Accumulation of Heavy Metals and Metalloid in Foodstuffs from Agricultural Soils around Tarkwa Area in Ghana, and Associated Human Health Risks
Abstract
:1. Introduction
2. Materials and Methods
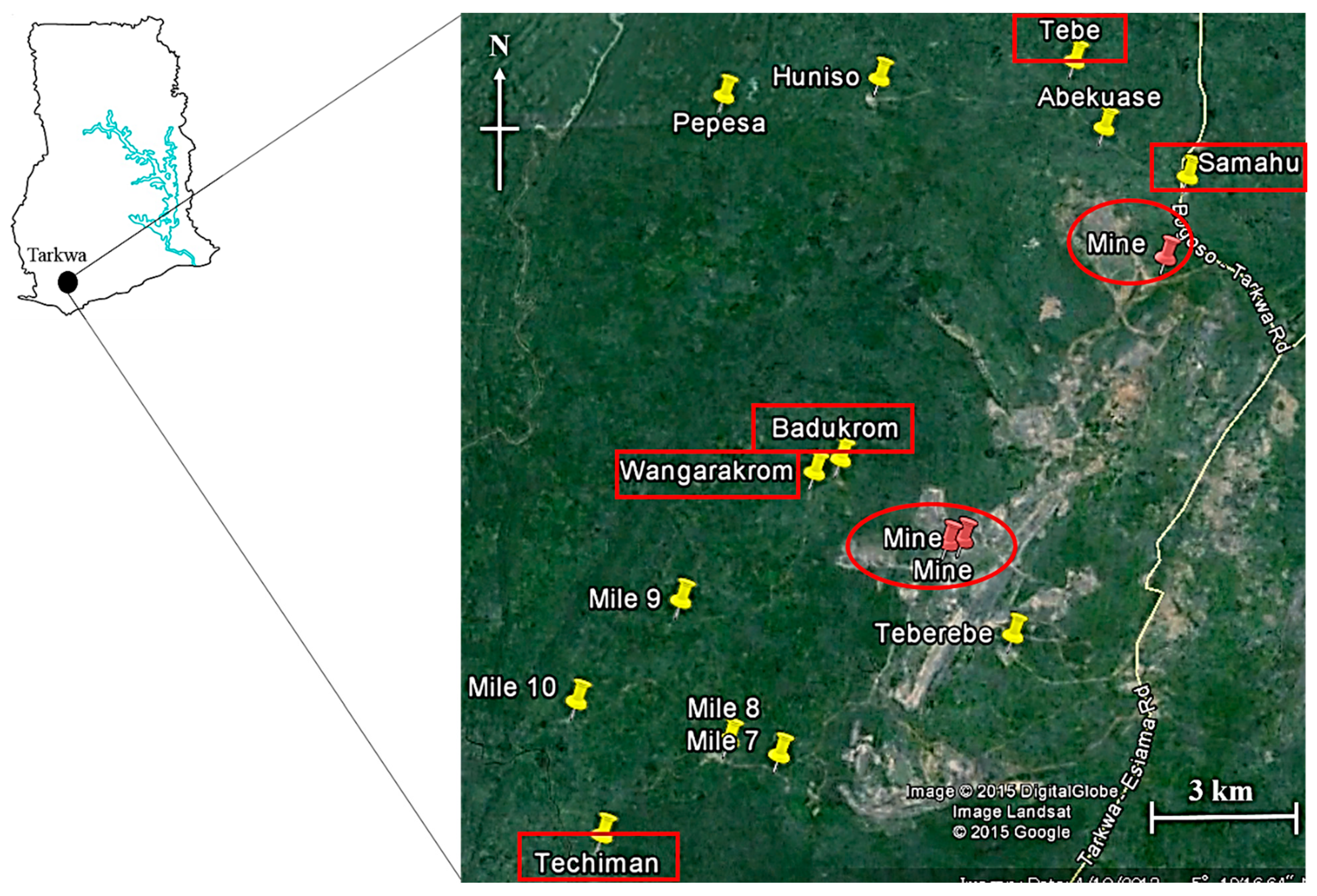
2.1. Study Area
2.2. Soil, Foodstuff Sampling and Processing
2.3. Heavy Metal Analyses
2.4. Quality Control and Quality Assurance
2.5. Data Analyses
2.5.1. Bioconcentration Factor (BCF)
2.5.2. Foodstuff Consumption-Associated Health Risk Assessment
2.5.3. Statistical Analysis
3. Results and Discussion
3.1. Heavy Metal Distribution in Agricultural Soils and Foodstuffs
| Sample Type | Sample Number | As | Cd | Co | Cr | Cu | Hg | Ni | Pb | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Soil # | 60 | Geometric Mean | 3.8 a | 0.026 a | 1.3 a | 16 a | 5.1 a | 0.49 a | 2.2 a | 4.8 a | 37 a |
| SD | 3.4 | 0.013 | 0.74 | 10 | 1.9 | 0.85 | 1.0 | 3.5 | 24 | ||
| Median | 2.9 | 0.022 | 1.2 | 12 | 5.5 | 0.13 | 1.9 | 3.8 | 32 | ||
| Minimum | 1.0 | 0.010 | 0.37 | 8.1 | 2.8 | 0.030 | 1.1 | 1.6 | 9.8 | ||
| Maximum | 13 | 0.052 | 3.0 | 38 | 8.9 | 2.4 | 4.5 | 13 | 86 | ||
| Cassava | 33 | Geometric Mean | 0.0090 b | 0.0070 b | 0.024 b | 0.050 b | 2.1 b | 0.0040 a | 3.7 b | 0.18 b | 7.6 b |
| SD | 0.0050 | 0.0050 | 0.013 | 0.054 | 0.83 | 0.0030 | 0.68 | 0.17 | 2.2 | ||
| Median | 0.0070 | 0.0050 | 0.024 | 0.038 | 2.3 | 0.0030 | 3.6 | 0.14 | 6.8 | ||
| Minimum | 0.0030 | 0.0020 | 0.010 | 0.010 | 1.0 | 0.0010 | 2.7 | 0.017 | 4.7 | ||
| Maximum | 0.017 | 0.016 | 0.051 | 0.20 | 3.3 | 0.010 | 5.4 | 0.45 | 12 | ||
| Plantain | 32 | Geometric Mean | 0.012 b | 0.0010 b | 0.0054 b | 0.025 b | 3.4 b | 0.0010 a | 3.6 b | 0.0070 b | 7.3 b |
| SD | 0.0060 | 0.0010 | 0.0030 | 0.014 | 1.0 | 0.0010 | 0.33 | 0.0060 | 1.9 | ||
| Median | 0.011 | 0.0010 | 0.0050 | 0.023 | 3.4 | 0.0010 | 3.7 | 0.0050 | 6.6 | ||
| Minimum | 0.0060 | nd | 0.0017 | 0.0090 | 1.9 | 0.0010 | 2.9 | 0.0022 | 4.8 | ||
| Maximum | 0.026 | 0.0020 | 0.011 | 0.056 | 5.1 | 0.0045 | 4.1 | 0.020 | 10 | ||
| ANOVA p value | ˂0.01 | ˂0.01 | ˂0.01 | ˂0.01 | ˂0.01 | ≤0.05 | ˂0.01 | ˂0.01 | ˂0.01 |
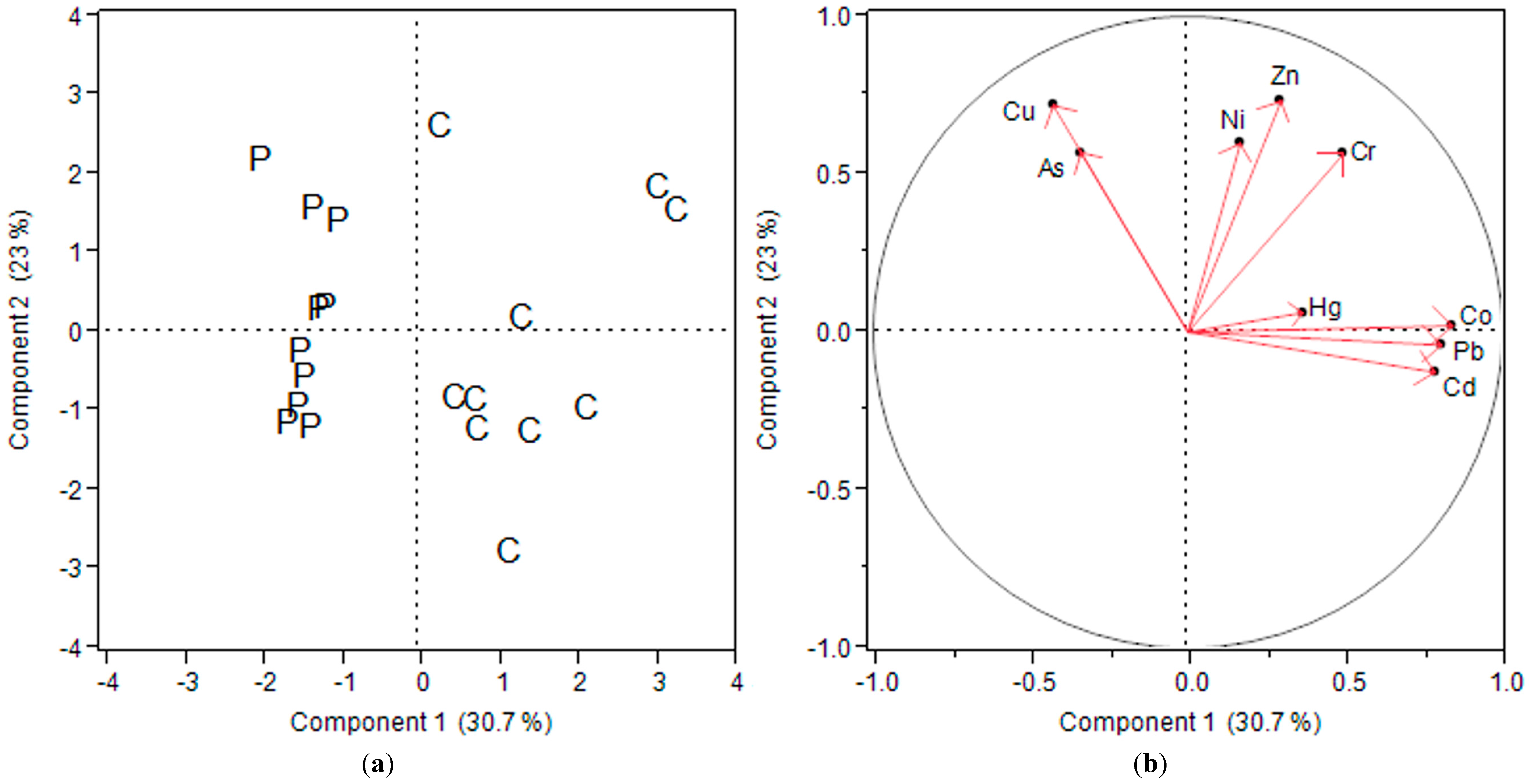
3.2. Bioaccumulation of Heavy Metals in Foodstuffs from Soil
| Sample Site | n | Soil pH | SOM | WC | As | Cd | Co | Cr | Cu | Hg | Ni | Pb | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | Mile 7 | 3 | 7.3 | 1.8 | 0.91 | 0.0058 | 1.1 | 0.019 | 0.004 | 0.64 | 0.067 | 3.2 | 0.13 | 0.70 |
| Mile 8 | 3 | 7.1 | 1.8 | 0.88 | 0.0043 | 0.25 | 0.035 | 0.001 | 0.49 | 0.021 | 2.3 | 0.020 | 0.34 | |
| Techiman | 3 | 6.3 | 1.8 | 0.66 | 0.0034 | 0.24 | 0.005 | 0.001 | 0.19 | 0.015 | 1.1 | 0.16 | 0.15 | |
| Wangarakrom | 3 | 6.9 | 2.8 | 0.66 | 0.0031 | 0.57 | 0.018 | 0.013 | 0.68 | 0.002 | 1.1 | 0.15 | 0.45 | |
| Badukrom | 3 | 6.9 | 2.0 | 2.5 | 0.00034 | 1.2 | 0.11 | 0.001 | 0.55 | 0.001 | 2.8 | 0.055 | 0.22 | |
| Samahu | 3 | 7.1 | 2.4 | 2.1 | 0.0016 | 0.24 | 0.041 | 0.002 | 0.45 | 0.020 | 1.8 | 0.049 | 0.34 | |
| Abekuase | 3 | 7.0 | 2.5 | 2.1 | 0.0048 | 0.14 | 0.009 | 0.007 | 0.87 | 0.15 | 2.9 | 0.013 | 0.22 | |
| Tebe | 3 | 6.9 | 2.9 | 1.9 | 0.0038 | 0.39 | 0.013 | 0.003 | 0.41 | 0.070 | 1.6 | 0.073 | 0.55 | |
| Huniso | 3 | 7.3 | 1.5 | 0.99 | 0.010 | 0.20 | 0.031 | 0.004 | 0.36 | 0.006 | 2.8 | 0.001 | 0.073 | |
| Pepesa | 3 | 7.3 | 1.9 | 1.9 | 0.0011 | 0.041 | 0.036 | 0.002 | 0.38 | 0.049 | 1.8 | 0.004 | 0.083 | |
| Teberebe | 3 | 7.6 | 2.5 | 1.1 | 0.0036 | 0.11 | 0.0035 | 0.001 | 0.11 | 0.023 | 0.59 | 0.003 | 0.14 | |
| Average | 7.1 | 2.2 | 1.4 | 0.004 | 0.40 | 0.029 | 0.0035 | 0.47 | 0.039 | 2.0 | 0.060 | 0.29 | ||
| SD | 0.34 | 0.46 | 0.67 | 0.003 | 0.39 | 0.030 | 0.0036 | 0.22 | 0.045 | 0.87 | 0.061 | 0.20 | ||
| Minimum | 6.3 | 1.5 | 0.66 | 0.00034 | 0.041 | 0.0035 | 0.0010 | 0.11 | 0.0014 | 0.60 | 0.0012 | 0.07 | ||
| Maximum | 7.6 | 2.9 | 2.5 | 0.010 | 1.2 | 0.11 | 0.013 | 0.87 | 0.15 | 3.2 | 0.16 | 0.70 | ||
| P | Mile 7 | 3 | 7.3 | 1.8 | 0.91 | 0.0085 | 0.12 | 0.0062 | 0.0024 | 1.3 | 0.033 | 3.7 | 0.0052 | 1.1 |
| Mile 9 | 3 | 5.8 | 2.1 | 0.50 | 0.0030 | 0.011 | 0.0043 | 0.00057 | 0.91 | 0.0061 | 2.3 | 0.0011 | 0.49 | |
| Mile 10 | 3 | 7.2 | 1.3 | 0.44 | 0.0093 | 0.024 | 0.0068 | 0.00073 | 1.5 | 0.023 | 3.8 | 0.0012 | 0.21 | |
| Techiman | 3 | 6.3 | 1.8 | 0.66 | 0.0075 | 0.031 | 0.0044 | 0.0023 | 0.93 | 0.0053 | 1.2 | 0.0018 | 0.32 | |
| Wangarakrom | 4 | 6.9 | 2.8 | 0.66 | 0.0026 | 0.070 | 0.0024 | 0.0022 | 0.99 | 0.0023 | 1.1 | 0.0060 | 0.35 | |
| Badukrom | 3 | 6.9 | 2.0 | 2.5 | 0.00093 | 0.011 | 0.015 | 0.0014 | 0.39 | 0.00041 | 2.8 | 0.00096 | 0.17 | |
| Abekuase | 3 | 7.0 | 2.5 | 2.1 | 0.0035 | 0.024 | 0.0060 | 0.0031 | 0.89 | 0.018 | 1.9 | 0.0019 | 0.21 | |
| Tebe | 3 | 6.9 | 2.9 | 1.9 | 0.0092 | 0.13 | 0.0024 | 0.0044 | 0.84 | 0.014 | 1.6 | 0.0048 | 0.46 | |
| Huniso | 3 | 7.3 | 1.5 | 0.99 | 0.0077 | 0.005 | 0.015 | 0.0020 | 0.73 | 0.0054 | 2.5 | 0.00031 | 0.074 | |
| Pepesa | 4 | 7.3 | 1.9 | 1.9 | 0.0033 | 0.030 | 0.0020 | 0.0012 | 0.28 | 0.0040 | 1.5 | 0.00053 | 0.082 | |
| Average | 6.9 | 2.1 | 1.3 | 0.0056 | 0.045 | 0.0065 | 0.0021 | 0.88 | 0.011 | 2.2 | 0.0024 | 0.35 | ||
| SD | 0.49 | 0.53 | 0.76 | 0.0032 | 0.044 | 0.005 | 0.0011 | 0.37 | 0.011 | 0.96 | 0.0021 | 0.29 | ||
| Minimum | 5.8 | 1.3 | 0.44 | 0.00093 | 0.0051 | 0.0020 | 0.00057 | 0.28 | 0.00041 | 1.1 | 0.00031 | 0.07 | ||
| Maximum | 7.3 | 2.9 | 2.5 | 0.0093 | 0.13 | 0.015 | 0.0044 | 1.4 | 0.033 | 3.8 | 0.0060 | 1.1 |
| Adult | Child | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sample Site | As | Cd | Hg | Pb | As | Cd | Hg | Pb | |
| C | Mile 7 | 0.21 | 0.13 | 0.07 | 0.60 | 0.28 | 0.17 | 0.090 | 0.81 |
| Mile 8 | 0.19 | 0.05 | 0.13 | 0.13 | 0.26 | 0.07 | 0.18 | 0.18 | |
| Techiman | 0.12 | 0.05 | 0.09 | 1.3 | 0.16 | 0.07 | 0.12 | 1.7 | |
| Wangarakrom | 0.57 | 0.12 | 0.11 | 1.1 | 0.77 | 0.17 | 0.14 | 1.4 | |
| Badukrom | 0.15 | 0.16 | 0.12 | 0.39 | 0.20 | 0.22 | 0.16 | 0.52 | |
| Samahu | 0.25 | 0.06 | 0.07 | 1.2 | 0.34 | 0.08 | 0.10 | 1.6 | |
| Abekuase | 0.52 | 0.03 | 0.25 | 0.14 | 0.71 | 0.05 | 0.34 | 0.19 | |
| Tebe | 0.37 | 0.04 | 0.12 | 0.86 | 0.50 | 0.05 | 0.16 | 1.2 | |
| Huniso | 0.50 | 0.10 | 0.03 | 0.05 | 0.67 | 0.14 | 0.036 | 0.064 | |
| Pepesa | 0.19 | 0.02 | 0.32 | 0.06 | 0.26 | 0.02 | 0.44 | 0.079 | |
| Teberebe | 0.33 | 0.04 | 0.05 | 0.05 | 0.44 | 0.06 | 0.07 | 0.066 | |
| P | Mile 7 | 0.19 | 0.0086 | 0.021 | 0.014 | 0.21 | 0.0095 | 0.023 | 0.015 |
| Mile 9 | 0.11 | 0.00082 | 0.016 | 0.0039 | 0.13 | 0.00090 | 0.018 | 0.0043 | |
| Mile 10 | 0.18 | 0.0029 | 0.014 | 0.0045 | 0.20 | 0.0032 | 0.016 | 0.0049 | |
| Techiman | 0.16 | 0.0039 | 0.018 | 0.0091 | 0.18 | 0.0042 | 0.020 | 0.010 | |
| Wangarakrom | 0.29 | 0.0093 | 0.092 | 0.027 | 0.32 | 0.010 | 0.10 | 0.029 | |
| Badukrom | 0.25 | 0.00091 | 0.021 | 0.0042 | 0.27 | 0.0010 | 0.022 | 0.0045 | |
| Abekuase | 0.23 | 0.0036 | 0.018 | 0.013 | 0.26 | 0.0039 | 0.020 | 0.014 | |
| Tebe | 0.54 | 0.0080 | 0.014 | 0.035 | 0.60 | 0.0087 | 0.015 | 0.038 | |
| Huniso | 0.24 | 0.0016 | 0.014 | 0.0074 | 0.26 | 0.0018 | 0.016 | 0.0082 | |
| Pepesa | 0.33 | 0.0076 | 0.016 | 0.0052 | 0.36 | 0.0084 | 0.018 | 0.0057 |
3.3. Potential Human Health Risk Associated with Consumption of Foodstuffs
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Zhuang, P.; Mcbridge, B.B.; Xia, H.P.; Li, N.Y.; Li, Z.A. Health risk from heavy metals via consumption of food crops in the vicinity of Dabaoshan mine, South China. Sci. Total. Environ. 2009, 407, 1551–1561. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.B.; Chen, J.J.; Zhang, J.; Zhang, H.; Qiao, L.; Men, Y. Heavy metals in rice and garden vegetables and their potential health risks to inhabitants in the vicinity of an industrial zone in Jiangsu, China. J. Environ. Sci. 2010, 22, 1792–1799. [Google Scholar] [CrossRef]
- Komárek, M.; Chrastný, V.; Mihaljevič, M. Lead isotopes in environmental sciences: A review. Environ. Int. 2008, 34, 562–577. [Google Scholar] [CrossRef] [PubMed]
- Akoto, O.; Bortey-Sam, N.; Nakayama, S.; Ikenaka, Y.; Baidoo, E.; Yohannes, Y.B.; Mizukawa, H.; Ishizuka, M. Distribution of heavy metals in organs of sheep and goat reared in Obuasi: A gold mining town in Ghana. Int. J. Environ. Sci. Toxicol. 2014, 2, 81–89. [Google Scholar]
- Asante, K.A.; Agusa, T.; Subramanian, A.; Ansa-Asare, O.D.; Biney, C.A.; Tanabe, S. Contamination status of arsenic and other trace elements in drinking water and residents from Tarkwa, a historic mining township in Ghana. Chemosphere 2007, 66, 1513–1522. [Google Scholar] [CrossRef] [PubMed]
- Bortey-Sam, N.; Nakayama, S.M.M.; Ikenaka, Y.; Akoto, O.; Yohannes, Y.B.; Baidoo, E.; Mizukawa, H.; Ishizuka, M. Human health risks from metals and metalloid via consumption of food animals near Gold Mines in Tarkwa, Ghana: Estimation of the daily intakes and target hazard quotients (THQs). Ecotoxicol. Environ. Saf. 2015, 111, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Obiri, S. Determination of heavy metals in water from boreholes in Dumasi in the Wassa West District of the Western Region of the Republic of Ghana. Environ. Monit. Assess. 2007, 130, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behavior and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Wang, X.L.; Sato, T.; Xing, B.S.; Tao, S. Health risks of heavy metals to the general public in Tianjin, China via consumption of vegetables and fish. Sci. Total Environ. 2005, 350, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Uzu, G.; Sobanska, S.; Aliouane, Y.; Pradere, P.; Dumat, C. Study of lead phytoavailability for atmospheric industrial micronic and sub-micronic particles in relation with lead speciation. Environ. Pollut. 2009, 157, 1178–1185. [Google Scholar] [CrossRef] [PubMed]
- Seregin, I.V.; Ivanov, V.B. Physiological aspects of cadmium and lead toxic effects on higher plants. Russ. J. Plant Physiol. 2001, 48, 523–544. [Google Scholar] [CrossRef]
- Tung, G.; Temple, P.J. Uptake and localization of lead in corn (Zea mays L.) seedlings, a study by histochemical and electron microscopy. Sci. Total Environ. 1996, 188, 71–85. [Google Scholar] [CrossRef]
- Arshad, M.; Silvestre, J.; Pinelli, E.; Kallerhoff, J.; Kaemmerer, M.; Tarigo, A.; Shahid, M.; Guiresse, M.; Pradere, P.; Dumat, C. A field study of lead phytoextraction by various scented Pelargonium cultivars. Chemosphere 2008, 71, 2187–2192. [Google Scholar] [CrossRef] [PubMed]
- Chukwuma, C., Sr. A comparative study of cadmium, lead, zinc, pH and bulk density from the Enyigba lead and zinc mine in two different seasons. Ecotoxicol. Environ. Saf. 1995, 31, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Nganje, T.N.; Adamu, C.I.; Ukpong, E.E. Heavy metal concentrations in soils and plants in the vicinity of Arufu lead-zinc mine, Middle Benue Trough, Nigeria. Chin. J. Geochem. 2010, 29, 167–174. [Google Scholar]
- Onyedika, G.O.; Nwosu, G.U. Lead, zinc and cadmium in root crops from mineralized galena-sphalerite mining areas and environment. Pakistan J. Nutr. 2008, 7, 418–420. [Google Scholar] [CrossRef]
- Amonoo-Neizer, E.H.; Nyamah, D.; Bakiamoh, S.B. Mercury and arsenic pollution in soil and biological samples around the mining town of Obuasi, Ghana. Water Air Soil Pollut. 1996, 91, 363–373. [Google Scholar] [CrossRef]
- Golow, A.A.; Adzei, E.A. Zinc in the surface soil and cassava crop in the vicinity of an alluvial goldmine at Dunkwa-on-Offin, Ghana. Bull. Environ. Contam. Toxicol. 2002, 69, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Kríbek, B.; Majer, V.; Knésl, I.; Nyambe, I.; Mihaljevic, M.; Ettler, V.; Sracek, O. Concentrations of arsenic, copper, cobalt, lead and zinc in cassava (Manihot esculenta Crantz) growing on uncontaminated and contaminated soils of the Zambian Copperbelt. J. Afr. Earth Sci. 2014, 99, 713–723. [Google Scholar] [CrossRef]
- US EPA. Supplementary Guidance for Conducting Health Risk Assessment of Chemical Mixtures, Risk Assessment Forum Technical Panel; US EPA: Washington, DC, USA, 2000; EPA/630/R-00/002.
- Itoh, H.; Iwasaki, M.; Sawada, N.; Takachi, R.; Kasuga, Y.; Yokoyama, S.; Onuma, H.; Nishimura, H.; Kusama, R.; Yokoyama, K.; Tsugane, S. Dietary cadmium intake and breast cancer risk in Japanese women: A case-control study. Int. J. Hyg. Environ. Health 2014, 217, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.J.; Sunge, T.; Cheng, C.Y.; Guo, H.R. Arsenic levels in drinking water and mortality of liver cancer in Taiwan. J. Hazard Mater. 2013, 263, 1132–1138. [Google Scholar] [CrossRef] [PubMed]
- Fantke, P.; Friedrich, R.; Jolliet, O. Health impact and damage cost assessment of pesticides in Europe. Environ. Int. 2012, 49, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Akabzaa, T.; Darimani, A. Impact of Mining Sector Investment in Ghana: A Study of the Tarkwa Mining Region; Third World Network: Penang, Malaysia, 2001. [Google Scholar]
- Hayford, E.K.; Amin, A.; Osae, E.K.; Kutu, J. Impact of gold mining on soil and some staple foods collected from selected mining communities in and around Tarkwa Prestea Area. West African J. Appl. Ecol. 2008, 14, 1–12. [Google Scholar] [CrossRef]
- Essumang, D.K.; Dodoo, D.K.; Obiri, S.; Yaney, J.Y. Arsenic, cadmium, and mercury in Cocoyam (Xanthosoma sagititolium) and Watercocoyam (Colocasia esculenta) in Tarkwa a mining community. Bull. Environ. Contam. Toxicol. 2007, 79, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Bortey-Sam, N.; Nakayama, S.M.M.; Ikenaka, Y.; Akoto, O.; Baidoo, E.; Mizukawa, H.; Ishizuka, M. Health risk assessment of heavy metals and metalloid in drinking water from communities near gold mines in Tarkwa, Ghana. Environ. Monit. Assess. 2015, 187, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ghana Statistical Service. Population and Housing Census 2010. Available online: https://www.google.co.uk/#q=Ghana+Statistical+Service%2C+2010.+Population+and+Housing+census (accessed on 12 May 2013).
- Bortey-Sam, N.; Nakayama, S.M.M.; Ikenaka, Y.; Akoto, O.; Baidoo, E.; Mizukawa, H.; Ishizuka, M. Ecological risk of heavy metals and metalloid in agricultural soils in Tarkwa, Ghana. Int. J. Environ. Res. Publ. Health 2015. under review. [Google Scholar]
- Wang, G.; Su, M.Y.; Chen, Y.H.; Lin, F.F.; Luo, D.; Gao, S.F. Transfer characteristics of cadmium and lead from soil to the edible parts of six vegetable species in south eastern China. Environ. Pollut. 2006, 144, 127–135. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Human Health Risk Assessment Protocol for Hazardous Waste Combustion Facilities. 2005. Available online: http://www.epa.gov/region6/6pd/rcra_c/protocol/protocol.htm (accessed on 12 October 2013). [Google Scholar]
- Pandey, J.; Pandey, U. Accumulation of heavy metals in dietary vegetables and cultivated soil horizon in organic farming system in relation to atmospheric deposition in a seasonally dry tropical region of India. Environ. Monit. Assess. 2009, 148, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Ngole, V.M. Using soil heavy metal enrichment and mobility factors to determine potential uptake by vegetables. Plant Soil Environ. 2011, 57, 75–80. [Google Scholar]
- Kim, Y.Y.; Yang, Y.Y.; Lee, Y. Pb and Cd uptake in rice roots. Physiol. Plant 2002, 116, 368–372. [Google Scholar] [CrossRef]
- Tudorean, L.; Phillips, C.J.C. Modeling cadmium uptake and accumulation in plants. Adv. Agron. 2004, 84, 121–157. [Google Scholar]
- Speir, T.W.; Van Schaik, A.P.; Percival, H.J.; Close, M.E.; Pang, L.P. Heavy metals in soil, plants and groundwater following high-rate sewage sludge application to land. Water Air Soil Poll. 2003, 150, 319–358. [Google Scholar] [CrossRef]
- Chang, C.Y.; Yu, H.Y.; Chen, J.J.; Li, F.B.; Zhang, H.H.; Liu, C.P. Accumulation of heavy metals in leaf vegetables from agricultural soils and associated potential health risks in the Pearl River Delta, South China. Environ. Monit. Assess. 2014, 186, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Region 9, Preliminary Remediation Goals. 2002. Available online: http://www.epa.gov/region09/waste/sfund/prg (assessed on 12 October 2013). [Google Scholar]
- FAOSTAT (Food and Agriculture Organization of the United Nations). 2014. Available online: http://faostat3.fao.org/faostat-gateway/go/to/download/C/CC/E (assessed on 15 April 2014).
- Bennett, D.; Kastenberg, W.; McKone, T. A multimedia, multiple pathway risk assessment of atrazine: the impact of age differentiated exposure including joint uncertainty and variability. Reliab. Eng. Syst. Saf. 1999, 63, 185–198. [Google Scholar] [CrossRef]
- Wayne, R.O. A physical explanation of the lognormality of pollutant concentrations. J. Air Manag. Assoc. 1990, 40, 1378–1383. [Google Scholar]
- ATSDR. Priority List of Hazardous substances. Available online: www.atsdr.cdc.gov/cercla/07List.html (accessed on 1 October 2008).
- FAO/WHO. Joint FAO/WHO Food Standards Programme–Codex Alimentarius Commission. Report of the 33rd Session of the Codex Committee on Food Additives and Contaminants; FAO/WHO: Hague, The Netherlands, 2001. [Google Scholar]
- Grover, P.; Rekhadevi, P.; Danadevi, K.; Vuyyuri, S.; Mahboob, M.; Rahman, M. Genotoxicity evaluation in workers occupationally exposed to lead. Int. J. Hyg. Environ. Health 2010, 213, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Islam, E.; Liu, D.; Li, T.; Yang, X.; Jin, X.; Mahmood, Q.; Tian, S.; Li, J. Effect of Pb toxicity on leaf growth, physiology and ultrastructure in the two ecotypes of Elsholtzia argyi. J. Hazard. Mater. 2008, 154, 914–926. [Google Scholar] [CrossRef] [PubMed]
- Sammut, M.; Noack, Y.; Rose, J.; Hazemann, J.; Proux, O.; Depoux Ziebel, M.; Fiani, E. Speciation of Cd and Pb in dust emitted from sinter plant. Chemosphere 2010, 78, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.J.; Zhu, Y.G.; Zhai, R.H.; Chen, D.Y.; Huang, Y.Z.; Qiu, Y.; Liang, .J.Z. Transfer of metals from soil to vegetables in an area near a smelter in Nanning, China. Environ. Int. 2004, 30, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Dayton, E.A.; Basta, N.T.; Payton, M.E.; Bradham, K.D.; Schroder, J.L.; Lamno, R.P. Evaluating the contribution of soil properties to modifying lead phytoavailability and phytotoxicity. Environ. Toxicol. Chem. 2006, 25, 719–725. [Google Scholar] [CrossRef] [PubMed]
- European Environment and Health Information System (ENHIS). Exposure of Children to Chemical Hazards in Food; World Health Organization: Geneva, Switzerland, 2007; Fact Sheet No. 4.4, CODE: RPG4_Food_EX1. [Google Scholar]
- Wongsasuluk, P.; Chotpantarat, S.; Siriwong, W.; Robson, M. Heavy metal contamination and human health risk assessment in drinking water from shallow ground water wells in an agricultural area in Ubon Ratchathani province, Thailand. Environ. Geochem. Health 2014, 36, 169–182. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bortey-Sam, N.; Nakayama, S.M.M.; Akoto, O.; Ikenaka, Y.; Fobil, J.N.; Baidoo, E.; Mizukawa, H.; Ishizuka, M. Accumulation of Heavy Metals and Metalloid in Foodstuffs from Agricultural Soils around Tarkwa Area in Ghana, and Associated Human Health Risks. Int. J. Environ. Res. Public Health 2015, 12, 8811-8827. https://doi.org/10.3390/ijerph120808811
Bortey-Sam N, Nakayama SMM, Akoto O, Ikenaka Y, Fobil JN, Baidoo E, Mizukawa H, Ishizuka M. Accumulation of Heavy Metals and Metalloid in Foodstuffs from Agricultural Soils around Tarkwa Area in Ghana, and Associated Human Health Risks. International Journal of Environmental Research and Public Health. 2015; 12(8):8811-8827. https://doi.org/10.3390/ijerph120808811
Chicago/Turabian StyleBortey-Sam, Nesta, Shouta M. M. Nakayama, Osei Akoto, Yoshinori Ikenaka, Julius N. Fobil, Elvis Baidoo, Hazuki Mizukawa, and Mayumi Ishizuka. 2015. "Accumulation of Heavy Metals and Metalloid in Foodstuffs from Agricultural Soils around Tarkwa Area in Ghana, and Associated Human Health Risks" International Journal of Environmental Research and Public Health 12, no. 8: 8811-8827. https://doi.org/10.3390/ijerph120808811
APA StyleBortey-Sam, N., Nakayama, S. M. M., Akoto, O., Ikenaka, Y., Fobil, J. N., Baidoo, E., Mizukawa, H., & Ishizuka, M. (2015). Accumulation of Heavy Metals and Metalloid in Foodstuffs from Agricultural Soils around Tarkwa Area in Ghana, and Associated Human Health Risks. International Journal of Environmental Research and Public Health, 12(8), 8811-8827. https://doi.org/10.3390/ijerph120808811







