Variability and Stability in Daily Moderate-to-Vigorous Physical Activity among 10 Year Old Children
Abstract
:1. Introduction
2. Material and Methods
2.1. Sample
2.2. Anthropometry
2.3. Maturity Offset
2.4. Physical Activity
2.5. Data Analysis
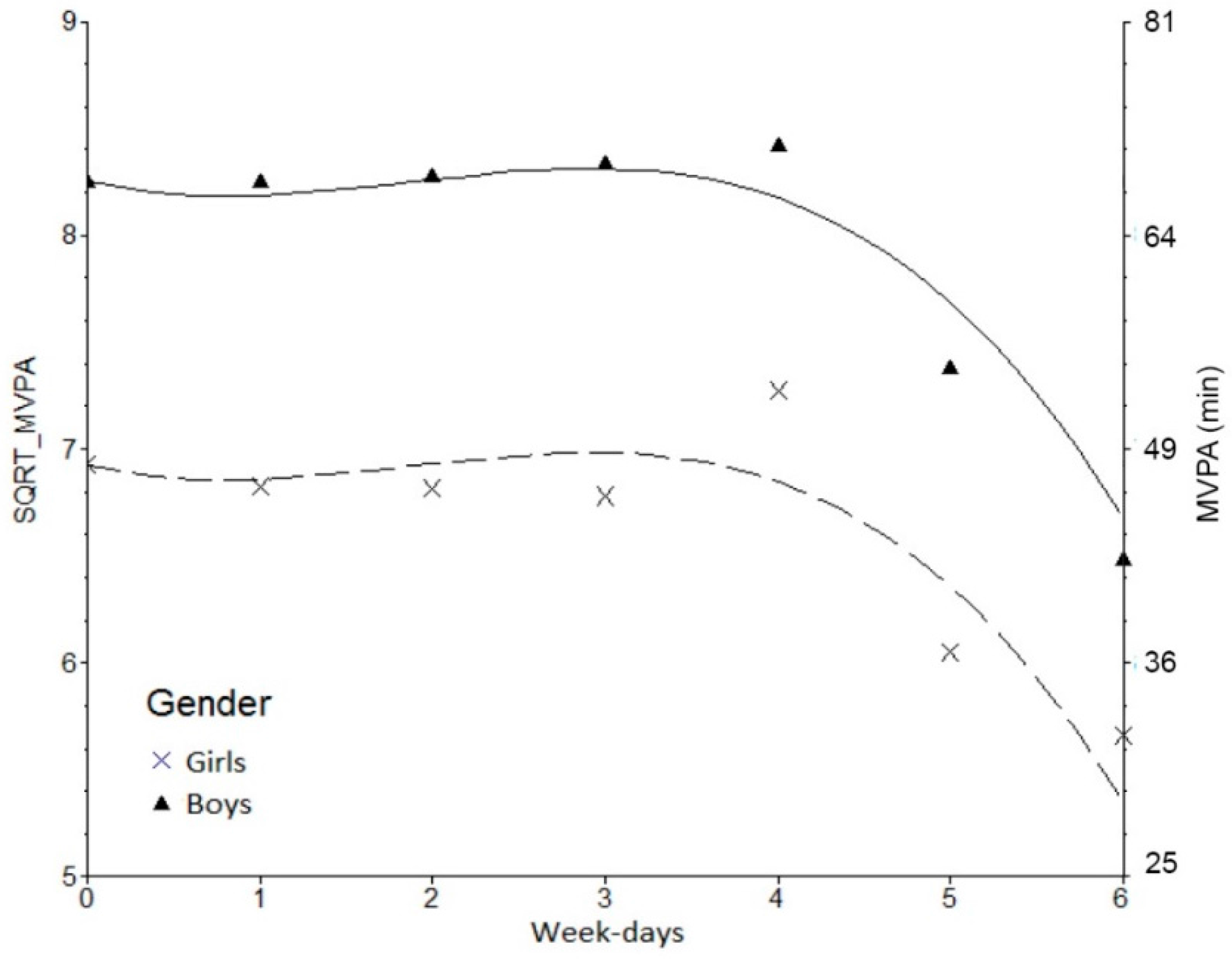
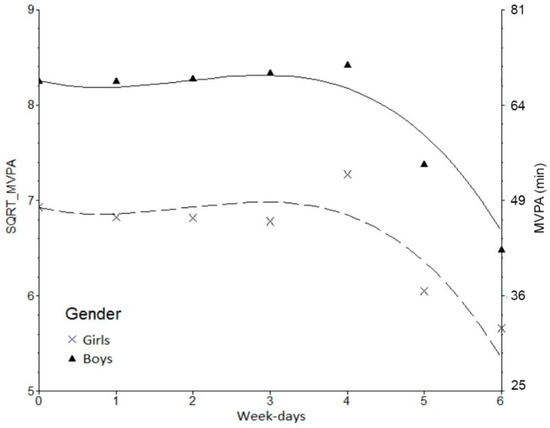
3. Results
| Boys M ± SD | Girls M ± SD | Total M ± SD | t | p-Value | |
|---|---|---|---|---|---|
| Height (cm) | 143.46 ± 6.42 | 143.49 ± 7.06 | 143.47 ± 6.78 | 0.60 | 0.952 |
| Weight (kg) | 40.52 ± 9.23 | 40.29 ± 9.23 | 40.39 ± 9.23 | −0.33 | 0.740 |
| BMI (kg·m−2) | 19.54 ± 3.36 | 19.41 ± 3.36 | 19.47 ± 3.40 | −0.51 | 0.610 |
| Maturity offset (years to PHV) | −2.73 ± 0.43 | −1.22 ± 0.53 | −1.90 ± 0.89 | 41.03 | <0.001 |
| Weight status | n (%) | n (%) | n (%) | χ2 | p-value |
| Normal weight | 151 (49.5%) | 221 (58.0%) | 372 (54.2%) | 2.899 | 0.004 |
| Overweight | 46 (15.1%) | 67 (17.6%) | 113 (16.5%) | ||
| Obese | 108 (35.4%) | 93 (24.4%) | 201 (29.3%) |
| Monday | Tuesday | Wednesday | Thursday | Friday | Saturday | Sunday | |
|---|---|---|---|---|---|---|---|
| Boys | |||||||
| Mdn (IQR) | 68.88 (49.38–91.88) | 67.00 (48.25–92.25) | 68.50 (48.50–91.00) | 69.50 (47.50–97.50) | 68.75 (49.75–94.50) | 50.00 (32.25–82.50) | 37.50 (22.50–63.50) |
| M (SD) | 71.88 (32.60) | 72.45 (35.60) | 72.63 (33.83) | 73.90 (35.26) | 74.52 (33.78) | 60.64 (38.87) | 47.36 (33.53) |
| CV | 0.45 | 0.49 | 0.47 | 0.48 | 0.45 | 0.64 | 0.71 |
| Girls | |||||||
| Mdn (IQR) | 48.88 (32.75–63.25) | 46.25 (31.75–63.25) | 46.75 (33.50–62.00) | 44.25 (30.50–64.25) | 53.00 (38.25–67.50) | 35.00 (22.25–51.25) | 28.75 (18.00–45.00) |
| M (SD) | 50.84 (24.17) | 49.52 (24.42) | 48.95 (22.24) | 48.87 (23.94) | 55.67 (25.77) | 40.04 (24.20) | 35.69 (25.44) |
| CV | 0.48 | 0.49 | 0.45 | 0.49 | 0.46 | 0.60 | 0.71 |
| %Rb | 60.8% | 59.5% | 60.5% | 59.2% | 60.5% | 42.1% | 28.4% |
| %Rg | 28.4% | 28.8% | 27.7% | 28.4% | 36.6% | 18.3% | 14.9% |
| Model 1 | Model 2 | Model 3 | |
|---|---|---|---|
| Fixed effects | |||
| Intercept | 7.515 ± 0075, p < 0.001 | 7.204 ± 0.189, p < 0.001 | 7.207 ± 0.194, p < 0.001 |
| Linear | −0.194 ± 0.093, p = 0.037 | −0.195 ± 0.092, p = 0.036 | −0.263 ± 0.010, p = 0.008 |
| Quadratic | 0.153 ± 0.037, p < 0.001 | 0.153 ± 0.037, p < 0.001 | 0.170 ± 0.038, p < 0.001 |
| Cubic | −0.027 ± 0.004, p < 0.001 | −0.027 ± 0.004, p < 0.001 | −0.027 ± 0.004, p < 0.001 |
| Sex | 1.588 ± 0.204, p < 0.001 | 1.580 ± 0.224, p < 0.001 | |
| Overweight | −0.077 ± 0.138, p = 0.574 | −0.079 ± 0.138, p = 0.567 | |
| Obese | −0.418 ± 0.130, p = 0.001 | −0.417 ± 0.130, p = 0.001 | |
| Maturity offset | 0.136 ± 0.116, p = 0.239 | 0.136 ± 0.116, p = 0.240 | |
| Linear-by-Sex | 0.155 ± 0.084, p = 0.067 | ||
| Quadratic-by-Sex | −0.038 ± 0.013, p = 0.004 | ||
| Variance components | |||
| Intercept | 1.492 ± 0.111, p < 0.001 | 1.017 ± 0.863, p < 0.001 | 1.14 ± 0.10, p < 0.001 |
| Linear | 0.015 ± 0.004, p = 0.051 | 0.019 ± 0.044, p < 0.001 | 0.02 ± 0.01, p = 0.110 |
| Residual | 2.462 ± 0.058 | 2.450 ± 0.058 | 2.39 ± 0.06 |
| Deviance | 18,690.16 | 18,506.24 | 18,489.41 |
| N° of estimated parameters | 7 | 11 | 13 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Recommendation on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Janssen, I.; LeBlanc, A.G. Systematic review of the health benefits of physical activity and fitness in school-aged children and youth. Int. J. Behav. Nutr. Phys. Activ. 2010, 7, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Hind, K.; Burrows, M. Review: Weight-bearing exercise and bone mineral accrual in children and adolescents: A review of controlled trials. Bone 2007, 40, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Ness, A.R.; Leary, S.D.; Mattocks, C.; Blair, S.N.; Reilly, J.J.; Wells, J.; Ingle, S.; Tilling, K.; Smith, G.D.; Riddoch, C. Objectively measured physical activity and fat mass in a large cohort of children. PLoS Med. 2007, 4, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.B.; Harro, M.; Sardinha, L.B.; Froberg, K.; Ekelund, U.; Brage, S.; Anderssen, S.A. Physical activity and clustered cardiovascular risk in children: A cross-sectional study (the European Youth Heart Study). Lancet 2006, 368, 299–304. [Google Scholar] [CrossRef]
- Ferreira, A.P.; Oliveira, C.E.R.; França, N.M. Metabolic syndrome and risk factors for cardiovascular disease in obese children: The relationship with insulin resistance (HOMA-IR). J. Pediatr. 2007, 83, 21–26. [Google Scholar] [CrossRef]
- Westerterp, K.R.; Speakman, J.R. Physical activity energy expenditure has not declined since the 1980s and matches energy expenditures of wild mammals. Int. J. Obes. (Lond.) 2008, 32, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Ekelund, U.; Tomkinson, G.; Armstrong, N. What proportion of youth are physically active? Measurement issues, levels and recent time trends. Brit. J. Sports Med. 2011, 45, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Brooke, H.L.; Corder, K.; Atkin, A.J.; van Sluijs, E.M. A systematic literature review with meta-analyses of within- and between-day differences in objectively measured physical activity in school-aged children. Sport. Med. 2014, 44, 1427–1438. [Google Scholar] [CrossRef] [PubMed]
- Strong, W.B.; Malina, R.M.; Blimkie, C.J.; Daniels, S.R.; Dishman, R.K.; Gutin, B.; Hergenroeder, A.C.; Must, A.; Nixon, P.A.; Pivarnik, J.M.; et al. Evidence based physical activity for school-age youth. J. Pediatr. 2005, 146, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Verloigne, M.; Van Lippevelde, W.; Maes, L.; Yildirim, M.; Chinapaw, M.; Manios, Y.; Androutsos, O.; Kovacs, E.; Bringolf-Isler, B.; Brug, J.; et al. Levels of physical activity and sedentary time among 10- to 12-year-old boys and girls across 5 European countries using accelerometers: An observational study within the Energy-Project. Int. J. Behav. Nutr. Phys. Act. 2012, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Basterfield, L.; Adamson, A.J.; Frary, J.K.; Parkinson, K.N.; Pearce, M.S.; Reilly, J.J. Longitudinal study of physical activity and sedentary behavior in children. Pediatrics 2011, 127, e24–e30. [Google Scholar] [CrossRef] [PubMed]
- Baptista, F.; Santos, D.A.; Silva, A.M.; Mota, J.; Santos, R.; Vale, S.; Ferreira, J.P.; Raimundo, A.M.; Moreira, H.; Sardinha, L.B. Prevalence of the Portuguese population attaining sufficient physical activity. Med. Sci. Sports Exerc. 2012, 44, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Prochaska, J.J.; Taylor, W.C. A review of correlates of physical activity of children and adolescents. Med. Sci. Sports Exerc. 2000, 32, 963–975. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, S.L.; Rhodes, R.E. Parental correlates of physical activity in children and early adolescents. Sports Med. 2006, 36, 79–97. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.; Van der Horst, K.; Wendel-Vos, W.; Kremers, S.; Van Lenthe, F.J.; Brug, J. Environmental correlates of physical activity in youth—A review and update. Obes. Rev. 2007, 8, 129–154. [Google Scholar] [CrossRef] [PubMed]
- Stanley, R.M.; Ridley, K.; Dollman, J. Correlates of children’s time-specific physical activity: A review of the literature. Int. J. Behav. Nutr. Phys. Act. 2012, 9, 50. [Google Scholar] [CrossRef] [PubMed]
- Erlandson, M.C.; Sherar, L.B.; Mosewich, A.D.; Kowalski, K.C.; Bailey, D.A.; Baxter-Jones, A.D. Does controlling for biological maturity improve physical activity tracking? Med. Sci. Sports Exerc. 2011, 43, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Anderssen, S.A.; Andersen, L.B.; Froberg, K.; Riddoch, C.; Sardinha, L.B.; Ekelund, U. Between- and within-day variability in physical activity and inactivity in 9- and 15-year-old European children. Scand. J. Med. Sci. Sports 2009, 19, 10–18. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, T.L.; Nader, P.R.; Strikmiller, P.K.; Yang, M.; Stone, E.J.; Perry, C.L.; Taylor, W.C.; Epping, J.N.; Feldman, H.A.; Luepker, R.V.; et al. School physical education: Effect of the child and adolescent trial for cardiovascular health. Prev. Med. 1996, 25, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, S.J.; Cardon, G.M.; De Clercq, D.L.; de Bourdeaudhuij, I.M. Increasing children’s physical activity levels during recess periods in elementary schools: The effects of providing game equipment. Eur. J. Public Health 2006, 16, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Ridgers, N.D.; Stratton, G.; Fairclough, S.J.; Twisk, J.W. Long-term effects of a playground markings and physical structures on children’s recess physical activity levels. Prev. Med. 2007, 44, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Telford, R.M.; Telford, R.D.; Cunningham, R.B.; Cochrane, T.; Davey, R.; Waddington, G. Longitudinal patterns of physical activity in children aged 8 to 12 years: The look study. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 81. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.; Hinkley, T.; Okely, A.D.; Salmon, J. Tracking physical activity and sedentary behavior in childhood: A systematic review. Am. J. Prev. Med. 2013, 44, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Dencker, M.; Tanha, T.; Wollmer, P.; Karlsson, M.K.; Andersen, L.B.; Thorsson, O. Tracking of physical activity with accelerometers over a 2-year time period. J. Phys. Act. Health 2013, 10, 241–248. [Google Scholar] [PubMed]
- Edwards, N.M.; Khoury, P.R.; Kalkwarf, H.J.; Woo, J.G.; Claytor, R.P.; Daniels, S.R. Tracking of accelerometer-measured physical activity in early childhood. Pediatr. Exerc. Sci. 2013, 25, 487–501. [Google Scholar] [PubMed]
- Katzmarzyk, P.T.; Barreira, T.V.; Broyles, S.T.; Champagne, C.M.; Chaput, J.P.; Fogelholm, M.; Hu, G.; Johnson, W.D.; Kuriyan, R.; Kurpad, A.; et al. The international study of childhood obesity, lifestyle and the environment (ISCOLE): Design and methods. BMC Publ. Health 2013, 13, 900. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Physical Status: The Use and Interpretation of Anthropometry. Report of a WHO Expert Committee. 1995. Available online: http://whqlibdoc.who.int/trs/WHO_TRS_854.pdf?ua=1 (accessed on 5 August 2015).
- Mirwald, R.L.; Baxter-Jones, A.D.; Bailey, D.A.; Beunen, G.P. An assessment of maturity from anthropometric measurements. Med. Sci. Sports Exerc. 2002, 34, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Vincent, S.D.; Pangrazi, R.P. An examination of the activity patterns of elementary school children. Pediatr. Exerc. Sci. 2002, 14, 432. [Google Scholar]
- Intille, S.S.; Lester, J.; Sallis, J.F.; Duncan, G. New horizons in sensor development. Med. Sci. Sports Exerc. 2012, 44, S24–S31. [Google Scholar] [CrossRef] [PubMed]
- Dossegger, A.; Ruch, N.; Jimmy, G.; Braun-Fahrlander, C.; Mader, U.; Hanggi, J.; Hofmann, H.; Puder, J.J.; Kriemler, S.; Bringolf-Isler, B. Reactivity to accelerometer measurement of children and adolescents. Med. Sci. Sports Exerc. 2014, 46, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Barreira, T.V.; Schuna, J.M., Jr.; Mire, E.F.; Katzmarzyk, P.T. Fully automated waist-worn accelerometer algorithm for detecting children’s sleep-period time separate from 24-h physical activity or sedentary behaviors. Appl. Physiol. Nutr. Metab. 2014, 39, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Barreira, T.V.; Schuna, J.M., Jr.; Mire, E.F.; Katzmarzyk, P.T.; Chaput, J.P.; Leduc, G.; Tudor-Locke, C. Identifying children’s nocturnal sleep using 24-h waist accelerometry. Med. Sci. Sports Exerc. 2014. [Google Scholar] [CrossRef] [PubMed]
- Evenson, K.R.; Catellier, D.J.; Gill, K.; Ondrak, K.S.; McMurray, R.G. Calibration of two objective measures of physical activity for children. J. Sport. Sci. 2008, 26, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Tabachnick, B.; Fidell, L.S. Using Multivariate Statistics; Pearson: New York, NY, USA, 2007. [Google Scholar]
- Foulkes, M.A.; Davis, C.E. An index of tracking for longitudinal data. Biometrics 1981, 37, 439–446. [Google Scholar] [CrossRef]
- Rogosa, D.; Ghandour, G.A. Timepath: Stastical Analysis of Individual Trajectories; Stanford University: Stanford, CA, USA, 1988. [Google Scholar]
- Rogosa, D. Individual trajectories as the starting point for longitudinal data analysis. Alzheimer Dis. Assoc. Disord. 1994, 8, S302–S307. [Google Scholar] [PubMed]
- Rogosa, D.; Floden, R.E.; Willet, J.B. Assessing the Stability of Teacher Behavior; East Lansing Institute for Research on Teaching, Michigan State University: East lansing, MI, USA, 1984. [Google Scholar]
- Hedeker, D.; Gibbons, R.; du Toit, M.; Cheng, Y. Supermix for Mixed Effects Models; Scientific Software International: Lincolnwood, IL, USA, 2008. [Google Scholar]
- Vale, S.; Silva, P.; Santos, R.; Soares-Miranda, L.; Mota, J. Compliance with physical activity guidelines in preschool children. J. Sports Sci. 2010, 28, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Laguna, M.; Ruiz, J.R.; Gallardo, C.; Garcia-Pastor, T.; Lara, M.T.; Aznar, S. Obesity and physical activity patterns in children and adolescents. J. Paediatr. Child Health 2013, 49, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Kettner, S.; Kobel, S.; Fischbach, N.; Drenowatz, C.; Dreyhaupt, J.; Wirt, T.; Koch, B.; Steinacker, J.M. Objectively determined physical activity levels of primary school children in South-West Germany. BMC Public Health 2013, 13, 895. [Google Scholar] [CrossRef] [PubMed]
- Trost, S.G.; Loprinzi, P.D.; Moore, R.; Pfeiffer, K.A. Comparison of accelerometer cut points for predicting activity intensity in youth. Med. Sci. Sports Exerc. 2011, 43, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- DuRant, R.H.; Thompson, W.O.; Johnson, M.; Baranowski, T. The relationship among television watching, physical activity, and body composition of 5- or 6-year-old children. Pediatr. Exerc. Sci. 1996, 8, 15–26. [Google Scholar]
- Sandercock, G.R.; Ogunleye, A.; Voss, C. Screen time and physical activity in youth: Thief of time or lifestyle choice? J. Phys. Act. Health 2012, 9, 977–984. [Google Scholar]
- Taverno Ross, S.; Dowda, M.; Saunders, R.; Pate, R. Double dose: The cumulative effect of TV viewing at home and in preschool on children’s activity patterns and weight status. Pediatr. Exerc. Sci. 2013, 25, 262–272. [Google Scholar] [PubMed]
- Corder, K.; Craggs, C.; Jones, A.P.; Ekelund, U.; Griffin, S.J.; van Sluijs, E.M. Predictors of change differ for moderate and vigorous intensity physical activity and for weekdays and weekends: A longitudinal analysis. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 69. [Google Scholar] [CrossRef] [PubMed]
- McMinn, A.M.; Griffin, S.J.; Jones, A.P.; van Sluijs, E.M. Family and home influences on children’s after-school and weekend physical activity. Eur. J. Public Health 2013, 23, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Edwardson, C.L.; Gorely, T. Review: Parental influences on different types and intensities of physical activity in youth: A systematic review. Psychol. Sport Exerc. 2010, 11, 522–535. [Google Scholar] [CrossRef]
- Lawman, H.G.; Wilson, D.K. A review of family and environmental correlates of health behaviors in high-risk youth. Obesity (Silver Spring) 2012, 20, 1142–1157. [Google Scholar] [CrossRef] [PubMed]
- Jago, R.; Stamatakis, E.; Gama, A.; Carvalhal, I.M.; Nogueira, H.; Rosado, V.; Padez, C. Parent and child screen-viewing time and home media environment. Am. J. Prev. Med. 2012, 43, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Vander Ploeg, K.A.; Kuhle, S.; Maximova, K.; McGavock, J.; Wu, B.; Veugelers, P.J. The importance of parental beliefs and support for pedometer-measured physical activity on school days and weekend days among Canadian children. BMC Public Health 2013, 13, 1132. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.; Hardman, K. Update on the state and status of physical education worldwide. Eur. Phys. Educ. Rev. 2000, 6, 203–229. [Google Scholar] [CrossRef]
- Hardman, K. Physical education in schools: A global perspective. Kinesiology 2008, 40, 5–28. [Google Scholar]
- Crespo, N.C.; Corder, K.; Marshall, S.; Norman, G.J.; Patrick, K.; Sallis, J.F.; Elder, J.P. An examination of multilevel factors that may explain gender differences in children’s physical activity. J. Phys. Act. Health 2013, 10, 982–992. [Google Scholar] [PubMed]
- Maia, J.A.; Thomis, M.; Beunen, G. Genetic factors in physical activity levels: A twin study. Am. J. Prev. Med. 2002, 23, 87–91. [Google Scholar] [CrossRef]
- Boyle, D.E.; Marshall, N.L.; Robeson, W.W. Gender at play: Fourth-grade girls and boys on the playground. Amer. Behav. Sci. 2003, 46, 1326–1345. [Google Scholar] [CrossRef]
- Carver, A.; Timperio, A.; Crawford, D. Perceptions of neighborhood safety and physical activity among youth: The CLAN study. J. Phys. Act. Health 2008, 5, 430–444. [Google Scholar] [PubMed]
- Niederer, I.; Kriemler, S.; Zahner, L.; Burgi, F.; Ebenegger, V.; Marques, P.; Puder, J.J. BMI group-related differences in physical fitness and physical activity in preschool-age children: A cross-sectional analysis. Res. Quart. Exercise Sport 2012, 83, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Rolland-Cachera, M.F.; Deheeger, M.; Bellisle, F.; Sempé, M.; Guilloud-Bataille, M.; Patois, E. Adiposity rebound in children: A simple indicator for predicting obesity. Am. J. Clin. Nutr. 1984, 39, 129–135. [Google Scholar] [PubMed]
- Malina, R.M. Top 10 research questions related to growth and maturation of relevance to physical activity, performance, and fitness. Res. Quart. Exercise Sport 2014, 85, 157–173. [Google Scholar] [CrossRef] [PubMed]
- D’Hondt, E.; Deforche, B.; Gentier, I.; de Bourdeaudhuij, I.; Vaeyens, R.; Philippaerts, R.; Lenoir, M. A longitudinal analysis of gross motor coordination in overweight and obese children versus normal-weight peers. Int. J. Obes. (Lond.) 2013, 37, 61–67. [Google Scholar] [CrossRef] [PubMed]
- D’Hondt, E.; Deforche, B.; de Bourdeaudhuij, I.; Lenoir, M. Relationship between motor skill and body mass index in 5- to 10-year-old children. Adapt. Phys. Act. Quart. 2009, 26, 21–37. [Google Scholar]
- Trost, S.G.; Kerr, L.M.; Ward, D.S.; Pate, R.R. Physical activity and determinants of physical activity in obese and non-obese children. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, S.I.; Stolk, R.P.; Liem, E.T.; Lemmink, K.A.; Corpeleijn, E. The role of fitness in the association between fatness and cardiometabolic risk from childhood to adolescence. Pediatr. Diabetes 2013, 14, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Wickel, E.E.; Eisenmann, J.C.; Welk, G.J. Maturity-related variation in moderate-to-vigorous physical activity among 9–14 year olds. J. Phys. Act. Health 2009, 6, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.; Baxter-Jones, A.D.; Mirwald, R.L.; Bailey, D.A. Comparison of physical activity in male and female children: Does maturation matter? Med. Sci. Sports Exerc. 2003, 35, 1684–1690. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, P.L.; Moller, N.C.; Korsholm, L.; Wedderkopp, N.; Andersen, L.B.; Froberg, K. Tracking of objectively measured physical activity from childhood to adolescence: The European Youth Heart Study. Scand. J. Med. Sci. Sports 2008, 18, 171–178. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, S.; Gomes, T.N.; Borges, A.; Santos, D.; Souza, M.; Dos Santos, F.K.; Chaves, R.N.; Katzmarzyk, P.T.; Maia, J.A.R. Variability and Stability in Daily Moderate-to-Vigorous Physical Activity among 10 Year Old Children. Int. J. Environ. Res. Public Health 2015, 12, 9248-9263. https://doi.org/10.3390/ijerph120809248
Pereira S, Gomes TN, Borges A, Santos D, Souza M, Dos Santos FK, Chaves RN, Katzmarzyk PT, Maia JAR. Variability and Stability in Daily Moderate-to-Vigorous Physical Activity among 10 Year Old Children. International Journal of Environmental Research and Public Health. 2015; 12(8):9248-9263. https://doi.org/10.3390/ijerph120809248
Chicago/Turabian StylePereira, Sara, Thayse Natacha Gomes, Alessandra Borges, Daniel Santos, Michele Souza, Fernanda K. Dos Santos, Raquel N. Chaves, Peter T. Katzmarzyk, and José A. R. Maia. 2015. "Variability and Stability in Daily Moderate-to-Vigorous Physical Activity among 10 Year Old Children" International Journal of Environmental Research and Public Health 12, no. 8: 9248-9263. https://doi.org/10.3390/ijerph120809248
APA StylePereira, S., Gomes, T. N., Borges, A., Santos, D., Souza, M., Dos Santos, F. K., Chaves, R. N., Katzmarzyk, P. T., & Maia, J. A. R. (2015). Variability and Stability in Daily Moderate-to-Vigorous Physical Activity among 10 Year Old Children. International Journal of Environmental Research and Public Health, 12(8), 9248-9263. https://doi.org/10.3390/ijerph120809248