Assessing Health Risk due to Exposure to Arsenic in Drinking Water in Hanam Province, Vietnam
Abstract
:1. Introduction
2. Methods
2.1. Study Site
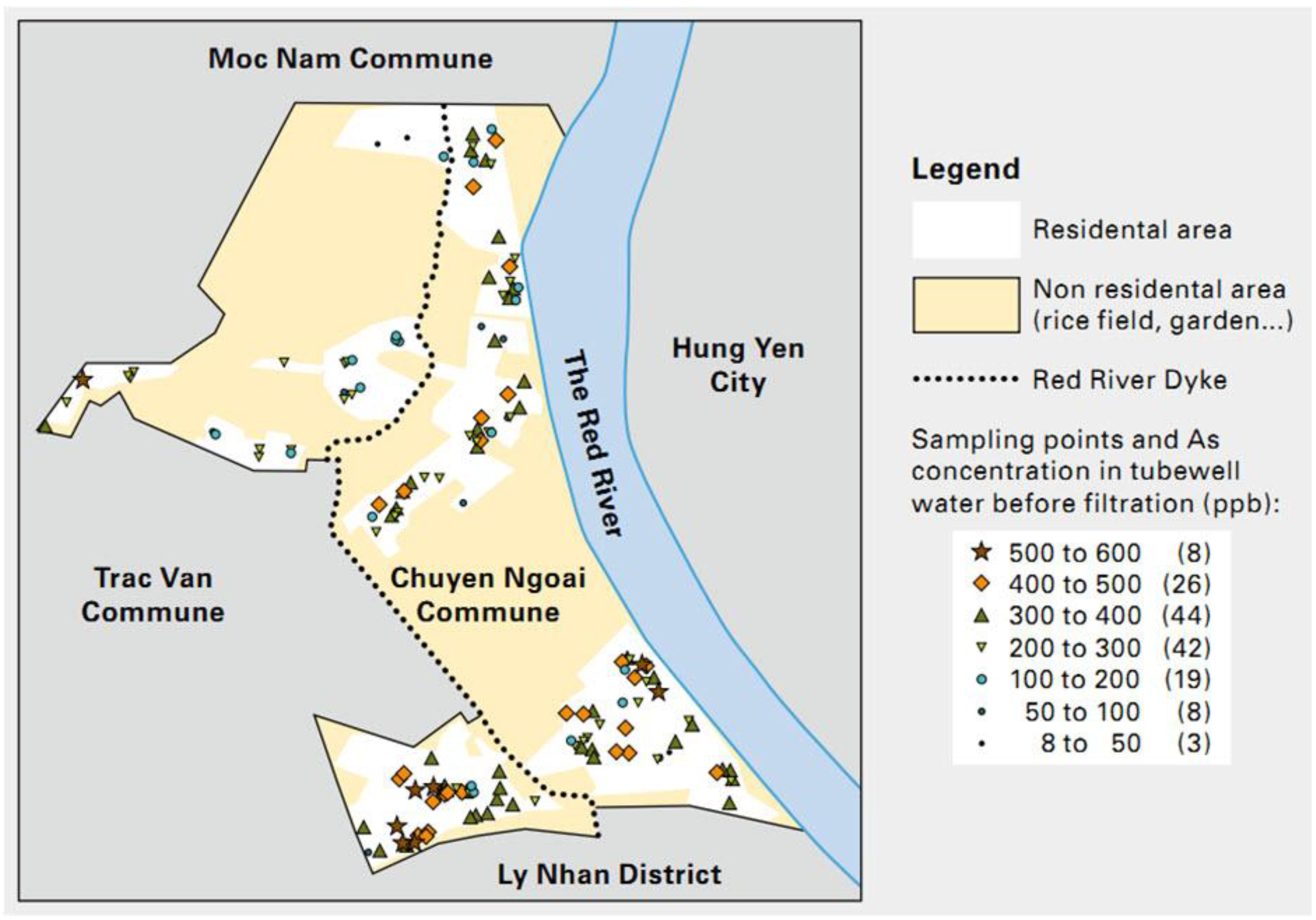
2.2. Environmental Health Risk Assessment
2.2.1. Issue Identification and Hazard Identification
2.2.2. Dose-Response Assessment
2.2.3. Exposure Assessment
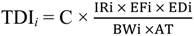
2.2.4. Risk Characterization
| Parameter | <2 years | 2–6 years | 6–16 years | >16 years | Source |
|---|---|---|---|---|---|
| IR, L/day | 1 | 1 | 2 | 2 | [21] |
| BW, kg | 10.5 | 18.2 | 27.5 | 50.5 | [22,23] |
| ADAF | 10 | 3 | 3 | 1 | [19] |
2.3. Statistical Analysis and Modeling
3. Results
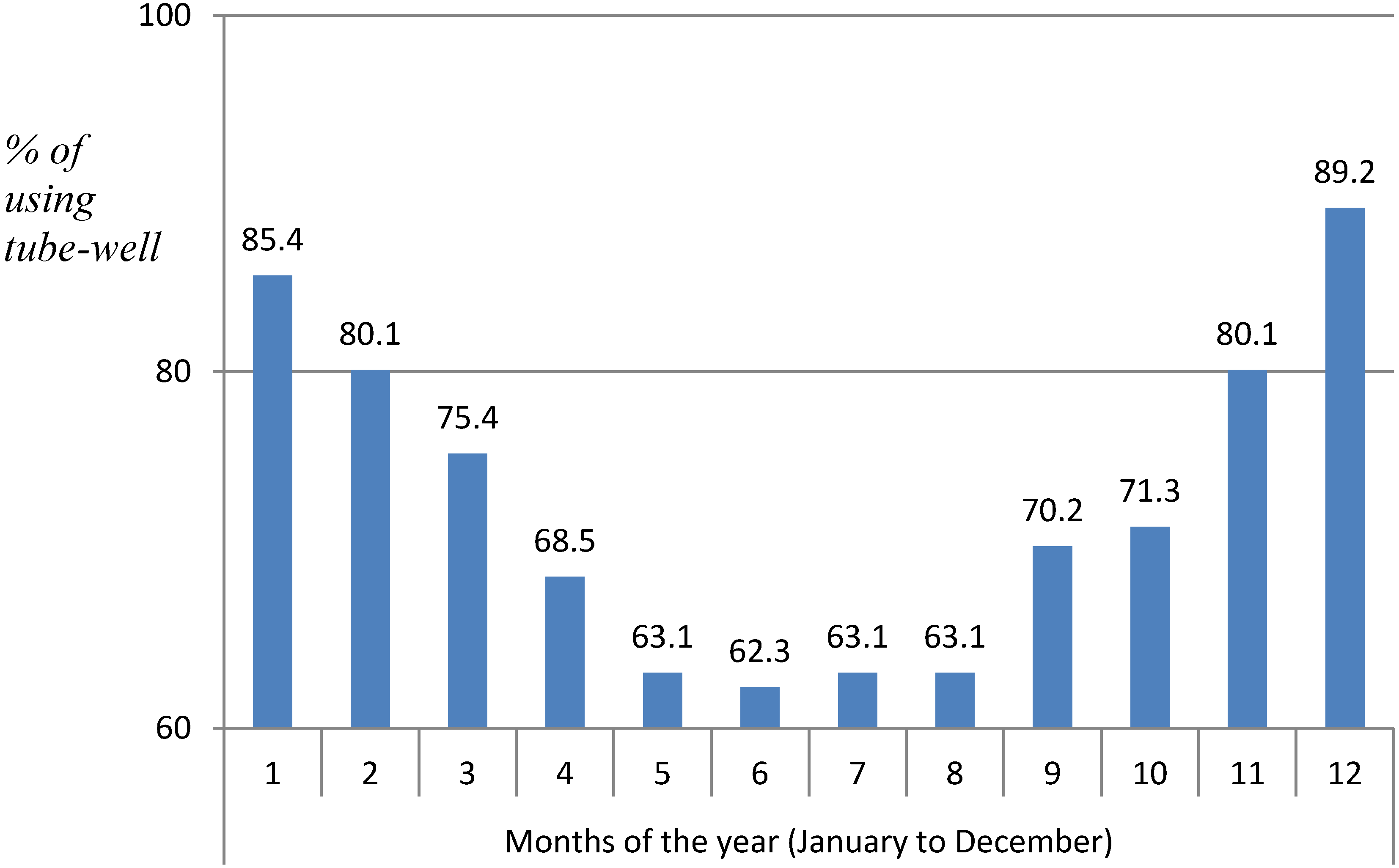
3.1. Issue Identification: Situation of Water Used for Drinking Purpose
3.2. Hazard Identification and Dose-Response Assessment
3.2.1. Hazard Identification
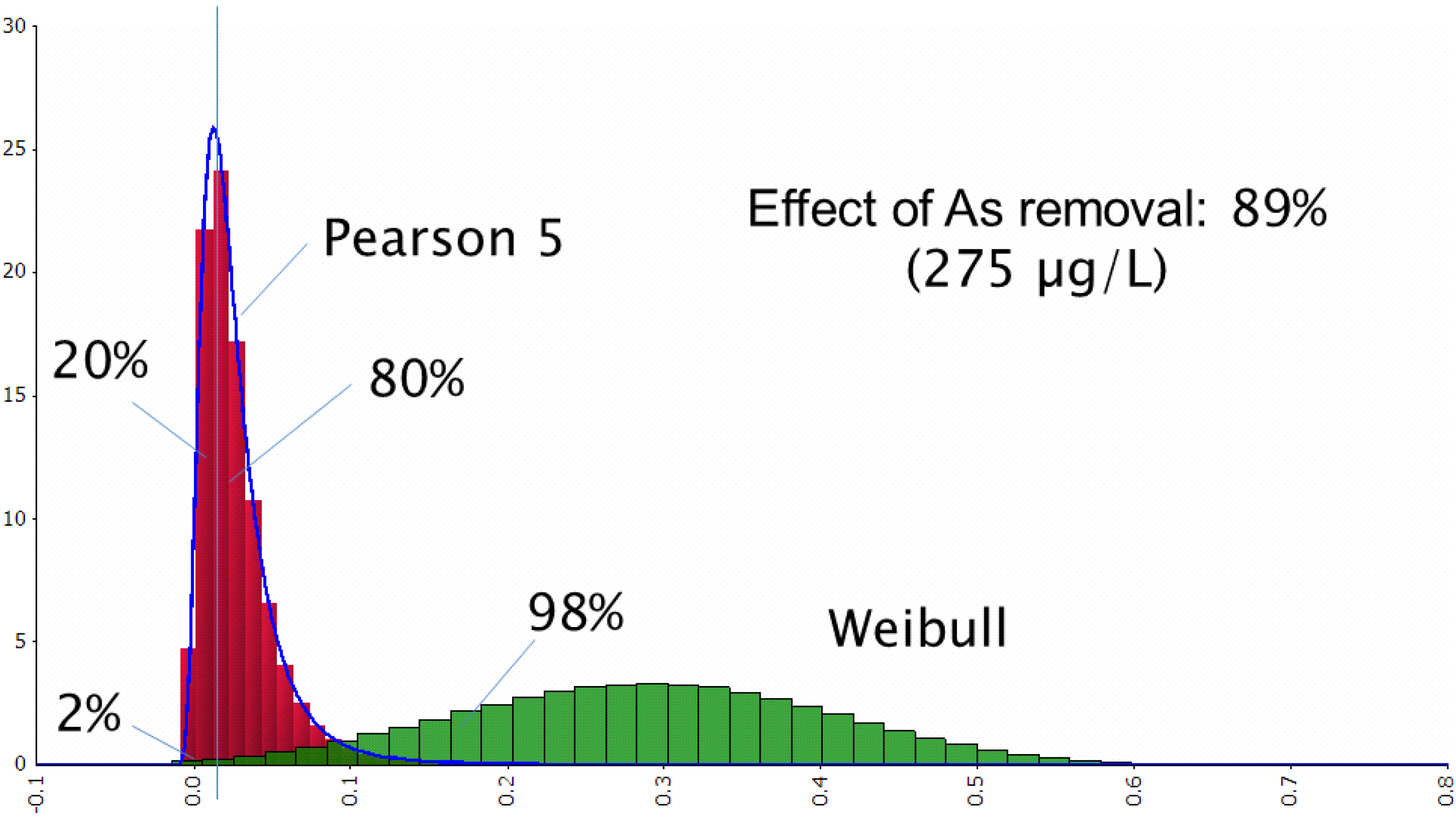
3.2.2. Arsenic Concentration in Tube-Well Water Before, after Sand Filtration and the Effect of Arsenic Removal
3.2.3. Dose-Response Assessment
3.3. Exposure Assessment
| No | Parameter | Group 1 | Group 2 | ||
|---|---|---|---|---|---|
| Function | Parameter | Function | Parameter | ||
| 1 | C1 | Weibull | RiskWeibull (3.5058, 0.40680, RiskShift (−0.077727)) | Weibull | RiskWeibull (5.0414, 0.59058, RiskShift (−0.22853)) |
| M = 0.289 | M = 0.314 | ||||
| SD = 0.116 | SD = 0.123 | ||||
| 2 | C2 | Pearson5 | M = 0.028 | Pearson5 | M = 0.0252 |
| SD = 0.0267 | SD = 0.0214 | ||||
| RiskPearson5(5.0000,0.18592, RiskShift (−0.018661)) | RiskPearson5(6.4116,0.24312, RiskShift (−0.019758)) | ||||
| 3 | EF/30 | NegBin | r = 3 | ||
| p = 0.46875 | |||||
| M = 3.4 | |||||
| SD = 2.693 | |||||
| RiskNegBin (3,0.46875) | |||||
| 4 | ED | NegBin | r = 6 | NegBin | r = 5 |
| p = 0.356 | p = 0.28 | ||||
| M = 10.867 | M = 12.854 | ||||
| SD = 5.527 | SD = 6.779 | ||||
3.4. Risk Characterization
| Group 1 | Group 2 | Group 1/ Group 2 | |
|---|---|---|---|
| R0 | 262.0 × 10−5 | 96.4 × 10−5 | 2.72 |
| R1 | 25.3 × 10−5 | 7.63 × 10−5 | 3.32 |
| R2 | 36.8 × 10−5 | 10.8 × 10−5 | 3.41 |
| R3 | 48.5 × 10−5 | 13.7 × 10−5 | 3.54 |
| R4 | 405.0 × 10·5 | 104.0 × 10−5 | 3.89 |
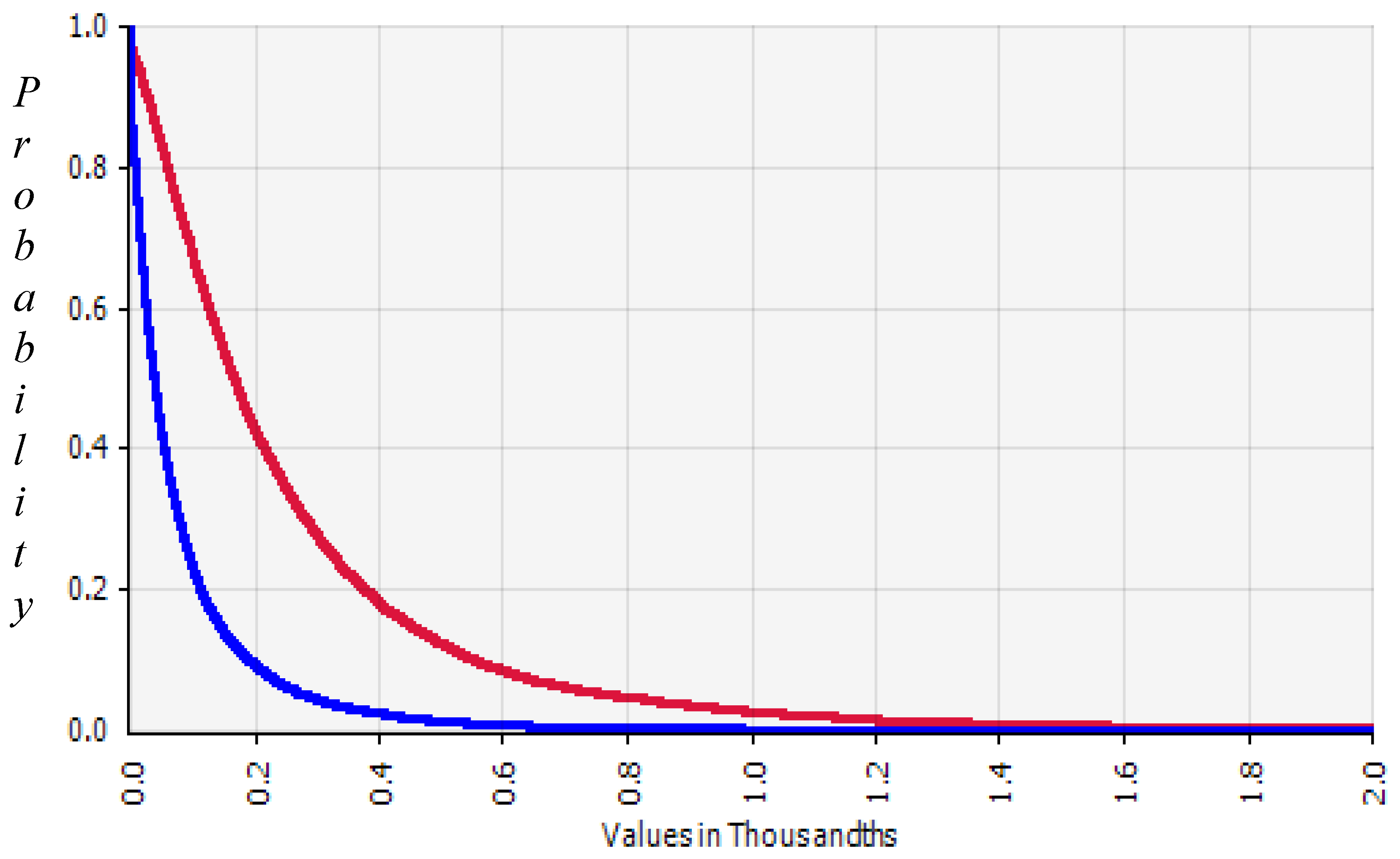
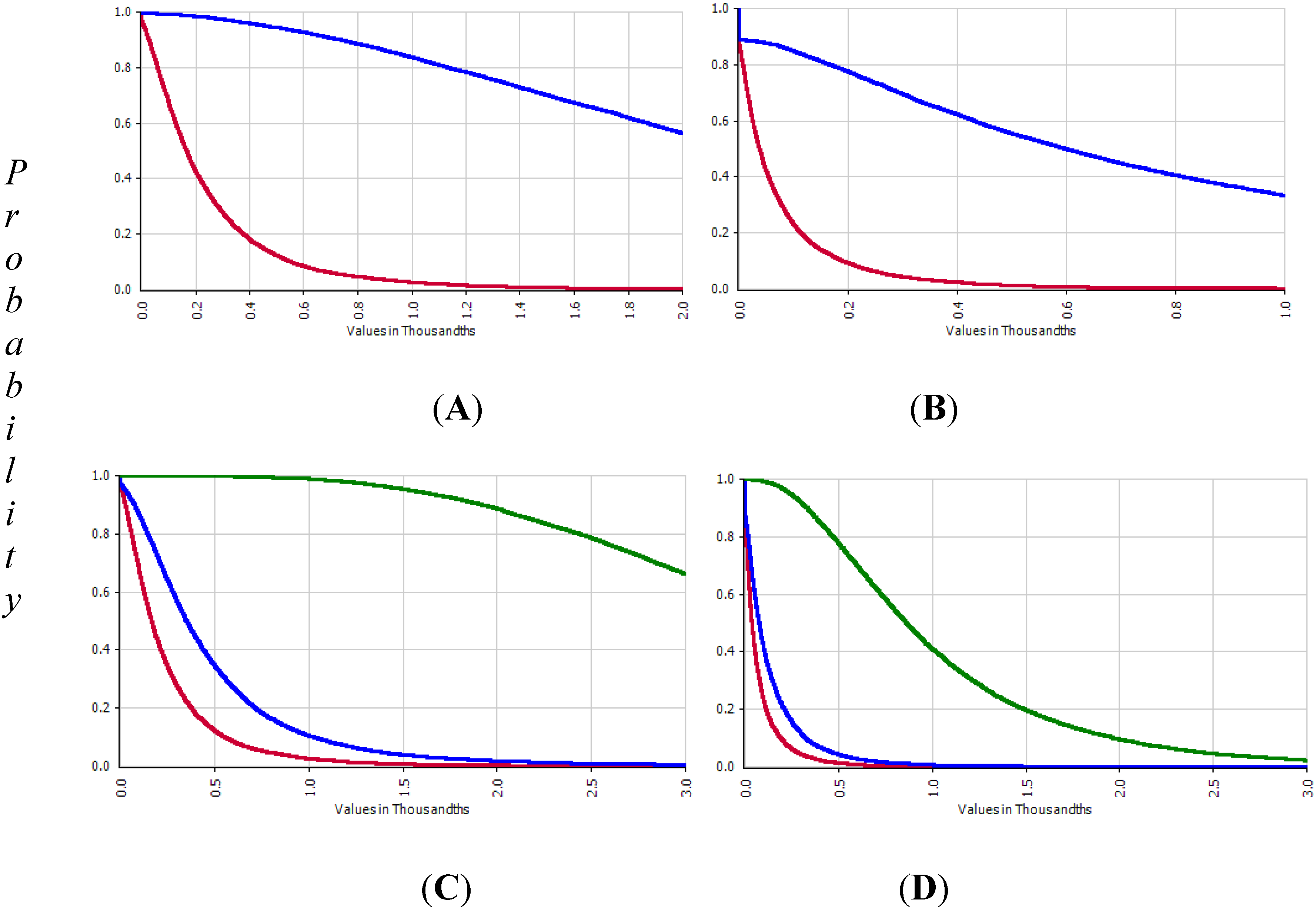
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for Arsenic; U.S. Department of Health and Human services: Atlanta, GA, USA, 2007. [Google Scholar]
- Brown, K.G.; Ross, G.L. Arsenic, drinking water, and health: A position paper of the American Council on science and health. Regulat. Toxicol. Pharmacol. 2002, 36, 162–174. [Google Scholar] [CrossRef]
- Nguyen, B.D. Cancer Pathology; Hanoi Medical Publisher: Hanoi, Vietnam, 2006. [Google Scholar]
- Chiou, H.Y.; Hsueh, Y.M.; Liaw, K.F.; Horng, S.F.; Chiang, M.H.; Pu, Y.S.; Lin, J.S.N.; Huang, C.H.; Chen, C.J. Incidence of internal cancers and ingested inorganic arsenic: A seven-year follow-up study in Taiwan. Cancer Res. 1995, 55, 1296–1300. [Google Scholar]
- Fewtrell, L.; Fuge, R.; Kay, D. An estimation of the global burden of disease due to skin lesions caused by arsenic in drinking water. J. Water Health 2005, 3, 101–107. [Google Scholar]
- Rahman, M.; Vahter, M.; Wahed, M.A.; Sohel, N.; Yunus, M.; Streatfield, P.K.; Arifeen, S.E.; Bhuiya, A.; Zaman, K.; Chowdhury, A.M.R.; Ekström, E.C.; Persson, L.Å. Prevalence of arsenic exposure and skin lesions. A population based survey in Matlab, Bangladesh. J. Epidemiol. Community Health 2006, 60, 242–248. [Google Scholar] [CrossRef]
- Basu, A.; Mahata, J.; Roy, A.K.; Sarkar, J.N.; Poddar, G.; Nandy, A.K.; Sarkar, P.K.; Dutta, P.K.; Banerjee, A.; Das, M.; Ray, K.; Roychaudhury, S.; Natarajan, A.T.; Nilsson, R.; Giri, A.K. Enhanced frequency of micronuclei in individuals exposed to arsenic through drinking water in West Bengal, India. Mutat. Res. 2002, 516, 29–40. [Google Scholar] [CrossRef]
- Smith, A.H.; Lingas, E.O.; Rahman, M. Contamination of drinking-water by arsenic in Bangladesh: A public health emergency. Bull. World Health Organ. 2000, 78, 1093–1103. [Google Scholar]
- Guha Mazumder, D.N.; Haque, R.; Ghosh, N.; De Binay, K.; Santra, A.; Chakraborty, D.; Smith, A.H. Arsenic levels in drinking water and the prevalence of skin lesions in West Bengal, India. Int. J. Epidemiol. 1998, 27, 871–877. [Google Scholar] [CrossRef]
- Arsenic Contamination in Drinking Water in Vietnam, an Overview of the Situation and the Necessary Mitigation Measures; University of Vietnam: Hanoi, Vietnam, 2004.
- Nguyen, K.H. Effects of Arsenic Contamination in Drinking Water on Human Health; Vietnam Medical Publishing House: Hanoi, Vietnam, 2006. [Google Scholar]
- Berg, M.; Stengel, C.; Pham, T.K.; Pham, H.V.; Sampson, M.L.; Leng, M.; Samreth, S.; Fredericks, D. Magnitude of arsenic pollution in the Mekong and Red River Deltas—Cambodia and Vietnam. Sci. Total Environ. 2007, 372, 413–425. [Google Scholar] [CrossRef]
- Winkel, L.H.; Pham, T.K.; Vi, M.L.; Stengel, C.; Amini, M.; Nguyen, T.H.; Pham, H.V.; Berg, M. Arsenic pollution of groundwater in Vietnam exacerbated by deep quifer exploitation for more than a century. Proc. Natl. Acad. Sci. USA 2010, 108, 1246–1251. [Google Scholar]
- Berg, M.; Luzi, S.; Trang, P.T.; Viet, P.H.; Giger, W.; Stuben, D. Arsenic removal from groundwater by household sand filters: Comparative field study, model calculations, and health benefits. Environ. Sci. Technol. 2006, 40, 5567–5573. [Google Scholar] [CrossRef]
- Environmental Health Risk Assessment: Guidelines for Assessing Human Health Risks from Environmental Hazards; Australian enHealth Council: Canberra, Australia, 2004.
- Lan, V.T.M. Determination of Arsenic in Biological Samples by Means of Atomic Absorption Spectroscopy (AAS) on the Basis of Samples by Microwave Processing; University of Natural Sciences, Vietnam National University: Hanoi, Vietnam, 2005. [Google Scholar]
- Trang, P.T.K. Studies Using Biochemical Indicators to Assess the Level of Arsenic Contamination in Water Wells and Correlation with Arsenic Exposure in Humans; University of Natural Sciences, Vietnam National University: Hanoi, Vietnam, 2006. [Google Scholar]
- National Census and Accommodation in Vietnam 2009: The Main Results; Steering National Committee for Census and Accommodation: Hanoi, Vietnam, 2010.
- United States Environmental Protection Agency (USEPA). Handbook for Implementing the Supplemental Cancer Guidance at Waste and Cleanup Sites, Cancer Risk Calculations. Available online: http://www.epa.gov/oswer/riskassessment/sghandbook/riskcalcs.htm (accessed on 4 July 2011).
- United States Environmental Protection Agency (USEPA). Integrated Risk Infomation System: Frequent Questions. Available online: http://www.epa.gov/iris/help_ques.htm (accessed on 4 July 2011).
- Nutrients in Drinking Water, Drinking Water Consumption; WHO: Geneva, Switzerland, 2005; p. 3.
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef]
- Vietnam National Institute of Nutrition. The Survey Results about Overweight—Obesity and Some Related Factors in the Group of 25–64 Age in Vietnam 2005–2006. Available online: http://viendinhduong.vn/news/vi/160/62/a/ket-qua-dieu-tra-thua-can---beo-phi-va-mot-so-yeu-to-lien-quan-o-nguoi-viet-nam-25--64-tuoi.aspx (7-7) (accessed on 18 January 2013).
- World Health Organization. Toxicological Evaluation of Certain Food Additives and Contaminants, Food Additive Series, No. 24; Cambridge University Press: Cambridge, UK, 1989. [Google Scholar]
- Lasky, T.; Sun, W.; Kadry, A.; Hoffman, M.K. Mean total arsenic concentrations in chicken 1989–2000 and estimated exposures for consumers of chicken. Environ. Health Perspect. 2004, 112, 18–21. [Google Scholar]
- Yu, W.H.; Harvey, C.M.; Harvey, C.F. Arsenic in groundwater in Bangladesh: A geostatistical and epidemiological framework for evaluating health effects and potential remedies. Water Resour. Res. 2003, 39. [Google Scholar] [CrossRef]
- Han, B.-C.; Jeng, W.-L.; Hung, T.-C.; Ling, Y.-C.; Shieh, M.-J.; Chien, L.-C. Estimation of metal and organochlorine pesticide exposures and potential health threat by consumption of oysters in Taiwan. Environ. Pollut. 2000, 109, 147–156. [Google Scholar] [CrossRef]
- Tao, S.S.; Bolger, P.M. Dietary arsenic intakes in the United States: FDA Total Diet Study, September 1991–December 1996. Food Addit. Contam. 1999, 16, 465–472. [Google Scholar] [CrossRef]
- Hsueh, Y.M.; Cheng, G.S.; Wu, M.M.; Yu, H.S.; Kuo, T.L.; Chen, C.J. Multiple risk factors associated with arsenic-induced skin cancer: Effects of chronic liver disease and malnutritional status. Br. J. Cancer 1995, 71, 109–114. [Google Scholar] [CrossRef]
- Lubin, J.H.; Pottern, L.M.; Stone, B.J.; Fraumeni, J.F., Jr. Respiratory cancer in a cohort of copper smelter workers: Results from more than 50 years of follow-up. Am. J. Epidemiol. 2000, 151, 554–565. [Google Scholar] [CrossRef]
- Kurttio, P.; Pukkala, E.; Kahelin, H.; Auvinen, A.; Pekkanen, J. Arsenic concentrations in well water and risk of bladder and kidney cancer in Finland. Environ. Health Perspect. 1999, 107, 705–710. [Google Scholar] [CrossRef]
- Rossman, T.G. Mechanism of arsenic carcinogenesis: An integrated approach. Mutat. Res. 2003, 533, 37–65. [Google Scholar] [CrossRef]
- Tchounwou, P.; Centeno, J.; Patlolla, A. Arsenic toxicity, mutagenesis, and carcinogenesis—A health risk assessment and management approach. Mol. Cell Biochem. 2004, 255, 47–55. [Google Scholar] [CrossRef]
- Johnston, A.M. Toxicological evaluation of certain veterinary residues. Presented on the 36th and 38th Meetings of Joint FAO/WHO Expert Committee on Food Additives (JECFA); WHO: Geneva, Switzerland; WHO Food Additives Series No. 29.
- Guidelines for Drinking Water Quality; WHO: Geneva, Switzerland, 2004.
- Smith, A.H.; Claudia, H.-R.; Michael, B.; Hellen, G.; Irva, P.; Heather, D.; Rose, W.; Michael, K.; Martyn, S. Cancer risks from arsenic in drinking water. Environ. Health Perspect. 1992, 97, 259–267. [Google Scholar] [CrossRef]
- Lamm, S.H.; Engel, A.; Penn, C.A.; Chen, R.; Feinleib, M. Arsenic cancer risk confounder in Southwest Taiwan data set. Environ. Health Perspet. 2006, 114, 1077–1082. [Google Scholar] [CrossRef]
- IARC Working Group. Some drinking-water disinfectants and contaminants, including Arsenic. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; WHO: Lyon, France, 2004; Volume 84. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huy, T.B.; Tuyet-Hanh, T.T.; Johnston, R.; Nguyen-Viet, H. Assessing Health Risk due to Exposure to Arsenic in Drinking Water in Hanam Province, Vietnam. Int. J. Environ. Res. Public Health 2014, 11, 7575-7591. https://doi.org/10.3390/ijerph110807575
Huy TB, Tuyet-Hanh TT, Johnston R, Nguyen-Viet H. Assessing Health Risk due to Exposure to Arsenic in Drinking Water in Hanam Province, Vietnam. International Journal of Environmental Research and Public Health. 2014; 11(8):7575-7591. https://doi.org/10.3390/ijerph110807575
Chicago/Turabian StyleHuy, Tung Bui, Tran Thi Tuyet-Hanh, Richard Johnston, and Hung Nguyen-Viet. 2014. "Assessing Health Risk due to Exposure to Arsenic in Drinking Water in Hanam Province, Vietnam" International Journal of Environmental Research and Public Health 11, no. 8: 7575-7591. https://doi.org/10.3390/ijerph110807575
APA StyleHuy, T. B., Tuyet-Hanh, T. T., Johnston, R., & Nguyen-Viet, H. (2014). Assessing Health Risk due to Exposure to Arsenic in Drinking Water in Hanam Province, Vietnam. International Journal of Environmental Research and Public Health, 11(8), 7575-7591. https://doi.org/10.3390/ijerph110807575




