A Better Antiviral Efficacy Found in Nucleos(t)ide Analog (NA) Combinations with Interferon Therapy than NA Monotherapy for HBeAg Positive Chronic Hepatitis B: A Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Study Design
2.4. Efficacy Measures
2.5. Data Extraction
2.6. Statistical analysis
| Study | Sample Size | Regimen | Detection Methods for HBsAg and HBeAg | Treatment Duration (Weeks) | Follow-up Period (Weeks) | HBV Genotype | Interferon | ||
|---|---|---|---|---|---|---|---|---|---|
| Comb/Mono | Comb | Mono | Comb | Mono | Type | ||||
| Yuan, 2014 [9] | 26/28 | ETV 0.5 mg/d + Peg-IFNα-2a 180 μg | ETV 0.5 mg/d | AIA1800 Chemiluminescence Analyzer, TOSOH Corporation, Tokyo, Japan | 48 | 52 | unknown | unknown | Peg-IFNα-2a |
| Zeng, 2013 [10] | 20/20 | ETV 0.5 mg/d + Peg-IFNα-2a 180 μg | ETV 0.5 mg/d | i2000 electro-chemiluminescence, Abbot Diagnostic Division, Sligo, UK | 24 | unknown | unknown | unknown | Peg-IFNα-2a |
| Yu, 2013 [11] | 90/45 | Lam 100 mg + IFN-α-2b 50 μg/Adv 10 mg + IFN-α-2b 50 μg/Adv 10 mg + Peg-IFNα-2a 180 μg | Lam 100 mg/ Adv 10 mg | Enzyme immunoassay, AxSym analyzer, Abbot Diagnostic Division | 96 | 24 | unknown | unknown | IFN-α-2b/Peg-IFNα-2a |
| Li, 2013 [12] | 17/22 | Adv 10 mg + Peg-IFNα-2a 180 μg | Adv 10 mg | unknown | 48 | unknown | unknown | unknown | Peg-IFNα-2a |
| Li, 2012 [13] | 44/51 | ETV 10 mg/d + IFNα-2b | ETV 10 mg/d | Cobass601 electro-chemiluminescence, Roche, Basel and Kaiseraugst, Switzerland | 48 | unknown | unknown | unknown | IFNα-2b |
| Wang, 2012 [14] | 30/31 | Adv 10 mg + IFNα-2b 50 μg | Adv 10 mg | IMx analyzer, Abbot Diagnostic Division | 48 | 24 | unknown | unknown | IFNα-2b |
| Chen, 2012 [15] | 19/35 | ETV 0.5 mg/d + Peg-IFNα-2a 180 μg | ETV 0.5 mg/d | Architect HBsAg QT, Abbot Diagnostic Division | 72 | 72 | B 12 (63%) C 7 (37%) | unknown | Peg-IFNα-2a |
| Ding, 2010 [16] | 22/23 | Adv 10 mg + Peg-IFNα-2a 180 μg | Adv 10 mg | unknown | 48 | unknown | unknown | unknown | Peg-IFNα-2a |
| Piratvisuth, 2008 [17] | 112/112 | Lam 100 mg + Peg-IFNα-2a 180 μg | Lam 100 mg | Microparticle enzyme immunoassay (AXSYM HBe 2.0, Abbott Laboratories, Abbott Park, IL, USA) | 48 | 48 | B 30% C 32% | 43% 49% | Peg-IFNα-2a |
| Li, 2006 [18] | 31/43 | Adv 10 mg + IFNα-2b 50 μg | Adv 10 mg | Shanghai GeneCore Biotechnologies Corporation, Shanghai, China | 104 | unknown | unknown | unknown | IFNα-2b |
| Janssen, 2005 [19] | 130/136 | Lam 100 mg + Peg-IFNα-2b 100 μg/w | Lam 100 mg | EIA (AxSYM, Abbott, Abbott Park, Chicago, IL, USA). | 52 | 24 | A 43 (33%) B 11 (9%) C 18 (14%) D 52 (40%) | 47 (35%) 12 (9%) 21 (15%) 51 (38%) | Peg-IFNα-2b |
| Amarapurkar, 2005 [20] | 20/5 | Lam 100 mg + Peg-IFNα-2b 180 μg | Lam 100 mg | Versant HBV DNA 1.0 assay, Bayer Corp, NewYork, NY, USA | 48 | 24 | unknown | unknown | Peg-IFNα-2b |
| Sarin, 2005 [21] | 38/37 | Lam 100 mg + IFN-α 5 MU | Lam 100 mg | Enzyme immunoassay | 52 | 24 | unknown | unknown | IFN-α |
| Chan, 2005 [22] | 48/47 | Lam 100 mg + Peg-IFNα-2b 100 μg | Lam 100 mg | Applied Biosystems, Foster City, CA, USA | 60 | 52 | B 15 (31%) C 30 (62%) | 16 (34%) 28 (60%) | Peg-IFNα-2b |
| Lau, 2005 [23] | 271/272 | Lam 100 mg + Peg-IFNα-2a 180 μg | Lam 100 mg | Central laboratory, AxSYM test (Abbott) | 48 | 24 | A 18 (7%) B 82 (30%) C 156 (58%) D 11 (4%) | 15 (6%) 73 (27%) 162 (60%) 17 (6%) | Peg-IFNα-2a |
| Song, 2004 [24] | 60/30 | Lam 100 mg + IFNα-2b 3 MU | Lam 100 mg | 71705 Biochemical analyzer, HITACHI, Tokyo, Japan | 24 | 60 | unknown | unknown | IFNα-2b |
| Deng, 2003 [25] | 32/24 | Lam 100 mg + IFNα-2b 5 MU | Lam 100 mg | ELISA, Shanghai Kehua Bio-engineering Corporation, Shanghai, China | 48 | 24 | unknown | unknown | IFNα-2b |
3. Results
3.1. Included Studies

3.2. HBeAg Loss
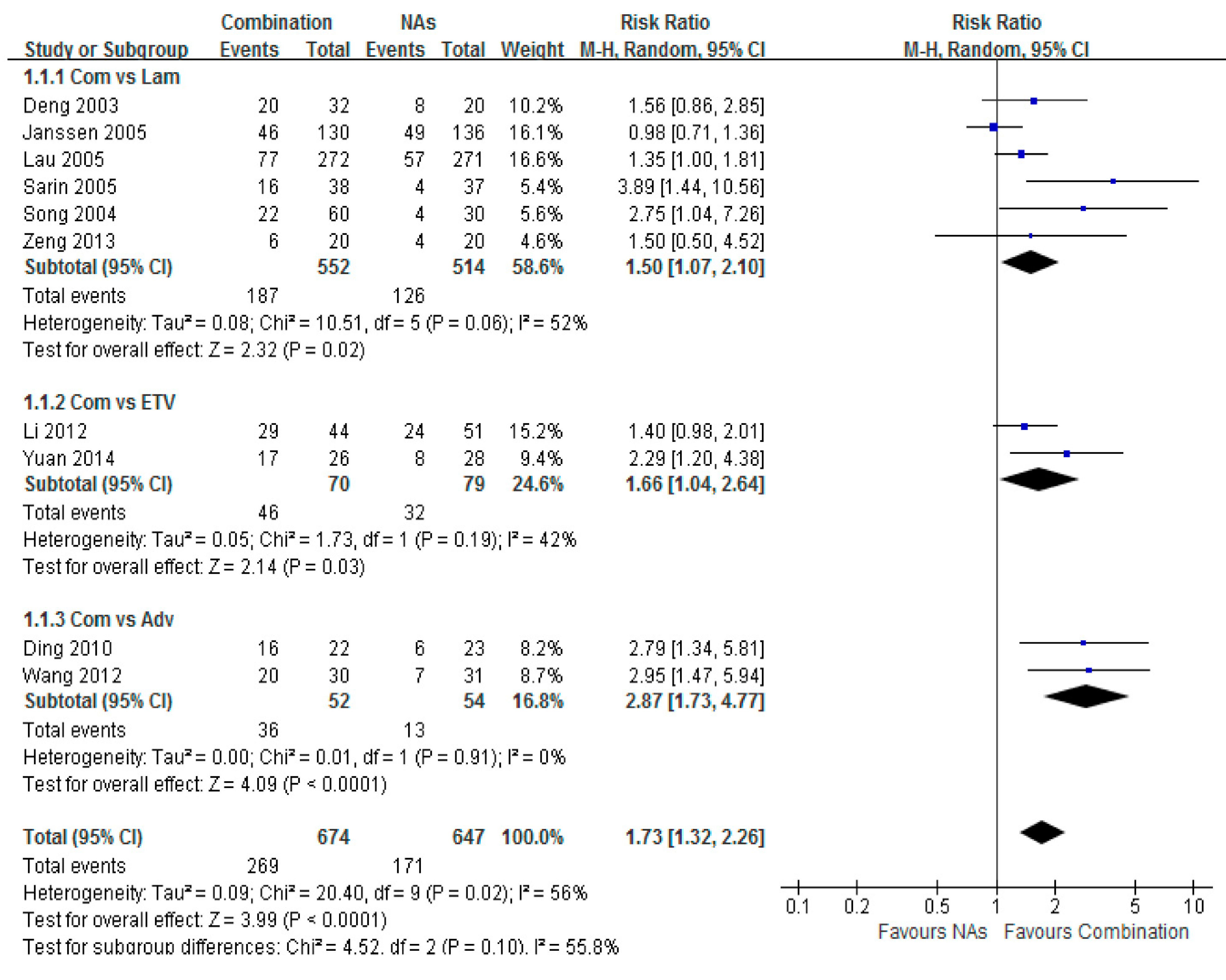
3.3. HBV-DNA Undetectable Rate
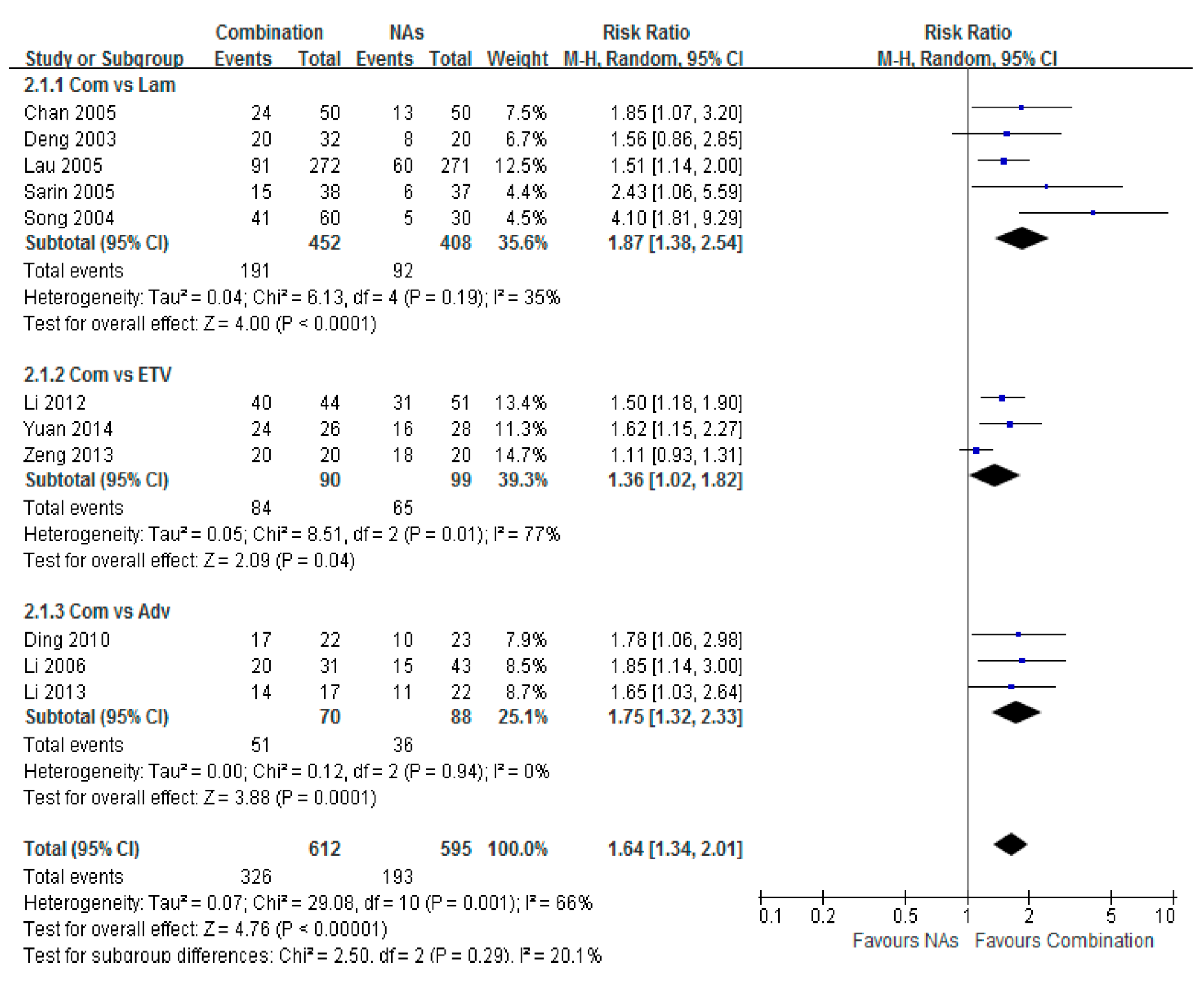
3.4. HBeAg Seroconversion
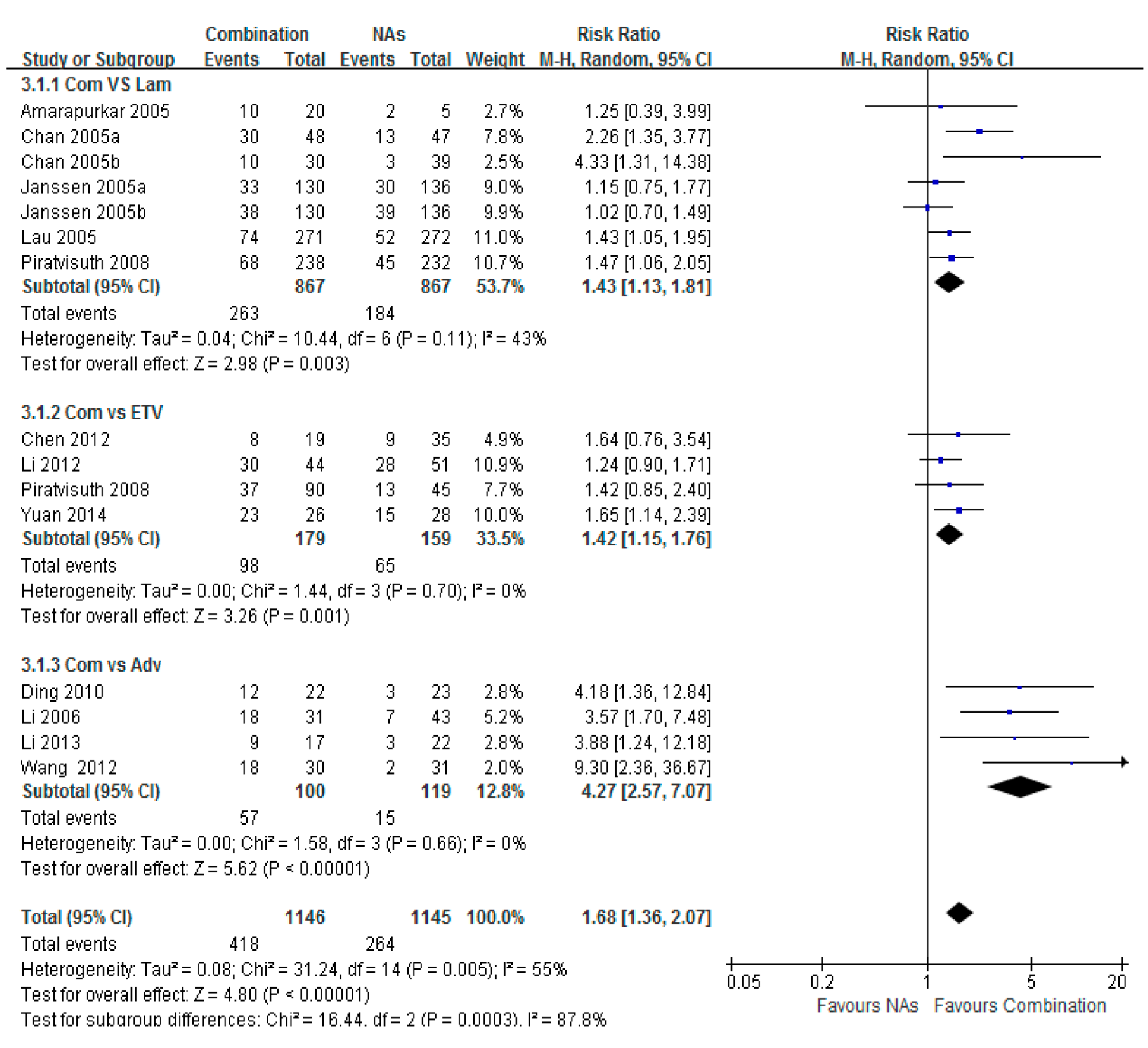
3.5. HBsAg Loss

3.6. HBsAg Seroconversion
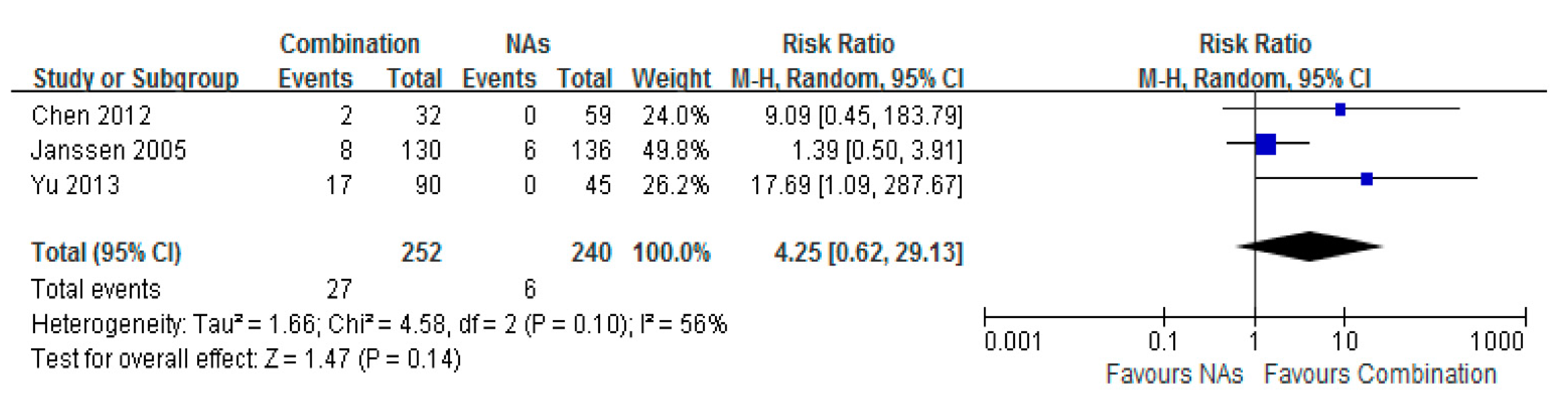
3.7. Histological Improvement
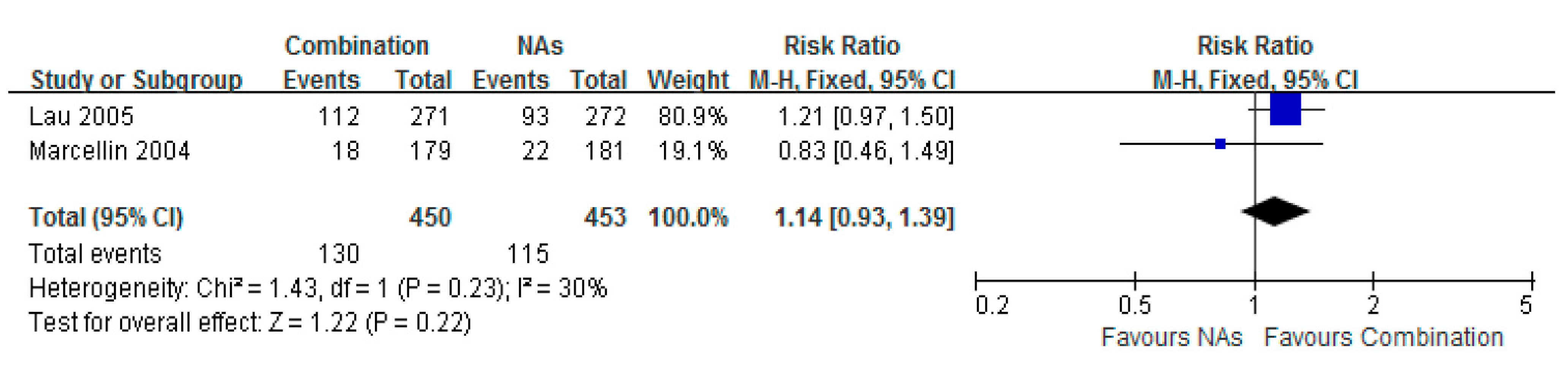
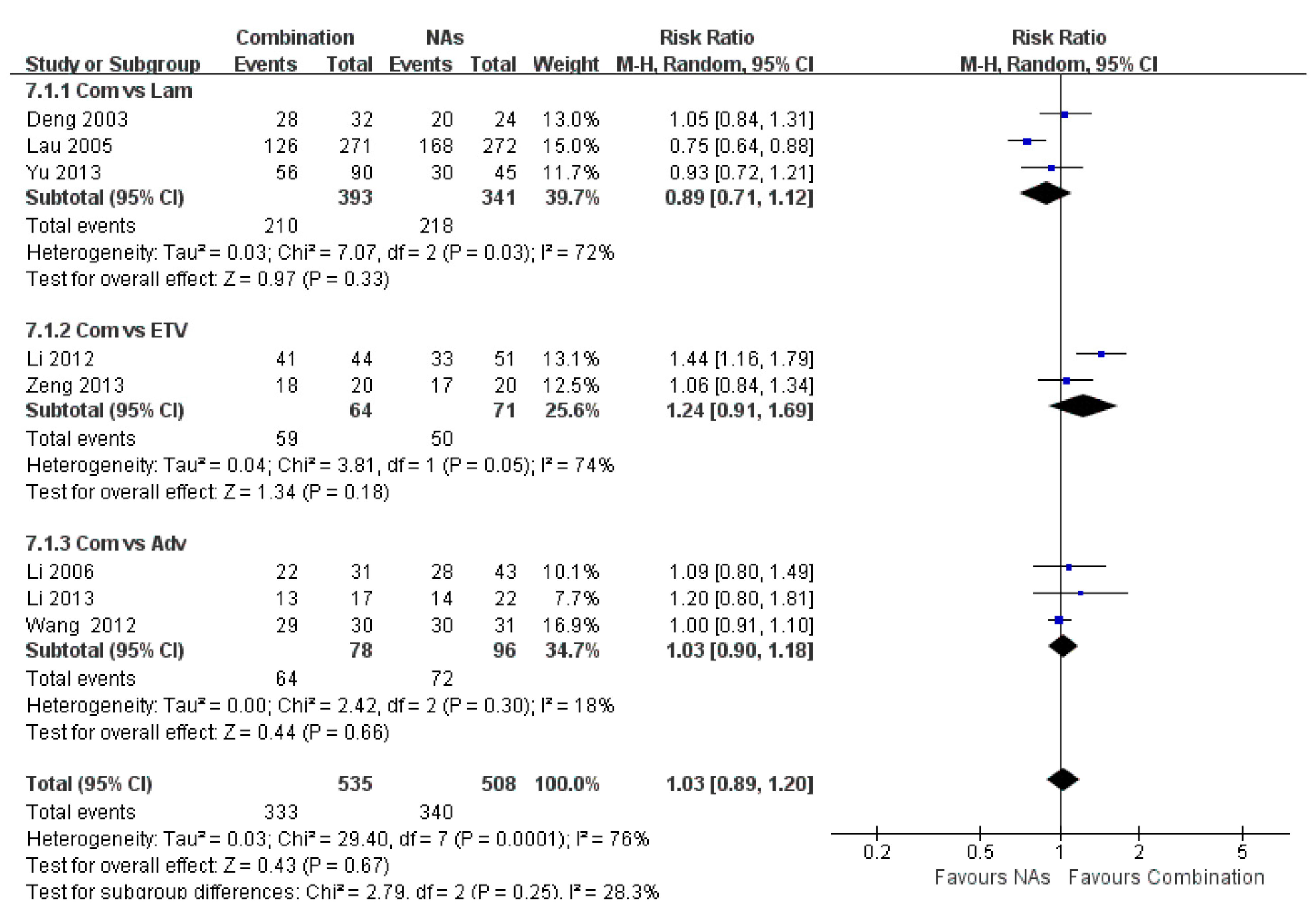
3.8. ALT Normalization
3.9. Safety
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- European Association for the Study of the Liver. EASL clinical practice guidelines: Management of chronic hepatitis B virus infection. J. Hepatol. 2012, 57, 167–185. [Google Scholar]
- Fung, J.; Lai, C.L.; Seto, W.K.; Yuen, M.-F. Nucleoside/nucleotide analogues in the treatment of chronic hepatitis B. J. Antimicrob. Chemother. 2011, 66, 2715–2725. [Google Scholar] [CrossRef] [PubMed]
- Ghany, M.G.; Doo, E.C. Antiviral resistance and hepatitis B therapy. Hepatology 2009, 49, S174–S184. [Google Scholar] [CrossRef] [PubMed]
- Guclu, E.; Tuna, N.; Karabay, O.; Akhan, S.; Bodur, H.; Ceylan, B.; Demirdal, T.; Demirdag, K.; Demirturk, N.; Ekerbicer, H.; et al. Predictors of response to pegylated interferon treatment in HBeAg-negative patients with chronic hepatitis B. J. Infect. Dev. Ctries. 2014, 8, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Masataka, T.; Eisuke, M.; Michio, I.; Abe, H.; Miki, D.; Hiraga, N.; Takahashi, S.; Ochi, H.; Hayes, N.; Ginba, H.; et al. Serum HBV RNA and HBeAg are useful markers for the safe discontinuation of nucleotide analogue treatments in chronic hepatitis B patients. J. Gastroenterol. 2013, 48, 1188–1204. [Google Scholar]
- Merican, I.; Guan, R.; Amarapuka, D.; Alexander, M.J.; Chutaputti, A.; Chien, R.N.; Hasnian, S.S.; Leunq, N.; Lesmana, L.; Phiet, P.H. Chronic hepatitis B virus infection in Asian countries. J. Gastroenterol. Hepatol. 2000, 15, 1356–1361. [Google Scholar] [CrossRef] [PubMed]
- Jeng, W.J.; Sheen, I.S.; Liaw, Y.F. Hepatitis B virus DNA level predicts hepatic decompensation in patients with acute exacerbation of chronic hepatitis B. Clin. Gastroenterol. Hepatol. 2010, 8, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Scaglione, S.J.; Lok, A.S. Effectiveness of hepatitis B treatment in clinical practice. Gastroenterology 2012, 142, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhu, X. Treatment efficacy of pegylated interferon for chronic hepatitis B patients with post-treatment of nucleoside analogues. J. Chengdu Med. Coll. 2014, 9, 80–82. (in Chinese). [Google Scholar]
- Zeng, W.; Yuan, J.; Liu, Y.X.; Zhang, Y.; Li, S.X.; Yao, S.M.; Lin, Y.M.; Chen, C.M.; Zhao, M.M.; Liu, J. Clinical research of pegylated interferon with ETV for high viral load HBeAg positive chronic hepatitis B. Chin. J. Exp. Clin. Virol. 2013, 27, 115–118. (in Chinese). [Google Scholar]
- Yu, S.L.; Guo, C.Y. A retrospective follow-up study of prolonged nucleos(t)ide analogue and interferon combination therapy in HBeAg-positive chronic hepatitis B patients. Zhonghua Gan Zang Bing Za Zhi 2013, 21, 267–270. (in Chinese). [Google Scholar] [PubMed]
- Li, W.B.; Ding, J.G.; Sun, Q.F.; Hong, L.; Fu, R.Q. Efficacy of pegylated interferon alfa-2a combined with adefovir dipivoxil in treatment of HBeAg positive chronic hepatitis B. Chin. J. Nosocomiol. 2013, 23, 1250–1252. (in Chinese). [Google Scholar]
- Li, J. Early efficacy of α-interferon combine with ETV for chronic hepatitis B. Chin. Hepatol. 2012, 17, 714–716. (in Chinese). [Google Scholar]
- Wang, L.; Zhang, X.Y.; Yu, H.Y.; Yang, J.M.; Cao, X.G.; Ding, Q.Y.; Pan, J. Clinical observation on of interferonα-2b combined with adefovir dipivoxil in the treatment of HBeAg positive chronic hepatitis B. Chin. J. Biochem. Pharm. 2012, 33, 849–851. (in Chinese). [Google Scholar]
- Chen, C.C.; Wang, P.C.; Chang, H.W.; Chen, C.F. Safety and efficacy of two-step peginterferon α-2a treatment in patients of chronic hepatitis B with acute exacerbation. J. Viral Hepat. 2012, 19, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.M. Peg-interferon combine with Adv for 22 chronic hepatitis B. Clin. J. Mod. Drug Appl. 2010, 4, 164–165. (in Chinese). [Google Scholar]
- Piratvisuth, T.; Lau, G.; Chao, Y.C.; Jin, R.; Chutaputti, A.; Zhang, Q.B.; Tanwandee, T.; Button, P.; Popescu, M. Sustained response to peginterferon alfa-2a (40 kD) with or without lamivudine in Asian patients with HBeAg-positive and HBeAg-negative chronic hepatitis B. Hepatol. Int. 2008, 2, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Li, S.B.; Xian, J. Adv alone and combined with interferon for HBeAg positive chronic hepatitis B. Clin. Hosp. Pharm. J. 2006, 26, 1538–1540. (in Chinese). [Google Scholar]
- Janssen, H.L.; Zonneveld, M.; Senturk, H.; Zeuzem, S.; Akarca, U.S.; Cakaloglu, Y.; Simon, C.; So, T.M.K.; Gerken, G.; de Man, R.A.; et al. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: A randomised trial. Lancet 2004, 365, 123–129. [Google Scholar] [CrossRef]
- Amarapurkar, D.N.; Patel, N.D. Combination of Peginterferon α-2b (12 kDa) and Lamivudine in difficult-to-treat chronichepatitis B—An Indian experience. Ann. Hepatol. 2005, 4, 56–59. [Google Scholar] [PubMed]
- Sarin, S.K.; Kumar, M.; Kumar, R.; Kazim, S.K.; Guptan, R.C.; Sakhuja, P.; Sharma, B.C. Higher efficacy of sequential therapy with interferon-alpha and lamivudine combination compared to lamivudine monotherapy in HBeAg positive chronichepatitis B patients. Am. J. Gastroenterol. 2005, 100, 2463–2471. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.Y.; Hui, A.Y.; Wong, V.; Chim, L.A.M.; Wong, M.-L.; Sung, J.J.-Y. Long term follow-up of peginterferon and lamivudine combination treatment in HBeAg-positive chronic hepatitis B. Hepatology 2005, 41, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.K.K.; Piratvisuth, T.; Luo, K.X.; Marcellin, P.; Thongsawat, S.; Cooksley, G.; Gane, E.; Fried, M.W.; Chow, W.C.; Paik, S.W.; et al. Peginterferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 2005, 352, 2682–2695. [Google Scholar] [CrossRef] [PubMed]
- Song, J.W.; Zhang, G.; Lin, J.G.; Tang, W.X.; Lin, J.S. Clinical study of lamivudine and interferon combinated administration to inhibit hepatitis B virus replication. Zhonghua Gan Zang BingZa Zhi 2004, 12, 593–596. (in Chinese). [Google Scholar]
- Deng, Q.W.; Wu, C.H.; Yang, J.; Tu, S.C.; Jiang, T.; Guo, Y. Clinical observation on the therapeutic efficacy of interferon-combined with lamivudine in the treatment of patients with chronic hepatitis B. J. Clin. Res. 2003, 20, 503–505. (in Chinese). [Google Scholar]
- Marcellin, P.; Lau, G.K.; Bonino, F.; Farci, P.; Hadziyannis, S.; Jin, R.; Lu, Z.M.; Piratvisuth, T.; Germanidis, G.; Yurdaydin, C.; et al. Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N. Engl. J. Med. 2004, 351, 1206–1217. [Google Scholar] [CrossRef] [PubMed]
- Lok, A.S.; Brian, J.M. Chronic hepatitis B: Update 2009. Hepatology 2009, 50, 661–662. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.A.; Lim, Y.S.; An, J.; Lee, D.; Shim, J.H.; Kim, K.M.; Lee, H.C.; Chung, Y.-H.; Lee, Y.S.; Suh, D.J. HBsAg seroclearance after nucleoside analogue therapy in patients with chronic hepatitis B: Clinical outcomes and durability. Gut 2014, 63, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Liaw, Y.F.; Chu, C.M. Hepatitis B virus infection. Lancet 2009, 373, 582–592. [Google Scholar] [CrossRef]
- Wang, L.C.; Chen, E.Q.; Cao, J.; Liu, L.; Zheng, L.; Li, D.-J.; Xu, L.; Lei, X.-Z.; Liu, C.; Tang, H. De novo combination of lamivudine and adefovir versus entecavir monotherapy for the treatment of naive HBeAg negative chronic hepatitis B patients. Hepatol. Int. 2011, 5, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Z.; Ji, D.; Chen, G.-F.; Feng, X.; Gong, L.-L.; Guo, J.; Li, Z.-W.; Chen, C.-F.; Zhao, B.-B.; et al. Regulation of T cell function by microRNA-720. Sci. Rep. 2015, 5, 12159. [Google Scholar] [CrossRef] [PubMed]
- Ning, Q.; Han, M.F.; Sun, Y.T.; Jiang, J.; Tan, D.; Hou, J.; Tang, H.; Sheng, J.; Zhao, M. Switching from entecavir to PegIFN alfa-2a in patients with HBeAg-positive chronic hepatitis B: A randomized open-label trial (OSST trial). J. Hepatol. 2014, 61, 777–784. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, W.; Wu, Q.; Zhou, J.; Kong, Y.; You, H. A Better Antiviral Efficacy Found in Nucleos(t)ide Analog (NA) Combinations with Interferon Therapy than NA Monotherapy for HBeAg Positive Chronic Hepatitis B: A Meta-Analysis. Int. J. Environ. Res. Public Health 2015, 12, 10039-10055. https://doi.org/10.3390/ijerph120810039
Wei W, Wu Q, Zhou J, Kong Y, You H. A Better Antiviral Efficacy Found in Nucleos(t)ide Analog (NA) Combinations with Interferon Therapy than NA Monotherapy for HBeAg Positive Chronic Hepatitis B: A Meta-Analysis. International Journal of Environmental Research and Public Health. 2015; 12(8):10039-10055. https://doi.org/10.3390/ijerph120810039
Chicago/Turabian StyleWei, Wei, Qinmei Wu, Jialing Zhou, Yuanyuan Kong, and Hong You. 2015. "A Better Antiviral Efficacy Found in Nucleos(t)ide Analog (NA) Combinations with Interferon Therapy than NA Monotherapy for HBeAg Positive Chronic Hepatitis B: A Meta-Analysis" International Journal of Environmental Research and Public Health 12, no. 8: 10039-10055. https://doi.org/10.3390/ijerph120810039
APA StyleWei, W., Wu, Q., Zhou, J., Kong, Y., & You, H. (2015). A Better Antiviral Efficacy Found in Nucleos(t)ide Analog (NA) Combinations with Interferon Therapy than NA Monotherapy for HBeAg Positive Chronic Hepatitis B: A Meta-Analysis. International Journal of Environmental Research and Public Health, 12(8), 10039-10055. https://doi.org/10.3390/ijerph120810039





