Sero-Prevalence and Risk Factors for Leptospirosis in Abattoir Workers in New Zealand
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design, Data Collection and Serological Testing
2.2. Case Definitions
2.3. Sample Size and Power Calculation
2.4. Data Analysis
2.4.1. Categories of Work Position and Personal Protective Equipment
| Species | Variable | Category | Adjusted OR | 95% CI | p-value |
|---|---|---|---|---|---|
| Sheep | Work position | Boning, chillers, office | Ref. | ||
| Offal, pet food | 6.5 | 1.4–29.8 | 0.017 | ||
| Gut removal, pulling kidneys | 8.2 | 2.1–32.7 | 0.003 | ||
| Yards, stunning, pelting | 10.4 | 2.8–38.8 | <0.001 | ||
| Gender | Female | Ref. | |||
| Male | 3.1 | 0.8–11.7 | 0.089* | ||
| Years worked at meat plant | (Continuous) | 1.1 | 1.0–1.1 | 0.011 | |
| Meat plant | Sheep 1 | Ref. | |||
| Sheep 2 | 4.5 | 1.2–16.3 | 0.022 | ||
| Sheep 3 | 6.3 | 1.8–22.4 | 0.004 | ||
| Sheep 4 | 2.1 | 0.7–6.3 | 0.201* | ||
| Deer | Work position | Boning, Chillers, Office | Ref. | ||
| Offal, pet food, gut removal, pulling kidneys, yards, stunning, pelting | 12.7 | 1.3–120.6 | 0.027 | ||
| Wear facemask, or safety glasses | Never or sometimes | Ref. | |||
| Often or always | 4.3 | 0.8–22.8 | 0.093* | ||
| Beef | Work position | Boning, chillers, office | Ref | ||
| Maintenance | 2.0 | 0.3–23.4 | 0.59* | ||
| Offal, pet food | 3.1 | 0.5–20.6 | 0.25* | ||
| Yards, stunning, pelting, gut, kidney removal & meat inspection | 2.2 | 0.5–10.8 | 0.32* | ||
| Age (years) | Continuous | 1.1 | 1.0–1.2 | 0.02 |
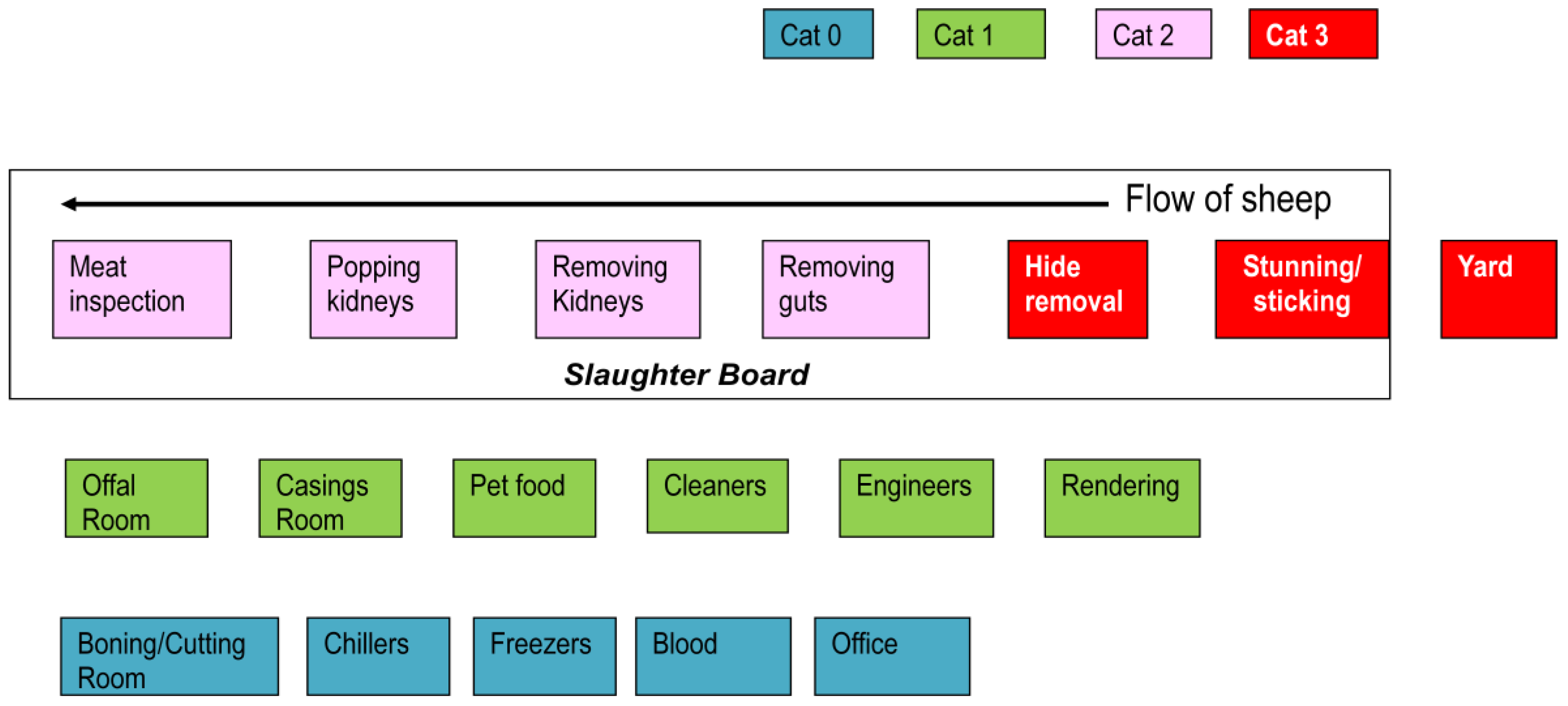
2.4.2. Multivariable Analysis
3. Results
3.1. Participants, Slaughter Plants and Study Population
| Abattoir | Total Number of Workers | Study Recruits (%) | Species Processed | Number of Animals Processed per Year | Regions of Animal Origin |
|---|---|---|---|---|---|
| Sheep 1 | 889 | 12 | Lamb, mutton, bobby calves | 1,797,809 | Hawke’s Bay, Waikato, Wairarapa, Bay of Plenty, Northland |
| Sheep 2 | 378 | 26 | Lamb, mutton, goats | 600,469 | Gisborne, Hawke’s Bay, Waikato, Bay of Plenty |
| Sheep 3 | 300 | 11 | Lamb | 780,000 | Central Hawke’s Bay, East Coast, Wairarapa, Manawatu |
| Sheep 4 | 180 | 51 | Lamb, mutton, bobby calves, goats | 488,546 | Wanganui, Manawatu, Taranaki, other |
| Deer 1 | 41 | 51 | Venison | 24,222 | Canterbury |
| Deer 2 | 59 | 61 | Farmed & feral a venison | 41,055 | South Waitaki River to Rakaia, North Canterbury |
| Beef 1 | 486 | 15 | Beef cattle, dairy cows | 93,837 | East Coast, West Coast, Waikato, Bay of Plenty, Northland |
| Beef 2 | 328 | 34 | Beef cattle | 159,347 | Taranaki, Waikato, Manawatu, Hawke’s Bay |
3.2. Sero-prevalence and Antibody Titres
| Abattoir | Participants | Prevalence (%) | |||||
|---|---|---|---|---|---|---|---|
| Pom (%) | 95% CI (%) | Har (%) | 95% CI (%) | Either (%) | 95% CI (%) | ||
| Sheep1 | 104 | 5 | 2–11 | 10 | 5–17 | 12 | 7–19 |
| Sheep2 | 97 | 8 | 4–16 | 4 | 2–10 | 11 | 6–19 |
| Sheep3 | 32 | 16 | 7–32 | 28 | 15–46 | 31 | 18–49 |
| Sheep4 | 92 | 5 | 2–12 | 7 | 3–14 | 10 | 5–18 |
| Deer1 | 21 | 5 | 1–27 | 19 | 7–41 | 19 | 7–41 |
| Deer2 | 36 | 6 | 11–20 | 11 | 4–26 | 17 | 8–32 |
| Beef1 | 73 | 3 | 1–10 | 4 | 1–12 | 5 | 2–14 |
| Beef2 | 112 | 1 | 0–6 | 5 | 2–11 | 5 | 2–11 |
| Total | 567 | 5 | 3–7 | 8 | 6–10 | 11 | 8–14 |
3.3. Disease
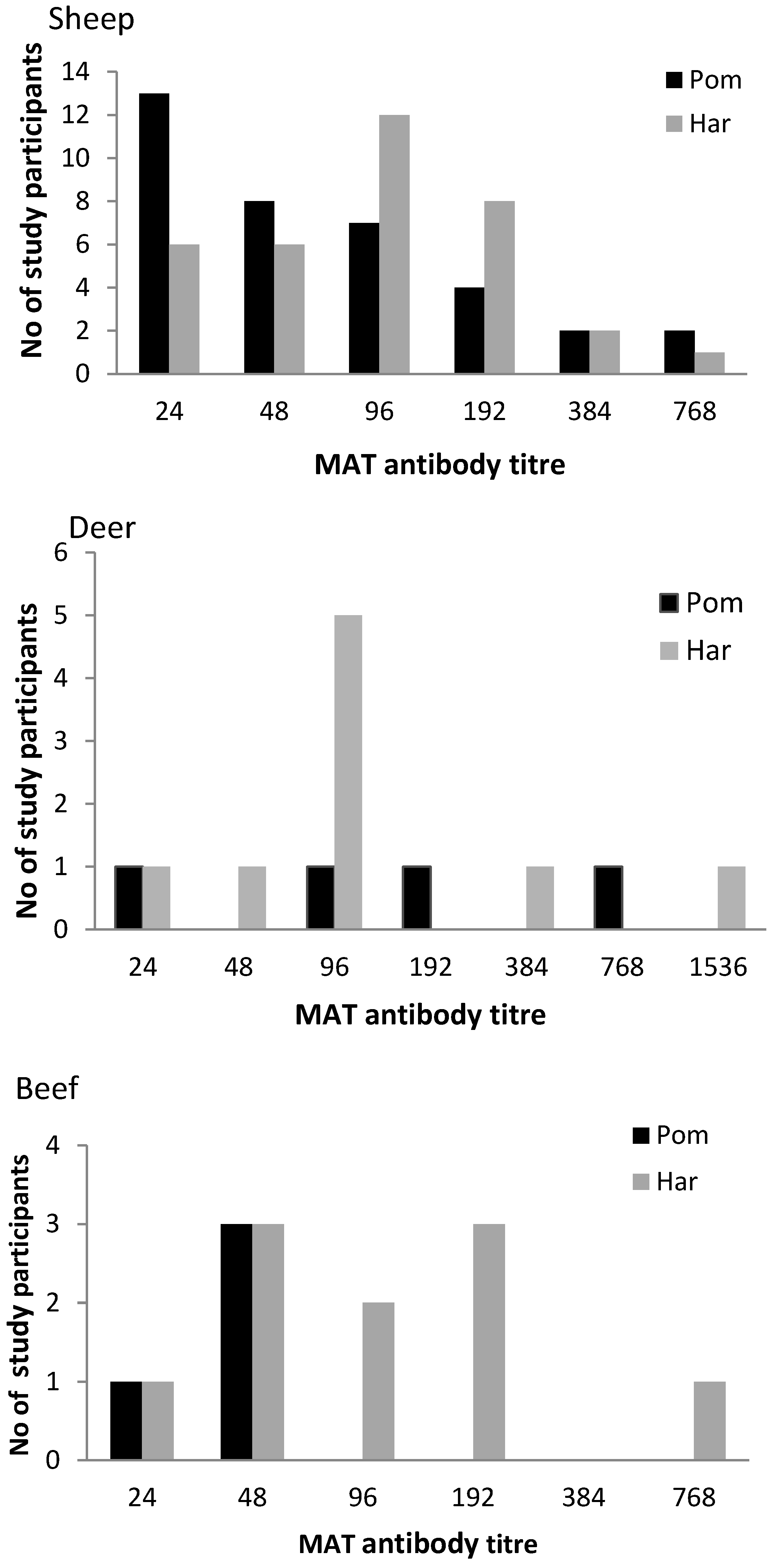
3.4. The Use of Personal Protective Equipment
3.5. Risk Factors for Sero-prevalence in Sheep Plants
3.6. Risk Factors for Sero-prevalence in Deer Abattoirs
3.7. Risk Factors for Sero-prevalence in Beef Abattoirs
4. Discussion and Conclusions
4.1. Sero-prevalence
4.2. Probable Leptospirosis
4.3. Risk Factors for Sero-positivity
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ayanegui-Alcerreca, M.A.; Wilson, P.R.; Mackintosh, C.G.; Collins-Emerson, J.M.; Heuer, C.; Midwinter, A.C.; Castillo-Alcala, F. Leptospirosis in farmed deer in new zealand: A review. New Zealand Veterinary Journal 2007, 55, 102–108. [Google Scholar] [CrossRef]
- Marshall, R.B.; Manktelow, B.W. Fifty years of leptospirosis research in new zealand: A perspective. New Zealand Veterinary Journal 2002, 50, 61–63. [Google Scholar] [CrossRef]
- Dreyfus, A.; Verdugo, C.; Benschop, J.; Collins-Emerson, J.; Wilson, P.; Heuer, C. Leptospirosis sero-prevalence and associated economic loss in new zealand livestock. In Proceedings of the food safety, animal welfare & biosecurity, epidemiology & animal health management, and industry branches of the nzva, VetLearn Foundation, Palmerston North, New zealand, 2011; pp. 3.12.11–13.12.10.
- Thornley, C.N.; Baker, M.G.; Weinstein, P.; Maas, E.W. Changing epidemiology of human leptospirosis in new zealand. Epidemiol. Infect. 2002, 128, 29–36. [Google Scholar]
- Ayanegui-Alcerreca, M.; Wilson, P.R.; Mackintosh, C.G.; Collins-Emerson, J.M.; Heuer, C.; Midwinter, A.C.; Castillo-Alcala, F. Regional seroprevalence of leptospirosis on deer farms in new zealand. New Zealand Veterinary Journal 2010, 58, 184–189. [Google Scholar] [CrossRef]
- Dorjee, S.; Heuer, C.; Jackson, R.; West, D.M.; Collins-Emerson, J.M.; Midwinter, A.C.; Ridler, A.L. Prevalence of pathogenic leptospira spp. In sheep in a sheep-only abattoir in new zealand. New Zealand Veterinary Journal 2008, 56, 164–170. [Google Scholar] [CrossRef]
- Dorjee, S.; Heuer, C.; Jackson, R.; West, D.M.; Collins-Emerson, J.M.; Midwinter, A.C.; Ridler, A.L. Assessment of occupational exposure to leptospirosis in a sheep-only abattoir. Epidemiol. Infect. 2011, 139, 797–806. [Google Scholar] [CrossRef]
- Victoriano, A.F.B.; Smythe, L.D.; Gloriani-Barzaga, N.; Cavinta, L.L.; Kasai, T.; Limpakarnjanarat, K.; Ong, B.L.; Gongal, G.; Hall, J.; Coulombe, C.A.; et al. Leptospirosis in the asia pacific region. Bmc Infectious Diseases 2009, 9. [Google Scholar]
- Institute of Environmental Science and Research (ESR) Annual surveillance summary: Notifiable and other diseases in new zealand: Annual report. Available online: https://surv.esr.cri.nz/surveillance/annual_surveillance.php.
- Dreyfus, A. Massey university human ethics application: Slaughter carcasses as a source for human infection with leptospira serotypes hardjo, pomona and ballum at abattoirs in new zealand. Southern a, application 09/08.; Massey University: Palmerston North, New Zealand, unpublished; 2009; p. 17. [Google Scholar]
- Faine, S.; Adler, B.; Bolin, C.; Perolat, P. Leptospira and leptospirosisMediSci, 2nd ed.; Melbourne, Australia, 1999; p. 272. [Google Scholar]
- McDermott, J.J.; Schukken, Y.H.; Shoukri, M.M. Study design and analytic methods for data collected from clusters of animals. Prev. Vet. Med. 1994, 18, 175–191. [Google Scholar] [CrossRef]
- Hosmer, D.W.; Lemeshow, S. Assessing the fit of the model. In Applied logistic regression, Second ed.; John Wiley & Sons Inc: New York, 2000; pp. 143–167. [Google Scholar]
- Hathaway, S.C. Leptospirosis in new zealand: An ecological view. New Zealand Veterinary Journal 1981, 29, 109–112. [Google Scholar]
- Bolin, C.A.; Alt, D.P. Use of a monovalent leptospiral vaccine to prevent venal colonization and urinary shedding in cattle exposed to leptospira borgpetersenii serovar hardjo. American Journal of Veterinary Research 2001, 62, 995–1000. [Google Scholar] [CrossRef]
- Marshall, R.; Schollum, L.M.; Dymock, B.L. Prevention of leptospira interrogans serovar pomona infection in cattle. New Zealand Veterinary Journal 1982, 30, 177–179. [Google Scholar] [CrossRef]
- Dreyfus, A.; Benschop, J.; Collins-Emerson, J.; Wilson, P.; Heuer, C. Leptospirosis sero-prevalence in meat workers - interim study report 1 EpiCentre. Massey University: Palmerston North, New Zealand, 2010; 1–10. [Google Scholar]
- Levett, P.N. Leptospirosis. Clinical Microbiology Reviews 2001, 14, 296–326. [Google Scholar] [CrossRef]
- Smythe, L.D.; Wuthiekanun, V.; Chierakul, W.; Suputtamongkol, Y.; Tiengrim, S.; Dohnt, M.F.; Symonds, M.L.; Slack, A.T.; Apiwattanaporn, A.; Chueasuwanchai, S.; et al. The microscopic agglutination test (mat) is an unreliable predictor of infecting leptospira serovar in thailand. Am. J. Trop. Med. Hyg. 2009, 81, 695–697. [Google Scholar] [CrossRef]
- Subharat, S.; Wilson, P.R.; Heuer, C.; Collins-Emerson, J.M. Evaluation of a syto9 real-time polymerase chain reaction assay to detect and identify pathogenic leptospira species in kidney tissue and urine of new zealand farmed deer. Journal of Veterinary Diagnostic Investigation 2011, 23, 743–752. [Google Scholar] [CrossRef]
- Subharat, S.; Wilson, P.R.; Heuer, C.; Collins-Emerson, J.M. Growth response and shedding of leptospira spp. In urine following vaccination for leptospirosis in young farmed deer. New Zealand Veterinary Journal 2012, 60, 14–20. [Google Scholar] [CrossRef]
- Shivakumar, S.; Krishnakumar, B. Diagnosis of leptospirosis - role of mat. Journal of the Association of Physicians of India 2006, 54. [Google Scholar]
- McBride, A.J.A.; Santos, B.L.; Queiroz, A.; Santos, A.C.; Hartskeerl, R.A.; Reis, M.G.; Ko, A.I. Evaluation of four whole-cell leptospira-based serological tests for diagnosis of urban leptospirosis. Clinical and Vaccine Immunology 2007, 14, 1245–1248. [Google Scholar] [CrossRef]
- Blackmore, D.K.; Schollum, L. The occupational hazards of leptospirosis in the meat industry. New Zealand Medical Journal 1982, 95, 494–497. [Google Scholar]
- Blackmore, D.K.; Bell, L.; Schollum, L. Leptospirosis in meat inspectors - preliminary-results of a serological survey. New Zealand Medical Journal 1979, 90, 415–418. [Google Scholar]
- Benschop, J.; Heuer, C.; Jaros, P.; Collins-Emerson, J.; Midwinter, A.; Wilson, P. Sero-prevalence of leptospirosis in workers at a new zealand slaughterhouse. The New Zealand Medical Journal 2009, 122, 39–47. [Google Scholar]
- Dorjee, S.; Ridler, A.; Collins, J.M.; Midwinter, A.; West, D.M.; Heuer, C.; Jackson, R. Leptospirosis in sheep in new zealand. In Proceedings of the society of sheep and beef cattle veterinarians of the new zealand veterinary association, Palmerston North, New Zealand, 2005; VetLearn Foundation; Vol. Annual Seminar 2005, Volume, pp. 19–31.
- Wilson, P.; McGhie, J.; Marshal, l.R.B.; Audige, L.J.M.; Collins-Emerson, J.; Quankai, W.; Alley, M.R. Observations of leptospirosis in farmed deer. New Zealand Veterinary Journal 1998, 46, 131–139. [Google Scholar] [CrossRef]
- Dreyfus, A. Leptospirosis in humans and pastoral livestock in new zealand. PhD, Massey University, Palmerston North, New Zealand, 2013. [Google Scholar]
- Blackmore, D.K.; Schollum, L.M.; Moriarty, K.M. The magnitude and duration of titers of leptospiral agglutinins in human-sera. New Zealand Medical Journal 1984, 97, 83–86. [Google Scholar]
- Heuer, C.; Dreyfus, A.; Wilson, P.R.; Benschop, J.; Subharat, S.; Ayanegui-Alcerreca, A.M.; Fang, F.; Collins-Emerson, J.M.; Midwinter, A.C. Epidemiology and control of leptospirosis in new zealand. In Conference proceedings of society for veterinary epidemiology and preventive medicine, 24-26 march, 2010; Alban, L., Kelly, L.A., Eds.; Society for Veterinary Epidemiology and Preventive Medicine: Nantes, France, 2010; pp. 174–185. [Google Scholar]
- Marshall, R.B. Vaccination of dairy cattle against leptospirosis as a means of preventing human infections. Surveillance 1996, 23, 27–28. [Google Scholar]
- New Zealand Pork Industry Board Animal status declaration (asd) form http://www.nzpork.co.nz/Portals/NZPib/Documents/Publications/NZPork%20ASDP%20Download%20FORM.pdf.
- Wilson, P.; Glossop, J.C.; van der Kroef, J.W.; Heuer, C.; Stringer, L. Disease and deer farm productivity and profitability, Proceedings of the Deer Branch of the New Zealand Veterinary Association, New Zealand, 2008; The Deer Branch New Zealand Veterinary Association: New Zealand; pp. 22–29.
- Keenan, B. Leptospirosis: Reducing the impact on new zealand workplaces. In Leptospirosis: Reducing the impact on new zealand workplaces; Department of Labour: Wellington, New Zealand, 2007; p. 55. [Google Scholar]
- Allen, J.D.; Meney, C.L.; Wilks, C.R. Evaluation of a hardjo-pomona vaccine to prevent leptospiruria in cattle exposed to a natural challenge with leptospira interrogans serovar hardjo. Aust. Vet. J. 1982, 58, 93–96. [Google Scholar] [CrossRef]
- Mackintosh, C.; Marshall, R.B.; Broughton, E.S. The use of a hardjo-pomona vaccine to prevent leptospiruria in cattle exposed to natural challenge with leptospira interrogans serovar hardjo. New Zealand Veterinary Journal 1980, 28, 174–177. [Google Scholar] [CrossRef]
- Marshall, R.; Broughton, E.S.; Hathaway, S.C. Protection of sheep by vaccination against artificial challenge with leptospira interrogans serovar hardjo. New Zealand Veterinary Journal 1979, 27, 195–196. [Google Scholar] [CrossRef]
- Subharat, S.; Wilson, P.R.; Heuer, C.; Collins-Emerson, J.M. Vaccination for leptospirosis improved the weaning percentage of 2-year-old farmed red deer hinds in new zealand. New Zealand Veterinary Journal 2011, 59, 191–196. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dreyfus, A.; Benschop, J.; Collins-Emerson, J.; Wilson, P.; Baker, M.G.; Heuer, C. Sero-Prevalence and Risk Factors for Leptospirosis in Abattoir Workers in New Zealand. Int. J. Environ. Res. Public Health 2014, 11, 1756-1775. https://doi.org/10.3390/ijerph110201756
Dreyfus A, Benschop J, Collins-Emerson J, Wilson P, Baker MG, Heuer C. Sero-Prevalence and Risk Factors for Leptospirosis in Abattoir Workers in New Zealand. International Journal of Environmental Research and Public Health. 2014; 11(2):1756-1775. https://doi.org/10.3390/ijerph110201756
Chicago/Turabian StyleDreyfus, Anou, Jackie Benschop, Julie Collins-Emerson, Peter Wilson, Michael G. Baker, and Cord Heuer. 2014. "Sero-Prevalence and Risk Factors for Leptospirosis in Abattoir Workers in New Zealand" International Journal of Environmental Research and Public Health 11, no. 2: 1756-1775. https://doi.org/10.3390/ijerph110201756
APA StyleDreyfus, A., Benschop, J., Collins-Emerson, J., Wilson, P., Baker, M. G., & Heuer, C. (2014). Sero-Prevalence and Risk Factors for Leptospirosis in Abattoir Workers in New Zealand. International Journal of Environmental Research and Public Health, 11(2), 1756-1775. https://doi.org/10.3390/ijerph110201756






