Interventions to Reduce Harm from Smoking with Families in Infancy and Early Childhood: A Systematic Review
Abstract
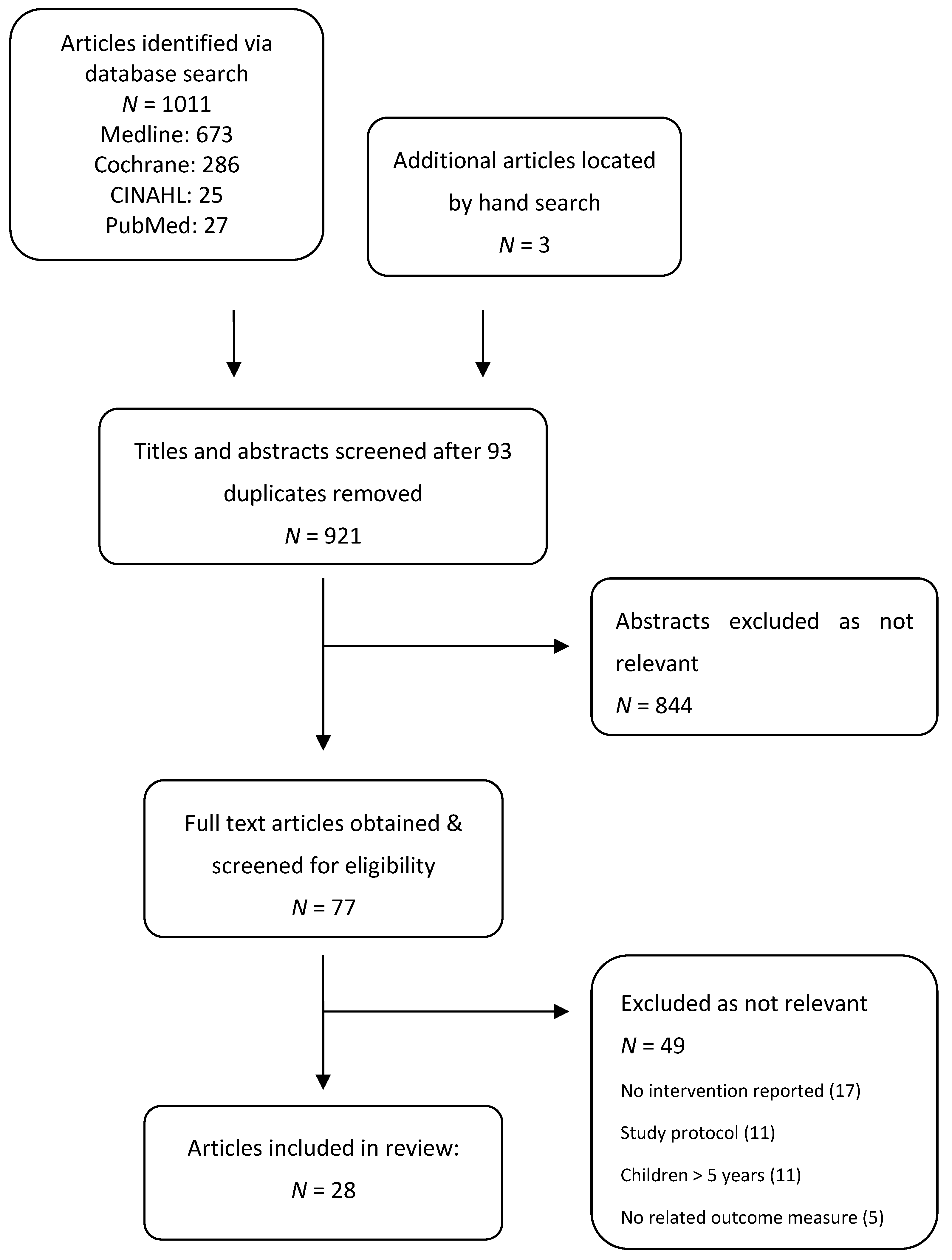
:1. Introduction
Objectives
2. Methods
2.1. Protocol
2.2. Eligibility Criteria
2.3. Information Sources
| Term set 1: Child * |
| Term set 2: Parent * OR father * OR mother * OR caregivers OR famil * OR school * OR communit * |
| Term set 3: Cigar * OR tobacco * OR smok * OR smoking cessation OR tobacco cessation OR tobacco smoke pollution OR smoking abstinence |
| Term set 4: prevent * OR control * |
| Term set 5: intervention OR clinical trial OR pilot study OR outcomes OR randomised control trial |
| Term set 6: 1 and 2 and 3 and 4 and 5 |
2.4. Study Selection
2.5. Data Collection Process and Data Items
2.6. Risk of Bias
2.7. Synthesis of Results
3. Results
3.1. Smoking Cessation Interventions
3.1.1. Target Populations
| Reference | Focus | Design | Internal Validity | External Validity |
|---|---|---|---|---|
| Phillips et al. 2012. USA [35] | Smoking relapse prevention | RCT | Good | Good |
| Hovell et al. 2009. USA [34] | Smoking cessation/SHS reduction | RCT | Good | Good |
| Kuiper et al. 2005. Schonberger et al. 2005. Netherlands [36,37] | Smoking cessation/SHS reduction | RCT | Fair | Good |
| Chan-Yeung et al. 2000; Becker et al. 2004, Chan-Yeung et al. 2005. Canada [55,56,57] | SHSe reduction | RCT | Fair | Good |
| Conway et al. 2004. USA [59] | SHSe reduction | RCT | Fair | Good |
| Joseph et al. 2014. USA [43] | Smoking cessation | Pilot Quasi-experimental | Fair | Fair |
| Jiminez-Muro et al. 2013. Spain [38] | Smoking cessation/relapse prevention | RCT | Fair | Fair |
| Storrø et al. 2010. Norway [42] | Smoking reduction | Cohort control trial with one year time difference | Fair | Fair |
| Winickoff et al. 2010. USA [40] | Smoking cessation/reduction | Quasi RCT | Fair | Fair |
| Hannover et al. 2009. Germany [39] | Smoking cessation/relapse prevention | Quasi RCT | Fair | Fair |
| Kallio et al. 2006. Finland [46] | Smoking cessation/reduction/SHS reduction | RCT (longitudinal) | Fair | Fair |
| Abdullah et al. 2005. Hong Kong [45] | Smoking cessation | RCT | Fair | Fair |
| Wiggins et al. 2005. UK [47] | Smoking cessation | RCT | Fair | Fair |
| Baheiraei et al. 2011. Iran [53] | SHSe reduction | RCT | Fair | Fair |
| Emmons et al. 2001. USA [52] | SHSe reduction | RCT | Fair | Fair |
| Kitzman et al. 2010. USA [61] | Smoking prevention | RCT (longitudinal) | Fair | Fair |
| Øien et al. 2008. Norway [44] | Smoking cessation | Control trial | Fair | Poor |
| Culp et al. 2007. USA [48] | Smoking cessation | Quasi-experimental | Fair | Poor |
| Wilson et al. 2013. Scotland [54] | SHSe reduction | Pilot RCT | Fair | Poor |
| Huang et al. 2013. Taiwan [60] | SHSe reduction | RCT | Poor | Fair |
| Harutyunyan et al. 2013. Armenia [50] | SHSe reduction | RCT | Poor | Fair |
| Fossum et al. 2004. Sweden [58] | SHSe reductions | CT | Poor | Fair |
| Zakarian et al. 2004. USA [51] | SHSe reduction | Quasi-experimental | Fair | Poor |
| Disantis et al. 2010. USA [41] | Smoking cessation/relapse prevention | Pilot 2 arm experimental | Poor | Poor |
| Yücel et al. 2014. Turkey [49] | SHSe reduction | RCT | Poor | Poor |
| Reference | Focus | Participants | Design | Outcomes/Results |
|---|---|---|---|---|
| Joseph et al. 2014. USA [43] | To investigate feasibility of screening serum cotinine with lead screening to increase parental smoking cessation and implementation of home smoking restrictions. | 80 smoking parents of children at well child clinics for 12 and 24 month checks. | Pilot Quasi-experimental | Parent smoking cessation: 74% engaged in smoking counselling and 24% accessed NRT. 7 day point prevalence abstinence at 8 weeks: IG 11/40 (29%) vs. CG 1/40 (p = 0.001). |
| Home smoking restrictions: High levels of smoking restriction at baseline in both groups, change not significant (IG full ban: 67.5% at baseline vs. 86.8% at 8 weeks; CG full ban 77.5% at baseline vs. 80% at 8 weeks). | ||||
| Jiminez-Muro et al. 2013. Spain [38] | To analyse the efficacy of a motivational interview intervention in postpartum women to prevent relapse in recent quitters and encourage behaviour change in those still smoking. | 412/626 postpartum women smokers. 64% Spanish, 34% immigrants. | RCT | Continuous abstinence: Probability of remaining abstinent at 12 week was 74% (IG) & 37% (CG) (p < 0.001). |
| Urine Cotinine: Only 49% of participants attended 3 month visit and therefore biochemical validation was not statistically significant (int 31%, control 23%, n.s.). | ||||
| Phillips et al. 2012. USA [35] | To reduce smoking relapse and prolong breastfeeding in mothers during the first 8 weeks postpartum. | 54 mothers of an infant in NICU. Mothers had a history of tobacco use during or within one year of pregnancy, but currently not smoking. | RCT | Maternal smoking status at 8 weeks postpartum: Significant decrease in smoking relapse at 8 weeks postpartum in the int gp (IG: 81% vs. CG: 46%, p < 0.001). |
| Salivary cotinine: A 94% agreement was found between salivary cotinine level and mothers reported smoking status. | ||||
| Disantis et al. 2010. USA [41] | To pilot a postpartum smoking intervention that combined postpartum smoking cessation & relapse prevention with breastfeeding counselling. | 31 low income women who were either current smokers or recent ex-smokers. Hispanic (50%), African-American (25%). Primiparous (45.8%). 62.5% completed high school or higher education. Years of smoking M = 6.96 years (SD = 5.67). Daily cigarettes M = 12.5 (SD = 7.7) 51% quit smoking prior to pregnancy. | Pilot 2 arm experimental | 7-day point prevalence: S + B: 50%; RP: 75%, not significant. |
| Days to relapse: related to duration of breastfeeding (r = 0.92, p = 0.08). | ||||
| S + B: mothers who quit before or during pregnancy had higher rates of smoking abstinence than those who smoked through pregnancy (x2 = 4.00, p < 0.05). | ||||
| Storrø et al. 2010. Norway [42] | To evaluate the impact of a primary prevention intervention program on risk behaviour for allergic disease in primary health care settings (increase cod liver and oily fish intake, reduce parental smoking, reduce indoor dampness). | 2860 pregnant women or women with a child <2 years of age. | Cohort control trial with one year time difference | Maternal smoking frequencies: Significant and stable decline in smoking from pregnancy to 2 years postnatal, not attributable to intervention. In addition, there was a statistically significant annual trend in the control cohort. (Baseline: IG 17.3% vs. CG 23.6%. OR 0.70, 95% CI 0.60-0.82. 6 weeks: IG 5.3% vs. CG 10.8%. OR 0.55, 95% CI 0.42-0.70. 2 years: IG 9.9% vs. CG 19%. OR 0.50, 95% CI 0.41–0.61). |
| Winickoff et al. 2010. USA [40] | To test an intervention to address maternal and paternal smoking during postpartum hospitalization. | 101/173 parents. 71% current smokers, 29% recent quitters. 67% female. | Quasi RCT | 7 day point prevalence of cotinine verified tobacco abstinence for 3 months: Self-reported 7 day abstinence not significant (IG: Decreased 31% to 25%; CG: 38% to 28%. Effect 9.4%, n.s.). Cotinine confirmed 7 day abstinence rate at follow up IG: 9% vs. CG: 3% (n.s.). |
| Self-reported 24 h quit attempts: IG: 64%; CG: 18%, p = 0.005. | ||||
| Hannover et al. 2009. Germany [39] | To test the efficacy of an intervention to aid cessation/relapse prevention for postpartum women. | 644 women from 6 hospitals with postpartum units. | Quasi RCT | Sustained abstinence (Still not smoking at 6 months or since birth): No statistically significant difference at follow up. |
| Repeated 4 week point prevalence (not smoking 4 weeks prior to follow up). | ||||
| No statistically significant difference in sustained abstinence at either follow up. Statistically significant 4 week point prevalence abstinence at 6 months only. | ||||
| Hovell et al. 2009. USA [34] | To test the effects of SHS and smoking counselling in high risk families. | 150/244 mothers of children aged less than 4 years exposed to minimum of 3 maternal cigarettes per day. | RCT | Reported SHS exposure: Decrease in both IG (80%) & CG (55%) in first 6 months. Group main effect 6–18 months significant for IG (p = 0.011). |
| Child urine cotinine: Decreased baseline to 6 months only (25% both gps). Only the group main effect significant for 6–18 months (p = 0.026). Controls higher throughout baseline & follow up. | ||||
| Maternal smoking (self-report): 6 months: IG decreased by 34%, CG decreased 5%. 6–18 months: IG decreased by 33% CG, 4.6%. | ||||
| Smoking cessations: 17% IG and 5.4% CG quit smoking for 7 days before one or more study measures. | ||||
| Øien et al. 2008. Norway [44] | Investigate parental smoking behaviour during pregnancy after introduction of a prenatal, structure, multidisciplinary smoking cessation intervention. | 3839 pregnant women attending primary health care settings. Estimated participation rate of 44% of eligible women in the location (Tondheim). Low smoking prevalence at inclusion (IG: 4.9%, CG: 7.1%). | Control trial | Self-reported smoking behaviour 6 weeks postnatal. No significant difference between IG and CG. |
| Culp et al. 2007. USA [48] | Evaluate health and safety intervention with first time mothers. | 355 pregnant women in rural south-western states (IG: n = 156, CG: n = 107). 61% smokers. | Quasi-experimental | Maternal smoking behaviour (no. of cigarettes/day): Baseline: n/s between IG and CG. Six months: IG smoking 2.4 fewer cigarettes per day (IG: M = 6.34; SD = 6.95 vs. CG: M = 8.72, SD 7.26, t (147) = 2.0, p = 0.023). Twelve months: IG smoking 2.1 fewer cigarettes per day (IG: M = 7.28, SD = 6.79 vs. CG: M = 9.41, SD = 7.09) t (147) = 1.82, p = 0.071.) |
| Knowledge of the effects of smoking on child development: e.g., Impaired brain development (IG 59.2% vs. CG 41.7%, p ≤ 0.01); lower mental health scores (IG 52.6% vs. CG 32.3%, p < 0.001). | ||||
| Kallio et al. 2006. Finland [46] | To determine whether repeated lifestyle counselling alters parental smoking and child exposure to tobacco smoke. | 1062/1105 parents of infants attending a well baby clinic. | RCT (longitudinal) | Parent smoking: Decreased across IG and CG over time. No significant difference between groups. |
| Serum cotinine of children: 46% of 8 year olds had been exposed to nicotine in last few days. None had high enough levels to confirm that they had smoked. Serum cotinine highest in children with both parent smokers. Serum cotinine higher in families where only father smoked than where only mother smoked. 24% of children from non-smoking families had cotinine higher than 1 ng/mL. | ||||
| Abdullah et al. 2005. Hong Kong [45] | To evaluate whether telephone counselling based on stages of change could help non-motivated smoking parents of young children to cease. | 952 smoking parents of Chinese children aged 5 years (85.3% fathers). | RCT | 7 point prevalence quite rate at 6 months: Higher in IG (15.3%: 68/444) than CG (7.4%: 34/459) p < 0.001. Absolute risk reduction 7.9% (95% CI: 3.78% to 12.01%). Number needed to treat 13 (95% CI: 8–26). |
| Kuiper et al. 2005. Schonberger et al. 2005. Netherlands [36,37] | To evaluate a multifaceted intervention strategy to reduce occurrence of severe asthma (smoking cessation, SHSE avoidance, dust mite avoidance, breastfeeding, timing of introduction of solid food). | Parents of 476 infants at high risk of severe asthma. | RCT | Self-report of SHSe at one year: No data reported. Authors state “No difference was found in the intervention compared with the control group concerning the exposure to tobacco smoke” (p. 329). |
| CO monitoring: No results reported. | ||||
| Wiggins et al. 2005. UK [47] | To evaluate the effect of two forms of postpartum social support (support health visitor (SVH) or community group support (CGS) on maternal and child health outcomes (maternal smoking). | 731 women with infants from culturally diverse and disadvantaged inner city areas of London. Approx 26%–30% smokers across groups. 14% non-English speakers. | RCT | Maternal smoking: not significantly reduced (SVH vs. CG: 95% CI 0.86 (0.62, 1.19); CGS vs. CG: 95% CI 0.97 (0.72, 1.33). |
| Yücel et al. 2014. Turkey [49] | To evaluate the effectiveness of an intensive intervention vs. a minimal intervention to reduce SHSe. | Parents of 182 children aged 1–5 years. | RCT | Urinary cotinine–pre and post intervention: Urine cotinine decreased across time in both groups. Decrease greater in intensive IG than minimal IG, but n.s. (p = 0.831). |
| Complete home smoking bans: Authors report that 30.6% of Intensive IG households who did not have a ban at baseline, did have a total ban at 3 months (p = 0.001). In the minimal IG, 10.5% more families had ban at 3 months, but n.s (p = 0.125). | ||||
| Wilson et al. 2013. Scotland [54] | To investigate feasibility of an intervention (REFRESH) to reduce SHSe for children in their homes. | 59/1693 smoking mothers with at least one child younger than 6 years. Maternal age M = 30 years; child age M = 3.5 years (range 1.2–5.7 years). | Pilot RCT | Difference in PM2.5 from visit 2 to visit 4: Greater reduction achieved for maximum PM. |
| Peak concentration of PM2.5: IG 67 vs. CG 148 (p = 0.006). | ||||
| The percentage of time when household PM2.5 concentrations exceeded 35 μ/m3: IG 0.49 vs. CG 3.6 (p = 0.017). | ||||
| Children’s salivary cotinine: No significant difference. | ||||
| Feasibility, acceptability and understanding of intervention: Qualitative data–intervention was acceptable and mothers were able to understand the data. | ||||
| Motivators and mechanisms of change: Personalised data made the concept of the dangers of SHSe more real to them and mothers reported a greater sense of motivation for change. | ||||
| Huang et al. 2013. Taiwan [60] | To evaluate the effectiveness of a transtheoretical model- based passive smoking prevention program for pregnant women and mothers of young children. | 294/335 women recruited from obstetrics and paediatric departments of four hospitals. IG: 48% pregnant. CG: 45% pregnant. Remainder mothers of children aged <3 years. | RCT | Stages of change: 73% were already in target stage at baseline. Less than 30% of the remaining changed stage. Distribution of stages of change statistically different after intervention between participant groups (mothers with children: F = 11.978, p = 0.003; pregnant women: F = 6.689, p = 0.035). |
| Knowledge: No significant difference between groups pre or post test. | ||||
| Frequency of avoiding passive smoking: Significant difference in intervention group (F = 5.115, p = 0.25) at post-test. | ||||
| Self-efficacy: No significant difference. | ||||
| Harutyunyan et al. 2013. Armenia [50] | To test an intense intervention to reduce child SHSe. | 250 households with children aged 2–6 years recruited via paediatrician primary health care clinics.Maternal age M = 30 years (SD 5.2 years). 53% employed, 36% had a university degree. Household smokers predominately fathers (80%). Child age M = 4 years (SD 1.2 years). Smoking was permitted in all households, some restrictions in approximately half of homes. | RCT | Child hair nicotine concentration: 17% lower in IG than CG although not significant (p = 0.239). Significantly decreased in IG from baseline to follow up (0.30 ng/mg to 0.23 ng/mg; p = 0.77). |
| Maternal knowledge of SHSe and smoking hazards: IG: From 9.5 at baseline to 11.3 at follow up. CG: From 9.8 to 10.5. 10% higher in IG than CG after controlling for baseline score (p = 0.006). | ||||
| Baheiraei et al. 2011. Iran [53] | To assess whether counselling both mother and father reduces infant SHSe. | 130 parents of health infants (<12 months) with at least one parent smoker. Families from predominately lower SES. | RCT | Urine cotinine: Decreased for both groups but significantly decreased in IG (Baseline: IG 48.72 vs. CG 40.83; 3 months IG: 28.68 vs. CG 3.32). p = 0.029). |
| Total daily cigarette consumption: Greater decrease in presence of child in IG (median = 0, interquartile range: 0, 2.71) than CG (median = 1, interquartile range: 0, 3.21) at the 3 month follow up (one tailed p, 0.3). No significant correlation between cigarettes consumed and reported level of SHSe. | ||||
| Home and car smoking bans: Increase in both IG & CG, but not significant in CG. Statistically significant between groups (p = 0.49). | ||||
| Fossum et al. 2004. Sweden [58] | To evaluate the effects of a counselling intervention (Smoke Free children). | 41 mothers of newborn infants attending child health clinics. | CT | Self reported smoking: More IG mothers reported smoking at baseline (M = 13.1, SD 6.5 than CG (M = 10.8, SD 5.7) and after intervention (M = 12.8, SD 5.9) than CG (M = 8.2, SD 4.3). |
| Maternal saliva cotinine: Cotinine levels increased by 40% in CG and decreased by 10% in IG (F = 5.501, df = 1, p = 0.027). | ||||
| Zakarian et al. 2004. USA [51] | To evaluate the effectiveness of a behavioural counselling program for reducing child SHSe. | 150 mothers of children aged less than 4 years attending a well-child community clinic. Most mothers were White, not employed, low education. Approximately 40% were single parents. | Quasi-experimental | Maternal report of child SHSe (number of maternal cigarettes child exposed to per week: Declined for baseline to 6 months post-test for both groups (IG: 18.89 at baseline to 5.41 at 12 months. CG: 13.25 at baseline to 5.23 at 12 months) (p < 0.001). Data presented in graph difficult to report exact results. Priest et al. (2008) reported data. Total exposure to cigarettes/week (IG 53.2 at baseline to 21.99 at 12 months. CG: l 54.48 atbaseline to 18.22 at 12 months) (p < 0.001). |
| No significant group x time differences. Number of counselling sessions completed was not a significant covariate. | ||||
| Children’s urinary cotinine concentration: No significant change over time in either group. No significant group x time or group differences. | ||||
| Maternal smoking rates: Similar to SHSe above, a sharp decline from baseline to post-test across both groups. | ||||
| Maternal smoking cessation: Self-reported 7-day quit status did not vary by experimental group at any time point. | ||||
| Chan-Yeung et al. 2000; Becker et al. 2004, Chan-Yeung et al. 2005. Canada [55,56,57] | Prevention of asthma in high-risk infants via multifaceted intervention program (house dust mite control, pet avoidance, avoidance of ETS, promotion of breastfeeding). | 545 infants at high risk for asthma and their families. 7% of mothers smoking at baseline (36/493). | RCT | Parental smoking cessation: No significant difference in proportion of mothers, fathers or others who gave up or acquired smoking at 12 months. |
| Conway et al. 2004. USA [59] | To evaluate the effectiveness of a lay delivered intervention to reduce ETS exposure in Latino children. | 143 Latino parent-child pairs. Child age 1–9 years (M = 4 years). | RCT | Child hair nicotine (log ng/mg): Baseline (IG: 0.25 vs. CG 0.23), post intervention (IG: 0.17 vs. CG: 0.19, 3 months (IG: 0.28 vs. CG 0.32), 12 months: (IG: 0.23 vs. CG: 0.23). No significant differences between groups over time. |
| Child hair cotinine (log ng/mg): Baseline (IG 0.05 vs. CG 0.05), post intervention (IG 0.03 vs. CG 0.03), 3 months (IG 0.04 vs. CG 0.04), 12 month (IG 0.02 vs. CF 0.04). No significant differences between groups, but time effect detected (p < 0.001). | ||||
| Parent report of number of cigarettes child exposed to in household over one month: Baseline (IG 1.75 vs. CG 1.85), post intervention (IG 1.42 vs. CG 1.62), 3 months (IG: 1.27 vs. CG 1.44), 12 months (IG: 1.06 vs. CG 1.27). No significant difference between groups, trending toward significance over time (p = 0.048). | ||||
| Confirmed reduction (dichotomous variable based on parent report and child hair biomarkers: Not significant. | ||||
| Emmons et al. 2001. USA [52] | Outcome evaluation of project KISS (Keep Infants Safe From Smoke). | 291 smoking low-income parent/caregivers. Children younger than 3 years. | RCT | Nicotine levels in household: significant time-by-treatment effect (F (2406) = 4.80, p < 0.01). IG: Levels at 3 & 6 months significantly lower than baseline (F (2200) = 4.36; p < 0.5). |
| Smoking cessation: Overall cessation 7.5% CG vs. 10.1% IG. No significant difference between groups. | ||||
| Kitzman et al. 2010. USA [61] | To test the effect of prenatal and infancy home visits by nurses on 12 year old first born children’s use of substances (cigarettes, alcohol, marijuana). | 1139 low SES African-American women pregnant with first child. | RCT (longitudinal) | Substance use by children: IG less likely to have used substances (CG: 5.1 vs. IG 1.7, OR 0.31, p = 0.04), to have used fewer of these substances (incidence ratio = 0.22, p = 0.02) and to have used these substances for fewer days (incidence ratio, 0.15, p = 0.02). |
3.1.2. Interventions
3.1.3. Outcome Measures
3.1.4. Effectiveness
| Author | Content | Delivery Personnel | Method of Communication | Intensity/Complexity | Environment | Conceptual Framework | Socio-Ecological Model | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Smoking cessation/relapse prevention | |||||||||||||||||||
| Joseph et al. 2014 [43] | Serum cotinine feedback, SHSe education, optional counselling, optional NRT | Trained tobacco advisor | Mail and phone | Weekly for 8 weeks | Home | MI, CBT | Intrapersonal | ||||||||||||
| Jiminez-Muro et al. 2013 [38] | Risks of smoking, health behaviours | Research student | Phone | 5 × 15 minute calls over 3 months | Home (phone) | MI | Intrapersonal | ||||||||||||
| Phillips et al. 2012 [35] | Newborn cues | Not stated. Partially self-administered | DVD Brochure | Not described | Hospital and home | Attachment theory | Intrapersonal | ||||||||||||
| Disantis et al. 2010 [41] | Smoking and breastfeeding counselling OR relapse prevention | Counsellor | Face to face Written materials | 15 minutes + written materials | Clinic | Not stated | Intrapersonal | ||||||||||||
| Storro et al. 2010 [42] | Brief 5As | GP or midwife | Face to face | At least 5 occasions | Clinic | Brief 5As | Intrapersonal Interpersonal | ||||||||||||
| Winickoff et al. 2010 [40] | Brief 5 As | Trained study staff | Face to face | 15 minutes + offer to enroll in Quitline | Hospital | Brief 5As | Intrapersonal Interpersonal | ||||||||||||
| Hannover et al. 2009 [39] | Relapse prevention/smoking cessation counselling | Trained study staff | Face to face + phone | Single interview + phone follow up × 2 | Home | MI | Intrapersonal | ||||||||||||
| Hovell et al. 2009 [34] | SHSe reduction and tailored smoking cessation including option of NRT | Study counsellor | Face to face + phone | 14 sessions over 7 weeks. Mean time/session: 23 minutes | Home | Learning theory | Intrapersonal Interpersonal | ||||||||||||
| Oien et al. 2008 [44] | Brief office intervention (Fiore et al. 2000) | Midwives, GP, nurses | Face to face | Not clear | Primary health care | Not stated | Intrapersonal | ||||||||||||
| Culp et al. 2007 [48] | Universal program, including smoking and effect of SHSe on infant growth and development | Visitors with child development degree level qualifications | Face to face | Average 10.9 visits before birth + 20.7 visits after birth (approx 1 h per visit) | Home | Not reported | Intrapersonal | ||||||||||||
| Kallio et al. 2006 [46] | Universal program including smoking | Paediatrician and dietician | Face to face | Paediatrician: every 1–3 months until 2 years Dietician: every 4–6 months until 2 years. Dietician and paediatrician every 6 months until 7 years | Clinic | Not reported | Intrapersonal | ||||||||||||
| Abdullah et al. 2005 [45] | Smoking cessation and SHSe reduction tailored to stage of change. No NRT information | Nurse | Phone + written materials | Three phone calls × 20–30 min | Home via phone | Transtheoretical model (stages of change) | Intrapersonal | ||||||||||||
| Kuiper et al. 2005. Schonberger et al. 2005 [36-37] | Smoking cessation and home bans on smoking | Research nurse | Face to face | Once | Not explained | Not explained | Intrapersonal Interpersonal | ||||||||||||
| Wiggens et al. 2005 [47] | Social support | Health visitor OR non-professional | Face to face | 1.5–10 h | Home OR community centre | Not explained. ? social support | Intrapersonal Interpersonal | ||||||||||||
| SHSe reduction interventions | |||||||||||||||||||
| Yucel et al. 2014 [49] | SHSe information, goal setting, use of resources, urine cotinine feedback | Researcher | Face to face Phone Written materials | Intensive group: Home visits at baseline, 1 & 3 months. Phone calls at 6 & 8 weeks. Minimal intensity group: Home visit at baseline and 3 months. Mail out urine cotinine result | Home | Not stated | Intrapersonal | ||||||||||||
| Wilson et al. 2013 [54] | 24 h measure on home air quality PM2.5 (particulate matter) & motivational interview | Research staff | Face to face | Four visits over a one month period | Home | MI | Intrapersonal | ||||||||||||
| Huang et al. 2013 [60] | Impact of passive smoking, avoiding passive smoke in public and at home. Sections tailored to stages of change. | Research staff | Face to face, audiovisual, written materials, phone | Time not stated. Included DVD, booklet, stickers, phone follow up at 2 weeks and 3 weeks post intervention | Home | Transtheoretical model (stages of change) | Intrapersonal | ||||||||||||
| Harutyunyan et al [50]. | Importance of healthy environment, dangers of smoking and SHSe, smoking cessation, smoke-free home, PM25 feedback, written materials. CG: written materials only | Research staff | Face to face Written materials Phone | 40 minute MI + 2 follow up phone calls (timeframe not specified) | Home | MI | Intrapersonal Interpersonal | ||||||||||||
| Baheiraei et al. 2011 [53] | Smoke free children (Fossum et al. 2004 [58]) | Research student | Face to face Phone Written materials | One face to face interview + two phone interviews (max. 20 min each) | Home | MI | Intrapersonal | ||||||||||||
| Chan-Yeung et al. 2000, Becker et al. 2004, Chan-Yeung et al. 2005 [55,56,57] | Counselled on smoking cessation and instructed to keep house smoke free | Research nurse | Face to face | Single prenatal visit | Home | Risk factors for asthma | Intrapersonal Interpersonal | ||||||||||||
| Conway et al. 2004 [59] | Problem solving aimed at lowering child ETS in the household | Lay bicultural and bilingual Latina community health advisors. All received 20 h training over 4 weeks | Face to face Phone | Six sessions over four months | Home | Not stated, but problem solving, positive reinforcement & social support described. | Intrapersonal Interpersonal | ||||||||||||
| Fossum et al. 2004 [58] | Counselling for effects of SHSe, monitoring SHSe, changing smoking habits, supporting non-smoking | Child health nurses | Face to face | Not explained | Child health clinic | Self-efficacy | Interpersonal | ||||||||||||
| Zakarian et al. 2004 [51] | Behavioural counselling including contracting to reduce SHSe, problem solving, goal setting and self-monitoring | Health educators Nurses Medical assistants | Face to face | Seven counselling sessions over 6 months | Clinic (× 3) Home via phone (× 4) | SLT (Bandura 1977) and behavioural ecological model (Hovell, Wahlgreen & Gehrman, 2002 [ref]) | Interpersonal | ||||||||||||
| Emmons et al. 2001 [52] | Choice, personal responsibility for change, sel-efficacy, feedback on CO level. Tailored to interest in quitting smoking or reducing SHSe | Health educator | Face to face Phone | One 30–45 motivational interview + four follow up phone calls | Home | MI | Interpersonal | ||||||||||||
| Anti-smoking socialisation | |||||||||||||||||||
| Kitzman et al. 2010 [61] | Nurse Family Partnership. Home visiting program during first two years of child’s life (health promotion, parenting support, developmental screening, planning for pregnancies, education and employment) | Nurse | Face to face | Mean visits during pregnancy = 7 (range 0–118). Mean visits during first two years = 26 visits (range 0–71) | Home | Family partnership model | Intrapersonal Interpersonal | ||||||||||||
3.2. Environmental Tobacco Smoke (ETS) Interventions
3.2.1. Target Populations
3.2.2. Interventions
3.2.3. Outcomes
3.2.4. Effectiveness
3.3. Anti-Smoking Socialisation Interventions
4. Discussion
Limitations
5. Conclusions
Author Contributions
Conflicts of Interest
References
- World Health Organization. WHO Report on the Global Tobacco Epidemic, 2013. Available online: http://www.who.int/tobacco/global_report/2013/en/ (accessed on 2 May 2014).
- Öberg, M.; Woodward, A.; Jaakkola, M.S.; Peruga, A.; Prüss-Ustün, A. Global Estimate of the Burden of Disease from Second-Hand Smoke; World Health Organisation: Geneva, Switzerland, 2010. [Google Scholar]
- Matt, G.E.; Quintana, P.J.E.; Destaillats, H.; Gundel, L.A.; Sleiman, M.; Singer, B.C.; Jacob, P., III; Benowitz, N.; Winickoff, J.P.; Rehan, V.; Talbot, P.; Schick, S.; Samet, J.; Yinsheng, W.; Bo, H.; Martins-Green, M.; Pankow, J.F.; Hovell, M. Thirdhand tobacco smoke: Emerging evidence and arguments for a multidisciplinary research agenda. Environ. Health Perspect. 2011, 119, 1218–1226. [Google Scholar]
- Protano, C.; Vitali, M. The new danger of thirdhand smoke: Why passive smoking does not stop at secondhand smoke. Environ. Health Perspect. 2011, 119, A422–A422. [Google Scholar] [CrossRef] [PubMed]
- Been, J.V.; Nurmatov, U.B.; Cox, B.; Nawrot, T.S.; van Schayck, C.P.; Sheikh, A. Effect of smoke-free legislation on perinatal and child health: A systematic review and meta-analysis. Lancet 2014, 383, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Leonardi-Bee, J.; Jere, M.L.; Britton, J. Exposure to parental and sibling smoking and the risk of smoking uptake in childhood and adolescence: A systematic review and meta-analysis. Thorax 2011, 66, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Loke, A.Y.; Wong, Y.P.I. Smoking among young children in Hong Kong: Influence of parental smoking. J. Adv. Nurs. 2010, 66, 2659–2670. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, A.V.; Shete, S.; Prokhorov, A.V.; Wilkinson, A.V.; Shete, S.; Prokhorov, A.V. The moderating role of parental smoking on their children’s attitudes toward smoking among a predominantly minority sample: A cross-sectional analysis. Subst. Abuse Treat. Prev. Policy 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Bricker, J.B.; Peterson, A.V., Jr.; Leroux, B.G.; Andersen, M.R.; Rajan, K.B.; Sarason, I.G. Prospective prediction of children’s smoking transitions: Role of parents’ and older siblings’ smoking. Addiction 2006, 101, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-Y.; Wu, W.-C.; Wu, C.-C.; Cheng, J.Y.; Hurng, B.-S.; Yen, L.-L. The incidence of experimental smoking in school children: An 8-year follow-up of the child and adolescent behaviors in long-term evolution (CABLE) study. BMC Public Health 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Vuolo, M.; Staff, J. Parent and child cigarette use: A longitudinal, multigenerational study. Pediatrics 2013, 132, 568–577. [Google Scholar] [CrossRef]
- Hahn, E.J.; Hall, L.A.; Rayens, M.K.; Burt, A.V.; Corley, D.; Sheffel, K.L. Kindergarten children’s knowledge and perceptions of alcohol, tobacco, and other drugs. J. School Health 2000, 70, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Dalton, M.A.; Bernhardt, A.M.; Gibson, J.J.; Sargent, J.D.; Beach, M.L.; Adachi-Mejia, A.M.; Titus-Ernstoff, L.T.; Heatherton, T.F. Use of cigarettes and alcohol by preschoolers while role-playing as adults: “Honey, have some smokes”. Arch. Pediatr. Adolesc. Med. 2005, 159, 854–859. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, R.N.; Engels, R.C.; Scholte, R.H. Parental smoking and pretend smoking in young children. Tob. Control 2010, 19, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Hruba, D.; Zaloudikova, I. Why to smoke? Why not to smoke? Major reasons for children’s decisions on whether or not to smoke. Cent. Eur. J. Public Health 2010, 18, 202–208. [Google Scholar] [PubMed]
- Jackson, C.; Dickinson, D. Enabling parents who smoke to prevent their children from initiating smoking: Results from a 3-year intervention evaluation. Arch. Pediatr. Adolesc. Med. 2006, 160, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Kumpfer, K.L.; Alvarado, R. Family-strengthening approaches for the prevention of youth problem behaviours. Amer. Psychol. 2003, 58, 457–465. [Google Scholar] [CrossRef]
- Huisman, M.; Van Lenthe, F.J.; Giskes, K.; Kamphuis, C.B.; Brug, J.; Mackenbach, J.P. Explaining socio-economic inequalities in daily smoking: A social-ecological approach. Eur. J. Public Health 2012, 22, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Scollo, M.; Winstanley, M. Tobacco in Australia: Facts and issues. In Melbourne: Cancer Council Victoria, 3rd ed.; Cancer Council Victoria: Melbourne, Australia, 2008; Available online: http://www.tobaccoinaustralia.org.au (accessed on 20 August 2014).
- Siahpush, M. Why is lone-motherhood so strongly associated with smoking? Aust. N. Zeal. J. Public Health 2004, 28, 37–42. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. National Aboriginal and Torres Strait Islander Social Survey, 2008. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/mf/4714.0/ (accessed on 11 October 2014).
- Lyvers, M.; Hall, T.; Bahr, M. Smoking and psychological health in relation to country of origin. Int. J. Psychol. 2009, 44, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Purnell, J.Q.; Peppone, L.J.; Alcaraz, K.; McQueen, A.; Guido, J.J.; Carroll, J.K.; Shacham, E.; Morrow, G.R. Perceived discrimination, psychological distress, and current smoking status: Results from the behavioral risk factor surveillance system reactions to race module, 2004–2008. Amer. J. Public Health 2012, 102, 844–851. [Google Scholar] [CrossRef]
- McLeroy, K.R.; Bibeau, D.; Steckler, A.; Glanz, K. An ecological perspective on health promotion programs. Health Educ. Behav. 1988, 15, 351–377. [Google Scholar] [CrossRef]
- Patnode, C.D.; O’Connor, E.; Whitlock, E.P.; Perdue, L.A.; Soh, C.; Hollis, J. Primary care–relevant interventions for tobacco use prevention and cessation in children and adolescents: A systematic evidence review for the U.S. preventive services task force. Ann. Intern. Med. 2013, 158, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Muller-Riemenschneider, F.; Bockelbrink, A.; Reinhold, T.; Rasch, A.; Greiner, W.; Willich, S.N. Long-term effectiveness of behavioural interventions to prevent smoking among children and youth. Tob. Control 2008, 17, 301–302. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Perera, R. School-based programmes for preventing smoking. Cochrane Database Syst. Rev. 2013, 8, 1616–2040. [Google Scholar]
- Thomas, R.E.; Baker, P.; Lorenzetti, D. Family-based programmes for preventing smoking by children and adolescents. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions. Available online: http://www.cochrane-handbook.org/ (accessed on 20 October 2013).
- Moher, D.; Liberati, A.; Tetziaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. PLoS Med. 2009, 6. [Google Scholar] [CrossRef]
- Abraham, C.; Michie, S. A taxonomy of behavior change techniques used in interventions. Health Psychol. 2008, 27, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Agency for Healthcare Research and Quality. U.S. Preventative Services Taskforce Procedures Manual. Available online: http://www.uspreventiveservicestaskforce.org/uspstf08/methods/procmanual.pdf (accessed on 6 February2014).
- Popay, J.; Roberts, H.; Sowden, A.; Petticrew, M.; Arai, L.; Rodgers, M.; Britten, N.; Roen, K.; Duffy, S. Guidance on the Conduct of Narrative Synthesis in Systematic Reviews; Lancaster University: New York, NY, USA, 2006. [Google Scholar]
- Hovell, M.F.; Zakarian, J.M.; Matt, G.E.; Liles, S.; Jones, J.A.; Hofstetter, C.R.; Larson, S.N.; Benowitz, N.L. Counseling to reduce children’s secondhand smoke exposure and help parents quit smoking: A controlled trial. Nicotine Tob. Res. 2009, 11, 1383–1394. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.M.; Merritt, T.A.; Goldstein, M.R.; Deming, D.D.; Slater, L.E.; Angeles, D.M. Prevention of postpartum smoking relapse in mothers of infants in the neonatal intensive care unit. J. Perinatol. 2012, 32, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Schonberger, H.J.A.M.; Dompeling, E.; Knottnerus, J.A.; Maas, T.; Muris, J.W.M.; van Weel, C.; van Schayck, C.P. The PREVASC study: The clinical effect of a multifaceted educational intervention to prevent childhood asthma. Eur. Respir. J. 2005, 25, 660–670. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, S.; Maas, T.; van Schayck, C.P.; Muris, J.W.M.; Schonberger, H.J.A.M.; Dompeling, E.; Gijsbers, B.; van Weel, C.; Knottnerus, J.A.; the PREVASC group. The primary prevention of asthma in children study: Design of a multifaceted prevention program. Pediatr. Allergy Immunol. 2005, 16, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Muro, A.; Nerin, I.; Samper, P.; Marqueta, A.; Beamonte, A.; Gargallo, P.; Oros, D.; Rodriguez, G. A proactive smoking cessation intervention in postpartum women. Midwifery 2013, 29, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Hannover, W.; Thyrian, J.R.; Roske, K.; Grempler, J.; Rumpf, H.-J.; John, U.; Hapke, U. Smoking cessation and relapse prevention for postpartum women: Results from a randomized controlled trial at 6, 12, 18 and 24 months. Addict. Behav. 2009, 34, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Winickoff, J.P.; Healey, E.A.; Regan, S.; Park, E.R.; Cole, C.; Friebely, J.; Rigotti, N.A. Using the postpartum hospital stay to address mothers’ and fathers’ smoking: The NEWS study. Pediatrics 2010, 125, 518–525. [Google Scholar] [CrossRef] [PubMed]
- DiSantis, K.I.; Collins, B.N.; McCoy, A.C.S. Associations among breastfeeding, smoking relapse, and prenatal factors in a brief postpartum smoking intervention. Acta Obstet. Gynecol. Scand. 2010, 89, 582–586. [Google Scholar] [CrossRef] [PubMed]
- Storro, O.; Oien, T.; Dotterud, C.K.; Jenssen, J.A.; Johnsen, R. A primary health-care intervention on pre- and postnatal risk factor behavior to prevent childhood allergy. The Prevention of Allergy among Children in Trondheim (PACT) study. BMC Public Health 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.; Murphy, S.; Thomas, J.; Okuyemi, K.S.; Hatsukami, D.; Wang, Q.; Briggs, A.; Doyle, B.; Winickoff, J.P. A pilot study of concurrent lead and cotinine screening for childhood tobacco smoke exposure: Effect on parental smoking. Amer. J. Health Promot. 2014, 28, 316–320. [Google Scholar] [CrossRef]
- Oien, T.; Storro, O.; Jenssen, J.A.; Johnsen, R. The impact of a minimal smoking cessation intervention for pregnant women and their partners on perinatal smoking behaviour in primary health care: A real-life controlled study. BMC Public Health 2008, 8. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.S.M.; Mak, Y.W.; Loke, A.Y.; Lam, T.-H. Smoking cessation intervention in parents of young children: A randomised controlled trial. Addiction 2005, 100, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Kallio, K.; Jokinen, E.; Hamalainen, M.; Kaitosaari, T.; Volanen, I.; Viikari, J.; Ronnemaa, T.; Simell, O. Impact of repeated lifestyle counselling in an atherosclerosis prevention trial on parental smoking and children’s exposure to tobacco smoke. Acta Paediatr. 2006, 95, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Wiggins, M.; Oakley, A.; Roberts, I.; Turner, H.; Rajan, L.; Austerberry, H.; Mujica, R.; Mugford, M. The social support and family health study: A randomised controlled trial and economic evaluation of two alternative forms of postnatal support for mothers living in disadvantaged inner-city areas. Health Technol. Assess. 2004, 8, 1–120. [Google Scholar] [CrossRef] [PubMed]
- Culp, A.M.; Culp, R.E.; Anderson, J.W.; Carter, S. Health and safety intervention with first-time mothers. Health Educ. Res. 2007, 22, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Yucel, U.; Ocek, Z.A.; Ciceklioglu, M. Evaluation of an intensive intervention programme to protect children aged 1–5 years from environmental tobacco smoke exposure at home in Turkey. Health Educ. Res. 2014, 29, 442–455. [Google Scholar] [CrossRef] [PubMed]
- Harutyunyan, A.; Movsisyan, N.; Petrosyan, V.; Petrosyan, D.; Stillman, F. Reducing children’s exposure to secondhand smoke at home: A randomized trial. Pediatrics 2013, 132, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Zakarian, J.M.; Hovell, M.F.; Sandweiss, R.D.; Hofstetter, C.R.; Matt, G.E.; Bernert, J.T.; Pirkle, J.; Hammond, S.K. Behavioral counseling for reducing children’s ETS exposure: Implementation in community clinics. Nicotine Tob. Res. 2004, 6, 1061–1074. [Google Scholar] [CrossRef] [PubMed]
- Emmons, K.M.; Hammond, S.K.; Fava, J.L.; Velicer, W.F.; Evans, J.L.; Monroe, A.D. A randomized trial to reduce passive smoke exposure in low-income households with young children. Pediatrics 2001, 108, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Baheiraei, A.; Kharaghani, R.; Mohsenifar, A.; Kazemnejad, A.; Alikhani, S.; Milani, H.S.; Mota, A.; Hovell, M.F. Reduction of secondhand smoke exposure among healthy infants in Iran: Randomized controlled trial. Nicotine Tob. Res. 2011, 13, 840–847. [Google Scholar] [CrossRef] [PubMed]
- Wilson, I.; Semple, S.; Mills, L.M.; Ritchie, D.; Shaw, A.; O’Donnell, R.; Bonella, P.; Turner, S.; Amos, A. REFRESH—Reducing families’ exposure to secondhand smoke in the home: A feasibility study. Tob. Control 2013. [Google Scholar] [CrossRef]
- Becker, A.; Watson, W.; Ferguson, A.; Dimich-Ward, H.; Chan-Yeung, M. The Canadian asthma primary prevention study: Outcomes at 2 years of age. J. Allergy Clin. Immunol. 2004, 113, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Chan-Yeung, M.; Ferguson, A.; Watson, W.; Dimich-Ward, H.; Rousseau, R.; Lilley, M.; Dybuncio, A.; Becker, A. The Canadian childhood asthma primary prevention study: Outcomes at 7 years of age. J. Allergy Clin. Immunol. 2005, 116, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Chan-Yeung, M.; Manfreda, J.; Dimich-Ward, H.; Ferguson, A.; Watson, W.; Becker, A. A randomized controlled study on the effectiveness of a multifaceted intervention program in the primary prevention of asthma in high-risk infants. Arch. Pediatr. Adolesc. Med. 2000, 154, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Fossum, B.; Arborelius, E.; Bremberg, S. Evaluation of a counseling method for the prevention of child exposure to tobacco smoke: An example of client-centered communication. Prev. Med. 2004, 38, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Conway, T.L.; Woodruff, S.I.; Edwards, C.C.; Hovell, M.F.; Klein, J. Intervention to reduce environmental tobacco smoke exposure in Latino children: Null effects on hair biomarkers and parent reports. Tob. Control 2004, 13, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-M.; Wu, H.-L.; Huang, S.-H.; Chien, L.-Y.; Guo, J.-L. Transtheoretical model-based passive smoking prevention programme among pregnant women and mothers of young children. Eur. J. Public Health 2013, 23, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Kitzman, H.J.; Olds, D.L.; Cole, R.E.; Hanks, C.A.; Anson, E.A.; Arcoleo, K.J.; Luckey, D.W.; Knudtson, M.D.; Henderson, C.R., Jr.; Holmberg, J.R. Enduring effects of prenatal and infancy home visiting by nurses on children: Follow-up of a randomized trial among children at age 12 years. Arch. Pediatr. Adolesc. Med. 2010, 164, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.J.; Myers, V.; Hovell, M.; Zucker, D.; Ben Noach, M. Meta-analysis of parental protection of children from tobacco smoke exposure. Pediatrics 2014, 133, 698–714. [Google Scholar] [CrossRef] [PubMed]
- Borland, R.; Partos, T.R.; Yong, H.-H.; Cummings, K.M.; Hyland, A. How much unsuccessful quitting activity is going on among adult smokers? Data from the International Tobacco Control Four Country cohort survey. Addiction 2012, 107, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Mumford, E.A.; Hair, E.C.; Yu, T.-C.; Liu, W. Women’s longitudinal smoking patterns from preconception through child’s kindergarten entry: Profiles of biological mothers of a 2001 U.S. birth cohort. Mater. Child Health J. 2014, 18, 810–820. [Google Scholar] [CrossRef]
- Greaves, L.; Hemsing, N. Women and tobacco control policies: Social-structural and psychosocial contributions to vulnerability to tobacco use and exposure. Drug Alcohol Depend. 2009, 104, S121–S130. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R. Spousal smoking as an indicator of total secondhand smoke exposure. Nicotine Tob. Res. 2009, 11, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Kayser, J.W.; Semenic, S. Smoking motives, quitting motives, and opinions about smoking cessation support among expectant or new fathers. J. Addict. Nurs. 2013, 24, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.J.; Noach, M.B.; Winickoff, J.P.; Hovell, M.F. Parental smoking cessation to protect young children: A systematic review and meta-analysis. Pediatrics 2012, 129, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Simons-Morton, B.G.; McLeroy, K.R.; Wendel, M.L. Behavior Theory in Health Promotion Practice and Research; Jones & Bartlett Learning: Burlington, MA, USA, 2012. [Google Scholar]
- Reitzel, L.; Kendzor, D.; Castro, Y.; Cao, Y.; Businelle, M.; Mazas, C.; Cofta-Woerpel, L.; Li, Y.; Cinciripini, P.; Ahluwalia, J.; et al. The relation between social cohesion and smoking cessation among black smokers, and the potential role of psychosocial mediators. Ann. Behav. Med. 2013, 45, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Letona, P.; Ramirez-Zea, M.; Caballero, B.; Gittelsohn, J. Formative research to develop a community-based intervention for chronic disease prevention in Guatemalan school-age children. BMC Public Health 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Carson, K.V.; Brinn, M.P.; Peters, M.; Veale, A.; Esterman, A.J.; Smith, B.J. Interventions for smoking cessation in Indigenous populations. Cochrane Database of Syst. Rev. 2012, 1. [Google Scholar] [CrossRef]
- Burgess, D.J.; Fu, S.S.; van Ryn, M. Potential unintended consequences of tobacco-control policies on mothers who smoke: A review of the literature. Am. J. Prev. Med. 2009, 37, S151–S158. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, N.; Luckett, T.; Davidson, P.M.; Di Giacomo, M. Interventions to Reduce Harm from Smoking with Families in Infancy and Early Childhood: A Systematic Review. Int. J. Environ. Res. Public Health 2015, 12, 3091-3119. https://doi.org/10.3390/ijerph120303091
Brown N, Luckett T, Davidson PM, Di Giacomo M. Interventions to Reduce Harm from Smoking with Families in Infancy and Early Childhood: A Systematic Review. International Journal of Environmental Research and Public Health. 2015; 12(3):3091-3119. https://doi.org/10.3390/ijerph120303091
Chicago/Turabian StyleBrown, Nicola, Tim Luckett, Patricia M. Davidson, and Michelle Di Giacomo. 2015. "Interventions to Reduce Harm from Smoking with Families in Infancy and Early Childhood: A Systematic Review" International Journal of Environmental Research and Public Health 12, no. 3: 3091-3119. https://doi.org/10.3390/ijerph120303091
APA StyleBrown, N., Luckett, T., Davidson, P. M., & Di Giacomo, M. (2015). Interventions to Reduce Harm from Smoking with Families in Infancy and Early Childhood: A Systematic Review. International Journal of Environmental Research and Public Health, 12(3), 3091-3119. https://doi.org/10.3390/ijerph120303091





