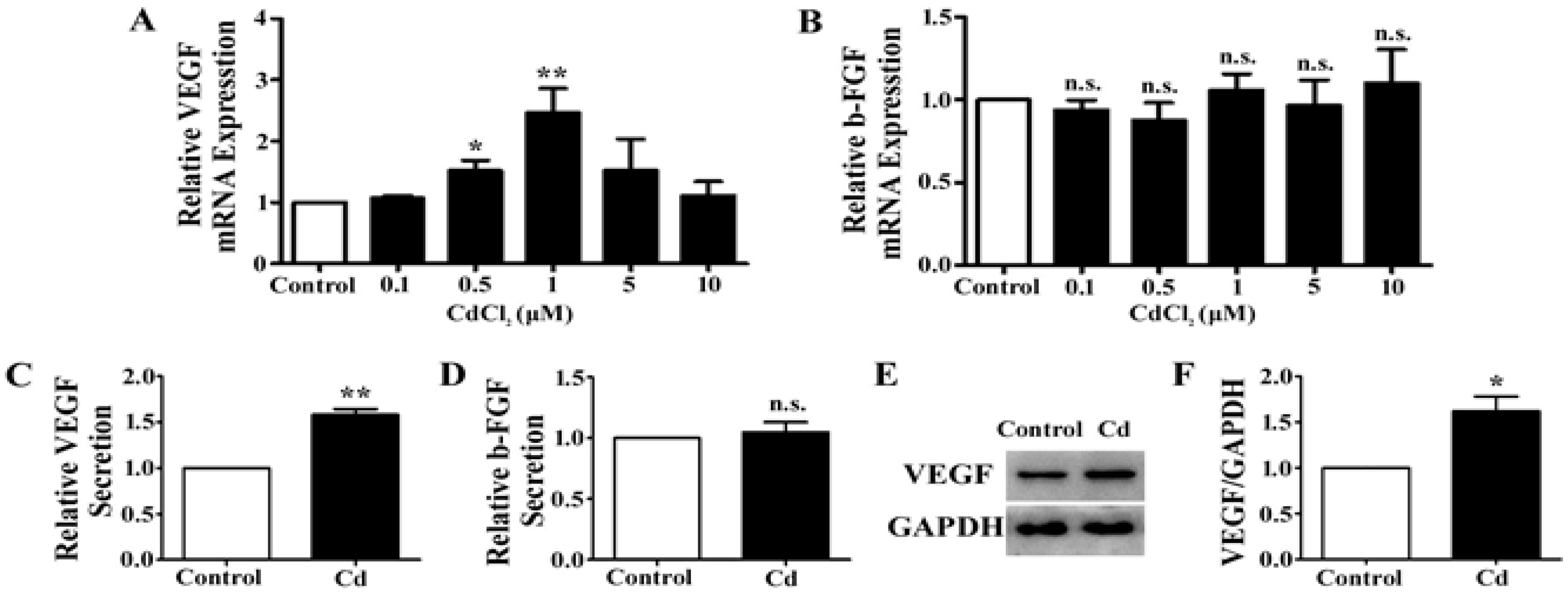
3.2. Low-Dose Cd Upregulates VEGF but Not b-FGF Expression in A549 Cells
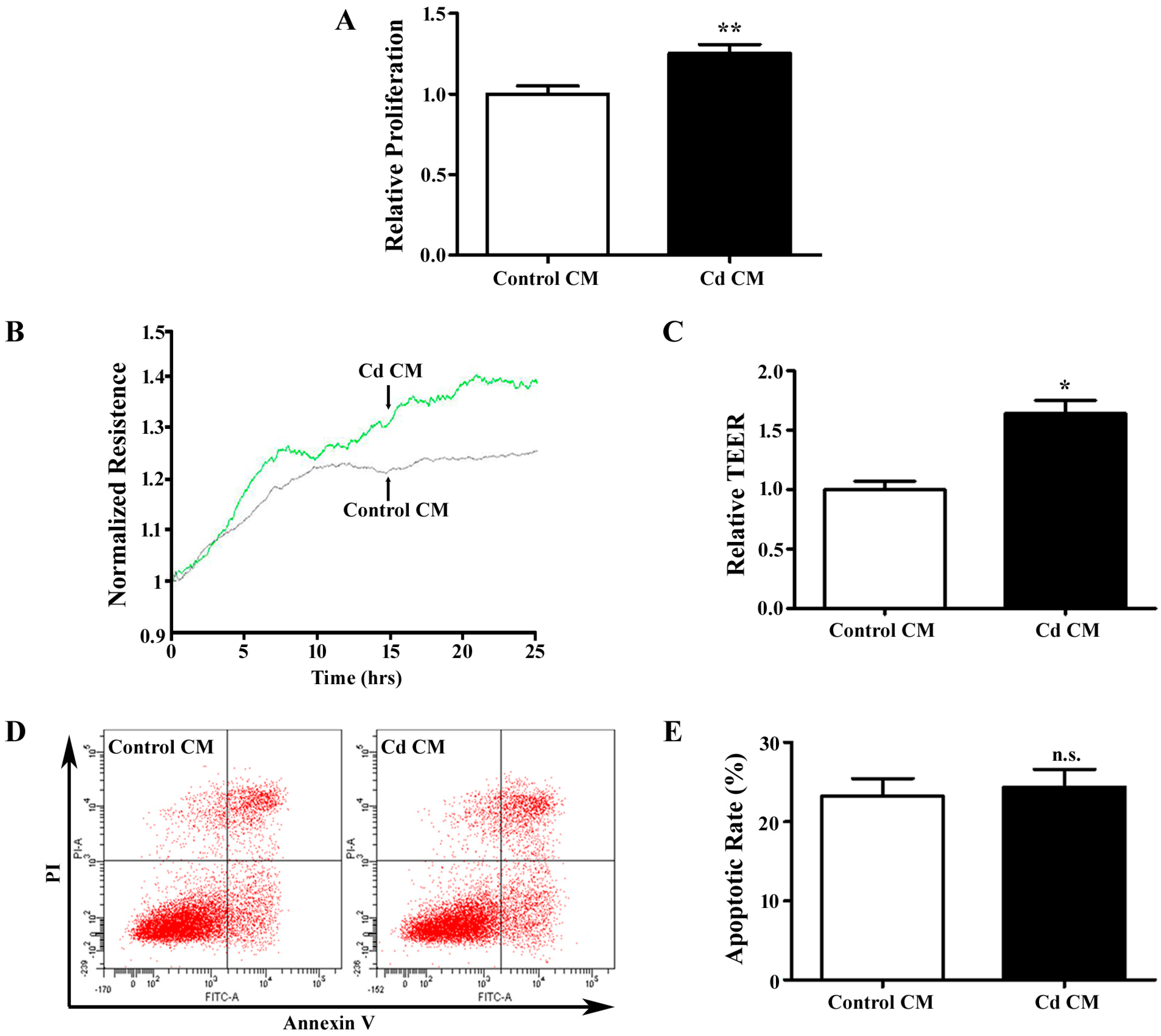
3.3. Conditioned Media Derived from Cd-Treated A549 Cells Promotes Endothelial Cell Proliferation and Migration
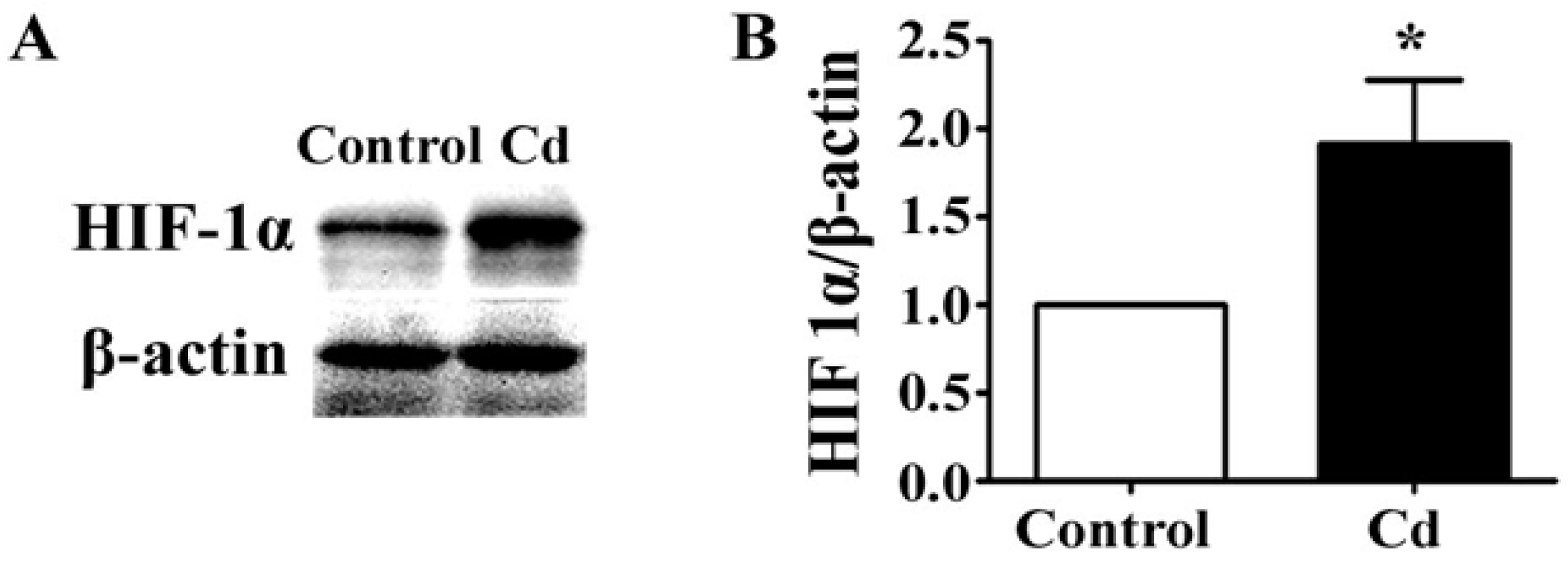
3.4. Low-Dose Cd Increases HIF-1α in A549 Cells
4. Discussion
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Kadara, H.; Kabbout, M.; Wistuba, II. Pulmonary adenocarcinoma: A renewed entity in 2011. Respirology 2012, 17, 50–65. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.; Ginsberg, R.J.; Ruckdeschel, J.C. Small-cell lung cancer. Chest Surg. Clin. N. Amer. 2001, 11, 165–188. [Google Scholar]
- Ren, Y.; Li, Y.; Tian, D. Role of the abce1 gene in human lung adenocarcinoma. Oncol. Rep. 2012, 27, 965–970. [Google Scholar] [PubMed]
- Jarup, L.; Berglund, M.; Elinder, C.G.; Nordberg, G.; Vahter, M. Health effects of cadmium exposure—A review of the literature and a risk estimate. Scand. J. Work Environ. Health 1998, 24, 1–51. [Google Scholar] [PubMed]
- International Agency for Research on Cancer. Cadmium and cadmium compounds. IARC Monogr. Eval. Carcinog. Risks Hum. 1993, 58, 119–237. [Google Scholar]
- Jarup, L.; Akesson, A. Current status of cadmium as an environmental health problem. Toxicol. Appl. Pharmacol. 2009, 238, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Liu, L.Z.; Jiang, Y.; Zhu, Y.; Guo, N.L.; Barnett, J.; Rojanasakul, Y.; Agani, F.; Jiang, B.H. Cadmium increases Hif-1 and Vegf expression through ros, erk, and akt signaling pathways and induces malignant transformation of human bronchial epithelial cells. Toxicol. Sci. 2012, 125, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Guo, F.; Li, L.; Guo, L.; Hou, Y.; Hao, E.; Yan, S.; Allen, T.D.; Liu, J. Cadmium induces vascular permeability via activation of the p38 MAPK pathway. Biochem. Biophys. Res. Commun. 2014, 450, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Garrett, S.H.; Sens, M.A.; Sens, D.A. Cadmium, environmental exposure, and health outcomes. Environ. Health Perspect. 2010, 118, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P. Mechanisms of cadmium carcinogenesis. Toxicol. Appl. Pharmacol. 2009, 238, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Waisberg, M.; Joseph, P.; Hale, B.; Beyersmann, D. Molecular and cellular mechanisms of cadmium carcinogenesis. Toxicology 2003, 192, 95–117. [Google Scholar] [CrossRef]
- Kundu, S.; Sengupta, S.; Bhattacharyya, A. Egfr upregulates inflammatory and proliferative responses in human lung adenocarcinoma cell line (A549), induced by lower dose of cadmium chloride. Inhal. Toxicol. 2011, 23, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. Tumor angiogenesis: Therapeutic implications. N. Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [PubMed]
- Eberhard, A.; Kahlert, S.; Goede, V.; Hemmerlein, B.; Plate, K.H.; Augustin, H.G. Heterogeneity of angiogenesis and blood vessel maturation in human tumors: Implications for antiangiogenic tumor therapies. Cancer Res. 2000, 60, 1388–1393. [Google Scholar] [PubMed]
- Rofstad, E.K.; Halsor, E.F. Vascular endothelial growth factor, interleukin 8, platelet-derived endothelial cell growth factor, and basic fibroblast growth factor promote angiogenesis and metastasis in human melanoma xenografts. Cancer Res. 2000, 60, 4932–4938. [Google Scholar] [PubMed]
- Shibuya, M. Structure and function of VEGF/VEGF-receptor system involved in angiogenesis. Cell. Struct. Funct. 2001, 26, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Yuan, L. Inhibition of p38 MAPK reduces tumor conditioned medium-induced angiogenesis in co-cultured human umbilical vein endothelial cells and fibroblasts. Biosci. Biotechnol. Biochem. 2007, 71, 1162–1169. [Google Scholar] [CrossRef] [PubMed]
- Kolluru, G.K.; Tamilarasan, K.P.; Geetha Priya, S.; Durgha, N.P.; Chatterjee, S. Cadmium induced endothelial dysfunction: Consequence of defective migratory pattern of endothelial cells in association with poor nitric oxide availability under cadmium challenge. Cell. Biol. Int. 2006, 30, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Helmestam, M.; Stavreus-Evers, A.; Olovsson, M. Cadmium chloride alters MRNA levels of angiogenesis related genes in primary human endometrial endothelial cells grown in vitro. Reprod. Toxicol. 2010, 30, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Pacini, S.; Punzi, T.; Morucci, G.; Gulisano, M.; Ruggiero, M. A paradox of cadmium: A carcinogen that impairs the capability of human breast cancer cells to induce angiogenesis. J. Environ. Pathol. Toxicol. Oncol. 2009, 28, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lim, W.; Ko, Y.; Kwon, H.; Kim, S.; Kim, O.; Park, G.; Choi, H.; Kim, O. The effects of cadmium on vegf-mediated angiogenesis in huvecs. J. Appl. Toxicol. 2012, 32, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Dong, F.; Hou, Y.; Cai, W.; Zhou, X.; Huang, A.L.; Yang, M.; Allen, T.D.; Liu, J. Dihydroartemisinin inhibits vascular endothelial growth factor-induced endothelial cell migration by a p38 mitogen-activated protein kinase-independent pathway. J. Pharmacol. Exp. Ther. 2014, 8, 1707–1712. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Zhou, X.; Li, C.; Yan, S.; Deng, X.; Cao, Z.; Li, L.; Tang, B.; Allen, T.D.; Liu, J. Dihydroartemisinin targets Vegfr2 via the NF-KAPPAB pathway in endothelial cells to inhibit angiogenesis. Cancer Biol. Ther. 2014, 15, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Duan, W.; Xu, L.; Song, S.; Zhu, C.; Wu, L. Biphasic effect of cadmium on cell proliferation in human embryo lung fibroblast cells and its molecular mechanism. Toxicol. In Vitro 2009, 23, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Siewit, C.L.; Gengler, B.; Vegas, E.; Puckett, R.; Louie, M.C. Cadmium promotes breast cancer cell proliferation by potentiating the interaction between eralpha and C-JUN. Mol. Endocrinol. 2010, 24, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Kapron, C.M. Differential induction of map kinase signalling pathways by cadmium in primary cultures of mouse embryo limb bud cells. Reprod. Toxicol. 2010, 29, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Dong, F.; Xu, D.; Du, L.; Yan, S.; Hu, H.; Lobe, C.G.; Yi, F.; Kapron, C.M.; Liu, J. Short-term, low-dose cadmium exposure induces hyperpermeability in human renal glomerular endothelial cells. J. Appl. Toxicol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Farhat, F.S.; Tfayli, A.; Fakhruddin, N.; Mahfouz, R.; Otrock, Z.K.; Alameddine, R.S.; Awada, A.H.; Shamseddine, A. Expression, prognostic and predictive impact of VEGF and BFGF in non-small cell lung cancer. Crc Crit. Rev. Oncol. Hematol. 2012, 84, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yuan, L.; Molema, G.; Regan, E.; Janes, L.; Beeler, D.; Spokes, K.C.; Okada, Y.; Minami, T.; Oettgen, P.; et al. Vascular bed-specific regulation of the von willebrand factor promoter in the heart and skeletal muscle. Blood 2011, 117, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, J.A.; Jiang, B.H.; Iyer, N.V.; Agani, F.; Leung, S.W.; Koos, R.D.; Semenza, G.L. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Meth. Mol. Cell. Biol. 1996, 16, 4604–4613. [Google Scholar]
- Semenza, G.L.; Agani, F.; Booth, G.; Forsythe, J.; Iyer, N.; Jiang, B.H.; Leung, S.; Roe, R.; Wiener, C.; Yu, A. Structural and functional analysis of hypoxia-inducible factor 1. Kidney Int. 1997, 51, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Jin, P.; Ringertz, N.R. Cadmium induces transcription of proto-oncogenes C-JUN and C-MYC in rat l6 myoblasts. J. Biol. Chem. 1990, 265, 14061–14064. [Google Scholar] [PubMed]
- Fang, M.Z.; Mar, W.; Cho, M.H. Cadmium affects genes involved in growth regulation during two-stage transformation of BALB/3t3 cells. Toxicology 2002, 177, 253–265. [Google Scholar] [CrossRef]
- Hida, K.; Kawamoto, T.; Ohga, N.; Akiyama, K.; Hida, Y.; Shindoh, M. Altered angiogenesis in the tumor microenvironment. Pathol. Int. 2011, 61, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Ilhan, N.; Ilhan, N.; Deveci, F. Functional significance of vascular endothelial growth factor and its receptor (Receptor-1) in various lung cancer types. Clin. Biochem. 2004, 37, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Vascular endothelial growth factor as a target for anticancer therapy. The oncologist 2004, 9, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of Vegf and its receptors. Nature Med. 2003, 9, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Alon, T.; Hemo, I.; Itin, A.; Pe’er, J.; Stone, J.; Keshet, E. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nature Med. 1995, 1, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Deutsch, U.; Jeong, J.; Lobe, C.G. Constitutive notch signaling in adult transgenic mice inhibits BFGF-induced angiogenesis and blocks ovarian follicle development. Genesis 2014, 52, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Shenberger, J.S.; Zhang, L.; Powell, R.J.; Barchowsky, A. Hyperoxia enhances VEGF release from a549 cells via post-transcriptional processes. Free Radical Biol. Med. 2007, 43, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Brekken, R.A.; Thorpe, P.E. Vascular endothelial growth factor and vascular targeting of solid tumors. Anticancer Res. 2001, 21, 4221–4229. [Google Scholar] [PubMed]
- Prozialeck, W.C.; Edwards, J.R.; Woods, J.M. The vascular endothelium as a target of cadmium toxicity. Life Sci. 2006, 79, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, F.; Wang, B.; Li, L.; Dong, F.; Chen, X.; Li, Y.; Dong, X.; Wada, Y.; Kapron, C.M.; Liu, J. Low-Dose Cadmium Upregulates VEGF Expression in Lung Adenocarcinoma Cells. Int. J. Environ. Res. Public Health 2015, 12, 10508-10521. https://doi.org/10.3390/ijerph120910508
Liu F, Wang B, Li L, Dong F, Chen X, Li Y, Dong X, Wada Y, Kapron CM, Liu J. Low-Dose Cadmium Upregulates VEGF Expression in Lung Adenocarcinoma Cells. International Journal of Environmental Research and Public Health. 2015; 12(9):10508-10521. https://doi.org/10.3390/ijerph120910508
Chicago/Turabian StyleLiu, Fuhong, Bei Wang, Liqun Li, Fengyun Dong, Xiaocui Chen, Yan Li, Xiuzhen Dong, Youichiro Wada, Carolyn M. Kapron, and Ju Liu. 2015. "Low-Dose Cadmium Upregulates VEGF Expression in Lung Adenocarcinoma Cells" International Journal of Environmental Research and Public Health 12, no. 9: 10508-10521. https://doi.org/10.3390/ijerph120910508
APA StyleLiu, F., Wang, B., Li, L., Dong, F., Chen, X., Li, Y., Dong, X., Wada, Y., Kapron, C. M., & Liu, J. (2015). Low-Dose Cadmium Upregulates VEGF Expression in Lung Adenocarcinoma Cells. International Journal of Environmental Research and Public Health, 12(9), 10508-10521. https://doi.org/10.3390/ijerph120910508






