The Association between Noise, Cortisol and Heart Rate in a Small-Scale Gold Mining Community—A Pilot Study
Abstract
:1. Introduction
2. Experimental Section
2.1. Overview
2.2. Site Description and Interview Process
2.3. Stress and Noise
2.4. Analysis
3. Results
3.1. Demographics
| Total | Female | Male | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||||
| 34.1 | 10.3 | 32.5 | 9.8 | 36.0 | 11.1 | ||||
| Years Lived in Kejetia | 10.5 | 6.7 | 10.1 | 6.9 | 11.0 | 6.7 | |||
| N | % | n | % | n | % | ||||
| Gender | 22 | - | 12 | 55% | 10 | 45% | |||
| Highest Level of Education Completed | |||||||||
| No School | 6 | 27% | 3 | 14% | 3 | 14% | |||
| Primary | 7 | 32% | 5 | 23% | 2 | 9% | |||
| Middle | 5 | 23% | 3 | 14% | 2 | 9% | |||
| Secondary | 3 | 14% | 0 | 0% | 3 | 14% | |||
| Post-Secondary | 0 | 0% | 0 | 0% | 0 | 0% | |||
| Missing | 1 | 5% | 1 | 5% | 0 | 0% | |||
| Occupation | |||||||||
| Current miner | 13 | 59% | 4 | 18% | 9 | 41% | |||
| Non-miner | 9 | 41% | 7 | 32% | 2 | 9% | |||
| Missing | 0 | 0% | 0 | 0% | 0 | 0 | |||
3.2. Perceived Stress
3.3. Salivary Cortisol
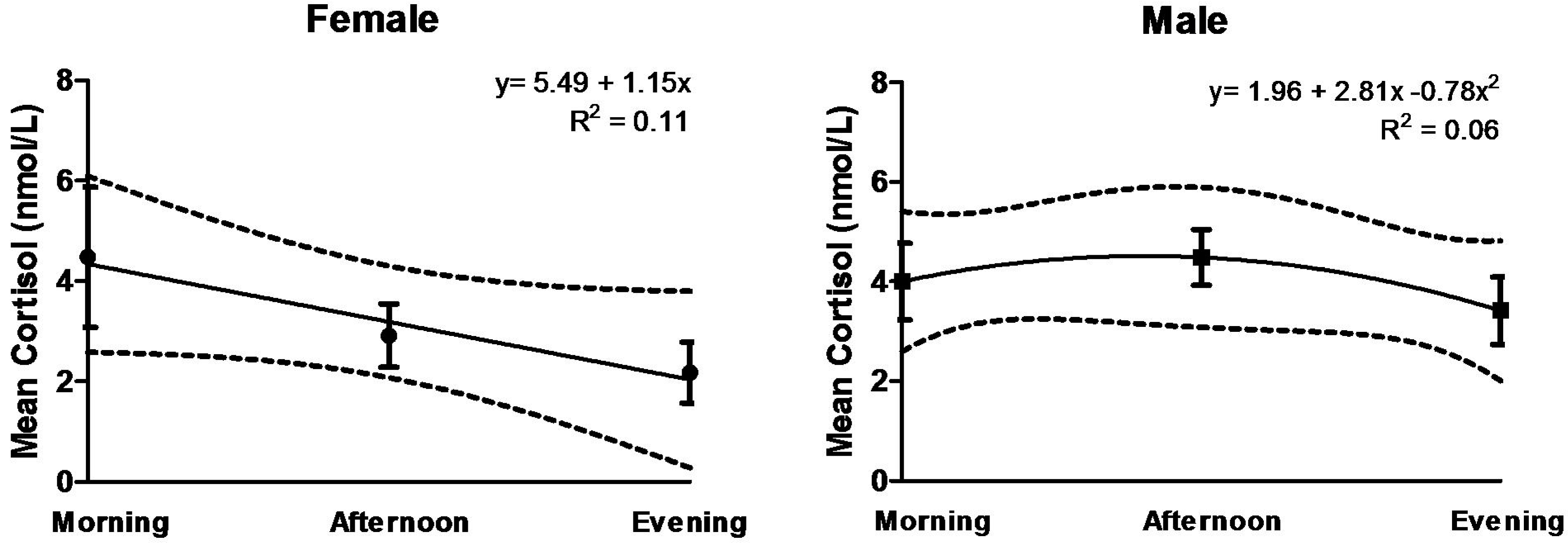
3.4. Heart Rate
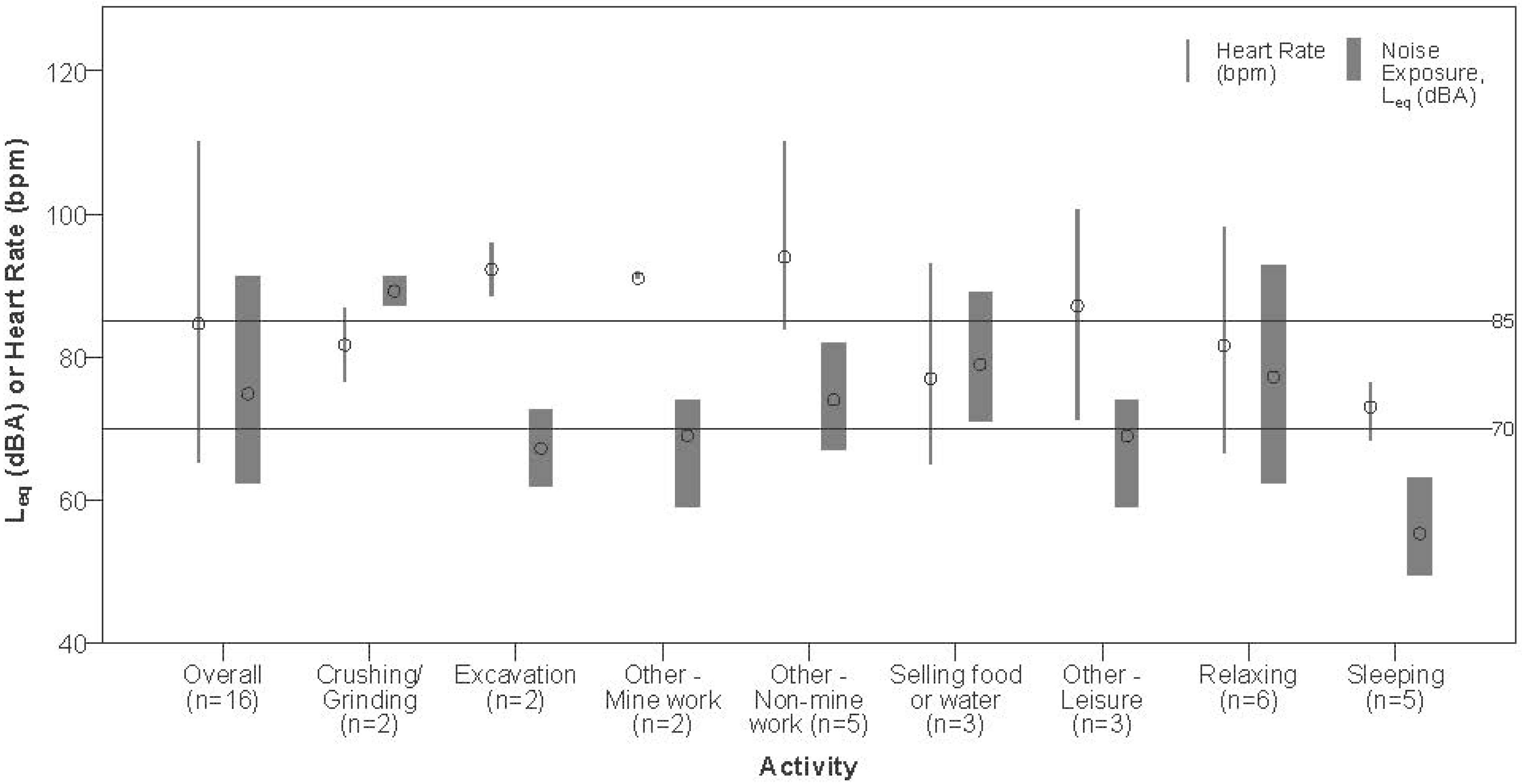
3.5. Noise
| Total | Female | Male | Miner | Non-miner | Dosimetry measurement | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leq (dBA) | Leq (dBA) | Leq (dBA) | Leq (dBA) | Leq (dBA) | Duration (hr) | |||||||||||||
| N | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | ||||||
| Daily | 22 | 82.2 | 7.3 | 82.2 | 4.4 | 82.3 | 10.0 | 82.8 | 8.8 | 81.5 | 4.6 | 22.2 | 1.9 | |||||
| Activity | ||||||||||||||||||
| Leisure | 17 | 81.9 * | 8.2 | 83.0 | 3.8 | 80.9 | 11.0 | 81.7 | 9.6 | 82.2 | 4.2 | 6.0 | 5.0 | |||||
| Work | 19 | 86.1 * | 5.2 | 85.2 | 5.5 | 87.3 | 5.6 | 87.6 | 5.2 | 83.9 | 5.5 | 10.3 | 5.0 | |||||
| Sleeping | 21 | 65.0 | 11.1 | 61.1 | 8.3 | 69.3 | 12.5 | 68.7 | 11.6 | 58.9 | 7.2 | 7.9 | 2.3 | |||||
| Non-mine work | 13 | 80.1 | 16.3 | 85.2 | 5.8 | 63.4 | 30.1 | 74.1 | 25.9 | 83.9 | 5.5 | 9.4 | 5.2 | |||||
| Mine work | 7 | 89.4 | 3.6 | 87.8 | 4.2 | 90.1 | 3.6 | 89.4 | 3.6 | - | - | 8.8 | 4.9 | |||||
| Grinding or crushing | 3 | 92.4 | 2.0 | - | - | 92.4 | 2.0 | 92.4 | 2.0 | - | - | 10.1 | 7.1 | |||||
| Sifting or shanking | 2 | 89.0 | 2.5 | 89.0 | 2.5 | - | - | 89.0 | 2.5 | - | - | 3.9 | 2.2 | |||||
| Excavation | 3 | 84.2 | 3.0 | - | - | 84.2 | 3.0 | 84.2 | 3.0 | - | - | 8.4 | 3.4 | |||||
| Variable | Unstandardized Coefficients | 95% CI |
|---|---|---|
| Intercept | −16.8 | −32.6–1.02 |
| Age (Years) | −0.06 | −0.15–0.02 |
| Sex (Male) | −0.51 | −2.37–1.35 |
| Smoker (Yes) | 0.71 | −2.21–3.63 |
| Times Between Cortisol Samples (Hours) | −0.34 | −0.76–0.09 |
| Leq (dBA) | 0.25 | 0.08–0.42 |
3.6. Salivary Cortisol and Noise
3.7. Heart Rate and Noise
| Variable | Unstandardized Coefficients | 95% CI |
|---|---|---|
| Intercept | 57.5 | 37.2–77.7 |
| Age (Years) | 0.24 | −0.31–0.78 |
| Sex (Male) | 5.37 | −16.5–6.84 |
| Leq (dBA) | 0.29 | 0.26–0.32 |
| Variable | Unstandardized Coefficients | 95% CI |
|---|---|---|
| Intercept | 3.1 | −5.4–11.8 |
| Age (Years) | 0.5 | −0.2–0.1 |
| Sex (Male) | 1.1 | −2.5–4.7 |
| Leq (dBA) | 0.5 | 0.04–0.9 |
4. Discussion
4.1. Noise
4.2. Stress
4.3. Limitations
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgements
Author Contributions
Conflicts of Interest
References and Notes
- Aryee, B.N.; Ntibery, B.K.; Atorkui, E. Trends in the small-scale mining of precious minerals in Ghana: A perspective on its environmental impact. J. Clean. Prod. 2003, 11, 131–140. [Google Scholar] [CrossRef]
- Grandjean, P.; Landrigan, P.J. Developmental neurotoxicity of industrial chemicals. Lancet 2006, 368, 2167–2178. [Google Scholar] [CrossRef]
- Basu, N.; Abare, M.; Buchanan, S.; Cryderman, D.; Nam, D.-H.; Sirkin, S.; Schmitt, S.; Hu, H. A combined ecological and epidemiologic investigation of metal exposures amongst Indigenous peoples near the Marlin Mine in Western Guatemala. Sci. Total Environ. 2010, 409, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Hilson, G. A Contextual Review of the Ghanaian Small-Scale Mining Industry; Imperial College Centre for Environmental Technology: London, UK, 2002; Vol. 76. [Google Scholar]
- UNEP Chemicals. Global Mercury Assessment; UNEP: Geneva, Switzerland, 2002. [Google Scholar]
- Awumbila, M.; Tsikata, D. Migration of Small Scale Gold Miners in North-Eastern Ghana: Implications for Sustainable Rural Livelihoods; University of Ghana: Accra, Ghana, 2004. [Google Scholar]
- Barry, M. Regularizing Informal Mining: A Summary of the Proceedings of the International Roundtable on Artisanal Mining; World Bank: Washington, DC, USA, 1996. [Google Scholar]
- Agyemang, I. Analysis of the socio-economic and cultural implications of environmental degradation in Northern Ghana using qualitative approach. Afr. J. Hist. Cult. 2011, 3, 113–122. [Google Scholar]
- Hoadley, M.; Limpitlaw, D. The artisanal and small scale mining sector and sustainable livelihoods. In Proceedings of Mintek Small Scale Mining Conference, Johanesburg, South Africa, 9 September 2004; pp. 1–9.
- Hinton, J.J.; Veiga, M.M.; Beinhoff, C. Women and artisanal mining : Gender roles and the road ahead. In The Socio-Economic Impacts of Artisonal and Small-Scale Mining in Developing Countries; Hilson, G.M., Ed.; A.A. Balkema, Swets & Zeitlinger Publishers: Lisse, Netherlands, 2003. [Google Scholar]
- Bairey Merz, C.N.; Dwyer, J.; Nordstrom, C.K.; Walton, K.G.; Salerno, J.W.; Schneider, R.H. Psychosocial stress and cardiovascular disease: Pathophysiological links. Behav. Med. 2002, 27, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Von Känel, R. Psychosocial stress and cardiovascular risk : Current opinion. Swiss Med. Wkly. 2012, 142. [Google Scholar] [CrossRef]
- Rosengren, A.; Hawken, S.; Ounpuu, S.; Sliwa, K.; Zubaid, M.; Almahmeed, W.; Blackett, K.N.; Sitthi-amorn, C.; Sato, H.; Yusuf, S. Association of psychosocial risk factors with risk of acute myocardial infarction in 11119 cases and 13648 controls from 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 953–962. [Google Scholar] [CrossRef]
- McEwen, B.S.; Gianaros, P.J. Central role of the brain in stress and adaptation: Links to socioeconomic status, health, and disease. Ann. NY Acad. Sci. 2010, 1186, 190–222. [Google Scholar] [CrossRef] [PubMed]
- Spruill, T.M. Chronic psychosocial stress and hypertension. Curr. Hypertens. Rep. 2010, 12, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Gouin, J.-P. Chronic stress, immune dysregulation, and health. Am. J. Lifestyle Med. 2011, 5, 476–485. [Google Scholar] [CrossRef]
- Bigert, C.; Bluhm, G.; Theorell, T. Saliva cortisol—A new approach in noise research to study stress effects. Int. J. Hyg. Environ. Health 2005, 208, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Basner, M.; Babisch, W.; Davis, A.; Brink, M. Auditory and non-auditory effects of noise on health. Lancet 2014, 383, 1325–1332. [Google Scholar] [CrossRef]
- Passchier-Vermeer, W.; Passchier, W.F. Noise exposure and public health. Environ. Health Perspect. 2000, 108 (Suppl. 1), S123–S131. [Google Scholar] [CrossRef]
- Evans, G.W.; Lercher, P.; Meis, M.; Ising, H.; Kofler, W.W. Community noise exposure and stress in children. J. Acoust. Soc. Am. 2001, 109, 1023–1027. [Google Scholar] [CrossRef] [PubMed]
- Kraus, U.; Schneider, A.; Breitner, S.; Hampel, R.; Rückerl, R.; Pitz, M.; Geruschkat, U.; Belcredi, P.; Radon, K.; Peters, A. Individual daytime noise exposure during routine activities and heart rate variability in adults: A repeated measures study. Environ. Health Perspect. 2013, 121, 607–612. [Google Scholar] [PubMed]
- Dickerson, S.S.; Kemeny, M.E. Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychol. Bull. 2004, 130, 355–391. [Google Scholar] [CrossRef] [PubMed]
- Renne, E.; Basu, N.; Gager, E.; Koomson, E.; Lee, B.; Lee, S.; Leeth, A.; Manigault, D., III; Rajaee, M.; Sajjad, A.; Smith, M.; Yee, A. Back to traditional Aboriginal food. Women Environments International Magazine 2011, 37, 10–13. [Google Scholar]
- Long, R.; Sun, K.; Neitzel, R.L. Injury risk factors in a small-scale gold mining community in Ghana’s Upper East Region. Int. J. Environ. Res. Public Health 2015, 12, 8744–8761. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Addo, A.A.; Marquis, G.S.; Lartey, A.A.; Pérez-escamilla, R.; Mazur, R.E.; Harding, K.B. Food insecurity and perceived stress but not HIV infection are independently associated with lower energy intakes among lactating Ghanaian women. Matern. Child Nutr. 2011, 7, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Smyth, N.; Hucklebridge, F.; Thorn, L.; Evans, P.; Clow, A. Salivary cortisol as a biomarker in social science research. Soc. Personal. Psychol. Compass 2013, 7, 605–625. [Google Scholar] [CrossRef]
- American National Standards Institute. American National Standard Specification for Personal Noise Dosimeters; American National Standards Institute: Washington, DC, USA, 1991; ANSI S1.25-1991 (R2007). [Google Scholar]
- Hornung, R.W. Statistical evaluation of exposure assessment strategies. Appl. Occup. Environ. Hyg. 1991, 6, 516–520. [Google Scholar] [CrossRef]
- Guidelines for Community Noise; Berglund, B.; Lindvall, T.; Schwela, D.H. (Eds.) World Health Organization: Geneva, Switzerland, 1999.
- Nelson, D.I.; Nelson, R.Y.; Concha-Barrientos, M.; Fingerhut, M. The global burden of occupational noise-induced hearing loss. Am. J. Ind. Med. 2005, 48, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Amedofu, G.K. Hearing-impairment among workers in a surface gold mining company in Ghana. Afr. J. Health Sci. 2002, 9, 91–97. [Google Scholar] [CrossRef] [PubMed]
- NIOSH. Criteria for a Recommended Standard: Occupational Noise Exposure, Revised Criteria 1998; NIOSH: Cincinnati, OH, USA, 1998. [Google Scholar]
- Saunders, J.E.; Jastrzembski, B.G.; Buckey, J.C.; Enriquez, D.; MacKenzie, T.; Karagas, M.R. Hearing loss and heavy metal toxicity in a Nicaraguan mining community: Audiological results and case reports. Audiol. Neurotol. 2013, 18, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Harding, A.-H.; Frost, G.; Tan, E.; Tsuchiya, A.; Mason, H.M. The cost of hypertension-related ill-health attributable to environmental noise. Noise Health 2013, 15, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Van Kamp, I.; Davies, H. Noise and health in vulnerable groups: A review. Noise Health 2013, 15, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Haines, M.M.; Stansfeld, S.; Job, R.F.S.; Berglund, B.; Head, J. Chronic aircraft noise exposure, stress responses, mental health and cognitive performance in school children. Psychol. Med. 2001, 31, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Babisch, W. Cardiovascular effects of noise. Noise Health 2011, 13, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Eisler, R. Health risks of gold miners: A synoptic review. Environ. Geochem. Health 2003, 25, 325–345. [Google Scholar] [CrossRef] [PubMed]
- Eller, N.H.; Netterstrøm, B.; Hansen, A.M. Psychosocial factors at home and at work and levels of salivary cortisol. Biol. Psychol. 2006, 73, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Melamed, S.; Ugarten, U.; Shirom, A.; Kahana, L.; Lerman, Y.; Froom, P. Chronic burnout, somatic arousal and elevated salivary cortisol levels. J. Psychosom. Res. 1999, 46, 591–598. [Google Scholar] [CrossRef]
- Selander, J.; Bluhm, G.; Theorell, T.; Pershagen, G.; Babisch, W.; Seiffert, I.; Houthuijs, D.; Breugelmans, O.; Vigna-Taglianti, F.; Antoniotti, M.C.; Velonakis, E.; Davou, E.; Dudley, M.-L.; Järup, L. Saliva cortisol and exposure to aircraft noise in six European countries. Environ. Health Perspect. 2009, 117, 1713–1717. [Google Scholar] [CrossRef] [PubMed]
- Fouladi, D.B.; Nassiri, P.; Monazzam, E.M.; Farahani, S.; Hassanzadeh, G.; Hoseini, M. Industrial noise exposure and salivary cortisol in blue collar industrial workers. Noise Health 2012, 14, 184–189. [Google Scholar] [PubMed]
- Maina, G.; Palmas, A.; Bovenzi, M. Salivary cortisol and psychosocial hazards at work. Am. J. Ind. Med. 2009, 260, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Miki, K.; Kawamorita, K.; Araga, Y.; Musha, T.; Sudo, A. Urinary and Salivary stress hormone levels while performing arithmetic calculationin a noisy environment. Ind. Health 1998, 36, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Sjödin, F.; Kjellberg, A.; Knutsson, A.; Landström, U.; Lindberg, L. Noise and stress effects on preschool personnel. Noise Health 2012, 14, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Stokholm, Z.A.; Hansen, Å.M.; Grynderup, M.B.; Bonde, J.P.; Christensen, K.L.; Frederiksen, T.W.; Lund, S.P.; Vestergaard, J.M.; Kolstad, H.A. Recent and long-term occupational noise exposure and salivary cortisol level. Psychoneuroendocrinology 2014, 39, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Kudielka, B.M.; Hellhammer, D.H.; Wüst, S. Why do we respond so differently? Reviewing determinants of human salivary cortisol responses to challenge. Psychoneuroendocrinology 2009, 34, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, S.J.; Viega, M.M. Global Impacts of Mercury Supply and Demand in Small-Scale Gold Mining: Report to the UNEP Governing Council meeting; UNEP: Nairobi, Kenya, 2006. [Google Scholar]
- Tschakert, P. Digging deep for justice: A radical re-imagination of the artisanal gold mining sector in Ghana. Antipode 2009, 41, 706–740. [Google Scholar] [CrossRef]
- Maconachie, R.; Hilson, G.M. Safeguarding livelihoods or exacerbating poverty? Artisanal mining and formalization in West Africa. Nat. Resour. Forum 2011, 35, 293–303. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Green, A.; Jones, A.D.; Sun, K.; Neitzel, R.L. The Association between Noise, Cortisol and Heart Rate in a Small-Scale Gold Mining Community—A Pilot Study. Int. J. Environ. Res. Public Health 2015, 12, 9952-9966. https://doi.org/10.3390/ijerph120809952
Green A, Jones AD, Sun K, Neitzel RL. The Association between Noise, Cortisol and Heart Rate in a Small-Scale Gold Mining Community—A Pilot Study. International Journal of Environmental Research and Public Health. 2015; 12(8):9952-9966. https://doi.org/10.3390/ijerph120809952
Chicago/Turabian StyleGreen, Allyson, Andrew D. Jones, Kan Sun, and Richard L. Neitzel. 2015. "The Association between Noise, Cortisol and Heart Rate in a Small-Scale Gold Mining Community—A Pilot Study" International Journal of Environmental Research and Public Health 12, no. 8: 9952-9966. https://doi.org/10.3390/ijerph120809952
APA StyleGreen, A., Jones, A. D., Sun, K., & Neitzel, R. L. (2015). The Association between Noise, Cortisol and Heart Rate in a Small-Scale Gold Mining Community—A Pilot Study. International Journal of Environmental Research and Public Health, 12(8), 9952-9966. https://doi.org/10.3390/ijerph120809952






