Extraction of 3,4,4′-Trichlorocarbanilide from Rat Fecal Samples for Determination by High Pressure Liquid Chromatography–Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Experimental Methods
2.1. Chemicals and Reagents
2.2. Animal Fecal Samples Collection and Preparation
2.3. Fecal Sample Extraction and Preparation
2.4. HPLC-MS/MS Determination
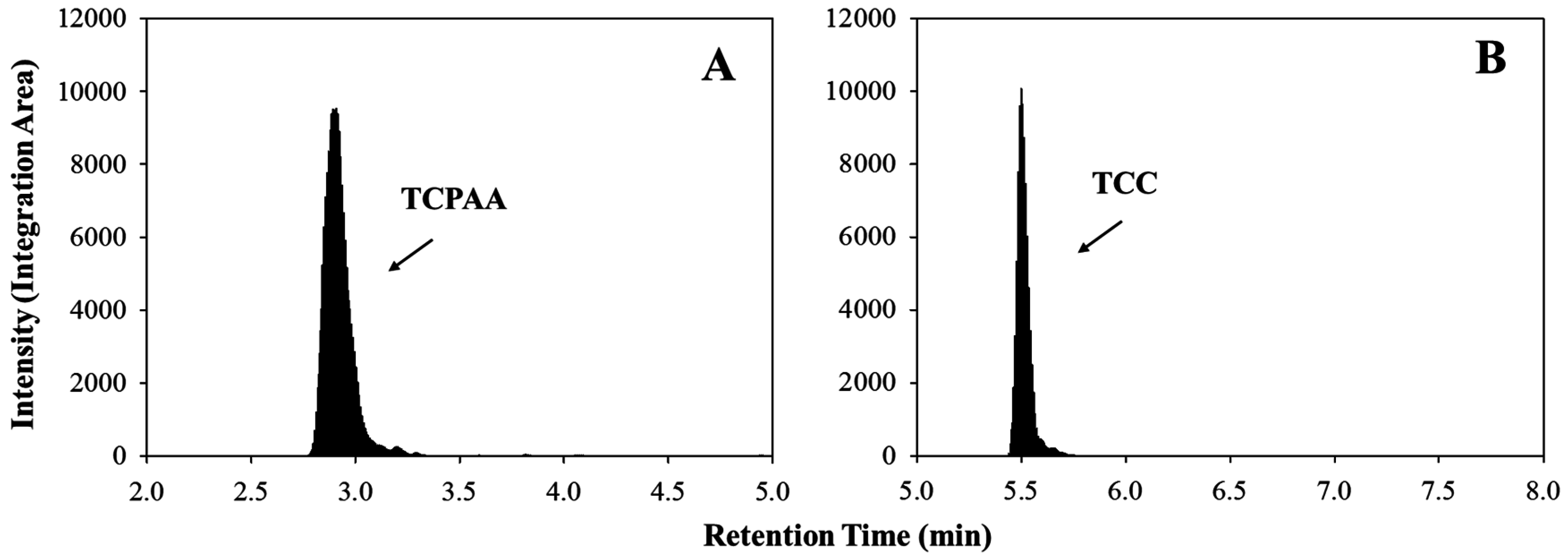
3. Results and Conclusions
3.1. Method Validation and Quality Control
| Spiked TCC * | Intra-variability | Inter-variability | |||
|---|---|---|---|---|---|
| Average (ng/m)L | % RSD ** | Average (ng/mL) | %RSD | ||
| 10 ng/mL | n = 4 | 8.77 | 22.4 | 7.35 | 30.6 |
| 350 ng/mL | n = 4 | 420 | 4.99 | 457 | 14.1 |
| Accuracy (%) | Precision (%RSD) | ||||
| 25 ng/mL | n = 15 | 98.0 | 12.8 | ||
| 100 ng/mL | n = 15 | 105 | 16.0 | ||
| Treatment Day | Control | 0.2% w/w TCC |
|---|---|---|
| 0 | <LOD | <LOD |
| 12 | <LOD | 2220 ± 150 |
| Washout Day | ||
| 2 | <LOD | 15.5 ± 3.50 |
| 8 | <LOD | 0.885 ± 0.377 |
| 28 | <LOD | 0.399 ± 0.178 |
3.2. Quantification of TCC in Feces Samples
4. Conclusions
Acknowledgement
Author Contributions
Conflicts of Interest
References
- Lozano, N.; Rice, C.P.; Ramirez, M.; Torrents, A. Fate of triclocarban, triclosan and methyltriclosan during wastewater and biosolids treatment processes. Water Res. 2013, 47, 4519–4527. [Google Scholar] [CrossRef] [PubMed]
- Schebb, N.H.; Inceoglu, B.; Ahn, K.C.; Morisseau, C.; Gee, S.J.; Hammock, B.D. Investigation of human exposure to triclocarban after showering and preliminary evaluation of its biological effects. Environ. Sci. Technol. 2011, 45, 3109–3115. [Google Scholar] [CrossRef] [PubMed]
- Pycke, B.F.; Geer, L.A.; Dalloul, M.; Abulafia, O.; Jenck, A.M.; Halden, R.U. Human fetal exposure to triclosan and triclocarban in an urban population from Brooklyn, New York. Environ. Sci. Technol. 2014, 48, 8831–8838. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, A.G.; Thomaidis, N.S.; Kannan, K. Widespread occurrence of bisphenol A diglycidyl ethers, p-hydroxybenzoic acid esters (parabens), benzophenone type-UV filters, triclosan, and triclocarban in human urine from Athens, Greece. Sci. Total Environ. 2014, 470–471, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Nielsen, J.K.; Mørck, T.A.; Hansen, P.W.; Jensen, J.F.; Nielsen, O.; Andersson, A.M.; Knudsen, L.E. Urinary excretion of phthalate metabolites, phenols and parabens in rural and urban Danish mother-child pairs. Int. J. Hyg. Environ. Health 2013, 216, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Kuehn, B.M. FDA pushes makers of antimicrobial soap to prove safety and effectiveness. JAMA 2014. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ahn, K.C.; Gee, N.A.; Ahmed, M.I.; Duleba, A.J.; Zhao, L.; Gee, S.J.; Hammock, B.D.; Lasley, B.L. Triclocarban enhances testosterone action: A new type of endocrine disruptor? Endocrinology 2008, 149, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.C.; Zhao, B.; Chen, J.; Cherednichenko, G.; Sanmarti, E.; Denison, M.S.; Lasley, B.; Pessah, I.N.; Kültz, D.; Chang, D.P.; Gee, S.J.; Hammock, B.D. In vitro biologic activities of the antimicrobials triclocarban, its analogs, and triclosan in bioassay screens: Receptor-based bioassay screens. Environ. Health Perspect. 2008, 116, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Duleba, A.J.; Ahmed, M.I.; Sun, M.; Gao, A.C.; Villanueva, J.; Conley, A.J.; Turgeon, J.L.; Benirschke, K.; Gee, N.A.; Chen, J.; Green, P.G.; Lasley, B.L. Effects of triclocarban on intact immature male rat: Augmentation of androgen action. Reprod. Sci. 2011, 18, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Ying, G.G.; Ma, Y.B.; Chen, Z.F.; Lai, H.J. Field dissipation of four personal care products in biosolids-amended soils in North China. Environ. Toxicol. Chem. 2014, 33, 2413–2421. [Google Scholar] [CrossRef] [PubMed]
- Mathews, S.; Henderson, S.; Reinhold, D. Uptake and accumulation of antimicrobials, triclocarban and triclosan, by food crops in a hydroponic system. Environ. Sci. Pollut. Res. Int. 2014, 21, 6025–6033. [Google Scholar] [CrossRef] [PubMed]
- Prosser, R.S.; Lissemore, L.; Topp, E.; Sibley, P.K. Bioaccumulation of triclosan and triclocarban in plants grown in soils amended with municipal dewatered biosolids. Environ. Toxicol. Chem. 2014, 33, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, R.C.; Menn, F.M.; Healy, L.; Fecteau, K.A.; Hu, P.; Bae, J.; Gee, N.A.; Lasley, B.L.; Zhao, L.; Chen, J. Early life triclocarban exposure during lactation affects neonate rat survival. Reprod. Sci. 2015, 22, 75–89. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liu, X.; Zhang, J.; Shao, B. Analysis of triclosan and triclocarban in human nails using isotopic dilution liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2013, 934, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Schebb, N.H.; Ahn, K.C.; Dong, H.; Gee, S.J.; Hammock, B.D. Whole blood is the sample matrix of choice for monitoring systemic triclocarban levels. Chemosphere 2012, 87, 825–827. [Google Scholar] [CrossRef] [PubMed]
- Birch, C.G.; Hiles, R.A.; Eichhold, T.H.; Jeffcoat, A.R.; Handy, R.W.; Hill, J.M.; Willis, S.L.; Hess, T.R.; Wall, M.E. Biotransformation products of 3,4,4'-trichlorocarbanilide in rat, monkey, and man. Drug Metab. Dispos. 1978, 6, 169–176. [Google Scholar] [PubMed]
- Nolen, G.A.; Dierckman, T.A. Reproduction and teratogenic studies of a 2:1 mixture of 3,4,4'-trichlorocarbanilide and 3-trifluoromethyl-4,4'-dichlorocarbanilide in rats and rabbits. Toxicol. Appl. Pharmacol. 1979, 51, 417–425. [Google Scholar] [CrossRef]
- Hiles, R.A. Metabolism and toxicity of halogenated carbanilides: Absorption, distribution and excretion of radioactivity from 3,4,4'-trichloro[14C]carbanilide (TCC) and 3-trifluoromethyl-4,4'-dichloro[14C]carbanilide (TFC) in rats. Food Cosmet. Toxicol. 1977, 15, 205–211. [Google Scholar] [CrossRef]
- Jeffcoat, A.R.; Handy, R.W.; Francis, M.T.; Willis, S.; Wall, M.E.; Birch, C.G.; Hiles, R.A. The metabolism and toxicity of halogenated carbanilides. Biliary metabolites of 3,4,4'-trichlorocarbanilide and 3-trifluoromethyl-4,4'-dichlorocarbanilide in the rat. Drug Metab. Dispos. 1977, 5, 157–166. [Google Scholar] [PubMed]
- USEPA Method 1694: Pharmaceutical and Personal Care Prodcuts in Water, Soil, Sediment, and Biosolids by HPLC/MS/MS; USEPA: Washington, DC, USA, 2007; EPA-821-R-08-002.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kennedy, R.C.; Fling, R.R.; Terry, P.D.; Menn, F.-M.; Chen, J.; Borman, C.J. Extraction of 3,4,4′-Trichlorocarbanilide from Rat Fecal Samples for Determination by High Pressure Liquid Chromatography–Tandem Mass Spectrometry. Int. J. Environ. Res. Public Health 2015, 12, 8125-8132. https://doi.org/10.3390/ijerph120708125
Kennedy RC, Fling RR, Terry PD, Menn F-M, Chen J, Borman CJ. Extraction of 3,4,4′-Trichlorocarbanilide from Rat Fecal Samples for Determination by High Pressure Liquid Chromatography–Tandem Mass Spectrometry. International Journal of Environmental Research and Public Health. 2015; 12(7):8125-8132. https://doi.org/10.3390/ijerph120708125
Chicago/Turabian StyleKennedy, Rebekah C., Russell R. Fling, Paul D. Terry, Fu-Min Menn, Jiangang Chen, and Christopher J. Borman. 2015. "Extraction of 3,4,4′-Trichlorocarbanilide from Rat Fecal Samples for Determination by High Pressure Liquid Chromatography–Tandem Mass Spectrometry" International Journal of Environmental Research and Public Health 12, no. 7: 8125-8132. https://doi.org/10.3390/ijerph120708125
APA StyleKennedy, R. C., Fling, R. R., Terry, P. D., Menn, F.-M., Chen, J., & Borman, C. J. (2015). Extraction of 3,4,4′-Trichlorocarbanilide from Rat Fecal Samples for Determination by High Pressure Liquid Chromatography–Tandem Mass Spectrometry. International Journal of Environmental Research and Public Health, 12(7), 8125-8132. https://doi.org/10.3390/ijerph120708125







