Enhancement of Lutein Production in Chlorella sorokiniana (Chorophyta) by Improvement of Culture Conditions and Random Mutagenesis
Abstract
:1. Introduction
2. Results
2.1. Screening of Different Species of Chlorophycean Microalgae for Lutein Production
2.2. Effect of Some Environmental and Nutritional Factors on Growth and Lutein Content in C. sorokiniana
2.2.1. Growth and Lutein Accumulation under Standard Conditions
2.2.2. Effect of Irradiance
2.2.3. Effect of Temperature
2.2.4. Effect of Nitrogen
2.2.5. Effect of NaCl
2.2.6. Effect of Acetate and Glucose (Mixotrophic Culture)
2.3. Isolation of High Lutein Yielding Mutants of C. sorokiniana
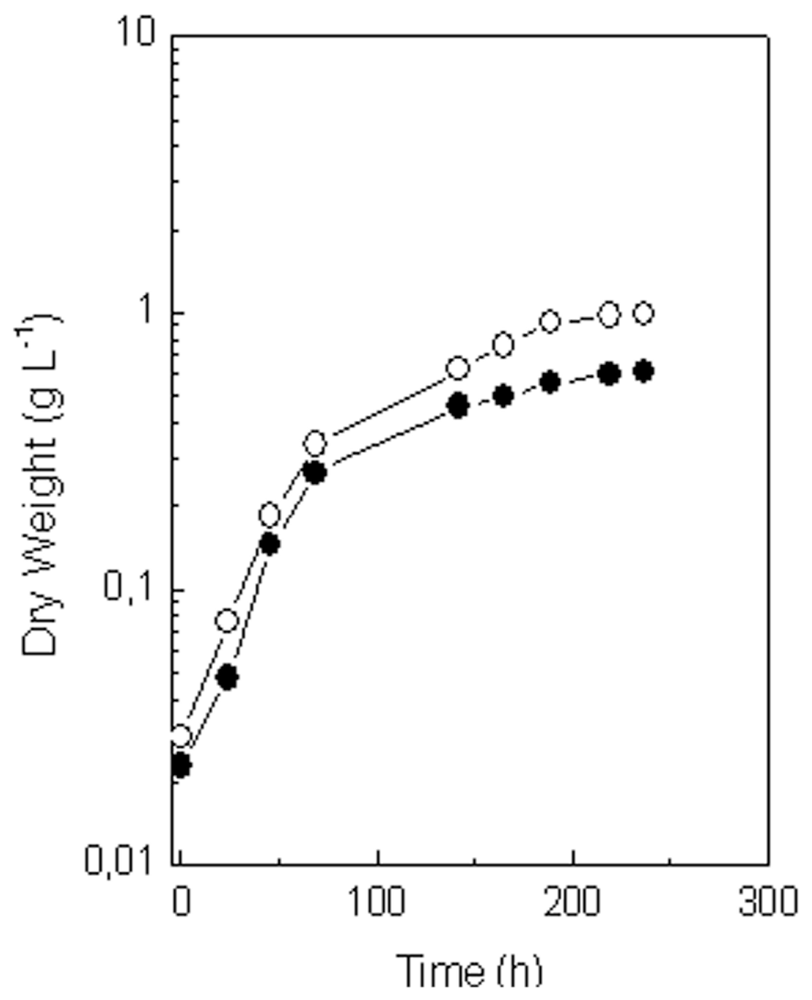
2.4. Comparison of Growth and Lutein Accumulation in the Selected Mutant (MR-16) and in the Wild Type of C. sorokiniana under Best Photoautotrophic and Mixotrophic Culture Conditions
3. Discussion
3.1. Effect of Several Environmental and Nutritional Factors on Lutein Production by C. sorokiniana under Photoautotrophic Conditions
3.2. Lutein Production by C. sorokiniana under Mixotrophic Conditions
3.3. Enhancement of Lutein Yield in C. sorokiniana by Random Mutagenesis
3.4. C. sorokiniana as a Promising Lutein Producer for Commercial Applications
4. Experimental Section
4.1. Organisms
4.2. Culture Conditions
4.2.1. Standard Culture Conditions
4.2.2. Mixotrophic Culture Conditions
4.3. Random Mutagenesis and Selection of High Lutein Yielding Mutants of C. sorokiniana
4.4. Analytical Methods
5. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Boussiba, S. Carotenogenesis in the green alga Haematococcus pluvialis: Cellular physiology and stress response. Physiol. Plant 2000, 108, 111–117. [Google Scholar]
- Demming-Adams, B; Adams, WW, III. Antioxidants in photosynthesis and human nutrition. Science 2002, 298, 2149–2153. [Google Scholar]
- Jin, E; Juergen, EWP; Hong, KL; Sang, MH; Man, C. Xanthophylls in microalgae: From biosynthesis to biotechnological mass production and application. J. Microbiol. Biotechnol 2003, 13, 165–174. [Google Scholar]
- Johnson, EA; Schroeder, WA. Advances in Biochemical Engineering, Biotechnology. In Microbial Carotenoids; Fiechter, A, Ed.; Springer: Berlin, Germany, 1995; Volume 53, pp. 119–178. [Google Scholar]
- Alves-Rodrigues, A; Shao, A. The science behing lutein. Toxicol. Lett 2004, 150, 57–83. [Google Scholar]
- Carpentier, S; Knaus, M; Such, M. Associations between lutein, zeaxanthin, and age-related macular degeneration: An overview. Crit. Rev. Food Sci. Nutr 2009, 49, 313–326. [Google Scholar]
- Chin, CJ; Taylor, A. Nutritional antioxidants and age-related cataract and macular degeneration. Exp. Eye Res 2007, 84, 229–245. [Google Scholar]
- Whitehead, AJ; Mares, JA; Danis, RP. Macular pigment: A review of current knowledge. Arch. Ophtalmol 2006, 124, 1038–1045. [Google Scholar]
- Arnal, E; Miranda, M; Almansa, I; Muriach, M; Barcia, JM; Romero, FJ; Díaz-Llopis, M; Bosch Moreer, F. Lutein prevents cataract development and progression in diabetic rats. Graefes Arch. Clin. Exp. Ophthalmol 2009, 247, 115–120. [Google Scholar]
- Dwyer, JH; Navab, M; Dwyer, KM; Hassan, K; Sun, P; Shircore, A; Hama-Levy, S; Hough, G; Wang, X; Drake, T; Merz, NB; Fogelman, AM. Oxygenated carotenoid lutein and the progression of early atherosclerosis. Circulation 2001, 103, 2922–2977. [Google Scholar]
- Kim, HW; Chew, BP; Wong, TS; Park, JS; Weng, BBC; Byrne, KM; Hayek, MG; Reinhart, GA. Modulation of humoral and cell-mediated immune responses by dietary lutein in cats. Vet. Immunol. Immunopathol 2000, 73, 331–341. [Google Scholar]
- Kim, HW; Chew, BP; Wong, TS; Park, JS; Weng, BBC; Byrne, KM; Hayek, MG; Reinhart, GA. Dietary lutein stimulates immune response in the canine. Vet. Immunol. Immunopathol 2000, 74, 315–327. [Google Scholar]
- Basu, HN; Del Vecchio, AJ; Flider, F; Orthoefer, FT. Nutritional and potential disease prevention properties of Carotenoids. J. Am. Oil Chem. Soc 2001, 78, 665–675. [Google Scholar]
- Del Campo, JA; Garcia-Gonzalez, M; Guerrero, MG. Outdoor cultivation of microalgae for carotenoid production: Current state and perspectives. Appl. Microbiol. Biotechnol 2007, 74, 1163–1174. [Google Scholar]
- Piccaglia, R; Marotti, M; Grandi, S. Lutein and lutein ester content in different types of Tagetes patula and T. erecta. Ind. Crops Prod 1998, 8, 45–51. [Google Scholar]
- Del Campo, JA; Moreno, J; Rodriguez, H; Vargas, MA; Rivas, J; Guerrero, MG. Carotenoid content of chlorophycean microalgae: Factors determining lutein accumulation in Muriellopsis sp. (Chlorophyta). J. Biotechnol 2000, 76, 51–59. [Google Scholar]
- Del Campo, JA; Rodriguez, H; Moreno, J; Vargas, MA; Rivas, J; Guerrero, MG. Lutein production by Muriellopsis sp. in an outdoor tubular photobioreactor. J. Biotechnol 2001, 85, 289–295. [Google Scholar]
- Del Campo, JA; Rodriguez, H; Moreno, J; Vargas, MA; Rivas, J; Guerrero, MG. Accumulation of astaxanthin and lutein in Chlorella zofingiensis (Chlorophyta). Appl. Microbiol. Biotechnol 2004, 64, 848–854. [Google Scholar]
- Casal, C; Cuaresma, M; Vega, JM; Vilchez, C. Enhanced productivity of a lutein-enriched novel acidophile microalga grown on urea. Mar. Drugs 2011, 9, 29–42. [Google Scholar]
- Sanchez, JF; Fernandez, JM; Acien, FG; Rueda, A; Perez-Parra, J; Molina, E. Influence of culture conditions on the productivity and lutein content of the new strain Scenedesmus almeriensis. Process Biochem 2008, 43, 398–405. [Google Scholar]
- Shi, XM; Jiang, Y; Chen, F. High-yield production of lutein by the green microalga Chlorella protothecoides in heterotrophic fed-batch culture. Biotechnol. Prog 2002, 18, 723–727. [Google Scholar]
- Fischer, R. Isolation of Mutants, a Key for the Analysis of Complex Pathways and for Strain Improvement. In Microbes for Health, Wealth and Sustainable Environment; Verma, A, Ed.; Malhotra Publishing House: New Delhi, India, 1998; pp. 739–751. [Google Scholar]
- Chen, Y; Li, D; Lu, W; Xing, J; Hui, B; Han, Y. Screening and characterization of astaxanthin-hyperproducing mutants of Haematococcus pluvialis. Biotechnol. Lett 2003, 25, 527–529. [Google Scholar]
- Tjahjono, AE; Hayama, Y; Kakizono, T; Terada, Y; Nishio, N; Nagai, S. Hyper-accumulation of astaxanthin in a green alga Haematococcus pluvialis at elevated temperatures. Biotechnol. Lett 1994, 16, 133–138. [Google Scholar]
- Jin, E; Melis, A. A mutant of the green alga Dunaliella salina constitutively accumulates zeaxanthin under all growth conditions. Biotechnol. Bioeng 2003, 81, 115–124. [Google Scholar]
- Bar, E; Rise, M; Vishkantsan, H; Arad, S. Pigment and structural changes in Chlorella zofingiensis upon light and nitrogen stress. J. Plant Physiol 1995, 146, 527–534. [Google Scholar]
- Bousiba, S; Vonshak, A. Astaxanthin accumulation in the green alga Haematococcus pluvialis. Plant Cell Physiol 1991, 32, 1077–1082. [Google Scholar]
- Fernandez-Sevilla, JM; Acien, FG; Molina, E. Biotechnological production of lutein and its applications. Appl. Microbiol. Biotechnol 2010, 86, 27–40. [Google Scholar]
- Shi, XM; Zhang, XW; Chen, F. Heterotrophic production of biomass and lutein by Chlorella protothecoides on various nitrogen sources. Enzym. Microb. Technol 2000, 27, 312–318. [Google Scholar]
- Harker, M; Tsavalos, AJ; Young, AJ. Factors responsible for astaxanthin formation in the chlorophyte Haematococcus pluvialis. Bioresour. Technol 1996, 55, 207–214. [Google Scholar]
- Vidhyavathi, R; Venkatachalam, L; Sarada, R; Ravishankar, GA. Regulation of carotenoid biosynthetic genes expression and carotenoid accumulation in the green alga Haematococcus pluvialis under nutrient stress conditions. J. Exp. Bot 2008, 59, 1409–1418. [Google Scholar]
- Borowitzka, MA; Huisman, JM; Osborn, A. Culture of the astaxanthin-producing green alga Haematococcus pluvialis. 1. Effects of nutrients on growth and cell type. J. Appl. Phycol 1991, 3, 295–304. [Google Scholar]
- Li, Y; Huang, J; Sandmann, G; Chen, F. Glucose sensing and the mitochondrial alternative pathway are involved in the regulation of astaxanthin biosynthesis in the dark-grown Chlorella zofingiensis (Chlorophyceae). Planta 2008, 228, 735–743. [Google Scholar]
- Huang, JC; Wang, Y; Sandmann, G; Chen, F. Isolation and characterization of a carotenoid oxygenase gene from Chlorella zofingiensis (Chlorophyta). Appl. Microbiol. Biotechnol 2006, 71, 473–479. [Google Scholar]
- Shi, XM; Liu, HJ; Zhang, XW; Chen, F. Production of biomass and lutein by Chlorella protothecoides at various glucose concentrations in heterotrophic cultures. Process Biochem 1999, 34, 341–347. [Google Scholar]
- Ishikawa, E; Sansawa, H; Abe, H. Isolation and characterization of a Chlorella mutant producing high amounts of chlorophyll and Carotenoids. J. Appl. Phycol 2004, 16, 385–393. [Google Scholar]
- Zhang, DH; Lee, YK. Enhanced accumulation of secondary carotenoids in a mutant of the green alga, Chlorococcum sp. J. Appl. Phycol 1997, 9, 459–463. [Google Scholar]
- Kamath, BS; Vidhyavathi, R; Sarada, R; Ravishankar, GA. Enhancement of carotenoids by mutation and stress induced carotenogenic genes in Haematococcus pluvialis mutants. Bioresour. Technol 2008, 99, 8667–8673. [Google Scholar]
- Arnon, DI; McSwain, BD; Tsujimoto, HY; Wada, K. Photochemical activity and components of membrane preparations from blue-green algae. I. Coexistence of two photosystems in relation to chlorophyll a and removal of phycocyanin. Biochim. Biophys. Acta 1974, 357, 231–245. [Google Scholar]
- Cordero, BF; Obraztsova, I; Martin, L; Couso, I; Leon, R; Vargas, MA; Rodriguez, H. Isolation and characterization of a lycopene β-cyclase gene from the astaxanthin-producing green alga Chlorella zofingiensis (Chlorophyta). J. Phycol 2010, 46, 1229–1238. [Google Scholar]


| Species | μ (h−1) | Biomass (g L−1) | Carotenoid Content (mg L−1)
| |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ant | Ast | C | α-c | β-c | L | V | Z | |||
| Chlamydomonas reinhardtii b | 0.05 | 4.5 | 0.4 | nd | nd | 0.1 | 3.0 | 12.4 | 1.0 | 0.2 |
| Chlorella fusca b | 0.04 | 8.5 | 1.5 | 6.5 | 1.2 | 0.3 | 4.6 | 22.0 | 1.5 | 6.0 |
| Chlorella luteoviridis b | 0.03 | 2.7 | 0.3 | nd | nd | nd | 0.5 | 5.8 | 0.2 | 0.6 |
| Chlorella sorokiniana a | 0.11 | 8.0 | 0.3 | nd | nd | 0.2 | 1.6 | 24.0 | 0.9 | 0.4 |
| Chlorella stigmatofora b | 0.02 | 4.1 | 0.1 | nd | nd | 0.1 | nd | 3.2 | 0.2 | 0.4 |
| Chlorella vulgaris c | 0.08 | 8.5 | 0.9 | nd | nd | 1.8 | 2.3 | 22.2 | 2.2 | 3.3 |
| Chlorella zofingiensis c | 0.03 | 7.6 | 0.9 | 5.6 | 1.2 | 0.4 | 3.0 | 20.0 | 1.3 | 3.0 |
| Chlorococcum sp. a | 0.03 | 7.5 | 0.8 | nd | 0.8 | 0.2 | 2.2 | 15.0 | 2.5 | 0.5 |
| Monoraphidium braunii c | 0.07 | 8.4 | 2.4 | nd | nd | 0.9 | 3.8 | 24.0 | 2.1 | 8.4 |
| Scenedesmus armatus b | 0.09 | 8.5 | 0.5 | nd | nd | 0.3 | 4.4 | 24.0 | 2.4 | nd |
| Scenedesmus quadricauda c | 0.06 | 8.4 | 0.6 | nd | nd | 0.3 | 3.0 | 22.0 | 5.0 | nd |
| Scenedesmus obliquus b | 0.08 | 8.0 | 0.5 | nd | nd | 0.2 | 2.5 | 15.0 | 1.5 | 1.2 |
| Scenedesmus vacuolatus c | 0.08 | 8.2 | 1.6 | 5.3 | 1.7 | 0.5 | 4.2 | 24.0 | 2.4 | 3.8 |
| Irradiance (μmol photons m−2 s−1) | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| 92 | 0.05 | 6.0 | 15.0 | 2.5 |
| 230 | 0.08 | 7.2 | 19.0 | 2.6 |
| 368 | 0.11 | 7.8 | 23.0 | 3.0 |
| 460 | 0.11 | 8.0 | 24.0 | 3.0 |
| 690 | 0.13 | 9.0 | 28.0 | 3.1 |
| 920 | 0.13 | 8.5 | 24.0 | 2.8 |
| 1495 | 0.13 | 8.5 | 20.0 | 2.4 |
| Temperature (°C) | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| 22 | 0.08 | 7.5 | 19.0 | 2.4 |
| 25 | 0.08 | 7.9 | 20.0 | 2.4 |
| 28 | 0.12 | 8.2 | 25.0 | 3.0 |
| 32 | 0.12 | 6.6 | 24.0 | 3.6 |
| 36 | 0.11 | 5.6 | 19.0 | 3.4 |
| 40 | 0.11 | 4.6 | 16.0 | 3.4 |
| NaNO3 concentration (mM) | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| 10 | 0.10 | 6.5 | 9.0 | 1.4 |
| 20 | 0.10 | 8.0 | 24.0 | 3.0 |
| 30 | 0.12 | 8.3 | 25.0 | 3.0 |
| 40 | 0.13 | 8.2 | 26.0 | 3.2 |
| 60 | 0.12 | 7.9 | 25.0 | 3.2 |
| 80 | 0.12 | 7.4 | 24.0 | 3.2 |
| 100 | 0.11 | 7.7 | 24.0 | 3.1 |
| 120 | 0.11 | 7.2 | 22.0 | 3.0 |
| Acetate concentration (mM) | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| 0 | 0.09 | 7.5 | 22.0 | 3.0 |
| 20 | 0.11 | 7.6 | 26.0 | 3.4 |
| 30 | 0.11 | 8.0 | 28.0 | 3.5 |
| 40 | 0.12 | 9.0 | 32.0 | 3.6 |
| 50 | 0.12 | 9.0 | 26.0 | 2.9 |
| 60 | 0.11 | 9.2 | 25.0 | 2.7 |
| Culture medium | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| AM | 0.12 | 8.8 | 30.0 | 3.4 |
| AM + 100 mM Glucose a | 0.11 | 13.5 | 35.0 | 2.6 |
| AM + 40 mM Acetate a | 0.11 | 9.0 | 29.0 | 3.2 |
| AM + 40 mM Nitrate a | 0.12 | 8.8 | 29.0 | 3.3 |
| AM + 60 mM Urea b | 0.10 | 8.5 | 23.0 | 2.7 |
| AM + 100 mM Glucose c | 0.11 | 14.0 | 33.5 | 2.4 |
| Strain | Volumetric Lutein Content (mg L−1) | Cellular Lutein Content (mg g−1 DW) |
|---|---|---|
| (% with respect to the wild strain) | ||
| MR-3 (NF) | 52 | 17 |
| MR-14 (NIC) | 63 | 29 |
| MR-16 (NIC) | 101 | 42 |
| DMR-4 (NIC) | 62 | 38 |
| DMR-11 (NIC) | 49 | 49 |
| DMR-5 (NF) | 29 | 55 |
| DMR-8 (NF) | 29 | 53 |
| Culture condition | μ (h−1) | Biomass (g L−1) | Lutein Content
| |
|---|---|---|---|---|
| mg L−1 | mg g−1 DW | |||
| Wild type | ||||
| Photoautotrophic | 0.11 | 8.4 | 25.0 | 3.0 |
| Mixotrophic | 0.11 | 13.0 | 33.0 | 2.6 |
| MR-16 mutant | ||||
| Photoautotrophic | 0.12 | 8.3 | 42.0 | 5.0 |
| Mixotrophic | 0.11 | 7.8 | 22.0 | 2.8 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cordero, B.F.; Obraztsova, I.; Couso, I.; Leon, R.; Vargas, M.A.; Rodriguez, H. Enhancement of Lutein Production in Chlorella sorokiniana (Chorophyta) by Improvement of Culture Conditions and Random Mutagenesis. Mar. Drugs 2011, 9, 1607-1624. https://doi.org/10.3390/md9091607
Cordero BF, Obraztsova I, Couso I, Leon R, Vargas MA, Rodriguez H. Enhancement of Lutein Production in Chlorella sorokiniana (Chorophyta) by Improvement of Culture Conditions and Random Mutagenesis. Marine Drugs. 2011; 9(9):1607-1624. https://doi.org/10.3390/md9091607
Chicago/Turabian StyleCordero, Baldo F., Irina Obraztsova, Inmaculada Couso, Rosa Leon, Maria Angeles Vargas, and Herminia Rodriguez. 2011. "Enhancement of Lutein Production in Chlorella sorokiniana (Chorophyta) by Improvement of Culture Conditions and Random Mutagenesis" Marine Drugs 9, no. 9: 1607-1624. https://doi.org/10.3390/md9091607
APA StyleCordero, B. F., Obraztsova, I., Couso, I., Leon, R., Vargas, M. A., & Rodriguez, H. (2011). Enhancement of Lutein Production in Chlorella sorokiniana (Chorophyta) by Improvement of Culture Conditions and Random Mutagenesis. Marine Drugs, 9(9), 1607-1624. https://doi.org/10.3390/md9091607





