Inactivation of Selected Bacterial Pathogens in Dairy Cattle Manure by Mesophilic Anaerobic Digestion (Balloon Type Digester)
Abstract
:1. Introduction
2. Materials and Methods
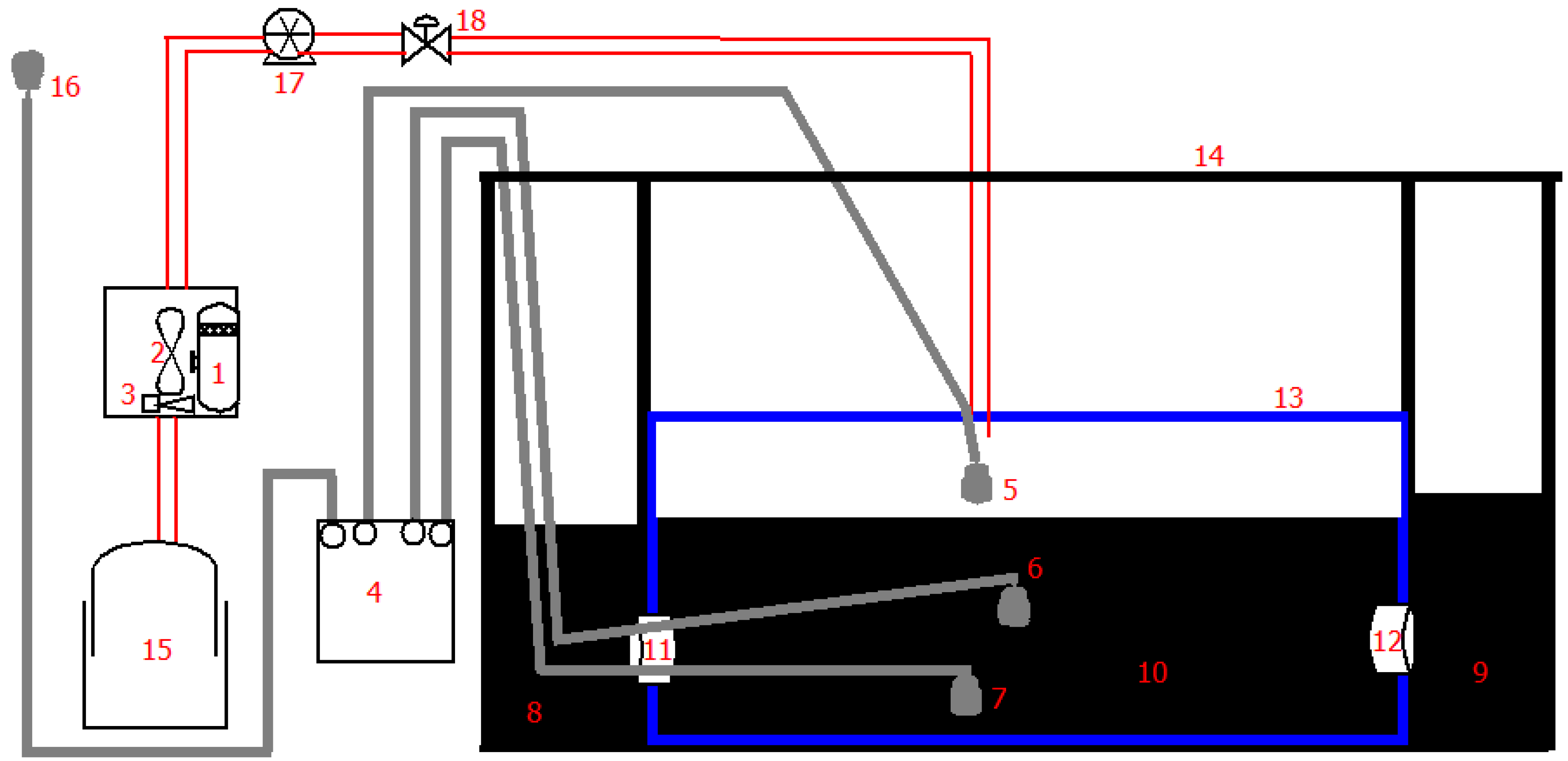
2.1. Experimental Set Up
2.2. Microbial Analysis
2.3. Statistical Analysis
3. Results

| Bacteria | Log of Count | X5 | X4 | X3 | X2 | X | Constant | R2 | p-value |
|---|---|---|---|---|---|---|---|---|---|
| ERN | Y | 0 | 0 | 0 | 0 | −0.61 | 5.2 | 0.976 | 0.957 |
| SMN | Y | 0 | −4.5 × 10−8 | 1 × 10−5 | −5.1 × 10−4 | −0.023 | 4.3 | 0.960 | 0.773 |
| CPL | Y | 3.8 × 10−3 | −0.015 | 0.21 | −1.2 | 2.1 | 4 | 0.991 | 0.723 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Grant, S.; Marshalleck, A. Energy Production and Pollution Mitigation from Broilers Houses on Poultry Farms in Jamaica and Pennsylvania. Int. J. Serv. Learn. Eng. 2008, 3, 41–52. [Google Scholar]
- Grewal, S.K.; Rajeev, S.; Sreevatsan, S.; Michel, F.C., Jr. Persistence of Mycobacterium avium subsp. Paratuberculosis and other zoonotic pathogens during simulated composting, manure packing and liquid storage of dairy manure. Appl. Environ. Microbiol. 2006, 72, 565–574. [Google Scholar] [CrossRef]
- Poudel, R.M.; Joshi, D.R.; Dhakal, N.R.; Karki, A.B. Anaerobic digestion of sewage sludge mixture for the reduction of indicator and pathogenic microorganisms. Sci. World 2010, 8, 47–50. [Google Scholar]
- Vanselow, B.A.; Hornitzky, M.A.; Bailey, G.D. Intensification of cattle production increases the risk of zoonotic campylobacteriosis. In Proceedings of the 11th International Symposium on Veterinary Epidemiology and Economics, Cairns, Australia, 6–11 August 2006.
- Hutchison, M.L.; Walters, L.D.; Avery, S.M.; Munro, F.; Moore, A. Analyses of livestock production, waste storage and pathogen levels and prevalences in farm manures. Appl. Environ. Microbiol. 2005, 71, 1231–1236. [Google Scholar] [CrossRef]
- Nicholson, F.A.; Groves, S.J.; Chambers, B.J. Pathogen survival during livestock manure storage and following land application. Bioresource Technol. 2005, 96, 135–143. [Google Scholar] [CrossRef]
- Strauch, D. Survival of pathogenic micro-organisms and parasites in excreta, manure and sewage sludge. Rev. Sci. Tech. Off. 1991, 10, 813–846. [Google Scholar]
- Hess, T.F.; Grdzelishvili, I.; Sheng, H.Q.; Hovde, C.J. Heat inactivation of E. coli during manure composting. Compost Sci. Utilization 2004, 12, 314–322. [Google Scholar]
- Martin, H. Manure composting as a pathogen reduction strategy. In Agricultural Engineering and Animal Science. Factsheet; Ministry of Agriculture and Food: Guelph, ON, Canada, 2005. [Google Scholar]
- Čempírková, R.; Šoch, M. The analysis of real microbiological risks for dissociated slurry. Agric. Trop. Subtrop. 2007, 40, 164–171. [Google Scholar]
- Lutge, B.; Standish, B. Assessing the potential for electricity generation from animal waste biogas on South African farms. Agrekon: Agric. Econ. Res. Policy Pract. South. Afr. 2013, 52, 1–24. [Google Scholar]
- Mukumba, P.; Makaka, G.; Mamphweli, S.; Simon, M.; Meyer, E. An insight into the status of biogas digesters technologies in South Africa with reference to the Eastern Cape Province. Fort Hare Pap. 2012, 19, 5–29. [Google Scholar]
- Gerba, C.P.; Smith, J.E., Jr. Sources of pathogenic microorganisms and their fate during land application of wastes. J. Environ. Qual. 2005, 34, 42–48. [Google Scholar]
- Manyi-Loh, C.E.; Mamphweli, S.N.; Meyer, E.L.; Okoh, A.I.; Makaka, G.; Simon, M. Micobial anaerobic digestion as an approach to the decontamination of animal wastes in pollution control and generation of renewable energy. Int. J. Environ. Res. Public Health 2013, 10, 4390–4417. [Google Scholar] [CrossRef]
- Yongabi, K.A.; Harris, P.L.; Lewis, D.M.; Agho, M.O. Preliminary study on the effect of anaerobically digested cow dung slurry on the antimicrobial activity of three medicinal plants. Afr. J. Micobiol. Res. 2009, 3, 168–174. [Google Scholar]
- Srijan, A.; Bodhidatta, L.; Mason, C.J.; Bunyarakyothin, G.; Jiarakul, W.; Vithayasai, N. Field evaluation of a transport medium and enrichment broth for the isolation of Campylobacter species from human diarrheal stool samples. Open J. Med. Microbiol. 2013, 3, 48–52. [Google Scholar] [CrossRef]
- Nyachoti, C.M.; Omogbenigun, F.O.; Rademacher, M.; Blank, G. Performance responses and indicators of gastrointestinal health in early-weaned pigs fed low-protein amino acid-supplemented diets. J. Anim. Sci. 2006, 84, 125–134. [Google Scholar]
- Cheesbrough, M. Part 2: Microbiology. In District Laboratory Practice in Tropical Country; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Campylobacter Jejuni and Campylobacter coli. In OIE Terrestrial Manual; World Organization for Animal Health: Paris, France, 2008; pp. 1185–1191.
- Côté, C.; Massé, D.I.; Quessy, S. Reduction of indicator and pathogenic microorganisms by psychrophilic anaerobic digestion in slurries. Bioresour. Technol. 2006, 97, 686–691. [Google Scholar] [CrossRef]
- Sutton, C. Accuracy of Plate Counts. Available online: http://www.microbiol.org/wp-content/uploads/2010/07/Sutton.jvt_.2011.17_3.pdf (accessed on 9 July 2014).
- Willey, J.; Sherwood, L.M.; Woolverton, C.J. Microbial growth. In Prescott’s Microbiology, 8th ed.; McGraw-Hill Companies Inc.: New York, NY, USA, 2011; pp. 162–189. [Google Scholar]
- Sahlström, L. A review of survival of pathogenic bacteria in organic waste used in biogas plants. Bioresour. Technol. 2003, 87, 161–166. [Google Scholar] [CrossRef]
- Ramanathan, V.; Feng, Y. Air pollution, greenhouse gases and climate change: Global and regional perspectives. Atmos. Environ. 2009, 43, 37–50. [Google Scholar]
- Strik, D.P.B.T.B.; Domnanovich, A.M.; Holubar, P.A. A pH-based control of ammonia in biogas during anaerobic digestion of artificial pig manure and maize silage. Process Biochem. 2006, 41, 1235–1238. [Google Scholar] [CrossRef]
- Willey, J.; Sherwood, L.M.; Woolverton, C.J. Microbial Taxonomy and Evolution of Diversity. In Prescott’s Microbiology, 8th ed.; McGraw-Hill Companies Inc.: New York, NY, USA, 2011; pp. 446–472. [Google Scholar]
- Babaee, A.; Shayegan, J. Effects of Organic Loading Rates (OLR) on production of methane from anaerobic digestion of vegetable waste. In Proceedings of the World Renewable Energy Congress, Linköping, Sweden, 8–13 May 2011; pp. 411–417.
- Anunputtikul, W.; Rodtong, S. Laboratory Scale Experiments for Biogas Production from Cassava Tubers. In Proceedings of the Joint International Conference on “Sustainable Energy and Environment (SEE)”, Hua Hin, Thailand, 1–3 December 2004; pp. 238–243.
- Bui, X.T.; Woff, A.; Madsen, M.; Bang, D.D. Fate and survival of Campylobacter coli in swine manure at various temperatures. Front. Microbiol. 2011, 2. [Google Scholar] [CrossRef]
- Carbone, S.R.; da Silva, F.M.; Tavares, C.R.G.; Dias Filho, B.P. Bacterial population of a two-phase anaerobic digestion process treating effluent of cassava starch factory. Environ. Technol. 2002, 23, 591–597. [Google Scholar] [CrossRef]
- Blumer-Schuette, S.E.; Kataeva, I.; Westpheling, J.; Adams, M.W.W.; Kelly, R.M. Extremely thermophilic microorganisms for biomass conversion: Status and prospects. Curr. Opin. Biotechnol. 2008, 19, 210–217. [Google Scholar] [CrossRef]
- Wirth, R.; Kovács, E.; Maròti, G.; Bagi, Z.; Rakhely, G.; Kovács, K.L. Characterization of a biogas—Producing microbial community by short-read next generation DNA sequencing. Biotechnol. Biofuels 2012, 5. [Google Scholar] [CrossRef]
- Harrison, J.H.; Gay, J.M.; McClanahan, R.; Whitefield, E.; Saunders, O.; Fortuna, A.M. Managing Manure to Minimize Environmental Impact. In Proceeding of the 2011 Midwest Manure Summit, Lambeau Field, Green Bay, WI, USA, 15–16 February 2011.
- Cha, G.-C.; Chung, H.-K.; Kim, D.-J. Characteristics of temperature change on the substrate degradation and bacterial population in one-phase and two-phase anaerobic digestion. Environ. Eng. Res. 2001, 6, 99–108. [Google Scholar]
- Demirel, B.; Scherer, P. The roles of acetotrophic and hydrogenotrophic methanogens during anaerobic conversion of biomass to methane: A review. Rev. Environ. Sci. Biotechnol. 2008, 7, 173–190. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Manyi-Loh, C.E.; Mamphweli, S.N.; Meyer, E.L.; Okoh, A.I.; Makaka, G.; Simon, M. Inactivation of Selected Bacterial Pathogens in Dairy Cattle Manure by Mesophilic Anaerobic Digestion (Balloon Type Digester). Int. J. Environ. Res. Public Health 2014, 11, 7184-7194. https://doi.org/10.3390/ijerph110707184
Manyi-Loh CE, Mamphweli SN, Meyer EL, Okoh AI, Makaka G, Simon M. Inactivation of Selected Bacterial Pathogens in Dairy Cattle Manure by Mesophilic Anaerobic Digestion (Balloon Type Digester). International Journal of Environmental Research and Public Health. 2014; 11(7):7184-7194. https://doi.org/10.3390/ijerph110707184
Chicago/Turabian StyleManyi-Loh, Christy E., Sampson N. Mamphweli, Edson L. Meyer, Anthony I. Okoh, Golden Makaka, and Michael Simon. 2014. "Inactivation of Selected Bacterial Pathogens in Dairy Cattle Manure by Mesophilic Anaerobic Digestion (Balloon Type Digester)" International Journal of Environmental Research and Public Health 11, no. 7: 7184-7194. https://doi.org/10.3390/ijerph110707184
APA StyleManyi-Loh, C. E., Mamphweli, S. N., Meyer, E. L., Okoh, A. I., Makaka, G., & Simon, M. (2014). Inactivation of Selected Bacterial Pathogens in Dairy Cattle Manure by Mesophilic Anaerobic Digestion (Balloon Type Digester). International Journal of Environmental Research and Public Health, 11(7), 7184-7194. https://doi.org/10.3390/ijerph110707184






