Extricating Sex and Gender in Air Pollution Research: A Community-Based Study on Cardinal Symptoms of Exposure
Abstract
:1. Introduction
2. Methods
2.1. Sampling and Exposure Assessment
| Sample (n = 804) | Females (n = 440) | Males (n = 364) | Sex Difference | Pollution Factor | |
|---|---|---|---|---|---|
| Categorical Variables | % | % | % | Pearson χ2 | Mann-Whitney Test (2-tailed. z) |
| 5+ Cardinal Symptoms | 19.3 | 21.9 | 16.2 | 4.44 * | −2.77 ** |
| Occupational Exposure | 46.3 | 27.4 | 68.8 | 137.22 *** | −1.44 |
| Hay Fever or Allergies | 29.0 | 35.1 | 21.7 | 17.09 *** | −0.52 |
| Asthma | 9.5 | 10.5 | 8.4 | 1.02 | −2.29 * |
| Cancer | 4.6 | 3.5 | 5.8 | 2.45 | −1.54 |
| Kidney Disease | 4.8 | 4.0 | 5.7 | 1.45 | −0.08 |
| Skin Condition | 13.9 | 16.9 | 10.4 | 7.27 ** | −1.17 |
| Hypertension | 22.8 | 22.5 | 23.1 | 0.05 | −0.73 |
| Heart Disease | 7.9 | 5.9 | 10.3 | 5.24 * | −1.44 |
| Low Income | 10.5 | 10.6 | 10.3 | 0.01 | −5.39 *** |
| Continuous Variables | μX | μX | μX | Independent t-Test | Pearson r (2-tailed) |
| NO2 (ppb) | 13.85 | 13.75 | 13.96 | 1.86 | 0.84 *** |
| SO2 (ppb) | 3.18 | 3.17 | 3.19 | 0.14 | 0.76 *** |
| BTEX (mg/m3) | 3.76 | 3.73 | 3.81 | 0.77 | 0.77 *** |
2.2. Outcome Measurement
2.3. Gendered Stratification Variables
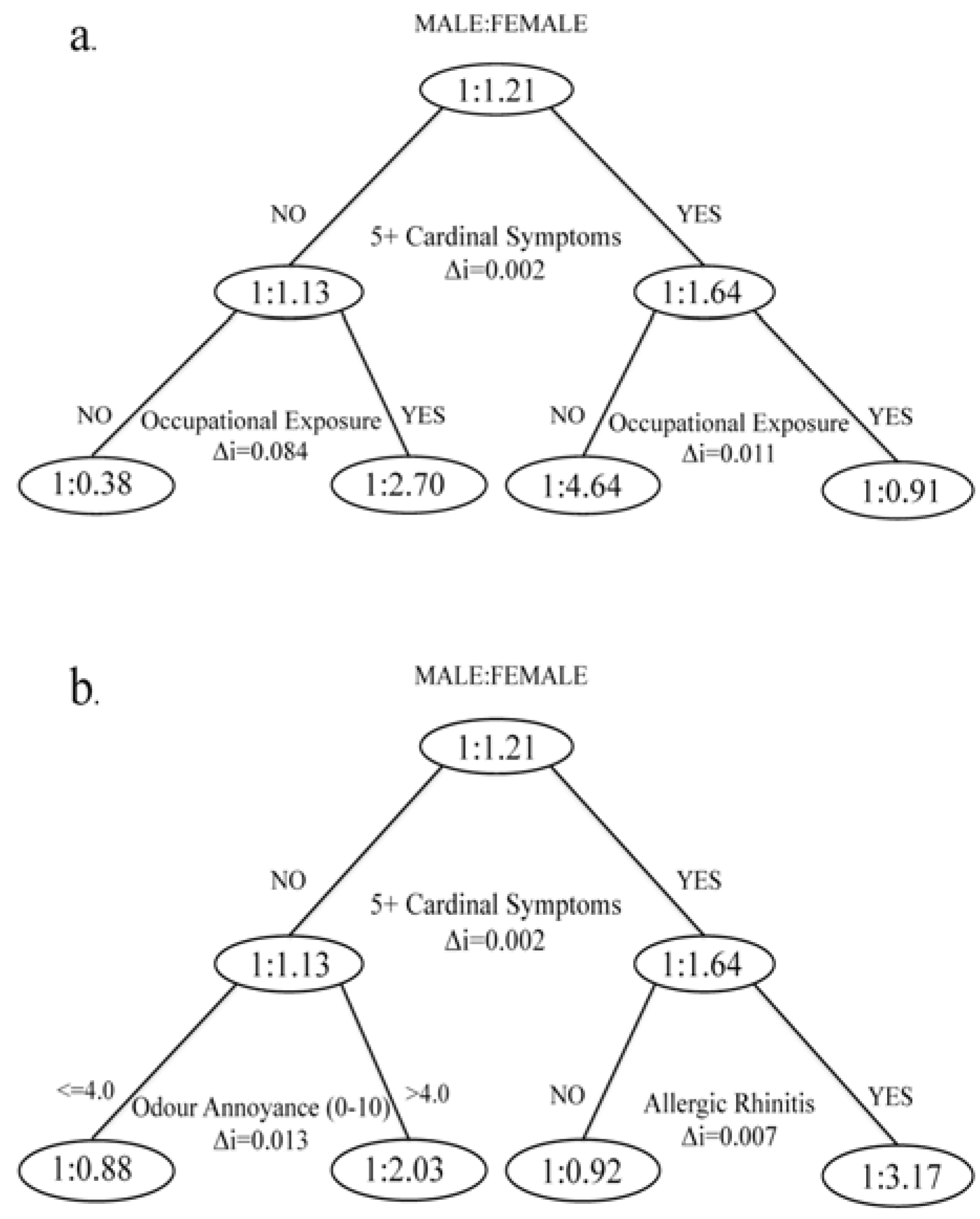
2.4. Analysis
3. Results
Sample Characteristics
4. Cardinal Symptoms of Exposure
4.1. Complete Sample
4.2. Occupational Exposure Stratification
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 | |
|---|---|---|---|---|---|---|
| Pollution Factor | 1.282 ** | 1.343 * | 1.351 * | 1.321 ** | 1.286 * | 1.254 * |
| Indoor Exposure | 1.181 | 1.187 | 1.192 | 1.225 | 1.286 * | |
| Sex (54.3% female) | 1.479 * | 1.520 * | 1.512 * | 1.523 * | ||
| Age Group (Reference 18–24) | ** | ** | *** | |||
| 25–44 | 0.947 | 1.021 | 0.705 | |||
| 45–64 | 0.559 * | 0.594 | 0.302 *** | |||
| 65+ | 0.381 ** | 0.398 ** | 0.106 *** | |||
| Low Income | 1.820 * | 1.794 * | ||||
| Asthma | 5.048 *** | |||||
| Cancer | 1.765 | |||||
| Kidney Disease | 2.973 ** | |||||
| Skin Condition | 2.602 *** | |||||
| Hypertension | 2.426 *** | |||||
| Heart Disease | 1.724 | |||||
| Hosmer & Lemeshow χ2 (df), significance | 16.71(8), 0.03 | 15.97(8), 0.43 | 1.09(8), 0.99 | 13.04(8), 0.11 | 6.17(8), 0.63 | 8.54(8), 0.38 |
| Nagelkerke R2 | 0.014 | 0.019 | 0.028 | 0.058 | 0.067 | 0.207 |
| % Correctly Classified | 80.7 | 80.7 | 80.7 | 80.7 | 80.7 | 81.1 |
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 | |
|---|---|---|---|---|---|---|
| Pollution Factor | 1.347 * | 1.525 ** | 1.570 ** | 1.560 ** | 1.488 * | 1.384 |
| Indoor Exposure | 1.535 ** | 1.570 ** | 1.577 * | 1.679 ** | 1.773 ** | |
| Sex (73.4% female) | 2.268 * | 2.388 * | 2.347 * | 2.389 * | ||
| Age Group (Reference 18–24) | * | |||||
| 25–44 | 1.057 | 1.311 | 0.953 | |||
| 45–64 | 0.827 | 1.009 | 0.576 | |||
| 65+ | 0.535 | 0.639 | 0.178 ** | |||
| Low income | 2.675 * | 2.617 * | ||||
| Asthma | 5.173 *** | |||||
| Cancer | 1.883 | |||||
| Kidney Disease | 3.348 * | |||||
| Skin Condition | 3.818 *** | |||||
| Hypertension | 1.964 | |||||
| Heart Disease | 2.924 | |||||
| Hosmer & Lemeshow χ2 (df), significance | 8.98(8), 0.34 | 5.29(8), 0.73 | 5.37(8), 0.72 | 12.41(8), 0.13 | 5.63(8), 0.69 | 11.30(8), 0.19 |
| Nagelkerke R2 | 0.018 | 0.047 | 0.07 | 0.082 | 0.106 | 0.267 |
| % Correctly Classified | 85.3 | 85.3 | 85.3 | 85.3 | 85.3 | 85.9 |
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 | |
|---|---|---|---|---|---|---|
| Pollution Factor | 1.280 | 1.257 | 1.260 | 1.191 | 1.162 | 1.194 |
| Indoor Exposure | 0.931 | 0.887 | 0.879 | 0.891 | 0.948 | |
| Sex (26.6% female) | 2.369 *** | 2.316 *** | 2.381 *** | 2.327 ** | ||
| Age Group (Reference 18–24) | ** | ** | *** | |||
| 25–44 | 0.576 | 0.583 | 0.393 * | |||
| 45–64 | 0.288 ** | 0.285 ** | 0.132 *** | |||
| 65+ | 0.242 ** | 0.232 ** | 0.051 *** | |||
| Low Income | 1.802 | 2.085 | ||||
| Asthma | 5.622 *** | |||||
| Cancer | 1.898 | |||||
| Kidney Disease | 2.500 | |||||
| Skin Condition | 1.916 | |||||
| Hypertension | 2.924 ** | |||||
| Heart Disease | 1.129 | |||||
| Hosmer & Lemeshow χ2 (df), significance | 16.79(8), 0.03 | 11.20(8), 0.19 | 8.84(8), 0.36 | 12.41(8), 0.13 | 11.27(8), 0.18 | 9.18(8), 0.33 |
| Nagelkerke R2 | 0.015 | 0.016 | 0.063 | 0.115 | 0.122 | 0.248 |
| % Correctly Classified | 75.3 | 75.3 | 75.3 | 75.3 | 75.3 | 75.9 |
4.3. Allergic Disease Stratification
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 | |
|---|---|---|---|---|---|---|
| Pollution Factor | 1.241 | 1.356 * | 1.356 * | 1.284 | 1.237 | 1.211 |
| Indoor Exposure | 1.390 * | 1.388 * | 1.421 * | 1.500 ** | 1.577 ** | |
| Sex (49.6% female) | 0.938 | 0.964 | 0.938 | 0.910 | ||
| Age Group (Reference 18x2013;24) | *** | *** | *** | |||
| 25x2013;44 | 0.506 * | 0.576 | 0.442 * | |||
| 45x2013;64 | 0.215 *** | 0.238 *** | 0.135 *** | |||
| 65+ | 0.288 ** | 0.314 ** | 0.102 *** | |||
| Low Income | 2.386 ** | 2.764 ** | ||||
| Asthma | 3.422 ** | |||||
| Cancer | 1.607 | |||||
| Kidney Disease | 1.152 | |||||
| Skin Condition | 3.895 *** | |||||
| Hypertension | 2.453 ** | |||||
| Heart Disease | 2.072 | |||||
| Hosmer & Lemeshow χ2 (df), significance | 8.07(8), 0.43 | 5.76(8), 0.76 | 9.56(8), 0.30 | 3.41(8), 0.91 | 9.99(8), 0.27 | 9.88(8), 0.27 |
| Nagelkerke R2 | 0.01 | 0.028 | 0.028 | 0.09 | 0.108 | 0.215 |
| % Correctly Classified | 85.9 | 85.9 | 85.9 | 85.9 | 85.9 | 85.9 |
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 | |
|---|---|---|---|---|---|---|
| Pollution Factor | 1.354 | 1.310 | 1.355 | 1.287 | 1.273 | 1.279 |
| Indoor Exposure | 0.899 | 0.904 | 0.845 | 0.850 | 0.891 | |
| Sex (50.4% female) | 2.212 * | 2.272 * | 2.266 * | 2.754 ** | ||
| Age Group (Reference 18–24) | ** | ** | ** | |||
| 25–44 | 3.667 * | 3.724 * | 2.310 | |||
| 45–64 | 3.250 | 3.258 | 1.473 | |||
| 65+ | 0.915 | 0.909 | 0.179 * | |||
| Low Income | 1.382 | 1.587 | ||||
| Asthma | 6.305 *** | |||||
| Cancer | 2.728 | |||||
| Kidney Disease | 11.493 ** | |||||
| Skin Condition | 1.383 | |||||
| Hypertension | 2.019 | |||||
| Heart Disease | 0.895 | |||||
| Hosmer & Lemeshow χ2 (df), significance | 8.88 (8), 0.35 | 6.85 (8), 0.55 | 12.89 (8), 0.12 | 3.20 (8), 0.92 | 8.22 (8), 0.41 | 5.27 (8), 0.73 |
| Nagelkerke R2 | 0.023 | 0.025 | 0.063 | 0.138 | 0.140 | 0.322 |
| % Correctly Classified | 67.8 | 67.8 | 67.8 | 67.8 | 67.8 | 73.8 |
5. Discussion
6. Conclusions
Acknowledgements
Conflicts of Interest
References
- Clougherty, J.E. A growing role for gender analysis in air pollution epidemiology. Environ. Health Perspect. 2010, 118, 167–176. [Google Scholar] [CrossRef]
- Luginaah, I.N.; Fung, K.Y.; Gorey, K.M.; Webster, G.; Wills, C. Association of ambient air pollution with respiratory hospitalization in a government-designated “area of concern”: The case of Windsor, Ontario. Environ. Health Perspect. 2005, 113, 290–296. [Google Scholar] [CrossRef]
- Granados-Canal, D.J.; Chardon, B.; Lefranc, A.; Gremy, I. Air pollution and respiratory hospital admissions in greater Paris: Exploring sex differences. Arch. Environ. Occup. Health 2005, 60, 307–313. [Google Scholar] [CrossRef]
- Kim, C.S.; Hu, S.C. Regional deposition of inhaled particles in human lungs: Comparison between men and women. J. Appl. Physiol. 1998, 84, 1834–1844. [Google Scholar] [CrossRef]
- Ernstgård, L.; Gullstrand, E.; Löf, A.; Johanson, G. Are women more sensitive than men to 2-propanol and m-xylene vapours? Occup. Environ. Med. 2002, 59, 759–767. [Google Scholar] [CrossRef]
- Becklake, M.R.; Kauffmann, F. Gender differences in airway behaviour over the human life span. Thorax 1999, 54, 1119–1138. [Google Scholar] [CrossRef]
- De Marco, R.; Locatelli, F.; Sunyer, J.; Burney, P. Differences in incidence of reported asthma related to age in men and women: A retrospective analysis of the data of the European respiratory health survey. Am. J. Respir. Crit. Care. Med. 2000, 162, 68–74. [Google Scholar] [CrossRef]
- Burra, T.A.; Moineddin, R.; Agha, M.M.; Glazier, R.H. Social disadvantage, air pollution, and asthma physician visits in Toronto, Canada. Environ. Res. 2009, 109, 567–574. [Google Scholar] [CrossRef]
- Boutin-Forzano, S.; Hammou, Y.; Gouitaa, M.; Charpin, D. Air pollution and atopy. Eur. Ann. Allergy Clin. Immunol. 2005, 37, 11–16. [Google Scholar]
- Hajat, S.; Haines, A.; Atkinson, R.W.; Bremner, S.A.; Anderson, H.R.; Emberlin, J. Association between air pollution and daily consultations with general practitioners for allergic rhinitis in London, United Kingdom. Am. J. Epidemiol. 2001, 153, 704–714. [Google Scholar] [CrossRef]
- Meding, B. Differences between the sexes with regard to work-related skin disease. Contact Dermat. 2000, 43, 65–71. [Google Scholar]
- Zeka, A.; Zanobetti, A.; Schwartz, J. Individual-level modifiers of the effects of particulate matter on daily mortality. Am. J. Epidemiol. 2006, 163, 849–859. [Google Scholar] [CrossRef]
- Jerrett, M.; Buzzelli, M.; Burnett, R.T.; DeLuca, P.F. Particulate air pollution, social confounders and mortality in small areas of an industrial city. Soc. Sci. Med. 2005, 60, 2845–2863. [Google Scholar] [CrossRef]
- Oiamo, T.H.; Luginaah, I.N.; Atari, D.O.; Gorey, K.M. Air pollution and general practitioner access and utilization: A population based study in Sarnia, “Chemical Valley”, Ontario. Environ. Health 2011, 10. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Kubzansky, L.D. A framework for examining social stress and susceptibility to air pollution in respiratory health. Environ. Health Perspect. 2009, 117, 1351–1358. [Google Scholar]
- Fung, K.Y.; Luginaah, I.N.; Gorey, K.M. Impact of air pollution on hospital admissions in Southwestern Ontario, Canada: Generating hypotheses in sentinel high-exposure places. Environ. Health 2007, 6. [Google Scholar] [CrossRef]
- Mackenzie, C.A.; Lockridge, A.; Keith, M. Declining sex ratio in a first nation community. Environ. Health Perspect. 2005, 113, 1295–1298. [Google Scholar] [CrossRef]
- Van Larebeke, N.A.; Sasco, A.J.; Brophy, J.T.; Keith, M.M.; Gilbertson, M.; Watterson, A. Sex ratio changes as sentinel health events of endocrine disruption. Int. J. Occup. Environ. Health 2008, 14, 138–143. [Google Scholar]
- Gilbertson, M.; Brophy, J. Community health profile of Windsor, Ontario, Canada: Anatomy of a Great Lakes area of concer. Environ. Health Perspect. 2001, 109, 827–843. [Google Scholar]
- Atari, D.O.; Luginaah, I.N. Assessing the distribution of volatile organic compounds using land use regression in Sarnia, “Chemical Valley”, Ontario, Canada. Environ. Health 2009, 8. [Google Scholar] [CrossRef]
- Atari, D.O.; Luginaah, I.; Xu, X.; Fung, K. Spatial variability of ambient nitrogen dioxide and sulfur dioxide in Sarnia, “Chemical Valley”, Ontario, Canada. J. Toxicol. Environ. Health 2008, 71, 1572–1581. [Google Scholar] [CrossRef]
- Hoek, G.; Beelen, R.; de Hoogh, K.; Vienneau, D.; Gulliver, J.; Fischer, P.; Briggs, D. A review of land-use regression models to assess spatial variation of outdoor air pollution. Atmos. Environ. 2008, 42, 7561–7578. [Google Scholar] [CrossRef]
- Miller, L.; Xu, X.H.; Luginaah, I. Spatial variability of volatile organic compound concentrations in Sarnia, Ontario, Canada. J. Toxicol. Environ. Health. 2009, 72, 610–624. [Google Scholar] [CrossRef]
- Zanobetti, A.; Schwartz, J. Race, gender, and social status as modifiers of the effects of PM10 on mortality. J. Occup. Environ. Med. 2000, 42, 469–474. [Google Scholar] [CrossRef]
- Boezen, H.M.; Vonk, J.M.; van der Zee, S.C.; Gerritsen, J.; Hoek, G.; Brunekreef, B.; Schouten, J.P.; Postma, D.S. Susceptibility to air pollution in elderly males and females. Eur. Respir. J. 2005, 25, 1018–1024. [Google Scholar] [CrossRef]
- Forsberg, B.; Stjernberg, N.; Wall, S. People can detect poor air quality well below guideline concentrations: A prevalence study of annoyance reactions and air pollution from traffic. Occup. Environ. Med. 1997, 54, 44–48. [Google Scholar] [CrossRef]
- Luginaah, I.N.; Taylor, S.M.; Elliott, S.J.; Eyles, J.D. A longitudinal study of the health impacts of a petroleum refinery. Soc. Sci. Med. 2000, 50, 1155–1166. [Google Scholar] [CrossRef]
- Lemon, S.C.; Roy, J.; Clark, M.A.; Friedmann, P.D.; Rakowski, W. Classification and regression tree analysis in public health: Methodological review and comparison with logistic regression. Ann. Behav. Med. 2003, 26, 172–181. [Google Scholar] [CrossRef]
- Low Income Cut-Offs for 2006 and Low Income Measures for 2005. Statistics Canada: Toronto, ON, Canada. Available online: http://www.statcan.gc.ca/pub/75f0002m/75f0002m2007004-eng.pdf. (accessed on 12 January 2013).
- Profile of Census Tracts in London, Sarnia and Windsor; Statistics Canada: Toronto, ON, Canada, 2009.
- Fagan, J.K.; Scheff, P.A.; Hryhorczuk, D.; Ramakrishnan, V.; Ross, M.; Persky, V. Prevalence of asthma and other allergic diseases in an adolescent population: Association with gender and race. Ann. Allergy Asthma Immunol. 2001, 86, 177–184. [Google Scholar] [CrossRef]
- Villeneuve, P.J.; Doiron, M.S.; Stieb, D.; Dales, R.; Burnett, R.T.; Dugandzic, R. Is outdoor air pollution associated with physician visits for allergic rhinitis among the elderly in Toronto, Canada? Allergy 2006, 61, 750–758. [Google Scholar] [CrossRef]
- Dong, G.H.; Chen, T.; Liu, M.M.; Wang, D.; Ma, Y.N.; Ren, W.H.; Lee, Y.L.; Zhao, Y.D.; He, Q.C. Gender differences and effect of air pollution on asthma in children with and without allergic predisposition: Northeast Chinese children health study. PLoS One 2011, 6, e22470. [Google Scholar] [CrossRef]
- Barrenas, F.; Andersson, B.; Cardell, L.O.; Langston, M.; Mobini, R.; Perkins, A.; Soini, J.; Ståhl, A.; Benson, M. Gender differences in inflammatory proteins and pathways in seasonal allergic rhinitis. Cytokine 2008, 42, 325–329. [Google Scholar] [CrossRef]
- Chen, W.; Mempel, M.; Schober, W.; Behrendt, H.; Ring, J. Gender difference, sex hormones, and immediate type hypersensitivity reactions. Allergy 2008, 63, 1418–1427. [Google Scholar] [CrossRef]
- Kraemer, U.; Sugiri, D.; Ranft, U.; Krutmann, J.; von Berg, A.; Berdel, D.; Behrendt, H.; Kuhlbusch, T.; Hochadel, M.; Wichmann, H.E.; et al. Eczema, respiratory allergies, and traffic-related air pollution in birth cohorts from small-town areas. J. Dermatol. Sci. 2009, 56, 99–105. [Google Scholar] [CrossRef]
- Kreutzer, R.; Neutra, R.R.; Lashuay, N. Prevalence of people reporting sensitivities to chemicals in a population-based survey. Am. J. Epidemiol. 1999, 150, 1–17. [Google Scholar] [CrossRef]
- Krewski, D.; Lemyre, L.; Turner, M.C.; Lee, J.E.C.; Dallaire, C.; Bouchard, L.; Brand, K.; Mercier, P. Public perception of population health risks in Canada: Health hazards and sources of information. Hum. Ecol. Risk Assess. 2006, 12, 626–644. [Google Scholar] [CrossRef]
- Abdel-Hafez, M.; Shimada, M.; Lee, P.Y.; Johnson, R.J.; Garin, E.H. Idiopathic nephrotic syndrome and atopy: Is there a common link? Am. J. Kidney Dis. 2009, 54, 945–953. [Google Scholar] [CrossRef]
- Siemiatycki, J.; Krewski, D.; Shi, Y.L.; Goldberg, M.S.; Nadon, L.; Lakhani, R. Controlling for potential confounding by occupational exposures. J. Toxicol. Environ. Health 2003, 66, 1591–1603. [Google Scholar] [CrossRef]
- Brook, R.D. Cardiovascular effects of air pollution. Clin. Sci. 2008, 115, 175–187. [Google Scholar] [CrossRef]
- Piro, F.N.; Madsen, C.; Næss, Ø.; Nafstad, P.; Claussen, B. A comparison of self reported air pollution problems and GIS-modeled levels of air pollution in people with and without chronic diseases. Environ. Health 2008, 7, 9–10. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Eisen, E.A.; Slade, M.D.; Kawachi, I.; Cullen, M.R. Gender and sex differences in job status and hypertension. Occup. Environ. Med. 2010, 68, 16–34. [Google Scholar]
- Edwards, R.D.; Schweizer, C.; Llacqua, V.; Lai, H.K.; Jantunen, M.; Bayer-Oglesby, L.; Kunzli, N. Time-activity relationships to VOC personal exposure factors. Atmos. Environ. 2006, 40, 5685–5700. [Google Scholar] [CrossRef]
- Keegan, T.H.M.; Hurley, S.; Goldberg, D.; Nelson, D.O.; Reynolds, P.; Bernstein, L.; Horn-Ross, P.L.; Gomez, S.L. The association between neighbourhood characteristics and body size and physical activity in the California teachers study cohort. Am. J. Public Health 2012, 102, 689–697. [Google Scholar] [CrossRef]
- Hu, W.B.; Mengerson, K.; McMichael, A.; Tong, S.L. Temperature, air pollution and total mortality during summers in Sydney, 1994–2004. Int. J. Biometeorol. 2008, 52, 689–696. [Google Scholar] [CrossRef]
- Forastiere, F.; Stafoggia, M.; Tasco, C.; Picciotto, S.; Agabiti, N.; Cesaroni, G.; Perucci, C.A. Socioeconomic status, particulate air pollution, and daily mortality: Differential exposure or differential susceptibility. Am. J. Ind. Med. 2007, 50, 208–216. [Google Scholar] [CrossRef]
- Peden, D.B. The epidemiology and genetics of asthma risk associated with air pollution. J. Allergy Clin. Immunol. 2005, 115, 213–219. [Google Scholar] [CrossRef]
- Larsson, U.; Taft, C.; Karlsson, J.; Sullivan, M. Gender and age differences in the relative burden of rhinitis and asthma on health-related quality of life—A Swedish population study. Respir. Med. 2007, 101, 1291–1298. [Google Scholar] [CrossRef]
- Butter, M.E. Are women more vulnerable to environmental pollution? J. Hum. Ecol. 2006, 20, 221–226. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oiamo, T.H.; Luginaah, I.N. Extricating Sex and Gender in Air Pollution Research: A Community-Based Study on Cardinal Symptoms of Exposure. Int. J. Environ. Res. Public Health 2013, 10, 3801-3817. https://doi.org/10.3390/ijerph10093801
Oiamo TH, Luginaah IN. Extricating Sex and Gender in Air Pollution Research: A Community-Based Study on Cardinal Symptoms of Exposure. International Journal of Environmental Research and Public Health. 2013; 10(9):3801-3817. https://doi.org/10.3390/ijerph10093801
Chicago/Turabian StyleOiamo, Tor H., and Isaac N. Luginaah. 2013. "Extricating Sex and Gender in Air Pollution Research: A Community-Based Study on Cardinal Symptoms of Exposure" International Journal of Environmental Research and Public Health 10, no. 9: 3801-3817. https://doi.org/10.3390/ijerph10093801
APA StyleOiamo, T. H., & Luginaah, I. N. (2013). Extricating Sex and Gender in Air Pollution Research: A Community-Based Study on Cardinal Symptoms of Exposure. International Journal of Environmental Research and Public Health, 10(9), 3801-3817. https://doi.org/10.3390/ijerph10093801





