The Changing Epidemiology of Kunjin Virus in Australia
Abstract
:1. Introduction
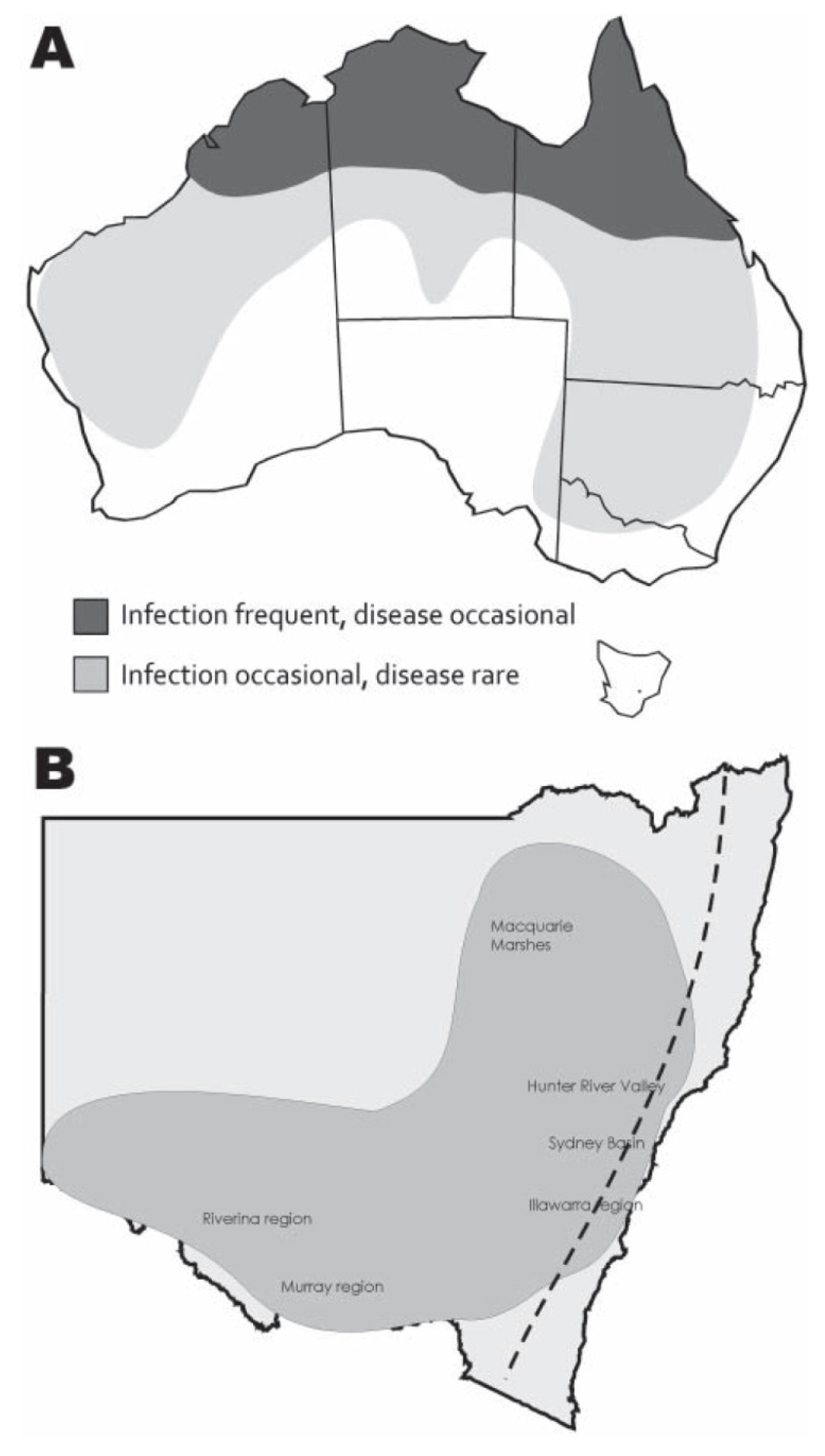
2. Ecology and Epidemiology
2.1. Vectors and Vertebrate Hosts
2.2. Surveillance Using Sentinel Chickens
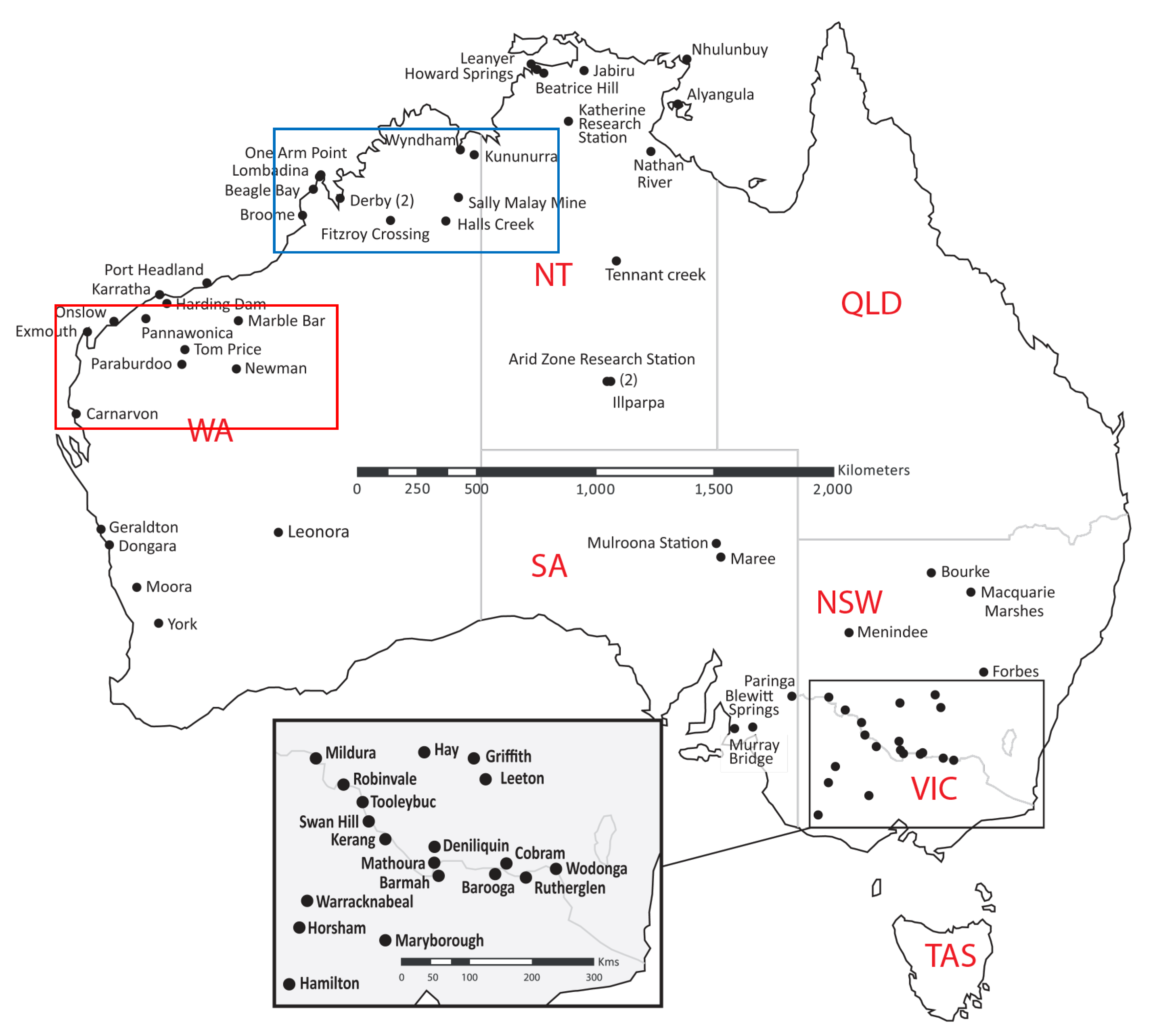
2.2. Epidemiology of Human Infections
| Year | MVEV | WNVKUN |
|---|---|---|
| Number of cases (location) | Number of cases (location) | |
| 2010–2011 * | 16 (2: NSW; 2: NT; 2: SA; 9: WA) | 1 (NT) |
| 2009–2010 # | 0 | 2 (1: NT; 1: QLD) |
| 2008–2009 € | 4 (2: NT; 2: WA) | 3 (1: NT; 2: QLD) |
| 2007–2008 $ | 2 (1: NSW; 1: WA) | 1 (VIC) |
| 2006–2007 ± | 0 | 0 |
| 2005–2006 ≠ | 1 (WA) | 2 (WA) |
| 2004–2005 § | 2 (1: NT; 1: QLD) | 4 (3: QLD; 1: VIC) |
3. Emergence of the First Virulent Strain in Australia to Cause an Outbreak
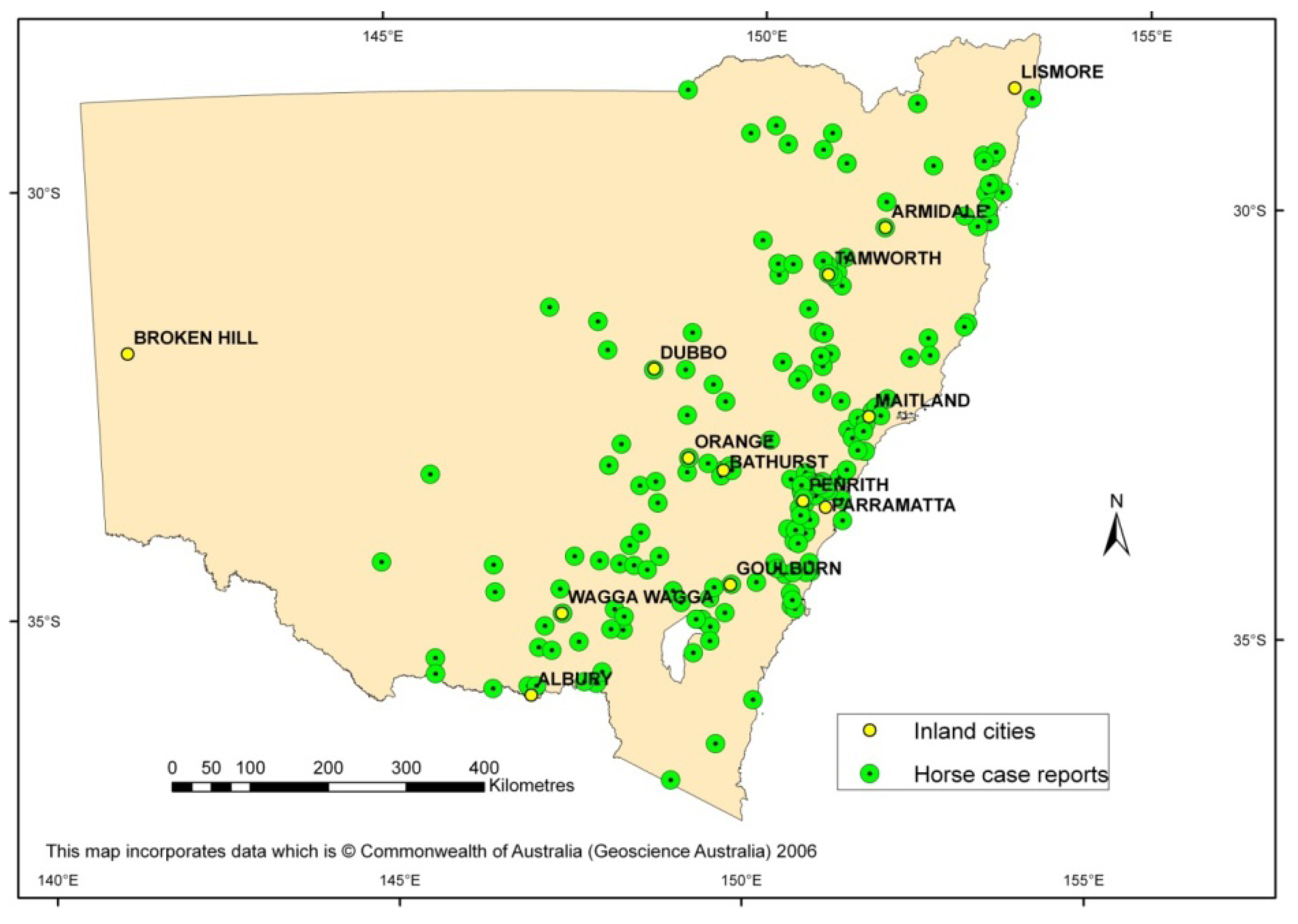
4. Evaluation of Virulence of Contemporary and Historical WNVKUN Strains
5. Discussion
6. Conclusions and Future Directions
Acknowledgments
Conflict of Interest
References
- Danis, K.; Papa, A.; Theocharopoulos, G.; Dougas, G.; Athanasiou, M.; Detsis, M.; Baka, A.; Lytras, T.; Mellou, K.; Bonovas, S.; et al. Outbreak of West Nile virus infection in Greece, 2010. Emerg. Infect. Dis. 2011, 17, 1868–1872. [Google Scholar]
- Frost, M.J.; Zhang, J.; Edmonds, J.H.; Prow, N.A.; Gu, X.; Davis, R.; Hornitzky, C.; Arzey, K.E.; Finlaison, D.; Hick, P.; et al. Characterization of virulent West Nile virus Kunjin strain, Australia, 2011. Emerg. Infect. Dis. 2012, 18, 792–800. [Google Scholar]
- Garcia-Bocanegra, I.; Jaén-Téllez, J.A.; Napp, S.; Arenas-Montes, A.; Fernández-Morente, M.; Fernández-Molera, V.; Arenas, A. West Nile fever outbreak in horses and humans, Spain, 2010. Emerg. Infect. Dis. 2011, 17, 2397–2399. [Google Scholar] [CrossRef]
- Kutasi, O.; Bakonyi, T.; Lecollinet, S.; Biksi, I.; Ferenczi, E.; Bahuon, C.; Sardi, S.; Zientara, S.; Szenci, O. Equine encephalomyelitis outbreak caused by a genetic lineage 2 West Nile virus in Hungary. J. Vet. Intern. Med. 2011, 25, 586–591. [Google Scholar] [CrossRef]
- Lanciotti, R.S.; Roehrig, J.T.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef]
- Mann, R.A.; Fegan, M.; O’Riley, K.; Motha, J.; Warner, S. Molecular characterization and phylogenetic analysis of Murray Valley encephalitis virus and West Nile virus (Kunjin subtype) from an arbovirus disease outbreak in horses in Victoria, Australia, in 2011. J. Vet. Diagn. Invest. 2013, 25, 35–44. [Google Scholar] [CrossRef]
- Murray, K.O.; Mertens, E.; Despres, P. West Nile virus and its emergence in the United States of America. Vet. Res. 2010, 41. [Google Scholar] [CrossRef]
- Roche, S.E.; Wicks, R.; Garner, M.G.; East, I.J.; Paskin, R.; Moloney, B.J.; Carr, M.; Kirkland, P. Descriptive overview of the 2011 epidemic of arboviral disease in horses in Australia. Aust. Vet. J. 2013, 91, 5–13. [Google Scholar] [CrossRef]
- Spissu, N.; Panichi, G.; Montisci, A.; Fiore, F. West Nile virus outbreak in Sardinia, Italy, in 2011. J. Infect. Dev. Ctries. 2013, 7, 6–9. [Google Scholar]
- Mackenzie, J.S.; Williams, D.T. The zoonotic flaviviruses of southern, south-eastern and eastern Asia, and Australasia: the potential for emergent viruses. Zoonoses Public Health 2009, 56, 338–356. [Google Scholar] [CrossRef]
- May, F.J.; Davis, C.T.; Tesh, R.B.; Barrett, A.D. Phylogeography of West Nile virus: from the cradle of evolution in Africa to Eurasia, Australia, and the Americas. J. Virol. 2011, 85, 2964–2974. [Google Scholar] [CrossRef]
- Badman, R.; Campbell, J.; Aldred, J. Arbovirus infection in horses—Victoria. Commun. Dis. Intell. 1984, 17, 5–6. [Google Scholar]
- Flynn, L.M.; Coelen, R.J.; Mackenzie, J.S. Kunjin virus isolates of Australia are genetically homogeneous. J. Gen. Virol. 1989, 70, 2819–2824. [Google Scholar] [CrossRef]
- Hall, R.A.; Broom, A.K.; Smith, D.W.; Mackenzie, J.S. The ecology and epidemiology of Kunjin virus. Curr. Top. Microbiol. Immunol. 2002, 267, 253–269. [Google Scholar] [CrossRef]
- Marshall, I. Murray Valley and Kunjin Encpehalitis. In The Arboviruses: Epidemiology and Ecology; Monath, T.P., Ed.; CRC Press: Boca Raton, FL, USA, 1988; pp. 151–189. [Google Scholar]
- Mackenzie, J.S.; Lindsay, M.D.; Coelen, R.J.; Broom, A.K.; Hall, R.A.; Smith, D.W. Arboviruses causing human disease in the Australasian zoogeographic region. Arch. Virol. 1994, 136, 447–467. [Google Scholar] [CrossRef]
- Prow, N.A.; Hall, R.A.; Lobigs, M. Murray Valley Encephalitis Virus. In Neuroviral Infections; Singh, S.K., Ruzek, D., Eds.; Taylor & Francis Group: Boca Raton, FL, USA, 2012; pp. 167–192. [Google Scholar]
- Knox, J.; Cowan, R.U.; Doyle, J.S.; Ligtermoet, M.K.; Archer, J.S.; Burrow, J.N.; Tong, S.Y.; Currie, B.J.; Mackenzie, J.S.; Smith, D.W.; et al. Murray Valley encephalitis: A review of clinical features, diagnosis and treatment. Med. J. Aust. 2012, 196, 322–326. [Google Scholar]
- Gordon, A.N.; Marbach, C.R.; Oakey, J.; Edmunds, G.; Condon, K.; Diviney, S.M.; Williams, D.T.; Bingham, J. Confirmed case of encephalitis caused by Murray Valley encephalitis virus infection in a horse. J. Vet. Diagn. Invest. 2012, 24, 431–436. [Google Scholar] [CrossRef]
- Holmes, J.M.; Gilkerson, J.R.; El Hage, C.M.; Slocombe, R.F.; Muurlink, M.A. Murray Valley encephalomyelitis in a horse. Aust. Vet. J. 2012, 90, 252–254. [Google Scholar] [CrossRef]
- Prow, N.A.; Tan, C.S.E.; Wang, W.; Hobson-Peters, J.; Kidd, L.; Barton, A.; Wright, J.; Hall, R.A.; Bielefeldt-Ohmann, H. Natural exposure of horses to mosquito-borne flaviviruses in South-East Queensland, Australia. Int. J. Environ. Res. Public Health 2013, 10, 4432–4443. [Google Scholar] [CrossRef]
- Russell, R.C. Arboviruses and their vectors in Australia: An update on the ecology and epidemiology of some mosquito-borne arboviruses. Rev. Med. Vet. Entomol. 1995, 83, 141–158. [Google Scholar]
- Broom, A.K.; Mackenzie, J.S.; Lindsay, M.D.; Wright, A.E. Epidemiology of Murray Valley encephalitis and Kunjin viruses in Western Australia. Arbovirus Res. Aust. 1989, 5, 14–18. [Google Scholar]
- Kay, B.H.; Boreham, P.F.; Fanning, I.D. Host-feeding patterns of Culex annulirostris and other mosquitoes (Diptera: Culicidae) at Charleville, southwestern Queensland, Australia. J. Med. Entomol. 1985, 22, 529–535. [Google Scholar]
- Australian Government Department of Health and Ageing. National Arbovirus and Malaria Advisory Committee (NAMAC) Annual Reports. Available online: http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-arboanrep.htm (accessed on 1 Aug 2013).
- Knope, K.; Whelan, P.; Smith, D.; Johansen, C.; Moran, R.; Doggett, S.; Sly, A.; Hobby, M.; Kurucz, N.; Wright, P.; et al. Arboviral diseases and malaria in Australia, 2010-11: Annual report of the National Arbovirus and Malaria Advisory Committee. Commun. Dis. Intell. Q. Rep. 2013, 37, E1–E20. [Google Scholar]
- Fitzsimmons, G.J.; Wright, P.; Johansen, C.A.; Whelan, P.I. Arboviral diseases and malaria in Australia, 2007/08: Annual report of the National Arbovirus and Malaria Advisory Committee. Commun. Dis. Intell. Q. Rep. 2009, 33, 155–169. [Google Scholar]
- Wright, P.; Fitzsimmons, G.J.; Johansen, C.A.; Whelan, P.I. Arboviral diseases and malaria in Australia, 2009–10: Annual report of the National Arbovirus and Malaria Advisory Committee. Commun. Dis. Intell. Q. Rep. 2012, 36, 70–81. [Google Scholar]
- Liu, C.; Begg, K.; Johansen, C.; Whelan, P.; Kurucz, N.; Melville, L. Communicable diseases network Australia National Arbovirus And Malaria Advisory Committee annual report, 2006-07. Commun. Dis. Intell. Q. Rep. 2008, 32, 31–47. [Google Scholar]
- Liu, C.; Johansen, C.; Kurucz, N.; Whelan, P. Communicable diseases network Australia National Arbovirus And Malaria Advisory Committee annual report, 2005–06. Commun. Dis. Intell. Q. Rep. 2006, 30, 411–429. [Google Scholar]
- Jacups, S.; Kurucz, N.; Whitters, R.; Whelan, P. Habitat modification for mosquito control in the Ilparpa Swamp, Northern Territory, Australia. J. Vector Ecol. 2011, 36, 292–299. [Google Scholar] [CrossRef]
- Forbes, J.A. Murray Valley Encephalitis 1974: Also the Epidemic Variance Since 1914 and Predisposing Rainfall Patterns; Australasian Medical Publishing Company: Sydney, Australia, 1978. [Google Scholar]
- Nicholls, N. A method for predicting Murray Valley encephalitis in southeast Australia using the Southern Oscillation. Aust. J. Exp. Biol. Med. Sci. 1986, 64, 587–594. [Google Scholar] [CrossRef]
- Schuster, G.; Ebert, E.E.; Stevenson, M.A.; Corner, R.J.; Johansen, C.A. Application of satellite precipitation data to analyse and model arbovirus activity in the tropics. Int. J. Health Geogr. 2011, 10. [Google Scholar] [CrossRef]
- Russell, R.C. Mosquito-borne arboviruses in Australia: The current scene and implications of climate change for human health. Int. J. Parasitol. 1998, 28, 955–969. [Google Scholar] [CrossRef]
- Kurucz, N.; Gray, T.J.; Burrow, J.; Whelan, P. A confirmed case of Kunjin virus disease encephalitis acquired in rural Darwin, NT—The mosquito story. North. Territ. Dis. Control Bull. 2010, 17, 5–10. [Google Scholar]
- Rogers, B.A.; Hueston, L.; Ratnam, I. Imported West Nile virus encephalitis in an Israeli tourist. Med. J. Aust. 2009, 191, 232–234. [Google Scholar]
- Fitzsimmons, G.J.; Wright, P.; Johansen, C.A.; Whelan, P.I. Arboviral diseases and malaria in Australia, 2008–09: Annual report of the National Arbovirus and Malaria Advisory Committee. Commun. Dis. Intell. Q. Rep. 2010, 34, 225–240. [Google Scholar]
- Liu, C.; Broom, A.K.; Kurucz, N.; Whelan, P.I. Communicable Diseases Network Australia: National Arbovirus and Malaria Advisory Committee annual report 2004-05. Commun. Dis. Intell. Q. Rep. 2005, 29, 341–357. [Google Scholar]
- Ostlund, E.N.; Crom, R.L.; Pedersen, D.D.; Johnson, D.J.; Williams, W.O.; Schmitt, B.J. Equine West Nile encephalitis, United States. Emerg. Infect. Dis. 2001, 7, 665–669. [Google Scholar]
- Beasley, D.W.; Li, L.; Suderman, M.T.; Barrett, A.D. Mouse neuroinvasive phenotype of West Nile virus strains varies depending upon virus genotype. Virology 2002, 296, 17–23. [Google Scholar] [CrossRef]
- Laurent-Rolle, M.; Boer, E.F.; Lubick, K.J.; Wolfinbarger, J.B.; Carmody, A.B.; Rockx, B.; Liu, W.; Ashour, J.; Shupert, W.L.; Holbrook, M.R.; et al. The NS5 protein of the virulent West Nile virus NY99 strain is a potent antagonist of type I interferon-mediated JAK-STAT signaling. J. Virol. 2010, 84, 3503–3515. [Google Scholar] [CrossRef]
- Bingham, J.; Lunt, R.A.; Green, D.J.; Davies, K.R.; Stevens, V.; Wong, F.Y. Experimental studies of the role of the little raven (Corvus mellori) in surveillance for West Nile virus in Australia. Aust. Vet. J. 2010, 88, 204–210. [Google Scholar] [CrossRef]
- Hall, R.A.; Tan, S.E.; Selisko, B.; Slade, R.; Hobson-Peters, J.; Canard, B.; Hughes, M.; Leung, J.Y.; Balmori-Melian, E.; Hall-Mendelin, S.; et al. Monoclonal antibodies to the West Nile virus NS5 protein map to linear and conformational epitopes in the methyltransferase and polymerase domains. J. Gen. Virol. 2009, 90, 2912–2922. [Google Scholar] [CrossRef]
- Walker, B. Kunjin virus neurological disease in horses. NSW Gov. 2012. Available online: http://www.dpi.nsw.gov.au/__data/assets/pdf_file/0004/417325/Kunjin-virus-neurological-disease-in-horses.pdf (accessed on 15 Aug 2013).
- Kay, B.H.; Fanning, I.D.; Carley, J.G. The vector competence of Australian Culex annulirostris with Murray Valley encephalitis and Kunjin viruses. Aust. J. Exp. Biol. Med. Sci. 1984, 62, 641–650. [Google Scholar] [CrossRef]
- Jansen, C.C.; Webb, C.E.; Northill, J.A.; Ritchie, S.A.; Russell, R.C.; Van den Hurk, A.F. Vector competence of Australian mosquito species for a North American strain of West Nile virus. Vector Borne Zoonotic Dis. 2008, 8, 805–811. [Google Scholar]
- Crabtree, M.B.; Sang, R.C.; Stollar, V.; Dunster, L.M.; Miller, B.R. Genetic and phenotypic characterization of the newly described insect flavivirus, Kamiti River virus. Arch. Virol. 2003, 148, 1095–1118. [Google Scholar] [CrossRef]
- Hoshino, K.; Isawa, H.; Tsuda, Y.; Yano, K.; Sasaki, T.; Yuda, M.; Takasaki, T.; Kobayashi, M.; Sawabe, K. Genetic characterization of a new insect flavivirus isolated from Culex pipiens mosquito in Japan. Virology 2007, 359, 405–414. [Google Scholar] [CrossRef]
- Cook, S.; Moureau, G.; Harbach, R.E.; Mukwaya, L.; Goodger, K.; Ssenfuka, F.; Gould, E.; Holmes, E.C.; de Lamballerie, X. Isolation of a novel species of flavivirus and a new strain of Culex flavivirus (Flaviviridae) from a natural mosquito population in Uganda. J. Gen. Virol. 2009, 90, 2669–2678. [Google Scholar] [CrossRef]
- Crabtree, M.B.; Nga, P.T.; Miller, B.R. Isolation and characterization of a new mosquito flavivirus, Quang Binh virus, from Vietnam. Arch. Virol. 2009, 154, 857–860. [Google Scholar] [CrossRef]
- Bolling, B.G.; Olea-Popelka, F.J.; Eisen, L.; Moore, C.G.; Blair, C.D. Transmission dynamics of an insect-specific flavivirus in a naturally infected Culex pipiens laboratory colony and effects of co-infection on vector competence for West Nile virus. Virology 2012, 427, 90–97. [Google Scholar] [CrossRef]
- Kent, R.J.; Crabtree, M.B.; Miller, B.R. Transmission of West Nile virus by Culex quinquefasciatus say infected with Culex Flavivirus Izabal. PLoS Negl. Trop. Dis. 2010, 4. [Google Scholar] [CrossRef]
- Hobson-Peters, J.; Yam, A.W.; Lu, J.W.; Setoh, Y.X.; May, F.J.; Kurucz, N.; Walsh, S.; Prow, N.A.; Davis, S.S.; Weir, R.; et al. A new insect-specific flavivirus from northern Australia suppresses replication of West Nile virus and Murray Valley encephalitis virus in co-infected mosquito cells. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Marshall, I.D.; Brown, B.K.; Keith, K.; Gard, G.P.; Thibos, E. Variation in arbovirus infection rates in species of birds sampled in a serological survey during an encephalitis epidemic in the Murray Valley of South-eastern Australia, February 1974. Aust. J. Exp. Biol. Med. Sci. 1982, 60, 471–478. [Google Scholar] [CrossRef]
- Russell, R.C.; Kay, B.H. Medical entomology: changes in the spectrum of mosquito-borne disease in Australia and other vector threats and risks, 1972–2004. Aust. J. Entomol. 2004, 43, 271–282. [Google Scholar] [CrossRef]
- Komar, N.; Langevin, S.; Hinten, S.; Nemeth, N.; Edwards, E.; Hettler, D.; Davis, B.; Bowen, R.; Bunning, M. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 2003, 9, 311–322. [Google Scholar] [CrossRef]
- Epstein, J.H.; McKee, J.; Shaw, P.; Hicks, V.; Micalizzi, G.; Daszak, P.; Kilpatrick, A.M.; Kaufman, G. The Australian White Ibis (Threskiornis molucca) as a reservoir of zoonotic and livestock pathogens. EcoHealth 2007, 3, 290–298. [Google Scholar] [CrossRef]
- Kay, B.H.; Hall, R.A.; Fanning, I.D.; Young, P.L. Experimental infection with Murray Valley encephalitis virus: Galahs, sulphur-crested cockatoos, corellas, black ducks and wild mice. Aust. J. Exp. Biol. Med. Sci. 1985, 63, 599–606. [Google Scholar]
- Tiawsirisup, S.; Platt, K.B.; Tucker, B.J.; Rowley, W.A. Eastern cottontail rabbits (Sylvilagus floridanus) develop West Nile virus viremias sufficient for infecting select mosquito species. Vector Borne Zoonotic Dis. 2005, 5, 342–350. [Google Scholar] [CrossRef]
- Suen, W; Prow, N.A.; Wang, W.; Broad, N.; Hall, R.A.; Kirkland, P.D.; Bielefeldt-Ohmann, H. The establishment of a rabbit model to elucidate the mechanism of neuroinvasion by an emergent Australian West Nile virus. Unpublished work.
- van den Hurk, A.F.; Smith, C.S.; Field, H.E.; Smith, I.L.; Northill, J.A.; Taylor, C.T.; Jansen, C.C.; Smith, G.A.; Mackenzie, J.S. Transmission of Japanese Encephalitis virus from the black flying fox, Pteropus alecto, to Culex annulirostris mosquitoes, despite the absence of detectable viremia. Am. J. Trop. Med. Hyg. 2009, 81, 457–462. [Google Scholar]
- Austgen, L.E.; Bowen, R.A.; Bunning, M.L.; Davis, B.S.; Mitchell, C.J.; Chang, G.J. Experimental infection of cats and dogs with West Nile virus. Emerg. Infect. Dis. 2004, 10, 82–86. [Google Scholar] [CrossRef]
- Prow, N.A.; Edmonds, J.H.; Setoh, Y.X.; Bielefeldt-Ohmann, H.; Suen, W.; van den Hurk, A.F.; Pyke, A.; Hall-Mendelin, S.; Warrilow, D.; et al. Investigating the origin of an emerging virulent strain of West Nile virus in Australia. Emerg. Infect. Dis. In preparation.
- Adams, S.C.; Broom, A.K.; Sammels, L.M.; Hartnett, A.C.; Howard, M.J.; Coelen, R.J.; Mackenzie, J.S.; Hall, R.A. Glycosylation and antigenic variation among Kunjin virus isolates. Virology 1995, 206, 49–56. [Google Scholar] [CrossRef]
- Hall, R.A.; Scherret, J.H.; Mackenzie, J.S. Kunjin virus: An Australian variant of West Nile? Ann. N. Y. Acad. Sci. 2001, 951, 153–160. [Google Scholar]
- Hobson-Peters, J.; Toye, P.; Sanchez, M.D.; Bossart, K.N.; Wang, L.F.; Clark, D.C.; Cheah, W.Y.; Hall, R.A. A glycosylated peptide in the West Nile virus envelope protein is immunogenic during equine infection. J. Gen. Virol. 2008, 89, 3063–3072. [Google Scholar] [CrossRef]
- Scherret, J.H.; Poidinger, M.; Mackenzie, J.S.; Broom, A.K.; Deubel, V.; Lipkin, W.I.; Briese, T.; Gould, E.A.; Hall, R.A. The relationships between West Nile and Kunjin viruses. Emerg. Infect. Dis. 2001, 7, 697–705. [Google Scholar]
- Williams, S.A.; Richards, J.S.; Faddy, H.M.; Leydon, J.; Moran, R.; Nicholson, S.; Perry, F.; Paskin, R.; Catton, M.; Lester, R.; Mackenzie, J.S. Low seroprevalence of Murray Valley Encephalitis and Kunjin viruses in an opportunistic serosurvey, Victoria 2011. Aust. N. Z. J. Public Health 2013, 37, 427–433. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. West Nile Virus. Available online: http://www.cdc.gov/westnile/statsMaps/final.html (accessed on 1 September 2013).
- Petersen, L.R.; Brault, A.C.; Nasci, R.S. West Nile virus: Review of the literature. JAMA 2013, 310, 308–315. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. . Mosquito Species in Which West Nile Virus has been Detected, United States, 1999–2012. 2012. Available online: http://www.cdc.gov/westnile/resources/pdfs/Mosquito%20Species%201999-2012.pdf (accessed on 1 September 2013).
- Kay, B.H.; Boreham, P.F.L.; Williams, G.M. Host preferences and feeding patterns of mosquitoes at Kowanyama, Cape York Peninsula, northern Queensland. Bull. Entomol. Res. 1979, 69, 441–457. [Google Scholar] [CrossRef]
- Russell, R.C. A review of the status and significance of the species within the Culex pipiens group in Australia. J. Am. Mosq. Control. Assoc. 2012, 28, 24–27. [Google Scholar] [CrossRef]
- Clark, L.; Hall, J.; McLean, R.; Dunbar, M.; Klenk, K.; Bowen, R.; Smeraski, C.A. Susceptibility of greater sage-grouse to experimental infection with West Nile virus. J. Wildl. Dis. 2006, 42, 14–22. [Google Scholar] [CrossRef]
- McLean, R.G.; Ubico, S.R.; Docherty, D.E.; Hansen, W.R.; Sileo, L.; McNamara, T.S. West Nile virus transmission and ecology in birds. Ann. N Y Acad. Sci. 2001, 951, 54–57. [Google Scholar]
- Moudy, R.M.; Meola, M.A.; Morin, L.L.; Ebel, G.D.; Kramer, L.D. A newly emergent genotype of West Nile virus is transmitted earlier and more efficiently by Culex mosquitoes. Am. J. Trop. Med. Hyg. 2007, 77, 365–370. [Google Scholar]
- Anderson, J.F.; Main, A.J.; Cheng, G.; Ferrandino, F.J.; Fikrig, E. Horizontal and vertical transmission of West Nile virus genotype NY99 by Culex salinarius and genotypes NY99 and WN02 by Culex tarsalis. Am. J. Trop. Med. Hyg. 2012, 86, 134–139. [Google Scholar] [CrossRef]
- McMullen, A.R.; May, F.J.; Li, L.; Guzman, H.; Bueno, R., Jr.; Dennett, J.A.; Tesh, R.B.; Barrett, A.D. Evolution of new genotype of West Nile virus in North America. Emerg. Infect. Dis. 2011, 17, 785–793. [Google Scholar] [CrossRef]
- Mann, B.R.; McMullen, A.R.; Guzman, H.; Tesh, R.B.; Barrett, A.D. Dynamic transmission of West Nile virus across the United States-Mexican border. Virology 2013, 436, 75–80. [Google Scholar]
- Pealer, L.N.; Marfin, A.A.; Petersen, L.R.; Lanciotti, R.S.; Page, P.L.; Stramer, S.L.; Stobierski, M.G.; Signs, K.; Newman, B.; Kapoor, H.; et al. Transmission of West Nile virus through blood transfusion in the United States in 2002. N Engl. J. Med. 2003, 349, 1236–1245. [Google Scholar] [CrossRef]
- Petersen, L.R.; Busch, M.P. Transfusion-transmitted arboviruses. Vox Sang. 2010, 98, 495–503. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Prow, N.A. The Changing Epidemiology of Kunjin Virus in Australia. Int. J. Environ. Res. Public Health 2013, 10, 6255-6272. https://doi.org/10.3390/ijerph10126255
Prow NA. The Changing Epidemiology of Kunjin Virus in Australia. International Journal of Environmental Research and Public Health. 2013; 10(12):6255-6272. https://doi.org/10.3390/ijerph10126255
Chicago/Turabian StyleProw, Natalie A. 2013. "The Changing Epidemiology of Kunjin Virus in Australia" International Journal of Environmental Research and Public Health 10, no. 12: 6255-6272. https://doi.org/10.3390/ijerph10126255
APA StyleProw, N. A. (2013). The Changing Epidemiology of Kunjin Virus in Australia. International Journal of Environmental Research and Public Health, 10(12), 6255-6272. https://doi.org/10.3390/ijerph10126255




