Hair Mercury Concentrations and Fish Consumption Patterns in Florida Residents
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Questionnaire Design and Administration
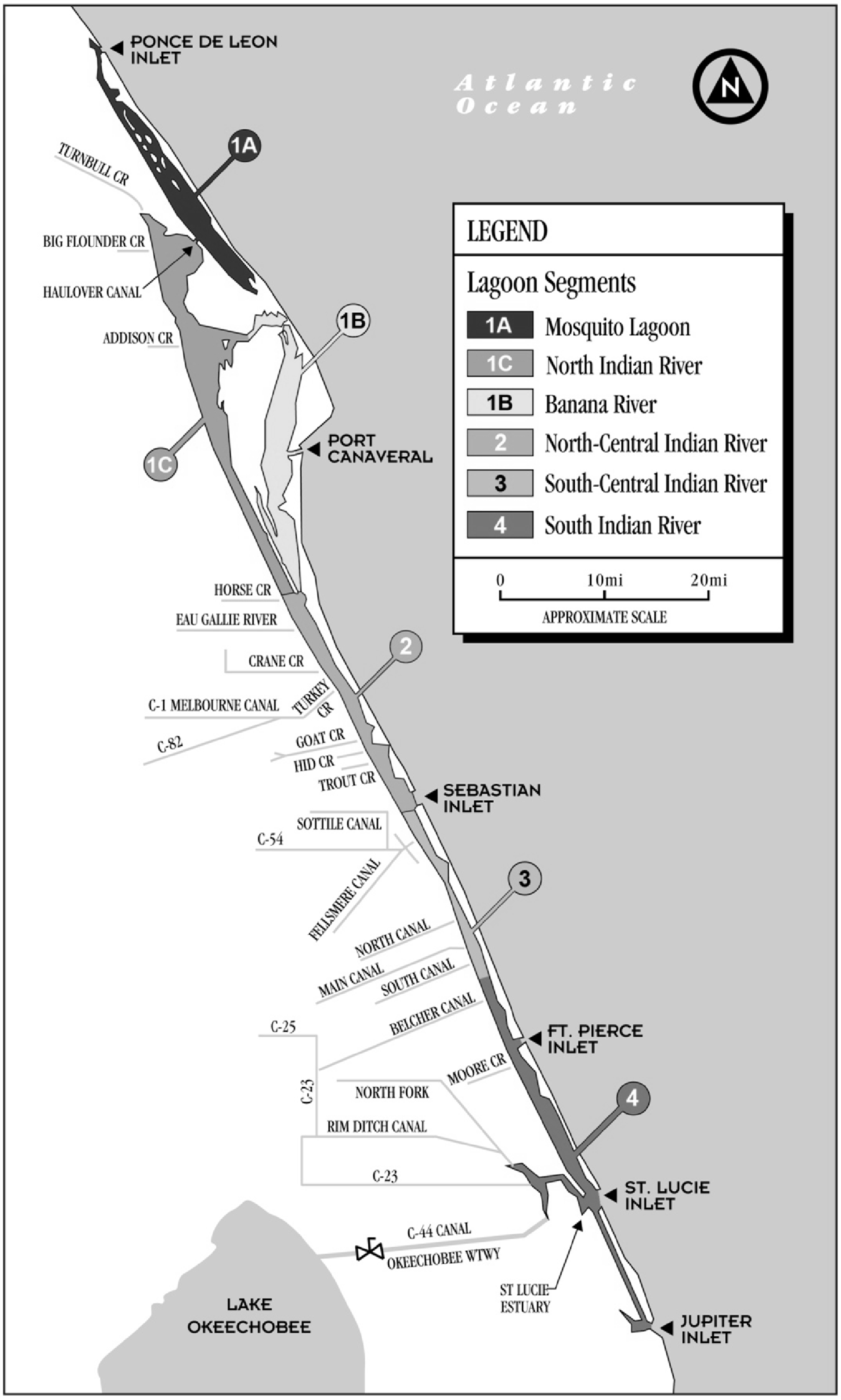
2.3. Hair Sample Collection and Mercury Analysis
2.4. Statistical Analysis
3. Results
3.1. Study Population Demographics and Fish Consumption Patterns
| Participant Group | Percentile | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| N | % | Mean ± SD | Range | Median | 75th | 90th | 95th | p-value 1 | |
| All | 135 | 1.53 ± 1.89 | 0.04–16.18 | 1.01 | 1.86 | 3.16 | 5.01 | ||
| Sex | <0.01 | ||||||||
| Male | 73 | 54% | 2.02 ± 2.38 | 0.07–16.18 | 1.17 | 2.81 | 4.74 | 6.06 | |
| Female | 62 | 46% | 0.96 ± 0.74 | 0.04–3.26 | 0.74 | 1.38 | 1.98 | 2.60 | |
| Age | 0.24 | ||||||||
| 18 – 40 | 29 | 22% | 1.07 ± 0.73 | 0.04–2.72 | 0.96 | 1.62 | 2.34 | 2.57 | |
| 41 – 60 | 53 | 39% | 1.75 ± 1.83 | 0.19–8.09 | 1.01 | 2.48 | 5.06 | 6.06 | |
| 61+ | 53 | 39% | 1.57 ± 2.33 | 0.07–16.18 | 1.07 | 1.84 | 3.21 | 4.30 | |
| Race/Ethnicity | 0.69 | ||||||||
| Caucasian | 129 | 95% | 1.56 ± 1.93 | 0.04–16.18 | 1.01 | 1.86 | 3.17 | 5.05 | |
| Other | 6 | 5% | 1.08 ± 0.90 | 0.31–2.58 | 0.77 | 1.90 | - | - | |
| Education | 0.31 | ||||||||
| High school or less | 28 | 21% | 1.78 ± 1.96 | 0.07–8.09 | 1.02 | 2.92 | 5.02 | 6.88 | |
| Some college | 31 | 23% | 1.09 ± 1.37 | 0.04–7.58 | 0.75 | 1.45 | 2.44 | 4.61 | |
| Graduated college or more | 76 | 56% | 1.63 ± 2.04 | 0.08–16.18 | 1.12 | 2.17 | 3.15 | 4.39 | |
| BMI (kg/m2) | 0.47 | ||||||||
| <24.9 | 54 | 40% | 1.26 ± 1.24 | 0.04–7.58 | 0.86 | 1.50 | 2.68 | 3.32 | |
| 25–29.9 | 55 | 42% | 1.99 ± 2.57 | 0.07–16.18 | 1.20 | 2.58 | 5.03 | 5.94 | |
| >30 | 25 | 18% | 1.16 ± 0.94 | 0.14–3.17 | 0.96 | 1.64 | 2.99 | 3.13 | |
| County of Residence | 0.03 | ||||||||
| Brevard | 33 | 25% | 1.88 ± 1.79 | 0.08–8.09 | 1.17 | 2.79 | 4.82 | 6.14 | |
| Indian river | 18 | 14% | 0.97 ± 0.79 | 0.07–2.94 | 0.78 | 1.50 | 2.39 | – | |
| St. Lucie | 22 | 17% | 1.07 ± 0.74 | 0.14–3.17 | 0.82 | 1.46 | 2.26 | 3.05 | |
| Martin | 38 | 28% | 2.08 ± 2.81 | 0.18–16.18 | 1.17 | 2.65 | 4.33 | 8.01 | |
| Palm beach | 5 | 4% | 1.34 ± 0.74 | 0.62–2.22 | 1.09 | 2.13 | – | – | |
| Other | 16 | 12% | 0.95 ± 1.38 | 0.04–5.41 | 0.50 | 1.02 | 3.70 | – | |
| Total Seafood Consumption | <0.01 | ||||||||
| Once or more per day | 9 | 7% | 2.14 ± 1.86 | 0.04–5.31 | 2.96 | 3.21 | – | – | |
| Three times per week | 66 | 49% | 1.95 ± 2.32 | 0.08–16.18 | 1.20 | 2.39 | 4.30 | 5.30 | |
| Once per week | 50 | 37% | 1.08 ± 1.16 | 0.09–7.58 | 0.73 | 1.41 | 2.02 | 2.84 | |
| Once per month or less | 10 | 7% | 0.49 ± 0.29 | 0.18–0.91 | 0.39 | 0.79 | 0.90 | – | |
| IRL Seafood Consumption | 0.11 | ||||||||
| Three times per week or more | 8 | 6% | 2.01 ± 1.47 | 0.79–4.98 | 1.19 | 3.02 | – | – | |
| Once per week | 17 | 13% | 1.71 ± 1.41 | 0.26–5.41 | 1.14 | 3.07 | 3.69 | – | |
| Once per month or less | 110 | 81% | 1.47 ± 1.99 | 0.04–16.18 | 0.89 | 1.73 | 2.96 | 4.71 | |
| Fish Sources | <0.01 | ||||||||
| All recreational | 28 | 21% | 2.53 ± 3.20 | 0.26–16.18 | 1.21 | 3.14 | 5.36 | 12.31 | |
| Most recreational | 17 | 13% | 2.46 ± 2.24 | 0.07–8.09 | 1.62 | 3.76 | 5.95 | – | |
| Half recreational | 13 | 10% | 1.65 ± 1.06 | 0.49–3.63 | 1.15 | 2.71 | 3.44 | – | |
| Most commercial | 24 | 18% | 1.20 ± 0.71 | 0.18–2.58 | 1.14 | 1.73 | 2.31 | 2.52 | |
| All commercial | 52 | 38% | 0.85 ± 0.73 | 0.04–2.96 | 0.60 | 1.11 | 1.85 | 2.72 | |
| Shellfish Sources | <0.01 | ||||||||
| All recreational | 14 | 10% | 3.37 ± 4.50 | 0.19–16.18 | 1.20 | 5.63 | 12.14 | – | |
| Most recreational | 10 | 7% | 2.54 ± 1.83 | 0.55–5.41 | 1.84 | 4.47 | 5.38 | – | |
| Half recreational | 5 | 4% | 2.77 ± 1.15 | 1.15–4.38 | 2.72 | 3.68 | – | – | |
| Most commercial | 19 | 15% | 1.10 ± 0.82 | 0.20–3.63 | 0.85 | 1.41 | 2.28 | – | |
| All commercial | 85 | 64% | 1.12 ± 1.00 | 0.04–5.31 | 0.75 | 1.60 | 2.74 | 3.08 | |
3.2. Hair Mercury Concentration
| Species | <Once a Week | ≥Once a Week | p-value 1 |
|---|---|---|---|
| Canned Tuna | 1.51 (1.97) | 1.63 (1.60) | 0.19 |
| Tuna Fillet or Steak 2 | 1.51 (1.95) | 1.80 (1.05) | 0.12 |
| Salmon | 1.52 (2.02) | 1.56 (1.48) | 0.39 |
| Snapper 2 | 1.38 (1.88) | 2.71 (1.55) | <0.01 |
| Mullet 2 | 1.49 (1.89) | 2.68 (1.78) | 0.06 |
| Sea Trout 2 | 1.49 (1.94) | 2.21 (0.87) | 0.02 |
| Cobia | 1.29 (1.23) | 4.99 (4.68) | <0.01 |
| Shark 2 | 1.54 (1.90) | - | - |
| Swordfish 2 | 1.51 (1.90) | 2.80 (0.24) | 0.13 |
| Grouper 2 | 1.43 (1.93) | 2.16 (1.55) | 0.02 |
| Shrimp and Crab | 1.72 (2.27) | 1.34 (1.39) | 0.19 |
| Shellfish other 2 | 1.40 (1.42) | 1.94 (2.86) | 0.12 |
| Sushi | 1.53 (1.97) | 1.56 (1.28) | 0.30 |
| Tilapia | 1.59 (1.95) | 0.83 (0.57) | 0.09 |
| Mahi 2 | 1.52 (1.99) | 1.63 (1.25) | 0.18 |
| Flounder 2 | 1.53 (1.90) | 1.80 (1.64) | 0.60 |
| Pompano | 1.53 (1.90) | 2.16 (1.40) | 0.32 |
| Redfish 2 | 1.53 (1.90) | 2.62 - | - |
| Cod | 1.54 (1.93) | 1.33 (0.62) | 0.64 |
| Sheepshead 2 | 1.54 (1.90) | 1.17 - | - |
| All Other Species | 1.44 (1.42) | 2.84 (5.15) | 0.91 |
| Variable | Β-Coefficient | 95% Confidence Interval | p-value 1 |
|---|---|---|---|
| Gender | |||
| Females | Ref | - | - |
| Males | 0.11 | −0.05–0.27 | 0.17 |
| Education | |||
| High school or less | Ref | - | - |
| Some college | 0.07 | −0.29–0.14 | 0.26 |
| Graduated college or more | 0.15 | 0.03–0.30 | 0.05 |
| Total Seafood Consumption | - | - | |
| ≤1x/month | Ref | - | - |
| 1x/week | 0.19 | 0.11–0.49 | 0.20 |
| ≥3x/week | 0.26 | 0.04–0.56 | 0.08 |
| Fish Sources | |||
| All commercial | Ref | - | - |
| Most commercial | 0.10 | −0.11–0.32 | 0.34 |
| Half recreational | 0.22 | −0.05–0.49 | 0.11 |
| Most recreational | 0.32 | 0.08–0.56 | 0.01 |
| All recreational | 0.32 | 0.11–0.53 | <0.01 |
| Variable | β-Coefficient | 95% Confidence Interval | p-value |
|---|---|---|---|
| Male Participants | |||
| Education | |||
| Less than college | Ref | - | - |
| Graduated from college or more | 0.10 | 0.11–0.32 | 0.34 |
| Total Seafood Consumption | |||
| ≤1x/month | Ref | - | - |
| 1x/week | −0.09 | −0.74–0.56 | 0.78 |
| ≥3x/week | 0.09 | −0.56–0.74 | 0.78 |
| Fish Sources | |||
| All commercial | Ref | - | - |
| Most commercial | 0.28 | 0.06–0.62 | 0.10 |
| Half recreational | 0.39 | 0.04–0.74 | 0.03 |
| Most recreational | 0.47 | 0.12–0.81 | 0.01 |
| All recreational | 0.50 | 0.21–0.80 | <0.01 |
| Female Participants | |||
| Education | |||
| Less than college | Ref | - | - |
| Graduated college or more | 0.19 | 0.03–0.40 | 0.08 |
| Total Seafood Consumption | |||
| ≤1x/month | Ref | - | - |
| 1x/week | 0.26 | −0.08–0.60 | 0.13 |
| ≥3x/week | 0.23 | −0.13–0.58 | 0.20 |
| Fish Sources | |||
| All commercial | Ref | - | - |
| Most commercial | 0.05 | −0.20–0.30 | 0.70 |
| Half recreational | 0.19 | −0.33–0.72 | 0.46 |
| Most recreational | 0.15 | 0.19–0.50 | 0.38 |
| All recreational | 0.23 | −0.11–0.57 | 0.18 |
| Risk Factor | < 1 µg/g (n = 67) | ≥ 1 µg/g (n = 68) | OR | 95% CI | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| Gender | |||||
| Male | 30 | 43 | 2.12 | 1.06 | 4.23 |
| Female | 37 | 25 | 1.00 | - | - |
| Total Seafood Consumption | |||||
| Once a day or more | 3 | 6 | 3.71 | 0.84 | 16.38 |
| Three times per week | 25 | 41 | 3.05 | 1.47 | 6.30 |
| Once per week or less | 39 | 21 | 1.00 | - | - |
| Fish Sources | |||||
| All or > 50% recreationally | 16 | 29 | 2.32 | 1.11 | 4.87 |
| Half or more commercially | 50 | 39 | 1.00 | - | - |
| Shellfish Sources | |||||
| All or > 50% recreationally | 7 | 17 | 2.98 | 1.14 | 7.75 |
| Half or more commercially | 60 | 49 | 1.00 | - | - |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Role of the Funding Source
IRB Approval Verification
References
- Harada, M. Minamata disease: Methylmercury poisoning in Japan caused by environmental pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef]
- Grandjean, P.; Weihe, P.; White, R.F. Cognitive deficit in 7-year-old children with prenatal exposure to methylmercury. Neurotoxicol. Teratol. 1997, 19, 417–428. [Google Scholar] [CrossRef]
- Davidson, P.W.; Myers, G.J.; Cox, C.; Shamlaye, C.F.; Marsh, D.O.; Tanner, M.A.; Berlin, M.; Sloane-Reeves, J.; Cernichiari, E.; Choisy, O.; et al. Longitudinal neurodevelopmental study of Seychellois children following in utero exposure to methylmercury from maternal fish ingestion: outcomes at 19 and 29 months. Neurotoxicology 1995, 16, 677–688. [Google Scholar]
- Daniels, J.L.; Longnecker, M.P.; Rowland, A.S.; Golding, J. Fish intake during pregnancy and early cognitive development of offspring. Epidemiology 2004, 4, 394–402. [Google Scholar]
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a US cohort. Environ. Health Perspect. 2005, 10, 1376–1380. [Google Scholar]
- National Research Council. Health Effects of Methylmercury. In Toxicology Effects of Methylmercury; National Academy Press: Washington, DC, USA, 2000; pp. 147–249. [Google Scholar]
- Karagas, M.R.; Choi, A.L.; Oken, E.; Horvat, M.; Schoeny, R.; Kamai, E.; Cowell, W.; Grandjean, P.; Korrick, S. Evidence on the human health effects of low-level methylmercury exposure. Environ. Health Perspect. 2012, 120, 799–806. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Sakamoto, M.; Nakai, K.; Oka, T.; Dakeishi, M.; Iwata, T.; Satoh, H.; Murata, K. Estimation of daily mercury intake from seafood in Japanese women: Akita cross-sectional study. Tohoku J. Exp. Med. 2003, 2, 67–73. [Google Scholar]
- Knobeloch, L.; Gliori, G.; Anderson, H. Assessment of methylmercury exposure in Wisconsin. Environ. Res. 2007, 2, 205–210. [Google Scholar] [CrossRef]
- Miranda, M.L.; Edwards, S.; Maxson, P.J. Mercury levels in an urban pregnant population in Durham County, North Carolina. Int. J. Environ. Res. Public Health 2011, 3, 698–712. [Google Scholar] [CrossRef]
- Lincoln, R.A.; Shine, J.P.; Chesney, E.J.; Vorhees, D.J.; Grandjean, P.; Senn, D.B. Fish consumption and mercury exposure among Louisiana recreational anglers. Environ. Health Perspect. 2011, 2, 245–251. [Google Scholar]
- Mahaffey, K.R.; Clickner, R.P.; Bodurow, C.C. Blood organic mercury and dietary mercury intake: National Health and Nutrition Examination Survey, 1999 and 2000. Environ. Health Perspect. 2004, 112, 562–570. [Google Scholar] [CrossRef]
- Degner, R.L.; Adams, C.M.; Moss, S.D.; Mack, S.K. Per Capita Fish and Shellfish Consumption in Florida; Florida Agricultural Market Research Center, Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2004. [Google Scholar]
- Knobeloch, L.; Anderson, H.; Imma, P.; Petersa, D.; Smith, A. Fish consumption, advisory awareness, and hair mercury levels among women of childbearing age. Environ. Res. 2005, 97, 220–227. [Google Scholar] [CrossRef]
- Warner, K. Mercury Levels in Hair of Coastal Alabama Anglers and Residents. Report Oceana: Washington, DC, 2007. Available online: http://oceana.org/sites/default/files/reports/Rodeo_Hair_Report_Final.pdf (accessed on 1 February 2014).
- Scudder, B.C.; Chaser, L.C.; Wentz, D.A.; Bauch, N.J.; Brigham, M.E.; Moran, W.; Krabbenhoft, D.P. Mercury in Fish, Bed Sediment, and Water from Streams across the United States, 1998–2005. U.S. Geological Survey, Scientific Investigations Report 2009–5109; U.S. Geological Survey: Middleton, WI, USA, 2009. [Google Scholar]
- Hsu-Kim, H.; Kucharzky, K.H.; Zhang, T.; Deshusses, M.A. Mechanisms regulating mercury bioavailability for methylating microorganisms in the aquatic environment: A critical review. Environ. Sci. Technol. 2013, 47, 2441–2456. [Google Scholar] [CrossRef]
- Stavros, H.C.; Bossart, G.D.; Hulsey, T.C.; Fair, P.A. Trace element concentrations in blood of free-ranging bottlenose dolphins (Tursiops truncatus): Influence of age, sex and location. Mar. Pollut. Bull. 2008, 56, 348–379. [Google Scholar] [CrossRef]
- Stavros, H.C.; Bossart, G.D.; Hulsey, T.C.; Fair, P.A. Trace element concentrations in skin of free-ranging bottlenose dolphins (Tursiops truncatus) from the southeast Atlantic coast. Sci. Total. Environ. 2007, 388, 300–315. [Google Scholar] [CrossRef]
- Schaefer, A.M.; Stavros, H.W.; Reif, J.S.; Fair, P.A.; Bossart, G.D. Effects of Mercury on Hepatic, Renal, Endocrine and Hematological Parameters in Atlantic Bottlenose Dolphins (Tursiops truncatus) along the Eastern Coast of Florida and South Carolina. Arch. Environ. Contam. 2011, 61. [Google Scholar] [CrossRef]
- Bryan, C.E.; Christopher, S.J.; Balmer, B.C.; Wells, R.S. Establishing baseline levels of trace elements in blood and skin of bottlenose dolphins in Sarasota Bay, Florida: Implications for non-invasive monitoring. Sci. Total. Environ. 2007, 388, 325–342. [Google Scholar] [CrossRef]
- Roditi-Elasar, M.; Kerem, D.; Hornung, H.; Kress, N.; Shohan-Frider, E.; Goffman, O.; Spanier, E. Heavy metal levels in bottlenose and striped dolphins off the Mediterranean coast of Israel. Mar. Pollut. Bull. 2003, 46, 491–521. [Google Scholar] [CrossRef]
- Bossart, G.D. Marine mammals as sentinel species for oceans and human health. Vet. Pathol. 2011, 48, 676–690. [Google Scholar] [CrossRef]
- Stavros, H.C.; Stolen, M.; Durden, W.N.; McFee, W.; Bossart, G.D.; Fair, P.A. Correlation and toxicological inference of trace elements in tissues from stranded and free-ranging bottlenose dolphins (Tursiops truncatus). Chemosphere 2011, 11, 1649–1661. [Google Scholar]
- Grandjean, P.; Jørgensen, P.J.; Weihe, P. Validity of Mercury Exposure Biomarkers. In Biomarkers of Environmentally Associated Disease; Wilson, S.H., Suk, W.A., Eds.; CRC Press/Lewis Publishers: Boca Raton, FL, USA, 2002; pp. 235–247. [Google Scholar]
- Zareba, G.; Cernichiari, E.; Goldsmith, L.A.; Clarkson, T.W. Validity of methlymercury hair analysis; Mercury monitoring in human scalp/nude mouse model. J. Appl. Toxicol. 2008, 28, 535–542. [Google Scholar] [CrossRef]
- Grandjean, P.; Budtz-Jorgensen, E. Total Imprecision of Exposure biomarkers: Implications for calculating exposure limits. Am. J. Ind. Med. 2010, 10, 712–719. [Google Scholar]
- Rice, D.C.; Schoeny, R.; Mahaffey, K. Methods and rationale for derivation of a reference dose for methylmercury by the U.S. EPA. Risk Anal. 2003, 23, 107–115. [Google Scholar] [CrossRef]
- McDowell, M.A.; Dillon, C.F.; Osterloh, J.; Bolger, P.M.; Pellizzari, E.; Fernando, R.; Montes de Oca, R.; Schober, S.E.; Sinks, T.; Jones, R.L.; et al. Hair mercury levels in U.S. children and women of childbearing age: Reference range data from NHANES 1999–2000. Environ. Health Perspect. 2004, 112, 1165–1171. [Google Scholar] [CrossRef]
- Kosatsky, T.; Przybysz, R.; Armstrong, B. Mercury exposure in Montrealers who eat St. Lawrence river sportfish. Environ. Res. 2000, 84, 36–43. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Fish Consumption Distribution Data 2009. Mercury Concentration and Market Share Data. In Draft Risk and Benefit Report. Report of Quantitative Risk and Benefit Assessment of Consumption of Commercial Fish Focusing on Fetal Neurodevelopmental Effects and on Coronary Heart Diseases and Stroke in the General Population; Center for Food Safety and Applied Nutrition, U.S. Food and Drug Administration: Silver Spring, MD, 2009; pp. 81–82, 126–137. [Google Scholar]
- Sunderland, E.M. Mercury exposure from domestic and imported estuarine and marine fish in the United States seafood market. Environ. Health Perspect. 2007, 115, 235–242. [Google Scholar] [CrossRef]
- Shimomura, S.; Kimura, A.; Nakagawa, H.; Takao, M. Mercury levels in human hair and sex factors. Environ. Res. 1980, 22, 22–30. [Google Scholar] [CrossRef]
- Holloman, E.L.; Newman, M.C. A community-based assessment of seafood consumption along the lower James river, Virginia, USA: Potential sources of dietary mercury exposure. Environ. Res. 2010, 110, 213–219. [Google Scholar] [CrossRef]
- Karouna-Renier, N.K.; Rao, K.R.; Lanza, J.J.; Rivers, S.D.; Wilson, P.A.; Hodges, D.K.; Levine, K.E.; Ross, G.T. Mercury levels and fish consumption practices in women of child-bearing age in the Florida panhandle. Environ. Res. 2008, 108, 320–326. [Google Scholar] [CrossRef]
- Canuel, R.; Grosbois, S.B.; Atikesse, L.; Lucotte, M.; Arp, P.; Ritchi, C. New evidence of variations of human body burden of methlymercury from fish consumption. Environ. Health Perspect. 2006, 114, 302–306. [Google Scholar] [CrossRef]
- McKelvey, W.; Gwynn, R.C.; Jeffery, N.; Kass, D.; Thorpe, L.E.; Garg, R.K. A biomonitoring study of lead, cadmium and mercury in the blood of New York City adults. Environ. Health Perspect. 2007, 115, 1435–1441. [Google Scholar]
- United States Census Bureau. 2010 Census Report; GPO: Washington, DC, USA, 2010.
- Barros, N.B.; Odell, D.K. Food Habits of Bottlenose Dolphins in the Southeastern United States. In The Bottlenose Dolphin; Leatherwood, S., Reeves, R.R., Eds.; Academic Press, Inc.: San Diego, CA, USA, 1990; pp. 309–328. [Google Scholar]
- Mahaffey, K.R.; Sunderland, E.M.; Chan, H.M.; Choi, A.L.; Grandjean, P.; Mariën, K.; Oken, E.; Sakamoto, M.; Schoeny, R.; Weihe, P.; et al. Balancing the benefits of n-3 polyunsaturated fatty acids and the risks of methylmercury exposure from fish consumption. Nutr. Rev. 2011, 69, 493–508. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schaefer, A.M.; Jensen, E.L.; Bossart, G.D.; Reif, J.S. Hair Mercury Concentrations and Fish Consumption Patterns in Florida Residents. Int. J. Environ. Res. Public Health 2014, 11, 6709-6726. https://doi.org/10.3390/ijerph110706709
Schaefer AM, Jensen EL, Bossart GD, Reif JS. Hair Mercury Concentrations and Fish Consumption Patterns in Florida Residents. International Journal of Environmental Research and Public Health. 2014; 11(7):6709-6726. https://doi.org/10.3390/ijerph110706709
Chicago/Turabian StyleSchaefer, Adam M., Emily L. Jensen, Gregory D. Bossart, and John S. Reif. 2014. "Hair Mercury Concentrations and Fish Consumption Patterns in Florida Residents" International Journal of Environmental Research and Public Health 11, no. 7: 6709-6726. https://doi.org/10.3390/ijerph110706709
APA StyleSchaefer, A. M., Jensen, E. L., Bossart, G. D., & Reif, J. S. (2014). Hair Mercury Concentrations and Fish Consumption Patterns in Florida Residents. International Journal of Environmental Research and Public Health, 11(7), 6709-6726. https://doi.org/10.3390/ijerph110706709




