Multiple Zoonotic Parasites Identified in Dog Feces Collected in Ponte de Lima, Portugal—A Potential Threat to Human Health
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
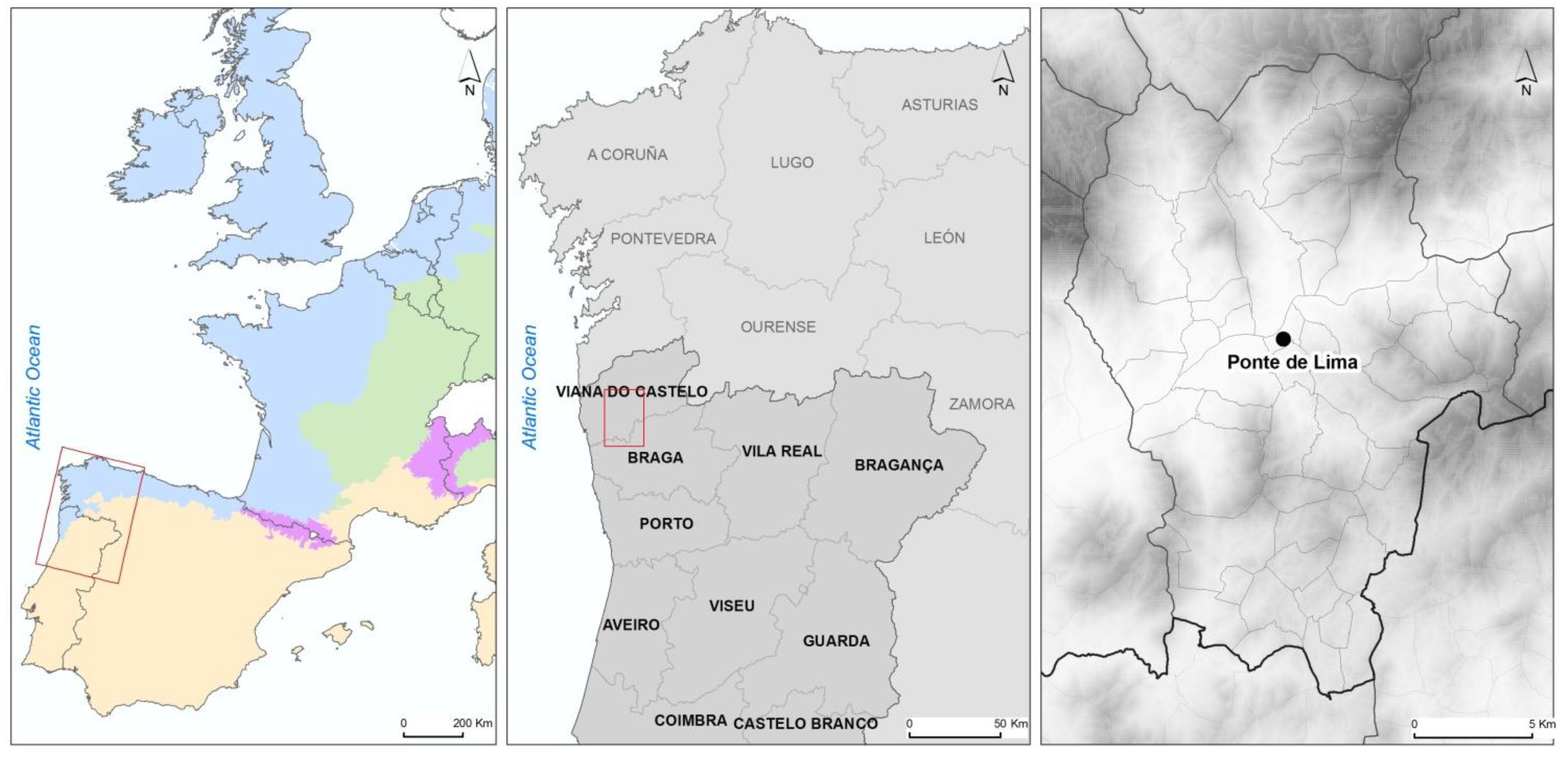
2.2. Fecal Sample Collection
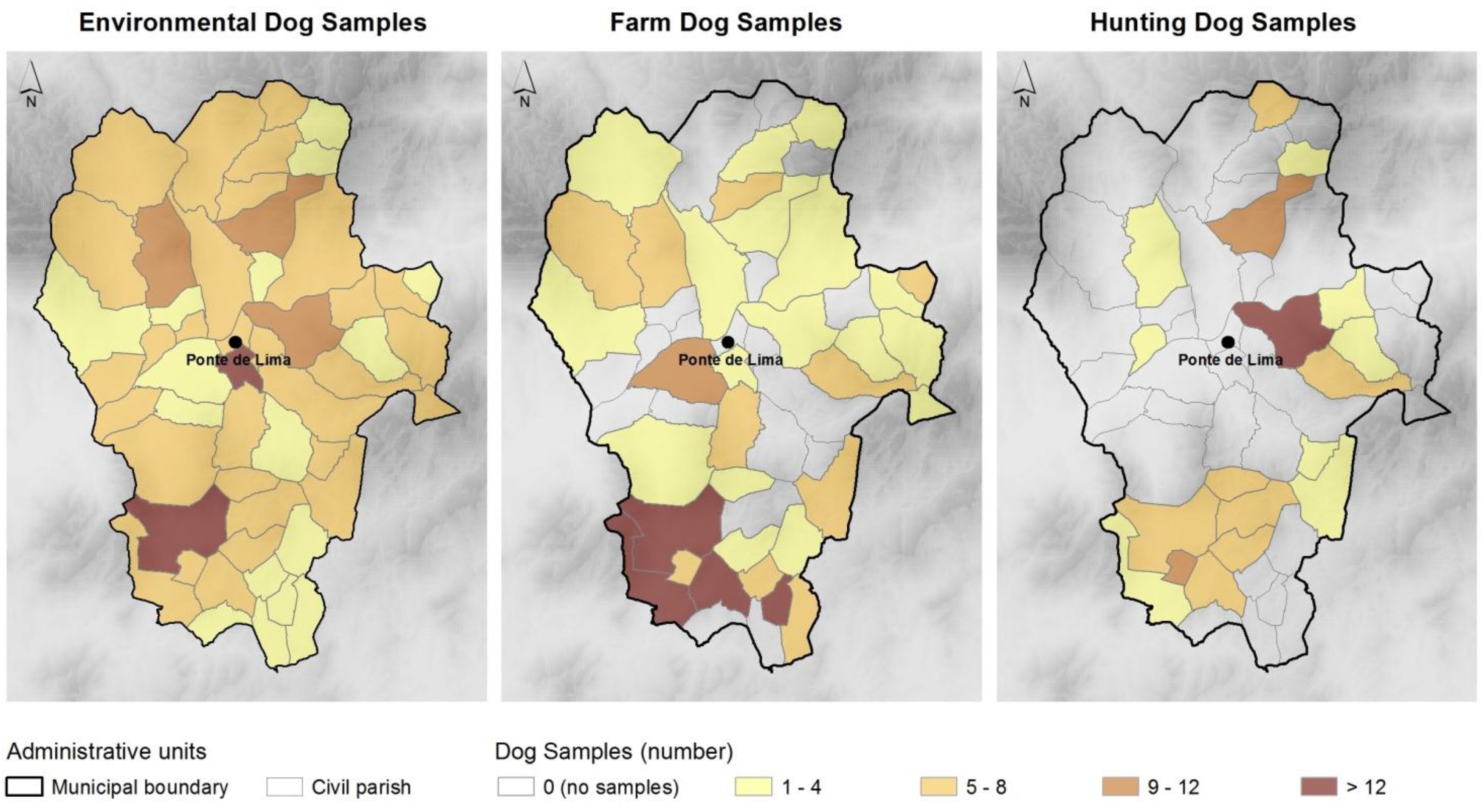
2.3. Coprological Methods Used
2.3.1. Qualitative Flotation Coprological Analysis
2.3.2. Percoll Fractionation of Fecal Samples
2.3.3. Molecular Detection of Echinococcus granulosus
2.4. Data Analysis
3. Results
3.1. Risk of Infection by Strata
| Presence of parasite | Environmental Dog Samples (n = 296) | Farm Dog Samples (n = 195) | Hunting Dog Samples (n = 101) | Total (n = 592) | ||||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | |
| Negative | 119 | 40.20 | 83 | 42.56 | 19 | 18.81 | 218 | 36.82 |
| Positive | 177 | 59.80 | 112 | 57.44 | 82 | 81.19 | 374 | 63.17 |
3.2. Diversity of Parasites Found and Individual Prevalence
| Parasite | Environmental Dog Samples (n = 296) (%) | Farm Dog Samples (n = 195) (%) | Hunting Dog Samples (n = 101) (%) |
|---|---|---|---|
| Ancylostomatidae | 44.59 | 31.28 | 70.30 |
| Trichuris spp. | 34.46 | 32.82 | 49.50 |
| Toxocara spp. | 7.43 | 11.28 | 10.89 |
| Toxascaris leonina | 0.68 | 0 | 0 |
| Dipylidium caninum | 0.68 | 1.02 | 0.99 |
| Taeniidae | 0.34 | 0.51 | 1.98 |
| Isospora spp. | 3.04 | 1.54 | 4.95 |
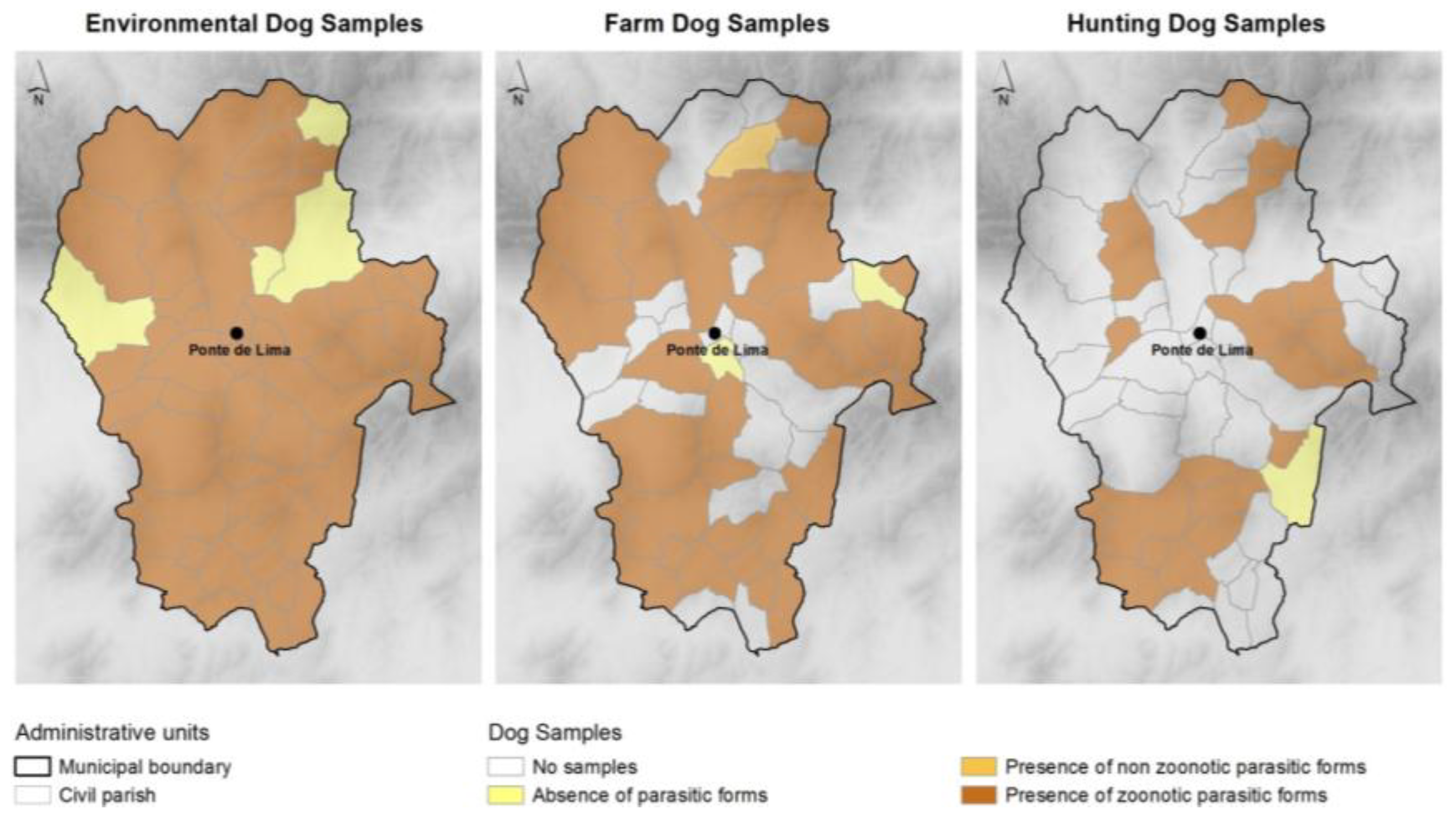
3.3. Parasite Associations and Infections
| Number of Different Parasites | Environmental Dog Samples (n = 296) (%) | Farm Dog Samples (n = 195) (%) | Hunting Dog Samples (n = 101) (%) |
|---|---|---|---|
| 0 | 40.2 a | 42.6 a | 18.8 a |
| 1 | 33.4 a | 39.0 a | 30.7 b |
| >1 | 26.4 a | 18.5 b | 50.5 c |
| Parasite | Environmental Dog Samples (n = 296) (%) | Farm Dog Samples (n = 195) (%) | Hunting Dog Samples (n = 101) (%) |
|---|---|---|---|
| Ancylostomatidae | 19.59 | 14.87 | 20.79 |
| Trichuris spp. | 11.49 | 16.41 | 5.94 |
| Toxocara spp. | 1.35 | 5.64 | 2.97 |
| Dipylidium caninum | 0.38 | 0 | 0.99 |
| Toxascaris leonina | 0.38 | 0 | 0 |
| Isospora spp. | 0.38 | 0 | 0 |
| Taeniidae | 0 | 0.51 | 0 |
| Ancylostomatidae + Trichuris spp. | 17.91 | 10.77 | 34.65 |
| Ancylostomatidae + Toxascaris leonina | 0 | 0 | 0.99 |
| Ancylostomatidae + Toxocara spp. | 2.03 | 2.56 | 2.97 |
| Trichuris spp. + Toxocara spp. | 1.01 | 2.56 | 0.99 |
| Ancylostomatidae + Dipylidium caninum | 0.38 | 0.51 | 0.99 |
| Ancylostomatidae + Isospora spp. | 0.38 | 0 | 0 |
| Ancylostomatidae + Trichuris spp. + Toxocara spp. | 1.69 | 0.51 | 1.98 |
| Ancylostomatidae + Trichuris spp. + Taeniidae | 0 | 0 | 0.99 |
| Ancylostomatidae + Toxocara spp. + Taeniidae | 0 | 0 | 0.99 |
| Ancylostomatidae + Trichuris spp. + Toxascaris leonina | 0 | 0 | 1.98 |
| Ancylostomatidae + Trichuris spp. + Isospora spp. | 1.35 | 1.54 | 1.98 |
| Ancylostomatidae + Isospora spp. + Toxocara spp. | 0.38 | 0 | 0 |
| Trichuris spp. + Toxocara spp. + Taeniidae | 0.38 | 0 | 0 |
| Dipylidiumcaninum + Toxocara spp. + Trichuris spp. | 0 | 0.51 | 0 |
| Ancylostomatidae + Isospora spp. + Toxocara spp. + Trichuris spp. | 0.38 | 0 | 0.99 |
| Ancylostomatidae + Isospora spp. + Toxocara spp. + Toxascaris leonina | 0.38 | 0 | 0.99 |

4. Discussion
4.1. Risk of Infection by Group
| Authors | Country | N | Dog Sample Origin | Overall Prevalence |
|---|---|---|---|---|
| Tarsitano et al. [5] | Italy | 152 | Environmental | 8.5% |
| Rinaldi et al. [19] | Italy | 415 | Environmental | 16.9% |
| Dubná et al. [20] | Czech Republic | 3780 | Environmental + Shelter Dogs | 17.6% |
| Martínez-Carrasco et al. [21] | Spain | 275 | Dogs presented to veterinary clinics + Shelter Dogs + Stray Dogs | 25.0% |
| Papazahariadou et al. [22] | Greece | 281 | Farm Dogs + Hunting Dogs | 26.0% |
| Gracenea et al. [23] | Spain | 505 | Shelter Dogs | 26.9% |
| Soriano et al. [24] | Argentina | 1944 | Environmental | 37.9% |
| Szabová et al. [1] | Slovak Republic | 752 | Environmental + Owned Dogs + Shelter Dogs | 45.7% |
| Balassiano et al. [25] | Brazil | 500 | Dogs presented to veterinary clinics | 46.4% |
| Fontanarrosa et al. [7] | Argentina | 2193 | Owned Dogs | 52.4% |
| Okoye et al. [26] | Nigeria | 413 | Stray Dogs | 52.6% |
| Benito et al. [27] | Spain | 1040 | Shelter Dogs | 53.6% |
| Katagiri and Oliveira-Sequeira [28] | Brazil | 254 | Owned Dogs | 54.3% |
| Beiromvand et al. [29] | Iran | 77 | Owned Dogs + Stray Dogs | 66.0% |
| Bajer et al. [30] | Poland | 108 | Sled Dogs | 68.0% |
| Ugbomoiko et al. [31] | Nigeria | 396 | Owned Dogs | 68.4% |
| Martínez-Moreno et al. [32] | Spain | 1800 | Shelter Dogs | 71.3% |
| Gingrich et al. [33] | Galapagos Island | 97 | Owned Dogs | 71.4% |
| Eguía-Aguilar et al. [34] | México | 120 | Stray Dogs | 85.0% |
| Mandarino-Pereira et al. [35] | Brazil | 81 | Environmental | 92.6% |
| Crespo and Jorge [36] | Portugal | 576 | Environmental | 17.9% |
| Cruz et al. [37] | Portugal | 49 | Environmental | 18.4% |
| Neves et al. [38] | Portugal | 368 | Dogs presented to veterinary clinics | 20.6% |
| Mateus et al. [39] | Portugal | 100 | Shelter Dogs | 41.0% |
| Crespo et al. [40] | Portugal | 548 | Environmental | 50.0% |
| Cardoso et al. [17] | Portugal | 301 | Farm Dogs | 58.8% |
4.2. Diversity of Parasites Found and Individual Prevalence
4.3. Parasite Associations and Infections
4.4. Limitations of the Study
4.5. “One Health” Approach Is Required
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Szabova, E.; Juris, P.; Miterpakova, M.; Antolova, D.; Papajova, I.; Sefcikova, H. Prevalence of important zoonotic parasites in dog populations from the Slovak Republic. Helminthologia 2007, 44, 170–176. [Google Scholar]
- Beck, A.M.; Meyers, N.M. Health enhancement and companion animal ownership. Annu. Rev. Public Health 1996, 17, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Overgaauw, P.A.M.; van Zutphen, L.; Hoek, D.; Yaya, F.O.; Roelfsema, J.; Pinelli, E.; van Knapen, F.; Kortbeek, L.M. Zoonotic parasites in fecal samples and fur from dogs and cats in The Netherlands. Vet. Parasitol. 2009, 163, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Chomel, B.B.; Ben, S. Zoonoses in the bedroom. Emerg. Infect. Dis. 2011, 17, 167–172. [Google Scholar] [CrossRef]
- Tarsitano, E.; Greco, G.; Decaro, N.; Nicassio, F.; Lucente, M.S.; Buonavoglia, C.; Tempesta, M. Environmental monitoring and analysis of faecal contamination in an urban setting in the city of Bari (Apulia Region, Italy): Health and hygiene implications. Int. J. Environ. Res. Public Health 2010, 7, 3972–3986. [Google Scholar] [CrossRef] [PubMed]
- Deplazes, P.; van Knapen, F.; Schweiger, A.; Overgaauw, P.A.M. Role of pet dogs and cats in the transmission of helminthic zoonoses in Europe, with a focus on echinococcosis and toxocarosis. Vet. Parasitol. 2011, 182, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Fontanarrosa, M.F.; Vezzani, D.; Basabe, J.; Eiras, D.F. An epidemiological study of gastrointestinal parasites of dogs from Southern Greater Buenos Aires (Argentina): Age, gender, breed, mixed infections, and seasonal and spatial patterns. Vet. Parasitol. 2006, 136, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Sowemimo, O.A. The prevalence and intensity of gastrointestinal parasites of dogs in ile-ife, Nigeria. J. Helminthol. 2009, 83, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Seimenis, A. Overview of the epidemiological situation on echinococcosis in the Mediterranean region. Acta Trop. 2003, 85, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Blaszkowska, J.; Kurnatowski, P.; Damiecka, P. Contamination of the soil by eggs of geohelminths in rural areas of Lodz District (Poland). Helminthologia 2011, 48, 67–76. [Google Scholar] [CrossRef]
- Habluetzel, A.; Traldi, G.; Ruggieri, S.; Attili, A.R.; Scuppa, P.; Marchetti, R.; Menghini, G.; Esposito, F. An estimation of toxocara canis prevalence in dogs, environmental egg contamination and risk of human infection in the Marche region of Italy. Vet. Parasitol. 2003, 113, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Himsworth, C.G.; Skinner, S.; Chaban, B.; Jenkins, E.; Wagner, B.A.; Harms, N.J.; Leighton, F.A.; Thompson, R.C.A.; Hill, J.E. Short report: Multiple zoonotic pathogens identified in canine feces collected from a remote canadian indigenous community. Am. J. Trop. Med. Hyg. 2010, 83, 338–341. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Estatísticas Demográficas; National Statistics Institute—Portugal Statistics: Lisboa, Portugal, 2011. [Google Scholar]
- Foreyt, W.J. Veterinary Parasitology; Blackwell Publishing: Iowa, USA, 2001; p. 246. [Google Scholar]
- Zajac, A.M.; Conboy, G.A. Veterinary Clinical Parasitology, 8th ed.; Wiley Blackwell: West Sussex, UK, 2012; p. 368. [Google Scholar]
- Thienpont, D.; Rochette, F.; Vanparijs, O.F.J. Diagnosing Helminthiasis by Coprological Examination. Coprological Examination, 2nd ed.; Janssen Research Foundation: Beerse, Belgium, 1986; p. 205. [Google Scholar]
- Cardoso, A.S.; Costa, I.M.H.; Figueiredo, C.; Castro, A.; Conceicao, M.A.P. The occurrence of zoonotic parasites in rural dog populations from Northern Portugal. J. Helminthol. 2014, 88, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, I.; Branzburg, A.; Campos-Ponce, M.; Abdel Hafez, S.K.; Raoul, F.; Craig, P.S.; Hamburger, J. Copro-diagnosis of echinococcus granulosus infection in dogs by amplification of a newly identified repeated DNA sequence. Am. J. Trop. Med. Hyg. 2003, 69, 324–330. [Google Scholar] [PubMed]
- Rinaldi, L.; Biggeri, A.; Carbone, S.; Musella, V.; Catelan, D.; Veneziano, V.; Cringoli, G. Canine faecal contamination and parasitic risk in the city of Naples (southern Italy). BMC Vet. Res. 2006, 2, 29–29. [Google Scholar] [CrossRef] [PubMed]
- Dubna, S.; Langrova, I.; Napravnik, J.; Jankovska, I.; Vadlejch, J.; Pekar, S.; Fechtner, J. The prevalence of intestinal parasites in dogs from Prague, rural areas, and shelters of the Czech Republic. Vet. Parasitol. 2007, 145, 120–128. [Google Scholar] [CrossRef]
- Martinez-Carrasco, C.; Berriatua, E.; Garijo, M.; Martinez, J.; Alonso, F.D.; de Ybanez, R.R. Epidemiological study of non-systemic parasitism in dogs in Southeast Mediterranean Spain assessed by coprological and post-mortem examination. Zoonoses Public Health 2007, 54, 195–203. [Google Scholar]
- Papazahariadou, A.; Founta, A.; Papadopoulos, E.; Chliounakis, S.; Antoniadou-Sotiriadou, K.; Theodorides, Y. Gastrointestinal parasites of shepherd and hunting dogs in the serres prefecture, northern Greece. Vet. Parasitol. 2007, 148, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Gracenea, M.; Soledad Gomez, M.; Torres, J. Prevalence of intestinal parasites in shelter dogs and cats in the metropolitan area of Barcelona (Spain). Acta Parasitol. 2009, 54, 73–77. [Google Scholar] [CrossRef]
- Soriano, S.V.; Pierangeli, N.B.; Pianciola, L.; Mazzeo, M.; Lazzarini, L.E.; Saiz, M.S.; Kossman, A.V.; Bergagna, H.F.J.; Chartier, K.; Basualdo, J.A. Molecular characterization of echinococcus isolates indicates goats as reservoir for Echinococcus Canadensis G6 genotype in Neuquen, Patagonia Argentina. Parasitol. Int. 2010, 59, 626–628. [Google Scholar] [CrossRef]
- Cramer Balassiano, B.C.; Campos, M.R.; Alves Alcantara de Menezes, R.D.C.; Salim Pereira, M.J. Factors associated with gastrointestinal parasite infection in dogs in Rio De Janeiro, Brazil. Prev. Vet. Med. 2009, 91, 234–240. [Google Scholar]
- Okoye, I.C.; Obiezue, N.R.; Okorie, C.E.; Ofoezie, I.E. Epidemiology of intestinal helminth parasites in stray dogs from markets in south-eastern Nigeria. J. Helminthol. 2011, 85, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Benito, A.; Carmena, D.; Postigo, I.; Estibalez, J.J.; Martinez, J.I.; Guisantes, J.A. Intestinal helminths in dogs in alava, north of Spain. Rev. Ibér. Parasitol. 2003, 63, 121–126. [Google Scholar]
- Katagiri, S.; Oliveira-Sequeira, T.C.G. Prevalence of dog intestinal parasites and risk perception of zoonotic infection by dog owners in sao paulo state, Brazil. Zoonoses Public Health 2008, 55, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Beiromvand, M.; Akhlaghi, L.; Massom, S.H.F.; Meamar, A.R.; Motevalian, A.; Oormazdi, H.; Razmjou, E. Prevalence of zoonotic intestinal parasites in domestic and stray dogs in a rural area of Iran. Prev. Vet. Med. 2013, 109, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Bajer, A.; Bednarska, M.; Rodo, A. Risk factors and control of intestinal parasite infections in sled dogs in Poland. Vet. Parasitol. 2011, 175, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Ugbomoiko, U.S.; Ariza, L.; Heukelbach, J. Parasites of importance for human health in Nigerian dogs: High prevalence and limited knowledge of pet owners. BMC Vet. Res. 2008, 4. [Google Scholar] [CrossRef]
- Martinez-Moreno, F.J.; Hernandez, S.; Lopez-Cobos, E.; Becerra, C.; Acosta, I.; Martinez-Moreno, A. Estimation of canine intestinal parasites in Cordoba (Spain) and their risk to public health. Vet. Parasitol. 2007, 143, 7–13. [Google Scholar]
- Gingrich, E.N.; Scorza, A.V.; Clifford, E.L.; Olea-Popelka, F.J.; Lappin, M.R. Intestinal parasites of dogs on the Galapagos Islands. Vet. Parasitol. 2010, 169, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Eguia-Aguilar, P.; Cruz-Reyes, A.; Maninez-Maya, J.J. Ecological analysis and description of the intestinal helminths present in dogs in Mexico City. Vet. Parasitol. 2005, 127, 139–146. [Google Scholar]
- Mandarino-Pereira, A.; de Souza, F.S.; Lopes, C.W.G.; Pereira, M.J.S. Prevalence of parasites in soil and dog feces according to diagnostic tests. Vet. Parasitol. 2010, 170, 176–181. [Google Scholar]
- Crespo, M.V.M.; Jorge, A.T. Contaminação parasitária por ovos de helmintes de alguns jardins e parques públicos das cidades de almeirim e do cartaxo. Acta Parasitol. Port. 2000, 7, 43–47. [Google Scholar]
- Cruz, A.; Santos, A.; Mateus, T.; Ramalho, F.; Sousa, S.; Madeira de Carvalho, L.M. Avaliação do grau de contaminação ambiental com formas parasitárias zoonóticas com origem em dejectos caninos em áreas públicas da cidade de coimbra. Acta Parasitol. Port. 2012, 19, 35–38. [Google Scholar]
- Neves, D.; Lobo, L.; Simoes, P.B.; Cardoso, L. Frequency of intestinal parasites in pet dogs from an urban area (Greater Oporto, Northern Portugal). Vet. Parasitol. 2014, 200, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Mateus, T.L.; Francisco, F.; Lima, N.R. Environmental contamination by dog faeces with parasitic forms in Ponte De Lima, Portugal. In International Congress on Environmental Health; Almeida, J., Ferreira, A., Paixão, S., Pinto, M.V., Sá, N.L., Santos, C., Simões, H., Eds.; Department of Environmental Health—College of Health Technology of Coimbra: Coimbra, Portugal, 2011; pp. 331–334. [Google Scholar]
- Crespo, M.V.; Rosa, F.; Almeida, J.P. Eliminação parasitária em fezes de canídeos no concelho de óbidos—Estudo geral. Acta Parasitol. Port. 2010, 17, 111. [Google Scholar]
- Ferreira, F.S.; Pereira-Baltasar, P.; Parreira, R.; Padre, L.; Vilhena, M.; Tavora Tavira, L.; Atouguia, J.; Centeno-Lima, S. Intestinal parasites in dogs and cats from the district of Evora, Portugal. Vet. Parasitol. 2011, 179, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, B.; Iorio, R.; Capelli, G.; Sparagano, O.A.E.; Giangaspero, A. Epidemiological scenario of giardiosis in dogs from central Italy. Anim. Biodivers. Emerg. Dis. 2008, 1149, 371–374. [Google Scholar]
- Oliveira-Sequeira, T.C.G.; Amarante, A.F.T.; Ferrari, T.B.; Nunes, L.C. Prevalence of intestinal parasites in dogs from São Paulo State, Brazil. Vet. Parasitol. 2002, 103, 19–27. [Google Scholar]
- Wolfe, A.; Hogan, S.; Maguire, D.; Fitzpatrick, C.; Vaughan, L.; Wall, D.; Hayden, T.J.; Mulcahy, G. Red foxes (Vulpes vulpes) in ireland as hosts for parasites of potential zoonotic and veterinary significance. Vet. Rec. 2001, 149, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Bowman, D.D.; Lynn, R.C.; Eberhard, M.L. Georgis Parasitologia Para Veterinários, 8th ed.; Elsevier: Madrid, España, 2004; p. 440. [Google Scholar]
- Chomel, B.B. Control and prevention of emerging parasitic zoonoses. Int. J. Parasitol. 2008, 38, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, E.J.; Schurer, J.M.; Gesy, K.M. Old problems on a new playing field: Helminth zoonoses transmitted among dogs, wildlife, and people in a changing northern climate. Vet. Parasitol. 2011, 182, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Mateus, T.L.; Barrocas, C. Wild Carnivores as a Source of Zoonotic Helminths in the Northern Portugal. In Proceedings of the Joint 61st Wildlife Disease Association/10th Biennial European Wildlife Disease Association Conference, Lyon, France, 22–27 July 2012; p. 135.
- Magalhães, R.; Mateus, T. Desparasitação interna de cães—O que sabem os proprietários de ponte de lima? Acta Parasitol. Port. 2012, 19, 253. [Google Scholar]
- Ramirez-Barrios, R.A.; Barboza-Mena, G.; Munoz, J.; Angulo-Cubillan, F.; Hernandez, E.; Gonzalez, F.; Escalona, F. Prevalence of intestinal parasites in dogs under veterinary care in Maracaibo, Venezuela. Vet. Parasitol. 2004, 121, 11–20. [Google Scholar]
- Xhaxhiu, D.; Kusi, I.; Rapti, D.; Kondi, E.; Postoli, R.; Rinaldi, L.; Dimitrova, Z.M.; Visser, M.; Knaus, M.; Rehbein, S. Principal intestinal parasites of dogs in Tirana, Albania. Parasitol. Res. 2011, 108, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Bwalya, E.C.; Nalubamba, K.S.; Hankanga, C.; Namangala, B. Prevalence of canine gastrointestinal helminths in urban Lusaka and rural Katete Districts of Zambia. Prev. Vet. Med. 2011, 100, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Riggio, F.; Mannella, R.; Ariti, G.; Perrucci, S. Intestinal and lung parasites in owned dogs and cats from central Italy. Vet. Parasitol. 2013, 193, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, D.J.; Tzannes, S.; Graham, P.A.; Wastling, J.M.; Pinchbeck, G.L.; German, A.J. Detection of endoparasites with zoonotic potential in dogs with gastrointestinal disease in the UK. Transbound. Emerg. Dis. 2008, 55, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Rubel, D.; Zunino, G.; Santillan, G.; Wisnivesky, C. Epidemiology of toxocara canis in the dog population from two areas of different socioeconomic status, Greater Buenos Aires, Argentina. Vet. Parasitol. 2003, 115, 275–286. [Google Scholar] [CrossRef]
- Bowman, D.D.; Montgomery, S.P.; Zajac, A.M.; Eberhard, M.L.; Kazacos, K.R. Hookworms of dogs and cats as agents of cutaneous larva migrans. Trends Parasitol. 2010, 26, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Traversa, D. Are we paying too much attention to cardiopulmonary nematodes and neglecting old-fashioned worms like trichuris vulpis? Parasites Vectors 2011, 4. [Google Scholar] [CrossRef]
- Dunn, J.J.; Columbus, S.T.; Aldeen, W.E.; Davis, M.; Carroll, K.C. Trichuris vulpis recovered from a patient with chronic diarrhea and five dogs. J. Clin. Microbiol. 2002, 40, 2703–2704. [Google Scholar] [CrossRef] [PubMed]
- Keegan, J.D.; Holland, C.V. A comparison of toxocara canis embryonation under controlled conditions in soil and hair. J. Helminthol. 2013, 87, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Gawor, J.; Borecka, A.; Zarnowska, H.; Marczynska, M.; Dobosz, S. Environmental and personal risk factors for toxocariasis in children with diagnosed disease in urban and rural areas of central Poland. Vet. Parasitol. 2008, 155, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Auer, H. Parasitoses of the human central nervous system. J. Helminthol. 2013, 87, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Ulukanligil, M.; Seyrek, A.; Aslan, G.; Ozbilge, H.; Atay, S. Environmental pollution with soil-transmitted helminths in Sanliurfa, Turkey. Mem. Inst. Oswaldo Cruz 2001, 96, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Deutz, A.; Fuchs, K.; Auer, H.; Kerbl, U.; Aspock, H.; Kofer, J. Toxocara-infestations in Austria: A study on the risk of infection of farmers, slaughterhouse staff, hunters and veterinarians. Parasitol. Res. 2005, 97, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Molina, C.P.; Ogburn, J.; Adegboyega, P. Infection by dipylidium caninum in an infant. Arch. Pathol. Lab. Med. 2003, 127, e157–e159. [Google Scholar] [PubMed]
- Robertson, I.D.; Thompson, R.C. Enteric parasitic zoonoses of domesticated dogs and cats. Microbes Infect. 2002, 4, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Dyachenko, V.; Pantchev, N.; Gawlowska, S.; Vrhovec, M.G.; Bauer, C. Echinococcus multilocularis infections in domestic dogs and cats from Germany and other European countries. Vet. Parasitol. 2008, 157, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Ziadinov, I.; Schweiger, A.; Schnyder, M.; Deplazes, P. Hair coat contamination with zoonotic helminth eggs of farm and pet dogs and foxes. Berl. Munch. Tierarztl. Wochenschr. 2011, 124, 503–511. [Google Scholar] [PubMed]
- Dakkak, A. Echinococcosis/hydatidosis: A severe threat in mediterranean countries. Vet. Parasitol. 2010, 174, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.-A.M.; Gray, D.J.; Clements, A.C.A.; Barnes, T.S.; McManus, D.P.; Yang, Y.R. Environmental changes impacting echinococcus transmission: Research to support predictive surveillance and control. Glob. Chang. Biol. 2013, 19, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Schantz, P.M. Intestinal parasites of dogs in Western Australia: Progress in control and new concerns. Vet. J. 1999, 157, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Samantaray, J.C.; Singh, N.; Das, G.B.; Verma, I.C. Trichuris-vulpis infection in an Indian tribal population. J. Parasitol. 1993, 79, 457–458. [Google Scholar] [CrossRef] [PubMed]
- Stull, J.W.; Peregrine, A.S.; Sargeant, J.M.; Weese, J.S. Pet husbandry and infection control practices related to zoonotic disease risks in Ontario, Canada. BMC Public Health 2013, 13, 520. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Wilkins, P.P. Toxocariasis: America’s most common neglected infection of poverty and a helminthiasis of global importance? PLoS Negl. Trop. Dis. 2009, 3. [Google Scholar] [CrossRef]
- Kersting, A.L.; Medeiros, L.C.; LeJeune, J.T. Zoonoses and the physicians’ role in educating farming patients. J. Agromed. 2009, 14, 306–311. [Google Scholar] [CrossRef]
- Grant, S.; Olsen, C.W. Preventing zoonotic diseases in immunocompromised persons: The role of physicians and veterinarians. Emerg. Infect. Dis. 1999, 5, 159–163. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, J.; Moore, T.A. Emerging helminth zoonoses. Int. J. Parasitol. 2000, 30, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mateus, T.L.; Castro, A.; Ribeiro, J.N.; Vieira-Pinto, M. Multiple Zoonotic Parasites Identified in Dog Feces Collected in Ponte de Lima, Portugal—A Potential Threat to Human Health. Int. J. Environ. Res. Public Health 2014, 11, 9050-9067. https://doi.org/10.3390/ijerph110909050
Mateus TL, Castro A, Ribeiro JN, Vieira-Pinto M. Multiple Zoonotic Parasites Identified in Dog Feces Collected in Ponte de Lima, Portugal—A Potential Threat to Human Health. International Journal of Environmental Research and Public Health. 2014; 11(9):9050-9067. https://doi.org/10.3390/ijerph110909050
Chicago/Turabian StyleMateus, Teresa Letra, António Castro, João Niza Ribeiro, and Madalena Vieira-Pinto. 2014. "Multiple Zoonotic Parasites Identified in Dog Feces Collected in Ponte de Lima, Portugal—A Potential Threat to Human Health" International Journal of Environmental Research and Public Health 11, no. 9: 9050-9067. https://doi.org/10.3390/ijerph110909050
APA StyleMateus, T. L., Castro, A., Ribeiro, J. N., & Vieira-Pinto, M. (2014). Multiple Zoonotic Parasites Identified in Dog Feces Collected in Ponte de Lima, Portugal—A Potential Threat to Human Health. International Journal of Environmental Research and Public Health, 11(9), 9050-9067. https://doi.org/10.3390/ijerph110909050






