Longitudinal Trajectories of Cholesterol from Midlife through Late Life according to Apolipoprotein E Allele Status
Abstract
:1. Introduction
1.1. Age Related Changes to Cholesterol
1.2. Apolipoprotein E and Cholesterol
2. Methods
2.1. Framingham Heart Study: Original Cohort
2.2. Measures of Serum Cholesterol
2.3. Statistical Analysis
2.4. Covariates
3. Results
3.1. Description of Framingham Heart Study: Original Cohort
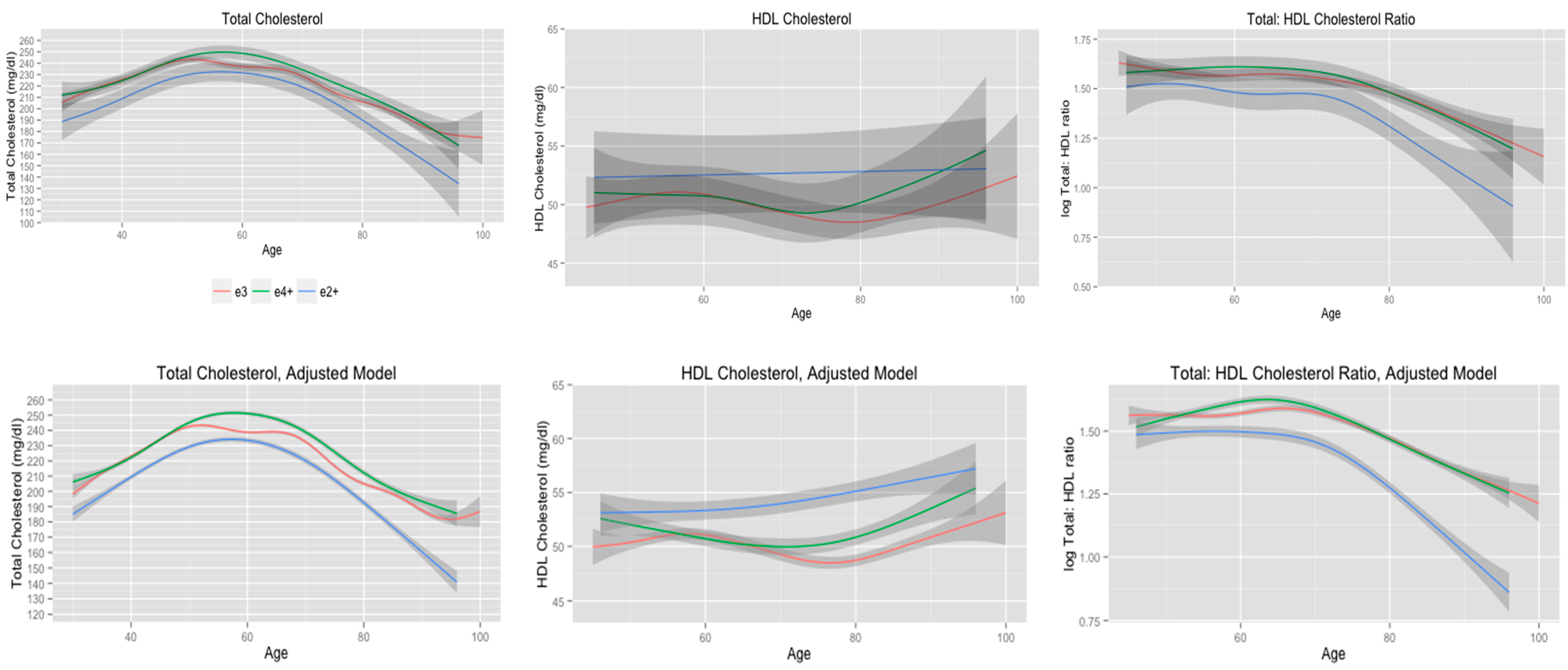
3.2. Total Cholesterol Stratified by APOE Allele Status
| Measure | Measure Collected | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Weight | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Height | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Diastolic BP | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Systolic BP | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Smoking | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | |||
| Diet | * | * | * | * | * | * | * | * | * | * | * | * | |||||||||||||||
| Estrogen use | * | * | * | * | * | * | * | * | * | * | * | ||||||||||||||||
| Menopause cause | * | * | * | * | * | * | * | * | * | * | * | * | * | * | |||||||||||||
| Menopause | * | * | * | * | * | * | * | * | * | * | * | * | * | * | |||||||||||||
| Cholesterol medications | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | ||||||
| Sex | * | ||||||||||||||||||||||||||
| Education | * | ||||||||||||||||||||||||||
| Blood sugar | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | |||||
| HDL cholesterol | * | * | * | * | * | * | * | * | * | * | |||||||||||||||||
| Total cholesterol | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | * | |||||||
| Clinical Exam | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 |
| Years | 1948 | 1950 | 1952 | 1954 | 1956 | 1968 | 1960 | 1962 | 1964 | 1966 | 1968 | 1971 | 1972 | 1975 | 1977 | 1979 | 1981 | 1983 | 1985 | 1986 | 1988 | 1990 | 1992 | 1995 | 1997 | 1999 | 2001 |
| Characteristic | Value |
|---|---|
| Avg. age, midlife (range, SD) | 38.3 (30.0–55.0, 5.5) |
| Gender, n (%) | |
| Men Women | 242 (40.6) 354 (59.4) |
| Educational attainment | |
| ≤High school >High school | 387 (66.2) 198 (33.8) |
| Smoking midlife, n (%), n = 577 | |
| Non-smoker Former Current | 282 (48.9) 11 (1.9) 284 (49.2) |
| Smoking, late life, n (%), n = 585 | |
| Non-smoker Former Current | 231 (39.5) 201 (34.3) 153 (26.1) |
| Use of cholesterol-lowering medications, n (%) | |
| No Yes | 487 (81.7) 109 (18.3) |
| * Avg. age cholesterol medication use (SD, range) | 69.7 (45.0–88.0, 11.4) |
| Avg. midlife blood sugar, mg/dL (SD, range), n = 590 | 79.4 (13.5, 51.0–173.0) |
| Avg. late life blood sugar, mg/dL (SD, range), n = 584 | 93.3 (24.2, 52.0–326.0) |
| Avg. midlife systolic blood pressure, mmHg (SD, range) | 128.4 (16.0, 90.0–195.0) |
| Avg. late life systolic blood pressure, mmHg (SD, range), n = 588 | 135.9 (19.5, 92.0–212) |
| Avg. midlife diastolic blood pressure, mmHg (SD, range) | 81.3 (10.4, 50.0–125.0) |
| Avg. late life diastolic blood pressure, mmHg (SD, range), n = 588 | 76.6 (9.8, 50.0–104.0) |
| Avg. midlife body mass index, Kg/M2 (SD, range) | 25.1 (3.8, 15.4–41.2) |
| Avg. late life body mass index, Kg/M2 (SD, range), n = 588 | 26.7 (4.3, 16.7–43.7 |
| ** Supplemental estrogen, n (%) | |
| No Yes | 315 (89.0) 39 (11.0) |
| Dietary restriction, n (%) | |
| No Yes | 345 (57.9) 251 (42.1) |
| APOE allele status, n (%) | |
| e2+ e3 e4+ | 61 (10.2) 406 (68.1) 129 (21.6) |
| Avg. number of cholesterol measures (range, SD) | |
| Total cholesterol HDL cholesterol | 14.4 (4.0–19.0, 2.5) 5.1 (2.0–10.0, 1.8) |
| Model | Total Cholesterol | HDL Cholesterol | Total: HDL Cholesterol | |||
|---|---|---|---|---|---|---|
| EDF | Adjusted R2 | EDF | Adjusted R2 | EDF | Adjusted R2 | |
| Unadjusted model | 0.12 | 0.01 | 0.06 | |||
| e2+ | 4.9 | 1.0 | 3.8 | |||
| e3 | 7.7 | 3.6 | 4.4 | |||
| e4+ | 5.7 | 2.8 | 3.3 | |||
| Adjusted model | 0.22 | 0.19 | 0.20 | |||
| e2+ | 4.9 | 1.0 | 3.7 | |||
| e3 | 7.5 | 3.7 | 4.7 | |||
| e4+ | 5.6 | 2.9 | 3.3 | |||
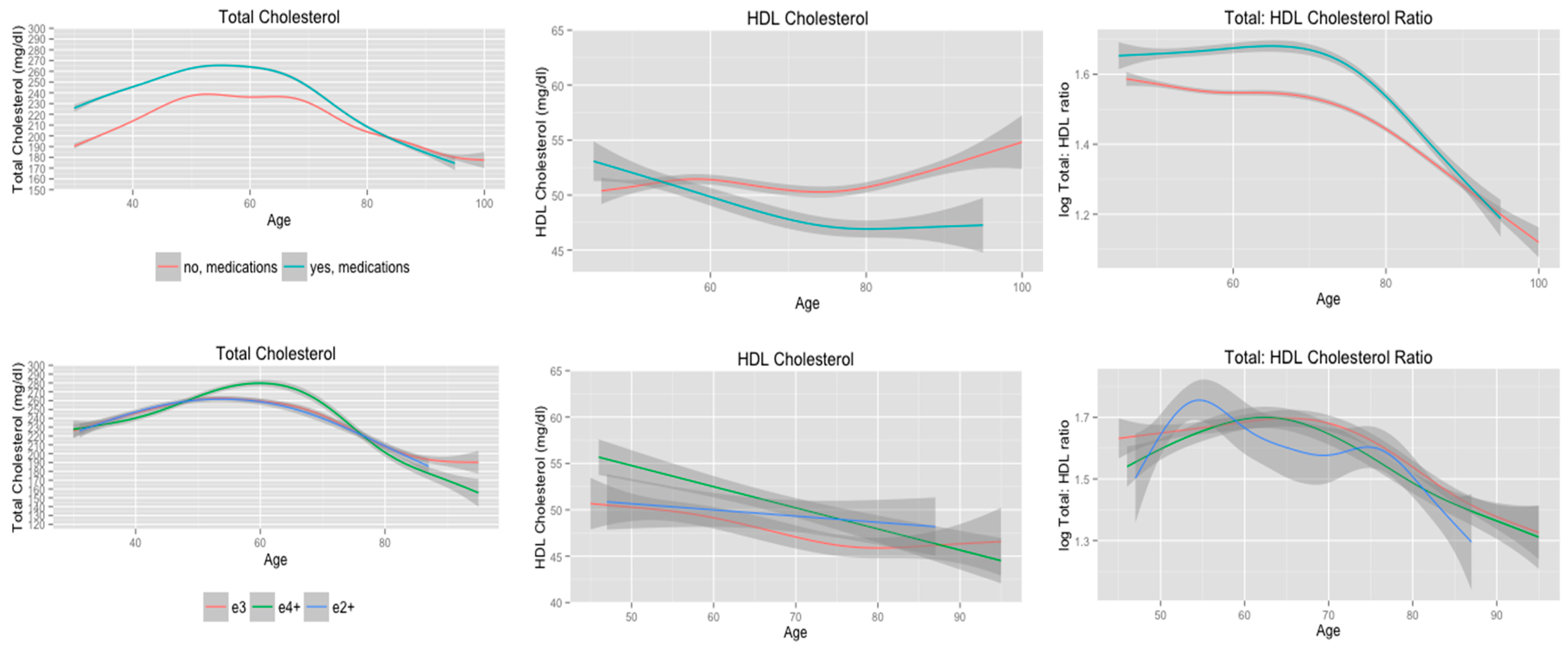
3.3. Cholesterol Trajectories according to Use of Cholesterol-Lowering Medications
| Model | Total Cholesterol | HDL Cholesterol | Total: HDL Cholesterol | |||
|---|---|---|---|---|---|---|
| EDF | Adjusted R2 | EDF | Adjusted R2 | EDF | Adjusted R2 | |
| Use of cholesterol-lowering medications | 0.22 | 0.19 | 0.16 | |||
| No | 7.2 | 3.7 | 4.0 | |||
| Yes | 5.4 | 2.5 | 3.7 | |||
| Adjusted model | 0.26 | 0.28 | 0.20 | |||
| e2+ | 3.4 | 1.0 | 3.8 | |||
| e3 | 4.6 | 1.5 | 3.3 | |||
| e4+ | 4.6 | 1.0 | 2.6 | |||
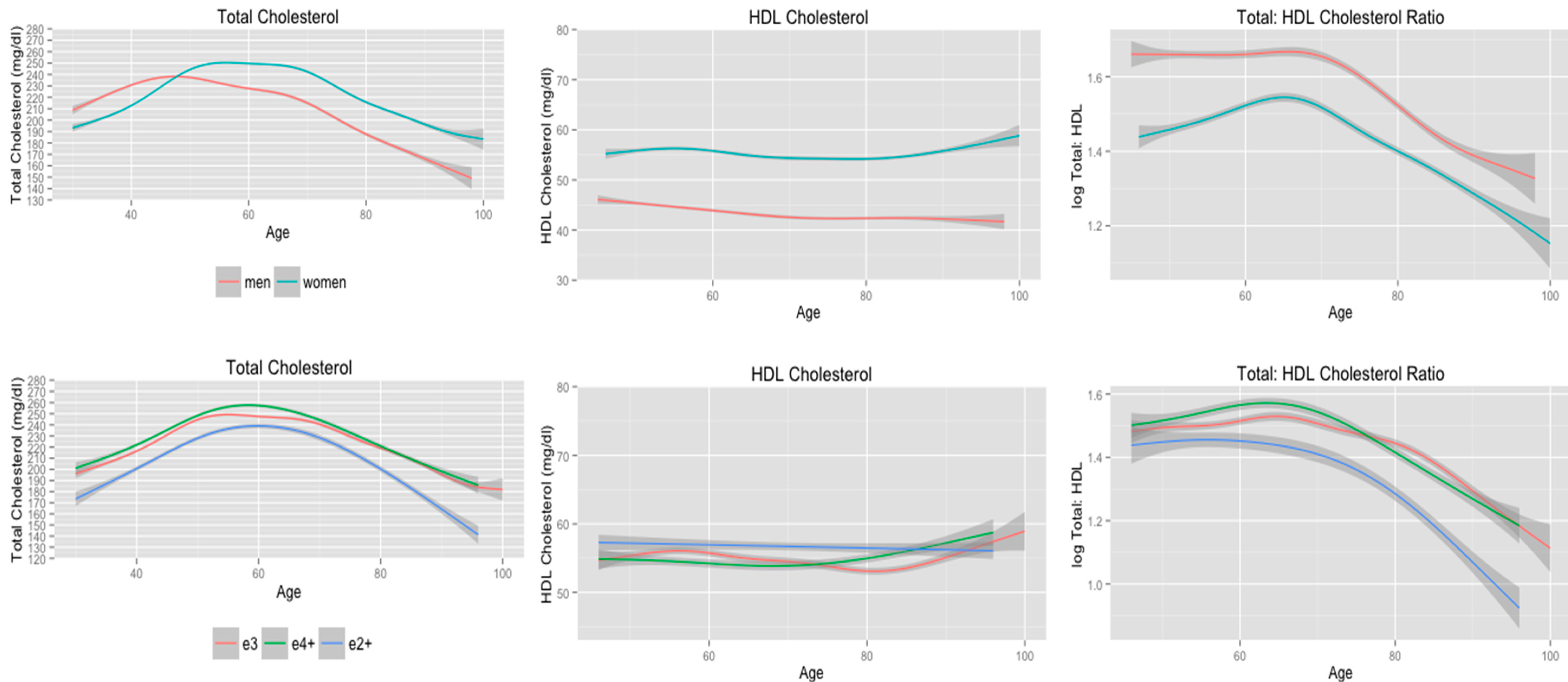
3.4. Cholesterol Trajectories Stratified by Gender, and APOE Allele Status Adjusting for Menopause
| Model | Total Cholesterol | HDL Cholesterol | Total: HDL Cholesterol | |||
|---|---|---|---|---|---|---|
| EDF | Adjusted R2 | EDF | Adjusted R2 | EDF | Adjusted R2 | |
| Gender | 0.25 | 0.19 | 0.20 | |||
| Male | 6.2 | 3.0 | 4.5 | |||
| Female | 7.3 | 4.2 | 4.3 | |||
| Adjusted | 0.25 | 0.08 | 0.15 | |||
| e2+ | 5.0 | 1.0 | 2.9 | |||
| e3 | 7.1 | 4.2 | 4.6 | |||
| e4+ | 5.5 | 2.6 | 3.1 | |||
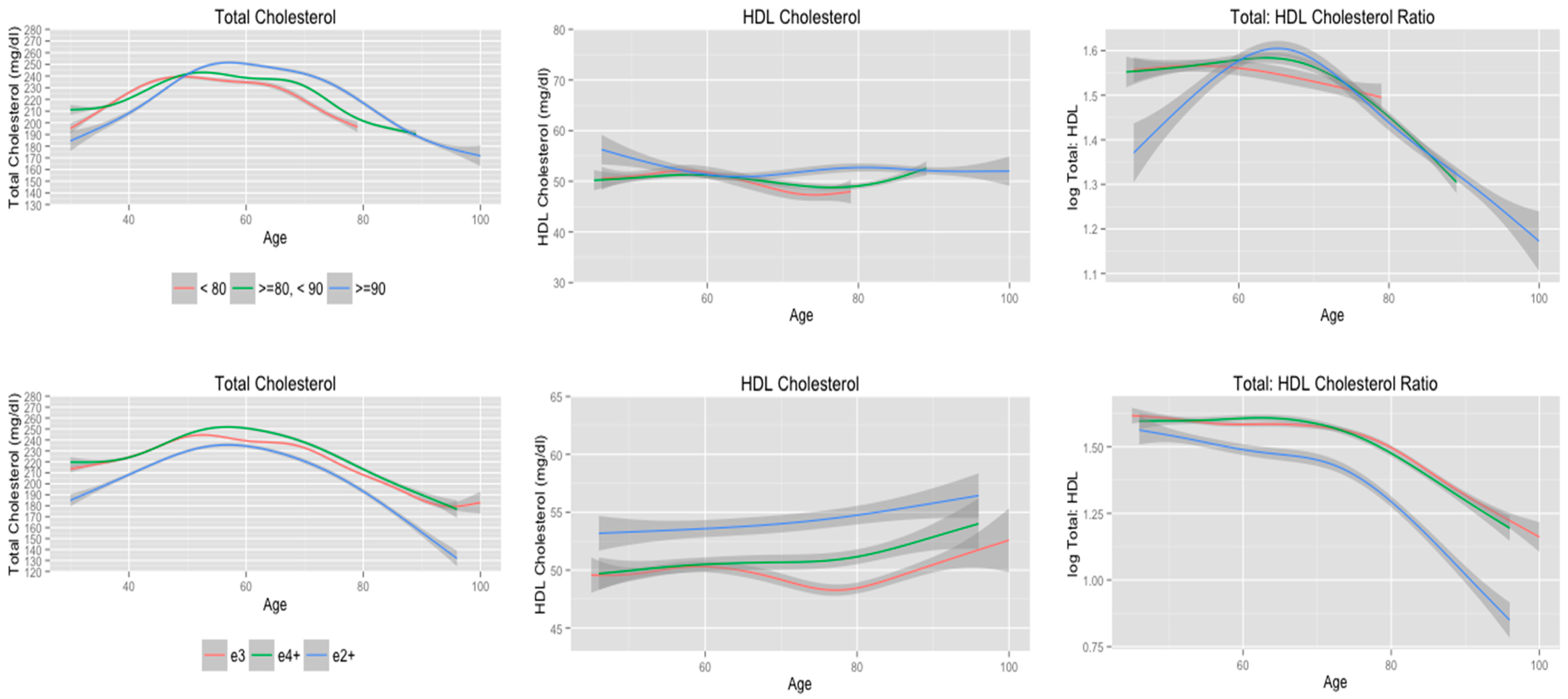
3.5. Cholesterol Trajectories Stratified according to Longevity
| Model | Total Cholesterol | HDL Cholesterol | Total: HDL Cholesterol | |||
|---|---|---|---|---|---|---|
| EDF | Adjusted R2 | EDF | Adjusted R2 | EDF | Adjusted R2 | |
| Longevity | 0.22 | 0.19 | 0.20 | |||
| <80 | 5.3 | 3.1 | 1.0 | |||
| ≥80 to <90 | 6.8 | 3.6 | 4.0 | |||
| ≥90 | 6.7 | 1.0 | 3.3 | |||
| APOE allele status | 0.22 | 0.16 | 0.16 | |||
| e2+ | 5.0 | 1.0 | 3.2 | |||
| e3 | 7.3 | 2.1 | 3.8 | |||
| e4+ | 5.8 | 1.0 | 3.1 | |||
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pancioli, A.M.; Broderick, J.; Kothari, R.; Brott, T.; Tuchfarber, A.; Miller, R.; Khoury, J.; Jauch, E. Public perception of stroke warning signs and knowledge of potential risk factors. JAMA 1998, 279, 1288–1292. [Google Scholar]
- Van der Wulp, M.Y.; Verkade, H.J.; Groen, A.K. Regulation of cholesterol homeostasis. Mol. Cell. Endocrinol. 2013, 368, 1–16. [Google Scholar]
- Ford, E.S.; Capewell, S. Trends in Total and low-density lipoprotein cholesterol among U.S. adults: Contributions of changes in dietary fat intake and use of cholesterol-lowering medications. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- De Meyer, F.; Smit, B. Effect of cholesterol on the structure of a phospholipid bilayer. Proc. Natl. Acad. Sci. USA 2009, 106, 3654–3658. [Google Scholar]
- Rezen, T.; Rozman, D.; Pascussi, J.M.; Monostory, K. Interplay between cholesterol and drug metabolism. Biochim. Biophys. Acta 2011, 1814, 146–160. [Google Scholar]
- Goldstein, J.L.; Brown, M.S. The LDL receptor. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 431–438. [Google Scholar]
- Hussain, M.M. A proposed model for the assembly of chylomicrons. Atherosclerosis 2000, 148, 1–15. [Google Scholar]
- Goldstein, J.L.; Brown, M.S. Progress in understanding the LDL receptor and HMG-CoA reductase, two membrane proteins that regulate the plasma cholesterol. J. Lipid Res. 1984, 25, 1450–1461. [Google Scholar]
- Havel, R.J. The formation of LDL: Mechanisms and regulation. J. Lipid Res. 1984, 25, 1570–1576. [Google Scholar]
- Fielding, C.J.; Fielding, P.E. Molecular physiology of reverse cholesterol transport. J. Lipid Res. 1995, 36, 211–228. [Google Scholar]
- Brown, M.S.; Kovanen, P.T.; Goldstein, J.L. Regulation of plasma cholesterol by lipoprotein receptors. Science 1981, 212, 628–635. [Google Scholar]
- Kinosian, B.; Glick, H.; Garland, G. Cholesterol and coronary heart disease: Predicting risks by levels and ratios. Ann. Int. Med. 1994, 121, 641–647. [Google Scholar]
- Natarajan, S.; Glick, H.; Criqui, M.; Horowitz, D.; Lipsitz, S.R.; Kinosian, B. Cholesterol measures to identify and treat individuals at risk for coronary heart disease. Am. J. Prev Med. 2003, 25, 50–57. [Google Scholar]
- Wang, T.D.; Chen, W.J.; Chien, K.L.; Seh-Yi Su, S.S.; Hsu, H.C.; Chen, M.F.; Liau, C.S.; Lee, Y.T. Efficacy of cholesterol levels and ratios in predicting future coronary heart disease in a Chinese population. Am. J. Cardiol. 2001, 88, 737–743. [Google Scholar]
- Wilson, P.W.; D’Agostino, R.B.; Levy, D.; Belanger, A.M.; Silbershatz, H.; Kannel, W.B. Prediction of coronary heart disease using risk factor categories. Circulation 1998, 97, 1837–1847. [Google Scholar]
- Grundy, S.M.; Cleeman, J.I.; Merz, C.N.; Brewer, H.B., Jr.; Clark, L.T.; Hunninghake, D.B.; Pasternak, R.C.; Smith, S.C., Jr.; Stone, N.J. Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines. J. Am. Coll Cardiol. 2004, 44, 720–732. [Google Scholar]
- Kuklina, E.V.; Carroll, M.D.; Shaw, K.M.; Hirsch, R. Trends in high LDL cholesterol, cholesterol-lowering medication use, and dietary saturated-fat intake: United States, 1976–2010. NCHS Data Brief. 2013, 117, 1–8. [Google Scholar]
- Fruchart, J.C.; Nierman, M.C.; Stroes, E.S.; Kastelein, J.J.; Duriez, P. New risk factors for atherosclerosis and patient risk assessment. Circulation 2004, 109, S15–S19. [Google Scholar]
- Carroll, M.D.; Lacher, D.A.; Sorlie, P.D.; Cleeman, J.I.; Gordon, D.J.; Wolz, M.; Grundy, S.M.; Johnson, C.L. Trends in serum lipids and lipoproteins of adults, 1960–2002. JAMA 2005, 294, 1773–1781. [Google Scholar]
- Ferrara, A.; Barrett-Connor, E.; Shan, J. Total, LDL, and HDL cholesterol decrease with age in older men and women. The Rancho Bernardo Study 1984–1994. Circulation 1997, 96, 37–43. [Google Scholar]
- Wilson, P.W.; Anderson, K.M.; Harris, T.; Kannel, W.B.; Castelli, W.P. Determinants of change in total cholesterol and HDL-C with age: The Framingham Study. J. Gerontol. 1994, 49, 252–257. [Google Scholar]
- Weijenberg, M.P.; Feskens, E.J.; Kromhout, D. Age-related changes in total and high-density-lipoprotein cholesterol in elderly Dutch men. Amer. J. Public Health 1996, 86, 798–803. [Google Scholar]
- Ettinger, W.H.; Wahl, P.W.; Kuller, L.H.; Bush, T.L.; Tracy, R.P.; Manolio, T.A.; Borhani, N.O.; Wong, N.D.; O’Leary, D.H. Lipoprotein lipids in older people. Results from the cardiovascular health study. The CHS collaborative research group. Circulation 1992, 86, 858–869. [Google Scholar]
- Garry, P.J.; Hunt, W.C.; Koehler, K.M.; VanderJagt, D.J.; Vellas, B.J. Longitudinal study of dietary intakes and plasma lipids in healthy elderly men and women. Amer. J. Clin. Nutr. 1992, 55, 682–688. [Google Scholar]
- Abbott, R.D.; Sharp, D.S.; Burchfiel, C.M.; Curb, J.D.; Rodriguez, B.L.; Hakim, A.A.; Yano, K. Cross-sectional and longitudinal changes in total and high-density-lipoprotein cholesterol levels over a 20-year period in elderly men: The Honolulu Heart Program. Ann. Epidemiol. 1997, 7, 417–424. [Google Scholar]
- Mahley, R.W.; Innerarity, T.L. Lipoprotein receptors and cholesterol homeostasis. Biochim. Biophys. Acta 1983, 737, 197–222. [Google Scholar]
- Kroll, J. Bile acids, chaperones, and mammalian longevity. Rejuvenation Res. 2012, 15, 210–212. [Google Scholar]
- Johansson, I.; Nilsson, L.M.; Stegmayr, B.; Boman, K.; Hallmans, G.; Winkvist, A. Associations among 25-year trends in diet, cholesterol and BMI from 140,000 observations in men and women in Northern Sweden. Nutr J. 2012, 11. [Google Scholar] [CrossRef]
- Lauer, R.M.; Lee, J.; Clarke, W.R. Factors affecting the relationship between childhood and adult cholesterol levels: The muscatine study. Pediatrics 1988, 82, 309–318. [Google Scholar]
- Sinaiko, A.R.; Donahue, R.P.; Jacobs, D.R., Jr.; Prineas, R.J. Relation of weight and rate of increase in weight during childhood and adolescence to body size, blood pressure, fasting insulin, and lipids in young adults. The Minneapolis Children’s Blood Pressure Study. Circulation 1999, 99, 1471–1476. [Google Scholar]
- Jousilahti, P.; Vartiainen, E.; Tuomilehto, J.; Puska, P. Sex, age, cardiovascular risk factors, and coronary heart disease: A prospective follow-up study of 14 786 middle-aged men and women in Finland. Circulation 1999, 99, 1165–1172. [Google Scholar]
- Matthews, K.A.; Crawford, S.L.; Chae, C.U.; Everson-Rose, S.A.; Sowers, M.F.; Sternfeld, B.; Sutton-Tyrrell, K. Are changes in cardiovascular disease risk factors in midlife women due to chronological aging or to the menopausal transition? J. Amer. Coll Cardiol. 2009, 54, 2366–2373. [Google Scholar]
- Stevenson, J.C.; Crook, D.; Godsland, I.F. Influence of age and menopause on serum lipids and lipoproteins in healthy women. Atherosclerosis 1993, 98, 83–90. [Google Scholar]
- Babischkin, J.S.; Grimes, R.W.; Pepe, G.J.; Albrecht, E.D. Estrogen stimulation of P450 cholesterol side-chain cleavage activity in cultures of human placental syncytiotrophoblasts. Biol. Reprod. 1997, 56, 272–278. [Google Scholar]
- Vehkavaara, S.; Silveira, A.; Hakala-Ala-Pietila, T.; Virkamaki, A.; Hovatta, O.; Hamsten, A.; Taskinen, M.R.; Yki-Jarvinen, H. Effects of oral and transdermal estrogen replacement therapy on markers of coagulation, fibrinolysis, inflammation and serum lipids and lipoproteins in postmenopausal women. Thromb. Haemost. 2001, 85, 619–625. [Google Scholar]
- Koh, K.K.; Cardillo, C.; Bui, M.N.; Hathaway, L.; Csako, G.; Waclawiw, M.A.; Panza, J.A.; Cannon, R.O., 3rd. Vascular effects of estrogen and cholesterol-lowering therapies in hypercholesterolemic postmenopausal women. Circulation 1999, 99, 354–360. [Google Scholar]
- Albert, M.A.; Glynn, R.J.; Buring, J.; Ridker, P.M. Impact of traditional and novel risk factors on the relationship between socioeconomic status and incident cardiovascular events. Circulation 2006, 114, 2619–2626. [Google Scholar]
- Benetou, V.; Chloptsios, Y.; Zavitsanos, X.; Karalis, D.; Naska, A.; Trichopoulou, A. Total cholesterol and HDL-cholesterol in relation to socioeconomic status in a sample of 11,645 Greek adults: The EPIC study in Greece. European Prospective Investigation into Nutrition and Cancer. Scand. J. Public Health 2000, 28, 260–265. [Google Scholar]
- Criqui, M.H.; Wallace, R.B.; Heiss, G.; Mishkel, M.; Schonfeld, G.; Jones, G.T. Cigarette smoking and plasma high-density lipoprotein cholesterol. The lipid research clinics program prevalence study. Circulation 1980, 62, 70–76. [Google Scholar]
- Castelli, W.P.; Anderson, K. A population at risk. Prevalence of high cholesterol levels in hypertensive patients in the Framingham Study. Amer. J. Med. 1986, 80, 23–32. [Google Scholar]
- Bjorkhem, I.; Eggertsen, G. Genes involved in initial steps of bile acid synthesis. Curr. Opin. Lipidol. 2001, 12, 97–103. [Google Scholar]
- Chawla, A.; Repa, J.J.; Evans, R.M.; Mangelsdorf, D.J. Nuclear receptors and lipid physiology: Opening the X-files. Science 2001, 294, 1866–1870. [Google Scholar]
- Genomes Project, C.; Abecasis, G.R.; Auton, A.; Brooks, L.D.; dePristo, M.A.; Durbin, R.M.; Handsaker, R.E.; Kang, H.M.; Marth, G.T.; McVean, G.A. An integrated map of genetic variation from 1092 human genomes. Nature 2012, 491, 56–65. [Google Scholar]
- Hauser, P.S.; Narayanaswami, V.; Ryan, R.O. Apolipoprotein E: From lipid transport to neurobiology. Prog. Lipid Res. 2011, 50, 62–74. [Google Scholar]
- Gylling, H.; Aalto-Setala, K.; Kontula, K.; Miettinen, T.A. Serum low density lipoprotein cholesterol level and cholesterol absorption efficiency are influenced by apolipoprotein B and E polymorphism and by the FH-Helsinki mutation of the low density lipoprotein receptor gene in familial hypercholesterolemia. Arterioscler. Thromb. 1991, 11, 1368–1375. [Google Scholar]
- Von Bergmann, K.; Lutjohann, D.; Lindenthal, B.; Steinmetz, A. Efficiency of intestinal cholesterol absorption in humans is not related to APOE phenotype. J. Lipid Res. 2003, 44, 193–197. [Google Scholar]
- Boerwinkle, E.; Utermann, G. Simultaneous effects of the apolipoprotein E polymorphism on apolipoprotein E, apolipoprotein B, and cholesterol metabolism. Am. J. Hum. Genet. 1988, 42, 104–112. [Google Scholar]
- Ehnholm, C.; Lukka, M.; Kuusi, T.; Nikkila, E.; Utermann, G. Apolipoprotein E polymorphism in the Finnish population: Gene frequencies and relation to lipoprotein concentrations. J. Lipid Res. 1986, 27, 227–235. [Google Scholar]
- Tan, C.E.; Tai, E.S.; Tan, C.S.; Chia, K.S.; Lee, J.; Chew, S.K.; Ordovas, J.M. APOE polymorphism and lipid profile in three ethnic groups in the Singapore population. Atherosclerosis 2003, 170, 253–260. [Google Scholar]
- De Beer, F.; Stalenhoef, A.F.; Hoogerbrugge, N.; Kastelein, J.J.; Gevers Leuven, J.A.; van Duijn, C.M.; Havekes, L.M.; Smelt, A.H. Expression of type III hyperlipoproteinemia in apolipoprotein E2 (Arg158→Cys) homozygotes is associated with hyperinsulinemia. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 294–299. [Google Scholar]
- Lahoz, C.; Schaefer, E.J.; Cupples, L.A.; Wilson, P.W.; Levy, D.; Osgood, D.; Parpos, S.; Pedro-Botet, J.; Daly, J.A.; Ordovas, J.M. Apolipoprotein E genotype and cardiovascular disease in the Framingham Heart Study. Atherosclerosis 2001, 154, 529–537. [Google Scholar]
- McCarron, M.O.; Delong, D.; Alberts, M.J. APOE genotype as a risk factor for ischemic cerebrovascular disease: A meta-analysis. Neurology 1999, 53, 1308–1311. [Google Scholar]
- Wilson, P.W.; Schaefer, E.J.; Larson, M.G.; Ordovas, J.M. Apolipoprotein E alleles and risk of coronary disease. A meta-analysis. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 1250–1255. [Google Scholar]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993, 261, 921–923. [Google Scholar]
- Corder, E.H.; Saunders, A.M.; Risch, N.J.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C., Jr.; Rimmler, J.B.; Locke, P.A.; Conneally, P.M.; Schmader, K.E.; et al. Protective effect of apolipoprotein E type 2 allele for late onset Alzheimer disease. Nat. Genet. 1994, 7, 180–184. [Google Scholar]
- Kivipelto, M.; Helkala, E.L.; Hallikainen, M. Elevated systolic blood pressure and high cholesterol levels at midlife are risk factors for late-life dementia. Neurobiol. Aging 2000, 21, S174. [Google Scholar]
- Lepara, O.; Valjevac, A.; Alajbegovic, A.; Zaciragic, A.; Nakas-Icindic, E. Decreased serum lipids in patients with probable Alzheimer’s disease. Bosn J. Basic Med. Sci. 2009, 9, 215–220. [Google Scholar]
- Reitz, C.; Tang, M.X.; Luchsinger, J.; Mayeux, R. Relation of plasma lipids to Alzheimer disease and vascular dementia. Arch. Neurol. 2004, 61, 705–714. [Google Scholar]
- Stewart, R.; White, L.R.; Xue, Q.L.; Launer, L.J. Twenty-six-year change in total cholesterol levels and incident dementia: The Honolulu-Asia Aging Study. Arch. Neurol. 2007, 64, 103–107. [Google Scholar]
- Dawber, T.R.; Meadors, G.F.; Moore, F.E.J. Epidemiological approaches to heart disease: The Framingham Study. Amer. J. Public Health Nations Health 1951, 41, 279–281. [Google Scholar]
- Abell, L.L.; Levy, B.B.; Brodie, B.B.; Kendall, F.E. A simplified method for the estimation of total cholesterol in serum and demonstration of its specificity. J. Biol. Chem. 1952, 195, 357–366. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar]
- Martin, S.S.; Blaha, M.J.; Elshazly, M.B.; Brinton, E.A.; Toth, P.P.; McEvoy, J.W.; Joshi, P.H.; Kulkarni, K.R.; Mize, P.D.; Kwiterovich, P.O.; et al. Friedewald-estimated vs. directly measured low-density lipoprotein cholesterol and treatment implications. J. Amer. Coll Cardiol. 2013, 62, 732–739. [Google Scholar]
- Lin, X.; Zhang, D. Inferencein generalized additive mixed models by using smoothing splines. J. R. Stat. Soc. 1999, 61, 381–400. [Google Scholar]
- Wood, S.; Scheipl, F. Gamm4: Generalized Additive Mixed Models using mgcv and lme4. Available online: http://cran.r-project.org/web/packages/gamm4/index.html (accessed on 11 October 2014).
- Wood, S. Thin plate regression splines. J. R. Stat. Soc. 2003, 65, 95–114. [Google Scholar]
- Wood, S. Generalized Additive Models: An Introduction with R; Chapman & Hall: Boca Raton, FL, USA, 2006. [Google Scholar]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning: with Applications in R; Springer: New York, NY, USA, 2013. [Google Scholar]
- Fitzmaurice, G.; Laird, N.; Ware, J. Applied Longitudinal Analysis, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Mendez, D.; Warner, K.E.; Courant, P.N. Has smoking cessation ceased? Expected trends in the prevalence of smoking in the United States. Amer. J. Epidemiol. 1998, 148, 249–258. [Google Scholar]
- Franklin, S.S.; Gustin, W.T.; Wong, N.D.; Larson, M.G.; Weber, M.A.; Kannel, W.B.; Levy, D. Hemodynamic patterns of age-related changes in blood pressure. The Framingham Heart Study. Circulation 1997, 96, 308–315. [Google Scholar]
- Livshits, G.; Malkin, I.; Williams, F.M.; Hart, D.J.; Hakim, A.; Spector, T.D. Longitudinal study of variation in body mass index in middle-aged UK females. Age 2012, 34, 1285–1294. [Google Scholar]
- Sui, X.; Jackson, A.S.; Church, T.S.; Lee, D.C.; O’Connor, D.P.; Liu, J.; Blair, S.N. Effects of cardiorespiratory fitness on aging: Glucose trajectory in a cohort of healthy men. Ann. Epidemiol. 2012, 22, 617–622. [Google Scholar]
- Kivipelto, M.; Helkala, E.L.; Hanninen, T.; Laakso, M.P.; Hallikainen, M.; Alhainen, K.; Soininen, H.; Tuomilehto, J.; Nissinen, A. Midlife vascular risk factors and late-life mild cognitive impairment: A population-based study. Neurology 2001, 56, 1683–1689. [Google Scholar]
- Whitmer, R.A.; Sidney, S.; Selby, J.; Johnston, S.C.; Yaffe, K. Midlife cardiovascular risk factors and risk of dementia in late life. Neurology 2005, 64, 277–281. [Google Scholar]
- Green, M.S.; Heiss, G.; Rifkind, B.M.; Cooper, G.R.; Williams, O.D.; Tyroler, H.A. The ratio of plasma high-density lipoprotein cholesterol to total and low-density lipoprotein cholesterol: Age-related changes and race and sex differences in selected North American populations. The Lipid Research Clinics Program Prevalence Study. Circulation 1985, 72, 93–104. [Google Scholar]
- Bilheimer, D.W. Clinical considerations regarding treatment of hypercholesterolemia in the elderly. Atherosclerosis 1991, 91, S35–S57. [Google Scholar]
- Tilvis, R.S.; Valvanne, J.N.; Strandberg, T.E.; Miettinen, T.A. Prognostic significance of serum cholesterol, lathosterol, and sitosterol in old age; a 17-year population study. Ann. Med. 2011, 43, 292–301. [Google Scholar]
- Silbernagel, G.; Fauler, G.; Hoffmann, M.M.; Lutjohann, D.; Winkelmann, B.R.; Boehm, B.O.; Marz, W. The associations of cholesterol metabolism and plasma plant sterols with all-cause and cardiovascular mortality. J. Lipid Res. 2010, 51, 2384–2393. [Google Scholar]
- Strandberg, T.E.; Gylling, H.; Tilvis, R.S.; Miettinen, T.A. Serum plant and other noncholesterol sterols, cholesterol metabolism and 22-year mortality among middle-aged men. Atherosclerosis 2010, 210, 282–287. [Google Scholar]
- Tobin, M.D.; Sheehan, N.A.; Scurrah, K.J.; Burton, P.R. Adjusting for treatment effects in studies of quantitative traits: Antihypertensive therapy and systolic blood pressure. Stat. Med. 2005, 24, 2911–2935. [Google Scholar]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Covas, M.I.; Fiol, M.; Gomez-Gracia, E.; Lopez-Sabater, M.C.; Vinyoles, E.; et al. Effects of a Mediterranean-style diet on cardiovascular risk factors: A randomized trial. Ann. Int. Med. 2006, 145, 1–11. [Google Scholar]
- Donini, L.M.; Savina, C.; Cannella, C. Eating habits and appetite control in the elderly: The anorexia of aging. Int. Psychogeriatr. 2003, 15, 73–87. [Google Scholar]
- Kaiser, M.J.; Bauer, J.M.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.S.; Charlton, K.E.; Maggio, M.; et al. Mini nutritional assessment international G. Frequency of malnutrition in older adults: A multinational perspective using the mini nutritional assessment. J. Amer. Geriatr. Soc. 2010, 58, 1734–1738. [Google Scholar]
- De Oliveira, E.S.E.R.; Foster, D.; McGee Harper, M.; Seidman, C.E.; Smith, J.D.; Breslow, J.L.; Brinton, E.A. Alcohol consumption raises HDL cholesterol levels by increasing the transport rate of apolipoproteins A-I and A-II. Circulation 2000, 102, 2347–2352. [Google Scholar]
- Halverstadt, A.; Phares, D.A.; Wilund, K.R.; Goldberg, A.P.; Hagberg, J.M. Endurance exercise training raises high-density lipoprotein cholesterol and lowers small low-density lipoprotein and very low-density lipoprotein independent of body fat phenotypes in older men and women. Metabolism 2007, 56, 444–450. [Google Scholar]
- Gordon, D.J.; Probstfield, J.L.; Garrison, R.J.; Neaton, J.D.; Castelli, W.P.; Knoke, J.D.; Jacobs, D.R., Jr.; Bangdiwala, S.; Tyroler, H.A. High-density lipoprotein cholesterol and cardiovascular disease. Four prospective American studies. Circulation 1989, 79, 8–15. [Google Scholar]
- Engstrom, G.; Lind, P.; Hedblad, B.; Stavenow, L.; Janzon, L.; Lindgarde, F. Effects of cholesterol and inflammation-sensitive plasma proteins on incidence of myocardial infarction and stroke in men. Circulation 2002, 105, 2632–2637. [Google Scholar]
- Schatz, I.J.; Masaki, K.; Yano, K.; Chen, R.; Rodriguez, B.L.; Curb, J.D. Cholesterol and all-cause mortality in elderly people from the Honolulu Heart Program: A cohort study. Lancet 2001, 358, 351–355. [Google Scholar]
- Raiha, I.; Marniemi, J.; Puukka, P.; Toikka, T.; Ehnholm, C.; Sourander, L. Effect of serum lipids, lipoproteins, and apolipoproteins on vascular and nonvascular mortality in the elderly. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 1224–1232. [Google Scholar]
- Bales, C.W.; Ritchie, C.S. Sarcopenia, weight loss, and nutritional frailty in the elderly. Annu. Rev. Nutr. 2002, 22, 309–323. [Google Scholar]
- Eichner, J.E.; Dunn, S.T.; Perveen, G.; Thompson, D.M.; Stewart, K.E.; Stroehla, B.C. Apolipoprotein E polymorphism and cardiovascular disease: A HuGE review. Am. J. Epidemiol. 2002, 155, 487–495. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Downer, B.; Estus, S.; Katsumata, Y.; Fardo, D.W. Longitudinal Trajectories of Cholesterol from Midlife through Late Life according to Apolipoprotein E Allele Status. Int. J. Environ. Res. Public Health 2014, 11, 10663-10693. https://doi.org/10.3390/ijerph111010663
Downer B, Estus S, Katsumata Y, Fardo DW. Longitudinal Trajectories of Cholesterol from Midlife through Late Life according to Apolipoprotein E Allele Status. International Journal of Environmental Research and Public Health. 2014; 11(10):10663-10693. https://doi.org/10.3390/ijerph111010663
Chicago/Turabian StyleDowner, Brian, Steven Estus, Yuriko Katsumata, and David W. Fardo. 2014. "Longitudinal Trajectories of Cholesterol from Midlife through Late Life according to Apolipoprotein E Allele Status" International Journal of Environmental Research and Public Health 11, no. 10: 10663-10693. https://doi.org/10.3390/ijerph111010663
APA StyleDowner, B., Estus, S., Katsumata, Y., & Fardo, D. W. (2014). Longitudinal Trajectories of Cholesterol from Midlife through Late Life according to Apolipoprotein E Allele Status. International Journal of Environmental Research and Public Health, 11(10), 10663-10693. https://doi.org/10.3390/ijerph111010663




