Delayed Numerical Chromosome Aberrations in Human Fibroblasts by Low Dose of Radiation
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Culture and Irradiation
2.2. The Cytokinesis-Block Micronucleus Assay
2.3. Fluorescent in Situ Hybridization (FISH) Using Pan-Centromeric Probe
2.4. FISH Using Centromeric Probes of Chromosomes 1 and 4
2.5. Microscopic Examination
2.6. Statistical Analysis
3. Results
3.1. Radiation Increased the Frequency of MN, but not Delayed MN Induction
| Time after Irradiation (h) | Dose (cGy) | No. of MNCB a Cells/1000 BN b Cells | Multi MNCB c/MNCB Cells (%) † | Total No. of MN d/1000 BN Cells |
|---|---|---|---|---|
| 28 (Direct) | Control | 28.0 ± 1.4 | 9.8 ± 2.5 | 30.5 ± 2.1 |
| 20 | 35.0 ± 0.0 | 6.1 ± 2.2 | 37.0 ± 2.8 | |
| 50 | 55.5 ± 0.7 | 19.1 ± 3.4 | 65.0 ± 2.1 | |
| 100 | 95.5 ± 3.5 | 18.9 ± 1.4 | 113.0 ± 7.0 * | |
| 88 (1 passage) | Control | 30.0 ± 0.0 | 5.3 ± 1.6 | 31.5 ± 0.7 |
| 20 | 33.0 ± 0.0 | 6.5 ± 1.7 | 35.5 ± 2.1 | |
| 50 | 57.0 ± 2.8 | 9.6 ± 1.4 | 62.0 ± 5.6 | |
| 100 | 99.5 ± 0.7 | 13.8 ± 3.5 | 114.5 ± 3.5 * | |
| 240 (5 passages) | Control | 31.0 ± 2.8 | 5.1 ± 1.9 | 32.5 ± 2.1 |
| 20 | 33.5 ± 2.1 | 9.8 ± 2.8 | 37.0 ± 2.8 | |
| 50 | 58.0 ± 4.2 | 9.4 ± 1.5 | 64.0 ± 8.4 | |
| 100 | 101.5 ± 0.7 | 8.0 ± 4.2 | 111.0 ± 4.2 * |
3.2. Induction of MNC+ and MNC− Did Not Differ with Number of Cell Passages After X-Irradiation
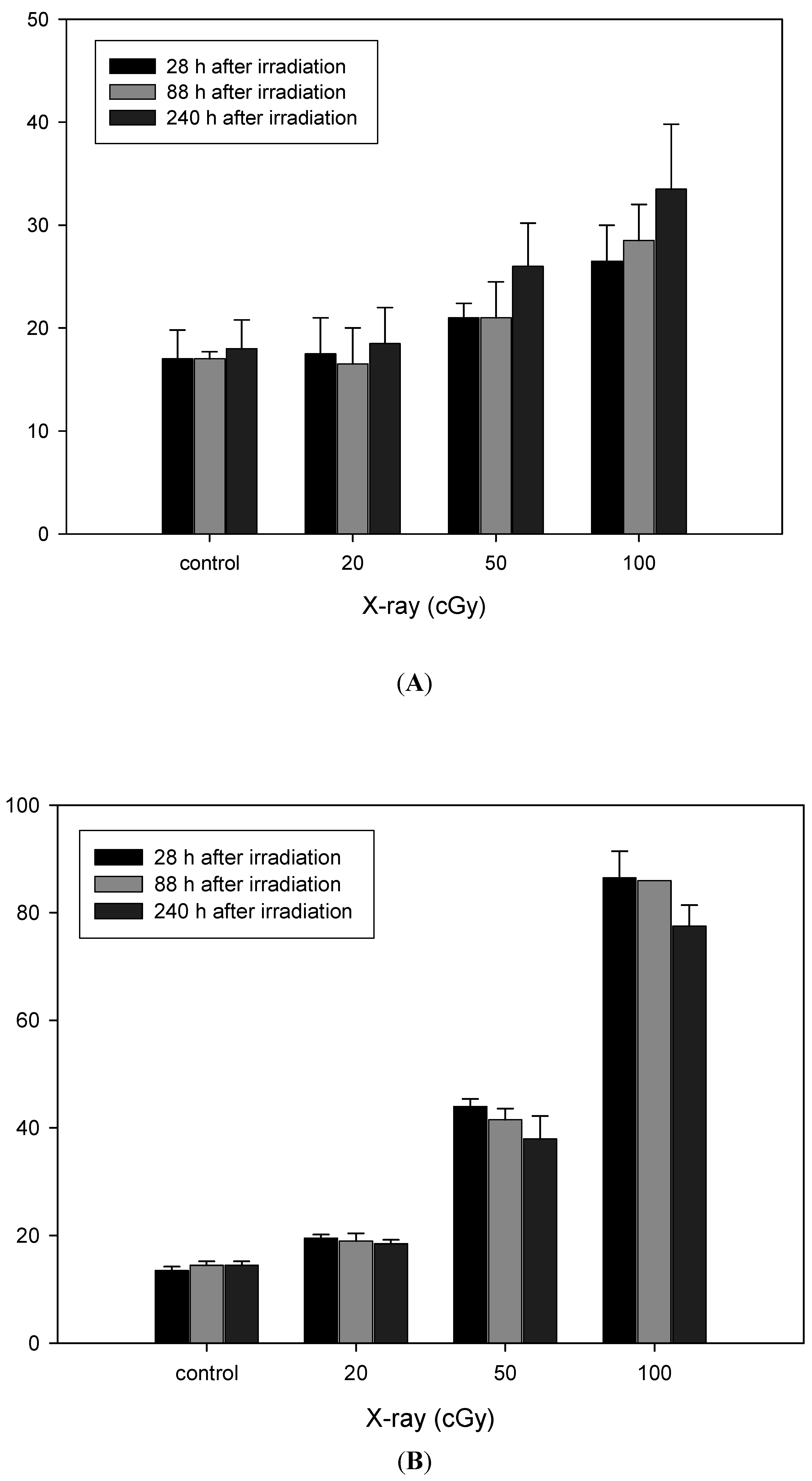
3.3. Radiation Induced Delayed Aneuploidy of Chromosomes 1 and 4 in Fibroblasts
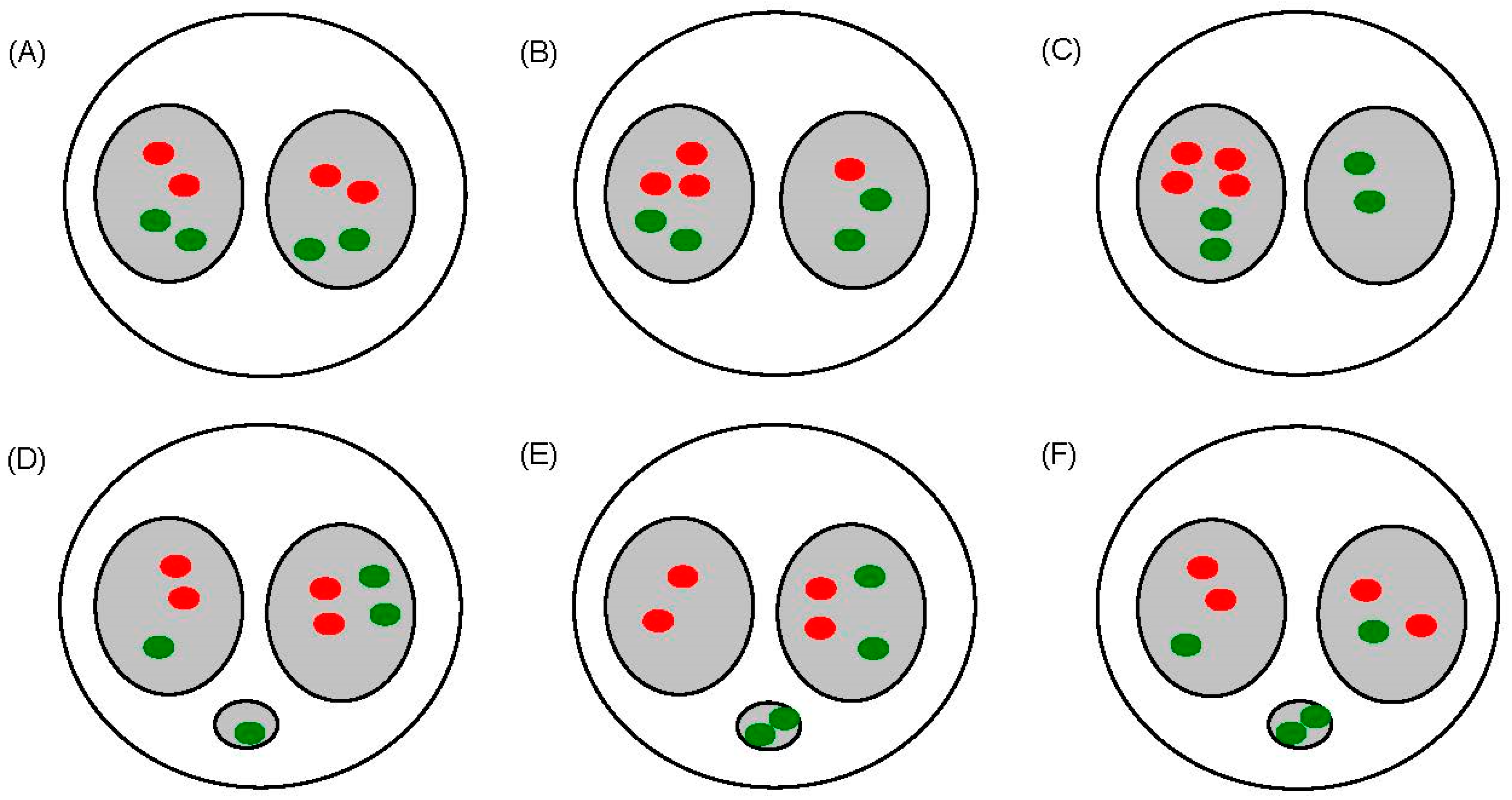
| Time after Irradiation (h) | Dose (cGy) | No. of BN a Cells Scored | Normal Cells b | Aneuploid Cells | Total Aneuploidy | |
|---|---|---|---|---|---|---|
| Non-Disjunction c | Chromosome Loss d | |||||
| 28 (Direct) | Control | 1000 | 997.0 ± 0.0 | 2.5 ± 0.7 | 0.5 ± 0.7 | 3.0 ± 0.0 |
| 20 | 1000 | 996.5 ± 0.7 | 3.0 ± 1.4 | 0.5 ± 0.7 | 3.5 ± 0.7 | |
| 50 | 1000 | 993.5 ± 2.1 | 5.0 ± 1.4 | 1.5 ± 0.7 | 6.5 ± 2.1 | |
| 100 | 1000 | 990.0 ± 4.2 | 7.5 ± 0.7 | 2.5 ± 2.1 | 10.0 ± 2.8 * | |
| 88 (1 passage) | Control | 1000 | 997.0 ± 1.4 | 3.0 ± 1.4 | 0.0 ± 0.0 | 3.0 ± 1.4 |
| 20 | 1000 | 996.0 ± 0.0 | 3.5 ± 0.7 | 0.5 ± 0.7 | 4.0 ± 0.0 | |
| 50 | 1000 | 995.0 ± 1.4 | 4.0 ± 1.4 | 1.5 ± 0.7 | 5.0 ± 1.4 | |
| 100 | 1000 | 991.5 ± 2.1 | 7.0 ± 2.8 | 1.5 ± 0.7 | 8.5 ± 2.1 * | |
| 240 (5 passages) | Control | 1000 | 997.0 ± 0.0 | 2.5 ± 0.7 | 0.5 ± 0.7 | 3.0 ± 0.0 |
| 20 | 1000 | 992.5 ± 0.7 | 6.0 ± 0.0 | 1.5 ± 0.7 | 7.5 ± 0.7 | |
| 50 | 1000 | 987.0 ± 1.4 | 11.0 ± 0.0 | 2.0 ± 1.4 | 13.0 ± 1.4 | |
| 100 | 1000 | 979.5 ± 2.1 | 15.0 ± 0.0 | 5.5 ± 2.1 | 21.0 ± 2.1 *† | |
| Time after Irradiation (h) | Dose (cGy) | No. of BN a Cells Scored | Normal Cells b | Aneuploid Cells | Total Aneuploidy | |
|---|---|---|---|---|---|---|
| Non-Disjunction c | Chromosome Loss d | |||||
| 28 (Direct) | Control | 1000 | 997.5 ± 0.7 | 2.5 ± 0.7 | 0.0 ± 0.0 | 2.5 ± 0.7 |
| 20 | 1000 | 997.0 ± 1.4 | 2.5 ± 0.7 | 0.5 ± 0.7 | 3.0 ± 1.4 | |
| 50 | 1000 | 995.5 ± 2.1 | 3.5 ± 0.7 | 1.0 ± 1.4 | 4.5 ± 2.1 | |
| 100 | 1000 | 993.5 ± 2.1 | 5.0 ± 1.4 | 1.5 ± 0.7 | 6.5 ± 2.1 * | |
| 88 (1 passage) | Control | 1000 | 997.0 ± 1.4 | 2.5 ± 0.7 | 0.5 ± 0.7 | 3.0 ± 1.4 |
| 20 | 1000 | 998.0 ± 1.4 | 2.0 ± 1.4 | 0.0 ± 0.0 | 2.0 ± 1.4 | |
| 50 | 1000 | 996.0 ± 1.4 | 4.0 ± 1.4 | 0.0 ± 0.0 | 4.0 ± 1.4 | |
| 100 | 1000 | 993.0 ± 1.4 | 5.0 ± 1.4 | 2.0 ± 0.0 | 7.0 ± 1.4 * | |
| 240 (5 passages) | Control | 1000 | 997.0 ± 0.0 | 3.0 ± 0.0 | 0.0 ± 00 | 3.0 ± 0.0 |
| 20 | 1000 | 995.5 ± 0.7 | 3.0 ± 1.4 | 1.5 ± 0.7 | 4.5 ± 0.7 | |
| 50 | 1000 | 991.5 ± 0.7 | 6.5 ± 0.7 | 2.0 ± 0.0 | 8.5 ± 0.7 | |
| 100 | 1000 | 988.0 ± 1.4 | 8.5 ± 1.4 | 3.5 ± 0.7 | 12.0 ± 1.4 * | |
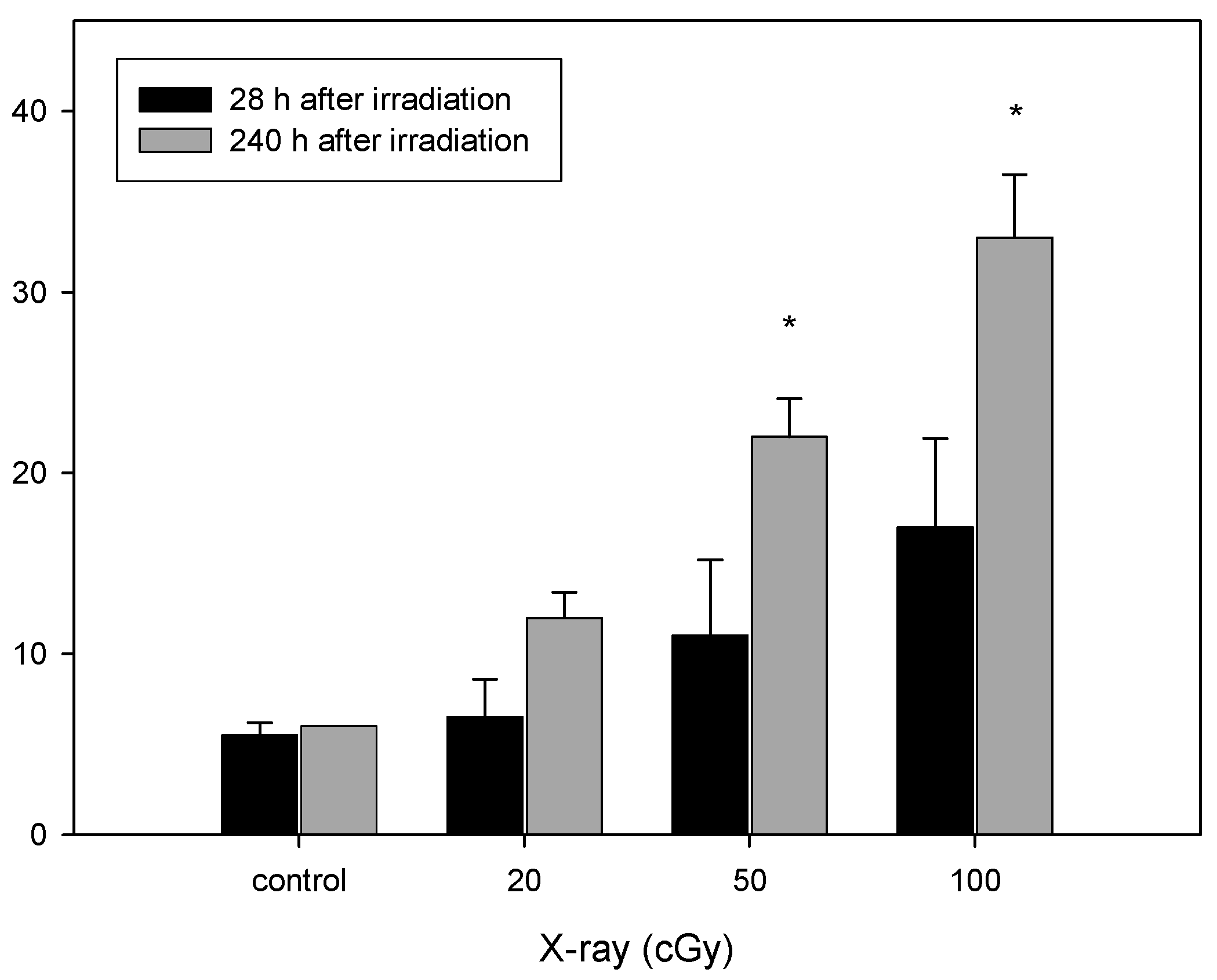
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ward, J.F. DNA damage produced by ionizing radiation in mammalian cells; Identities, mechanism of formation and reparability. Prog. Nucleic Acid Res. Mol. Biol. 1988, 35, 95–125. [Google Scholar] [PubMed]
- Morgan, W.F.; Day, J.P.; Kaplan, M.I.; McGhee, E.M.; Limoli, C.L. Genomic instability induced by ionizing radiation. Radiat. Res. 1996, 146, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Little, J.B. Radiation carcinogenesis. Carcinogenesis 2000, 21, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Lorimore, S.A.; Wright, E.G. Radiation-induced genomic instability and bystander effects: Related inflammatory-type responses to radiation-induced stress and injury? A review. Int. J. Radiat. Biol. 2003, 79, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Tomita, M.; Maeda, M. Mechanisms and biological importance of photon-induced bystander responses: Do they have an impact on low-dose radiation responses. J. Radiat. Res. 2015, 56, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Little, J.B. Genomic instability and bystander effects: A historical perspective. Oncogene 2003, 22, 6978–6987. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Seymour, C.B. Mechanisms and implications of genomic instability and other delayed effects of ionizing radiation exposure. Mutagenesis 1998, 13, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Ojima, M.; Kodama, S.; Watanabe, M. Radiaiton-induced DNA damage and delayed induced genomic instability. Oncogene 2003, 22, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kadhim, M.; Salomaa, S.; Wright, E.; Hildebrandt, G.; Belyakov, O.V.; Prise, K.M.; Little, M.P. Non-targeted effects of ionising radiation—Implications for low dose risk. Mutat. Res. 2013, 752, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Morgan, W.F.; Bair, W.J. Issues in low dose radiation biology: The controversy continues. A perspective. Radiat. Res. 2013, 179, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Hamada, N.; Maeda, M.; Otsuka, K.; Tomita, M. Signaling pathways underpinning the manifestations of ionizing radiation-induced bystander effects. Curr. Mol. Pharmacol. 2011, 4, 79–95. [Google Scholar] [CrossRef] [PubMed]
- Prise, K.M.; Schettino, G.; Vojnovic, B.; Belyakov, O.; Shao, C. Microbeam studies of the bystander response. J. Radiat. Res. 2009, 50, A1–A6. [Google Scholar] [CrossRef] [PubMed]
- Hall, E.J.; Hei, T.K. Genomic instability and bystander effects induced by high-LET radiation. Oncogene 2003, 22, 7034–7042. [Google Scholar] [CrossRef] [PubMed]
- Belyakov, O.V.; Malcolmson, A.M.; Folkard, M.; Prise, K.M.; Michael, B.D. Direct evidence for a bystander effect of ionizing radiation in primary human fibroblasts. Br. J. Cancer 2001, 84, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Kim, P.M.; Nickoloff, J.A.; Morgan, W.F. Targeted and nontargeted effects of low-dose ionizing radiation on delayed genomic instability in human cells. Cancer Res. 2007, 67, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, H.; Little, J.B. Induction of sister chromatid exchanges by extremely low doses of α-particles. Cancer Res. 1992, 52, 6394–6396. [Google Scholar] [PubMed]
- Kaplan, M.I.; Limoli, C.L.; Morgan, W.F. Perpetuating radiation-induced chromosomal instability. Radiat. Oncol. Investig. 1997, 5, 124–128. [Google Scholar] [CrossRef]
- Heim, R.A.; Lench, N.J.; Swift, M. Heterozygrous manifestations in our autosomal recessive human cancer-prone syndromes: Ataxia telangiectasia, xeroderma pigmentosum, Fanconi anemia and Bloom syndrome. Mutat. Res. 1992, 28, 25–36. [Google Scholar] [CrossRef]
- Kadhim, M.A.; Macdonal, D.A.; Goodhead, D.T.; Lorimre, S.A.; Marsden, S.J.; Wright, E.G. Transmission if chromosomal instability after plotonium alpha particle irradiation. Nature 1992, 355, 738–740. [Google Scholar] [CrossRef] [PubMed]
- Sen, S. Aneuploidy and cancer. Curr. Opin. Oncol. 2000, 12, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Malmanche, N.; Maia, A.; Sunkel, C.E. The spindle assembly checkpoint: Preventing chromosome mis-segregation during mitosis and meiosis. FEBS. Lett. 2006, 580, 2888–2895. [Google Scholar] [CrossRef] [PubMed]
- Yuen, K.W.; Montpetit, B.; Hieter, P. The kinetochore and cancer: What is the connection? Curr. Opin. Cell Biol. 2005, 17, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, H.; Nowak, M.A.; Vogelstein, B.; Lengauer, C. The significance of unstable chromosomes in colorectal cancer. Nat. Rev. Cancer 2003, 3, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Lengauer, C.; Kinzler, K.W.; Vogelstein, B. Genetic instabilities in human cancers. Nature 1998, 396, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Marx, J. Debate surges over the origins of genomic defects in cancer. Science 2002, 297, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Duesberg, P.; Rausch, C.; Rasnick, D.; Hehlmann, R. Genetic instability of cancer cells is proportional to their degree of aneuploidy. Proc. Natl. Acad. Sci. USA 1998, 95, 13692–13697. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Sonik, A.; Stindl, R.; Rasnick, D.; Duesberg, P. Aenuploidy vs. gene mutation hypothesis of cancer: Recent study claims mutation but is found to support aneuploidy. Proc. Natl. Acad. Sci. USA. 2000, 97, 3236–3241. [Google Scholar] [CrossRef] [PubMed]
- Olaharski, A.J.; Sotelo, R.; Solorza-Luna, G.; Gonsebatt, M.E.; Guzman, P.; Mohar, A.; Eastmond, D.A. Tetraploidy and chromosomal instability are early events during cervical carcinogenesis. Carcinogenesis 2006, 27, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Lucas, J.N.; Tenjin, T.; Straume, T.; Pinkel, D.; Moore, D., 2nd; Litt, M.; Gray, J.W. Rapid human chromosome aberration analysis using fluorescence in situ hybridization. Int. J. Radiat. Biol. 1989, 56, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.W.; Kang, S.J.; Kim, S.Y. A combination of the micronucleus assay and a FISH technique for evaluation of the genotoxicity of 1,2,4-benzenetriol. Mutat. Res. 2002, 516, 49–56. [Google Scholar] [CrossRef]
- Kryscio, A.; Ulrich Müller, W.U.; Wojcik, A.; Kotschy, N.; Grobelny, S.; Streffer, C. A cytogenetic analysis of the long-term effect of uranium mining on peripheral lymphocytes using the micronucleus-centromere assay. Int. J. Radiat. Biol. 2001, 77, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Barrios, L.; Miró, R.; Caballín, M.R.; Fuster, C.; Guedea, F.; Subías, A.; Egozcue, J. Cytogenetic effects of radiotherapy breakpoint distribution in induced chromosome aberrations. Cancer Genet. Cytogenet. 1989, 41, 61–70. [Google Scholar] [CrossRef]
- Chung, H.W.; Kim, S.Y.; Ha, S.W.; Kim, T.H.; Cho, C.K. Relationship between the DNA content of human chromosome and their contribution to radiation-induced chromosome aberration analyzed by fluorescence in situ hybridization (FISH). J. Korean Assoc. Radiat. Prot. 2001, 26, 101–111. [Google Scholar]
- Fenech, M. The in vitro micronucleus technique. Mutat. Res. 2000, 455, 81–95. [Google Scholar] [CrossRef]
- Sgura, A.; Antoccia, A.; Ramirez, M.J.; Marcos, R.; Tanzarella, C.; Degrassi, F. Micronuclei, centromere-positive micronuclei and chromosome nondisjunction in cytokinesis blocked human lymphocytes following mitomycin C or vincristine treatment. Mutat. Res. 1997, 392, 97–107. [Google Scholar] [CrossRef]
- Cho, Y.H.; Kim, Y.J.; An, Y.S.; Woo, H.D.; Choi, S.Y.; Kang, C.M.; Chung, H.W. Micronucleus-centromere assay and DNA repair gene polymorphism in lymphocytes of industrial radiographers. Mutat. .Res. 2009, 680, 17–24. [Google Scholar]
- Sari-Minodier, I.; Orsière, T.; Bellon, L.; Pompili, J.; Sapin, C.; Botta, A. Cytogenetic monitoring of industrial radiographers using the micronucleus assay. Mutat. Res. 2002, 521, 37–46. [Google Scholar] [CrossRef]
- Countryman, P.I.; Heddle, J.A. The production of micronuclei from chromosome aberrations in irradiated cultures of human lymphocytes. Mutat. Res. 1976, 41, 321–332. [Google Scholar] [CrossRef]
- Vig, B.K.; Swearngin, S.E. Sequence of centromere separation: Kinotochore formation in induced laggards and micronuclei. Mutagenesis 1986, 1, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Kriehuber, R.; Simko, M.; Schiffmann, D.; Trott, K.R. Delayed cytotoxic and genotoxic effects in a human cell line following X-irradiation. Int. J. Radiat. Biol. 1999, 75, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Limoli, C.L.; Kaplan, M.I.; Phillips, J.W.; Adair, G.M.; Morgan, W.F. Differential induction of chromosomal instability by DNA strand-breaking agents. Cancer Res. 1997, 57, 4048–4056. [Google Scholar] [PubMed]
- Chang, W.P.; Little, J.B. Evidence that DNA double-strand breaks initiate the phenotype of delayed reproductive death in Chinese hamster ovary cells. Radiat. Res. 1992, 131, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K. Multistep nature of X-ray-induced Neoplastic transformation in mammalian cells: Genetic alterations and instability. J. Radiat. Res. 1997, 38, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Grim, S.; Smith, L.E.; Kim, P.M.; Nickoloff, J.A.; Goloubeva, O.G.; Morgan, W.F. Ionizing radiation induces delayed hyperrecombination in mammalian cells. Mol. Cell. Biol. 2004, 24, 5060–5068. [Google Scholar] [CrossRef] [PubMed]
- Kadhim, M.A.; Hill, M.A. Non-targeted effects of radiation exposure: Recent advances and implications. Radiat. Prot. Dosimetry 2015, 166, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.; Zampetti-Bosseler, F. Relationships between chromosome damage, cell cycle delay and cell killing induced by bleomycin or X-rays. Mutat. Res. 1985, 151, 83–88. [Google Scholar] [CrossRef]
- Ford, J.H.; Schultz, C.J.; Correll, A.T. Chromosome elimination in micronuclei: A common cause of hypoploidy. Am. J. Hum. Genet. 1988, 43, 733–740. [Google Scholar] [PubMed]
- Dahle, J.; Kvam, E. Induction of delayed mutations and chromosomal instability in fibroblasts after UVA-, UVB- and X-radiation. Cancer Res. 2003, 63, 1464–1469. [Google Scholar] [PubMed]
- Purohit, A.; Tynan, S.H.; Vallee, R.; Doxsey, S.J. Direct interaction of pericentrin with cytoplasmic dynein light intermediate chain contributes to mitotic spindle organization. J. Cell Biol. 1999, 147, 481–492. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, Y.H.; Kim, S.Y.; Woo, H.D.; Kim, Y.J.; Ha, S.W.; Chung, H.W. Delayed Numerical Chromosome Aberrations in Human Fibroblasts by Low Dose of Radiation. Int. J. Environ. Res. Public Health 2015, 12, 15162-15172. https://doi.org/10.3390/ijerph121214979
Cho YH, Kim SY, Woo HD, Kim YJ, Ha SW, Chung HW. Delayed Numerical Chromosome Aberrations in Human Fibroblasts by Low Dose of Radiation. International Journal of Environmental Research and Public Health. 2015; 12(12):15162-15172. https://doi.org/10.3390/ijerph121214979
Chicago/Turabian StyleCho, Yoon Hee, Su Young Kim, Hae Dong Woo, Yang Jee Kim, Sung Whan Ha, and Hai Won Chung. 2015. "Delayed Numerical Chromosome Aberrations in Human Fibroblasts by Low Dose of Radiation" International Journal of Environmental Research and Public Health 12, no. 12: 15162-15172. https://doi.org/10.3390/ijerph121214979







