Socioeconomic Factors and Vulnerability to Outbreaks of Leptospirosis in Nicaragua
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Design and Data
| Variables | Sources | Scale | Range |
|---|---|---|---|
| Cases of Leptospirosis 2004–2010 | Ministry of Health. Nicaragua [26] | Number of cases | 0–176 |
| Population of Municipality 2005 | Nicaragua Census & annual projections [20] | Number of people | 4719–937,489 |
| UBN Quality of the Household | CELADE, CEPAL [25] | Percentage | 20.07–83.38 |
| UBN Access to Sanitary Services | CELADE, CEPAL [25] | Percentage | 5.27–55.04 |
| UBN Crowding | CELADE, CEPAL [25] | Percentage | 8.70–52.60 |
| UBN Access to Education | CELADE, CEPAL [25] | Percentage | 3.92–16.45 |
| People in Municipality Living in Extreme Poverty | Nicaragua Census [20] | Percentage | 15.7–53.5 |
| Population of 6-yr-olds and Over, Condition of Illiteracy in Municipality | Nicaragua Census [20] | Percentage | 7.91–26.25 |
| Household with Piped Water | CELADE, CEPAL [25] | Percentage | 0.66–80.01 |
| Household with Solid Waste Disposal | CELADE, CEPAL [25] | Percentage | 0.31–83.69 |
- Housing conditions were measured using two indicators: (1) UBN for quality of the household verifies the construction materials used in the floor, walls and roof; (2) UBN for crowding measures the size of the house in relation to the number of people living there.
- Access to sanitary services determines the availability of potable water and the system for elimination of human excreta.
- Access to education measures enrollment of school-age children in school.
2.2. Analysis
- Group I (low rates): 0.0 ≤ Zt ≤ 0.50
- Group II (medium rates): 0.51 ≤ Zt ≤ 1.00
- Group III (high rates): Zt > 1.00
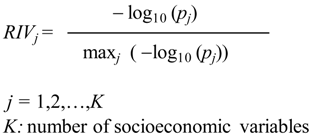
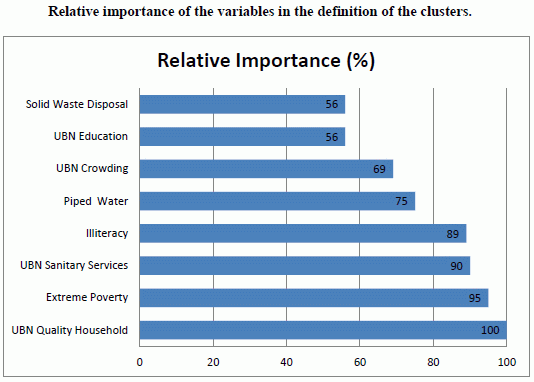
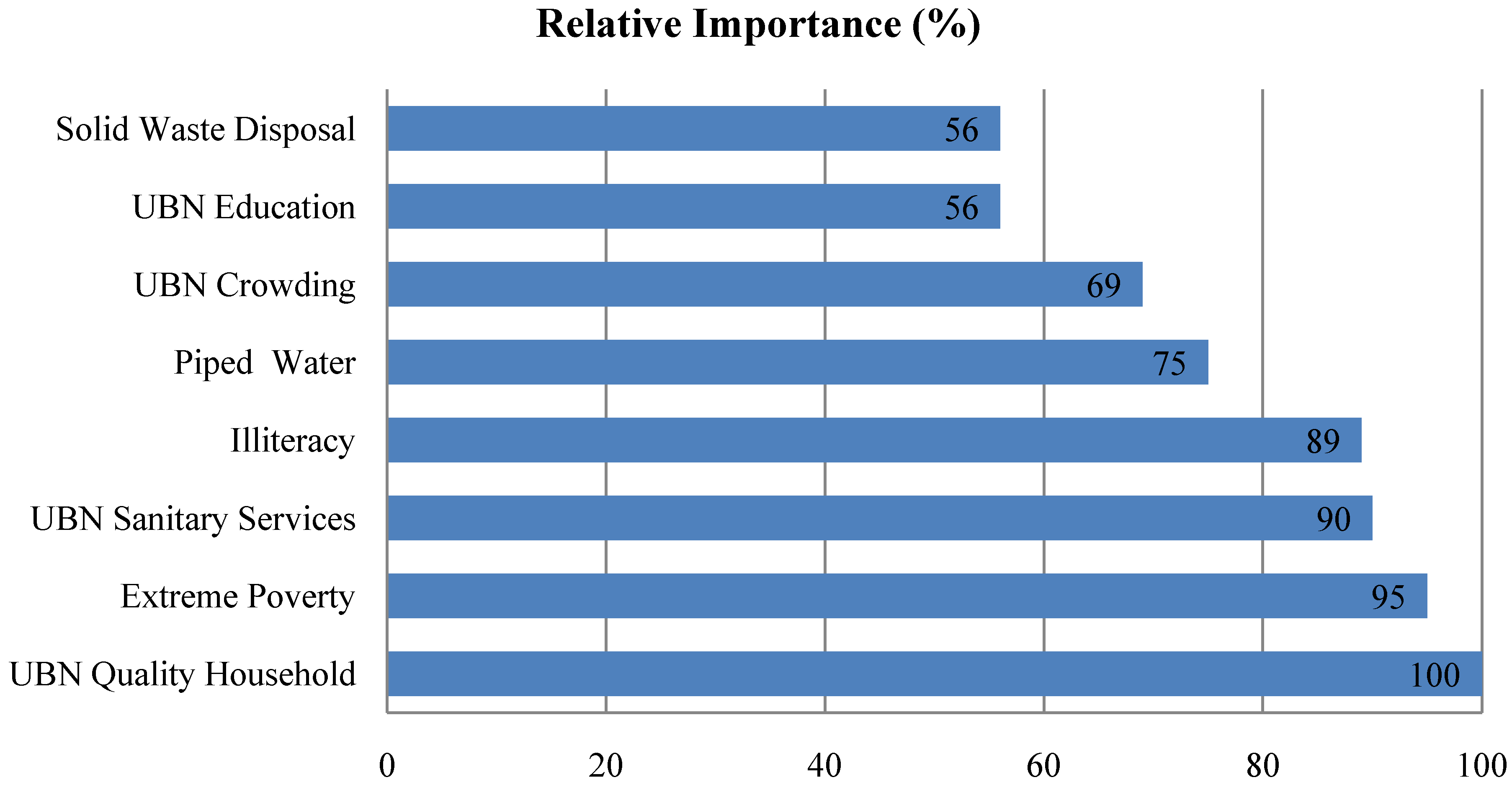
3. Results and Discussion
| Socioeconomic Variable | Pearson Correlation | p Value |
|---|---|---|
| UBN Quality Household | 0.525 | 0.002 |
| UBN Sanitary Services | 0.686 | 0.000 |
| UBN Crowding | 0.609 | 0.000 |
| UBN Education | 0.606 | 0.000 |
| Extreme Poverty | 0.610 | 0.000 |
| Illiteracy | 0.652 | 0.000 |
| Piped Water (*) | −0.630 | 0.000 |
| Solid Waste Disposal (*) | −0.586 | 0.000 |
| Number of Observations | Group Rates | UBN Quality Household | UBN Sanitary Services | UBN Crowding | UBN Education | Extreme Poverty | Illiteracy | Piped Water | Solid Waste Disposal | |
|---|---|---|---|---|---|---|---|---|---|---|
| N = 12 | Low Rates (0–0.48) | Mean | 36.8 | 15.9 | 22.2 | 6.7 | 24.6 | 10.2 | 50.3 | 34.3 |
| SD | 11.4 | 8.3 | 5.0 | 1.6 | 5.1 | 1.3 | 18.6 | 22.7 | ||
| N = 10 | Medium Rates (0.55–0.97) | Mean | 58.7 | 33.3 | 30.1 | 9.9 | 42.1 | 17.6 | 22.1 | 7.4 |
| SD | 12.2 | 11.5 | 13.8 | .8 | 5.5 | 1.4 | 15.4 | 6.5 | ||
| N = 10 | High Rates (1.11–2.51) | Mean | 64.8 | 37.2 | 37.7 | 11.4 | 42.9 | 20.3 | 15.4 | 3.0 |
| SD | 11.1 | 9.9 | 8.5 | 2.9 | 9.3 | 1.6 | 8.3 | 1.4 | ||
| Variables | Clusters of Municipalities | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| UBN Quality Household | 70.5% | 52.1% | 34.6% |
| UBN Sanitary Services | 42.6% | 27.1% | 14.1% |
| UBN Crowding | 42.1% | 24.3% | 21.6% |
| UBN Education | 11.7% | 9.3% | 6.6% |
| Extreme Poverty | 47.4% | 35.9% | 24.1% |
| Illiteracy | 20.0% | 17.0% | 11.0% |
| Piped Water | 10.6% | 27.4% | 53.5% |
| Solid Waste Disposal | 2.5% | 8.0% | 36.9% |
| Rates of Leptospirosis per 10 thousand | 3.1 | 1.4 | 0.2 |
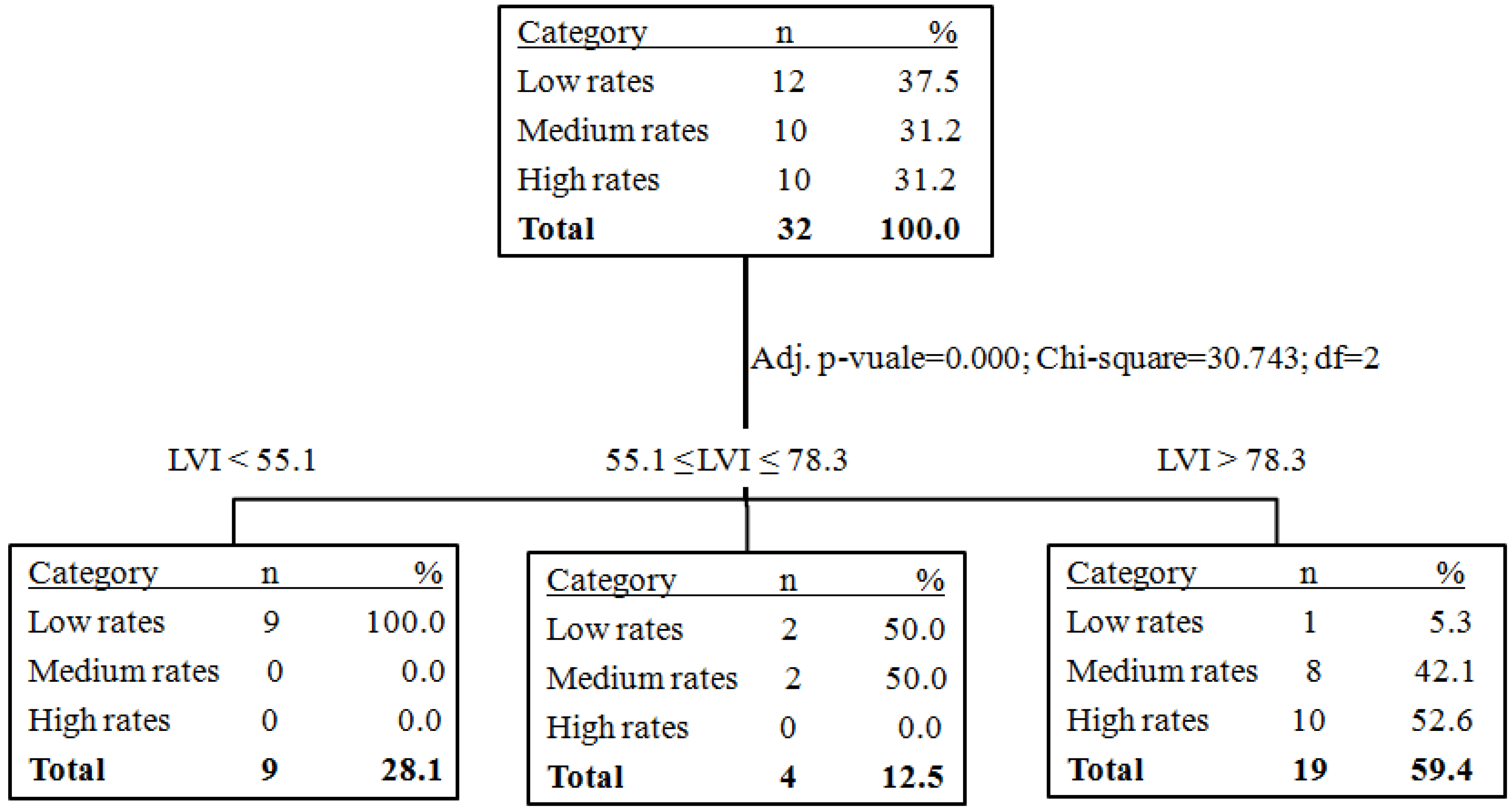
| Groups | Mean | N | Standard Deviation |
|---|---|---|---|
| Low rates | 36.7 | 12 | 46.5 |
| Medium rates | 134.4 | 10 | 48.9 |
| High rates | 158.4 | 10 | 35.6 |
| Total | 105.3 | 32 | 69.5 |




4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Report of the First Meeting of the Leptospirosis Burden Epidemiology Reference Group; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- World Health Organization. Human Leptospirosis: Guidance for Diagnosis, Surveillance and Control; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Lau, C.L.; Smythe, L.D.; Craig, S.B.; Weinstein, P. Climate change, flooding, urbanisation and Leptospirosis: Fuelling the fire? Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 631–638. [Google Scholar] [CrossRef]
- Schneider, M.C.; Nájera, P.; Aldighieri, S.; Bacallao, J.; Soto, A.; Marquiño, W.; Altamirano, L.; Saenz, C.; Marin, J.; Jimenez, E. Leptospirosis outbreaks in nicaragua: identifying critical areas and exploring drivers for evidence-based planning. Int. J. Environ. Res. Public Health 2012, 9, 3883–3910. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.J. Human Frontiers, Environments and Disease: Past Patterns, Uncertain Futures; Cambridge University Press: Cambridge, UK, 2001. [Google Scholar]
- Aron, J.L.; Patz, J.A. Ecosystem Change and Public Health: A Global Perspective; Johns Hopkins University Press: Baltimore, MD, USA, 2001. [Google Scholar]
- Kingscote, B.F. Correlation of bedrock type with the geography of Leptospirosis. Can. J. Comp. Med. 1970, 34, 31–37. [Google Scholar] [PubMed]
- Acha, P.; Szyfres, B. Zoonoses and Communicable Diseases Common to Man and Animals: Bacterioses and Mycoses, 3rd ed.; Pan American Health Organization: Washington, DC, USA, 2003; Volume 1. [Google Scholar]
- Lau, C.L.; Dobson, A.J.; Smythe, L.D.; Fearnley, E.J.; Skelly, C.; Clements, A.C.; Craig, S.B.; Fuimaono, S.D.; Weinstein, P. Leptospirosis in American Samoa 2010: Epidemiology, environmental drivers, and the management of emergence. Amer. J. Trop. Med. Hyg. 2012, 86, 309–319. [Google Scholar] [CrossRef]
- Reis, R.B.; Ribeiro, G.S.; Felzemburgh, R.D.; Santana, F.S.; Mohr, S.; Melendez, A.X.; Queiroz, A.; Santos, A.C.; Ravines, R.R.; Tassinari, W.S. Impact of environment and social gradient on Leptospira infection in urban slums. PLoS Negl. Trop. Dis. 2008, 2. [Google Scholar] [CrossRef]
- Barcellos, C.; Sabroza, P.C. The place behind the case: Leptospirosis risks and associated environmental conditions in a flood-related outbreak in Rio de Janeiro. Cadernos de Saúde Pública 2001, 17, S59–S67. [Google Scholar]
- Bhardwaj, P.; Kosambiya, J.; Desai, V. A case control study to explore the risk factors for acquisition of Leptospirosis in Surat city, after flood. Indian J. Med. Sci. 2008, 62, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Bovet, P.; Yersin, C.; Merien, F.; Davis, C.E.; Perolat, P. Factors associated with clinical Leptospirosis: A population-based case-control study in the Seychelles (Indian Ocean). Int. J. Epidemiol. 1999, 28, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.P.; Teixeira, M.G.; Costa, M.C.N.; Mendes, C.M.C.; Guimarães, P.; Reis, M.G.; Ko, A.; Barreto, M.L. Factors associated with Leptospira sp. infection in a large urban center in northeastern Brazil. Rev. Soc. Brasil. Med. Trop. 2007, 40, 499–504. [Google Scholar]
- World Health Organization. Closing the Gap: Policy into Practice on Social Determinants of Health. In Proceedings of the World Conference on Social Determinants of Health, Rio de Janeiro, Brazil, 19–21 October 2011.
- World Health Organization and Commission in Social Determinants of Health. Closing the Gap in a Generation: Health Equity through Action on the Social Determinants of Health; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- World Health Organization. Outcome of the World Conference on Social Determinants of Health. Resolution WHA65.8. In Proceedings of the Sixty-Fifth World Health Assembly, Geneva, Switzerland, 26 May 2012.
- World Health Organization. The Helsinki Statement on Health in All Policies. In Proceedings of the 8th Global Conference on Health Promotion, Helsinki, Finland, 12 June 2013.
- Smith, L.C.; Haddad, L.J. Explaining Child Malnutrition in Developing Countries: A Cross-country Analysis; International Food Policy Research Institute: Washington, DC, USA, 2000. [Google Scholar]
- Instituto Nacional de Información de Desarrollo (INIDE). VIII Censo de Población y IV de Vivienda. 2005. Available online: http://www.inide.gob.ni/censos2005/CifrasMun/tablas_cifras.htm (accessed on 10 May 2011).
- Pan American Health Organization. Impacto del Huracán Mitch en Centro América. Boletin Epidemiol. 1998, 19, 1–13. (in Spanish). [Google Scholar]
- Centers of Disease Control and Prevention. Outbreak of acute febrile illness and pulmonary hemorrhage—Nicaragua, 1995. Morbid. Mortal. Wkly. Rep. 1995, 44, 841–842. [Google Scholar]
- Trevejo, R.T.; Rigau-Pérez, J.G.; Ashford, D.A.; McClure, E.M.; Jarquín-González, C.; Amador, J.J.; José, O.; Gonzalez, A.; Zaki, S.R.; Shieh, W.-J. Epidemic Leptospirosis associated with pulmonary hemorrhage—Nicaragua, 1995. J. Infect. Dis. 1998, 178, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health of Nicaragua. Sistema Nicaragüense de Vigilancia Epidemiológica Nacional (SISNIVEN). Available online: http://www.minsa.gob.ni/index.php?option=com_remository&Itemid=52&func=select&id=1686 (accessed on 19 November 2013).
- Comisión Económica para América Latina y el Caribe (CEPAL). Indicadores de Pobreza: Necesidades Básicas Insatisfechas. Available online: http://www.eclac.cl/cgi-bin/getprod.asp?xml=/esalc/noticias/paginas/4/12754/P12754.xml&xsl=/esalc/tpl/p18f.xsl&base=/esalc/tpl/top-bottom.xsl (accessed on 12 December 2013).
- General Division of Public Health Surveillance, Ministry of Health of Nicaragua. General Information. Available online: http://www.minsa.gob.ni/ (accessed on 14 August 2012).
- Feres, J.C.; Mancero, X. El Método de las Necesidades Básicas Insatisfechas (NBI) y sus Aplicaciones en América Latina; Division de Estadística y Proyecciones Económicas, Naciones Unidas, CEPAL: Santiago de Chile, Chile, 2001. (in Spanish) [Google Scholar]
- Bacher, J. A probabilistic clustering model for variables of mixed type. Qual. Quant. 2000, 34, 223–235. [Google Scholar] [CrossRef]
- Watson, C.G.; Juba, M.P.; Manifold, V.; Kucala, T.; Anderson, P.E. The PTSD interview: Rationale, description, reliability, and concurrent validity of a DSM-III based technique. J. Clin. Psychol. 1991, 47, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Sudat, S.E.; Carlton, E.J.; Seto, E.Y.; Spear, R.C.; Hubbard, A.E. Using variable importance measures from causal inference to rank risk factors of schistosomiasis infection in a rural setting in China. Epidemiol. Perspect. Innov. 2010, 7. [Google Scholar] [CrossRef]
- Plan de Trabajo Interinstitucional de Abordaje Integral a la Leptospirosis; Ministerio de Salud de Nicaragua, Ministerio Agropecuario Forestal, Universidad Nacional Autónoma de Nicaragua, Organización Panamericana de la Salud: Managua, Nicaragua, 2011. (in Spanish)
- Sánchez, E. Epidemiological Situation of Leptospirosis in Nicaragua and the Intersectoral Plan. Available online: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&gid=19933&Itemid= (accessed on 13 June 2014).
- Moreno, G. Experience in Controlling Outbreaks of Leptospirosis in Leon. Available online: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&gid=19927&Itemid= (accessed on 13 June 2014).
- Chávez, O. Experience in Controlling Outbreaks of Leptospirosis in Chinandega. Available online: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&gid=19926&Itemid= (accessed on 13 June 2014).
- Soto, A. Preliminary Analysis of the Inter-Institutional Plan of Comprehensive Approach to Leptospirosis. Available online: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&gid=19924&Itemid= (accessed on 13 June 2014).
- Ashford, D.A.; Kaiser, R.M.; Spiegel, R.A.; Perkins, B.A.; Weyant, R.S.; Bragg, S.L.; Plikaytis, B.; Jarquin, C.; Reyes, J.D.L.; Amador, J.J. Asymptomatic infection and risk factors for Leptospirosis in Nicaragua. Amer. J. Trop. Med. Hyg. 2000, 63, 249–254. [Google Scholar]
- Muñoz-Zanzi, C.; Mason, M.; Encina, C.; Gonzalez, M.; Berg, S. Household characteristics associated with rodent presence and Leptospira infection in rural and urban communities from southern Chile. Amer. J. Trop. Med. Hyg. 2014, 90, 497–506. [Google Scholar]
- Hartskeerl, P.; Terpstra, W. Leptospirosis in wild animals. Vet. Quart. 1996, 18, 149–150. [Google Scholar] [CrossRef]
- Faria, M.T.; Calderwood, M.S.; Athanazio, D.A.; McBride, A.J.; Hartskeerl, R.A.; Pereira, M.M.; Ko, A.I.; Reis, M.G. Carriage of Leptospira interrogans among domestic rats from an urban setting highly endemic for leptospirosis in Brazil. Acta Tropica 2008, 108. [Google Scholar] [CrossRef]
- Porta, M. A Dictionary of Epidemiology, 5th ed.; Oxford University Press: New York, NY, USA, 2008. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bacallao, J.; Schneider, M.C.; Najera, P.; Aldighieri, S.; Soto, A.; Marquiño, W.; Sáenz, C.; Jiménez, E.; Moreno, G.; Chávez, O.; et al. Socioeconomic Factors and Vulnerability to Outbreaks of Leptospirosis in Nicaragua. Int. J. Environ. Res. Public Health 2014, 11, 8301-8318. https://doi.org/10.3390/ijerph110808301
Bacallao J, Schneider MC, Najera P, Aldighieri S, Soto A, Marquiño W, Sáenz C, Jiménez E, Moreno G, Chávez O, et al. Socioeconomic Factors and Vulnerability to Outbreaks of Leptospirosis in Nicaragua. International Journal of Environmental Research and Public Health. 2014; 11(8):8301-8318. https://doi.org/10.3390/ijerph110808301
Chicago/Turabian StyleBacallao, Jorge, Maria Cristina Schneider, Patricia Najera, Sylvain Aldighieri, Aida Soto, Wilmer Marquiño, Carlos Sáenz, Eduardo Jiménez, Gilberto Moreno, Octavio Chávez, and et al. 2014. "Socioeconomic Factors and Vulnerability to Outbreaks of Leptospirosis in Nicaragua" International Journal of Environmental Research and Public Health 11, no. 8: 8301-8318. https://doi.org/10.3390/ijerph110808301
APA StyleBacallao, J., Schneider, M. C., Najera, P., Aldighieri, S., Soto, A., Marquiño, W., Sáenz, C., Jiménez, E., Moreno, G., Chávez, O., Galan, D. I., & Espinal, M. A. (2014). Socioeconomic Factors and Vulnerability to Outbreaks of Leptospirosis in Nicaragua. International Journal of Environmental Research and Public Health, 11(8), 8301-8318. https://doi.org/10.3390/ijerph110808301