Yessotoxins, a Group of Marine Polyether Toxins: an Overview
Abstract
:1. Introduction
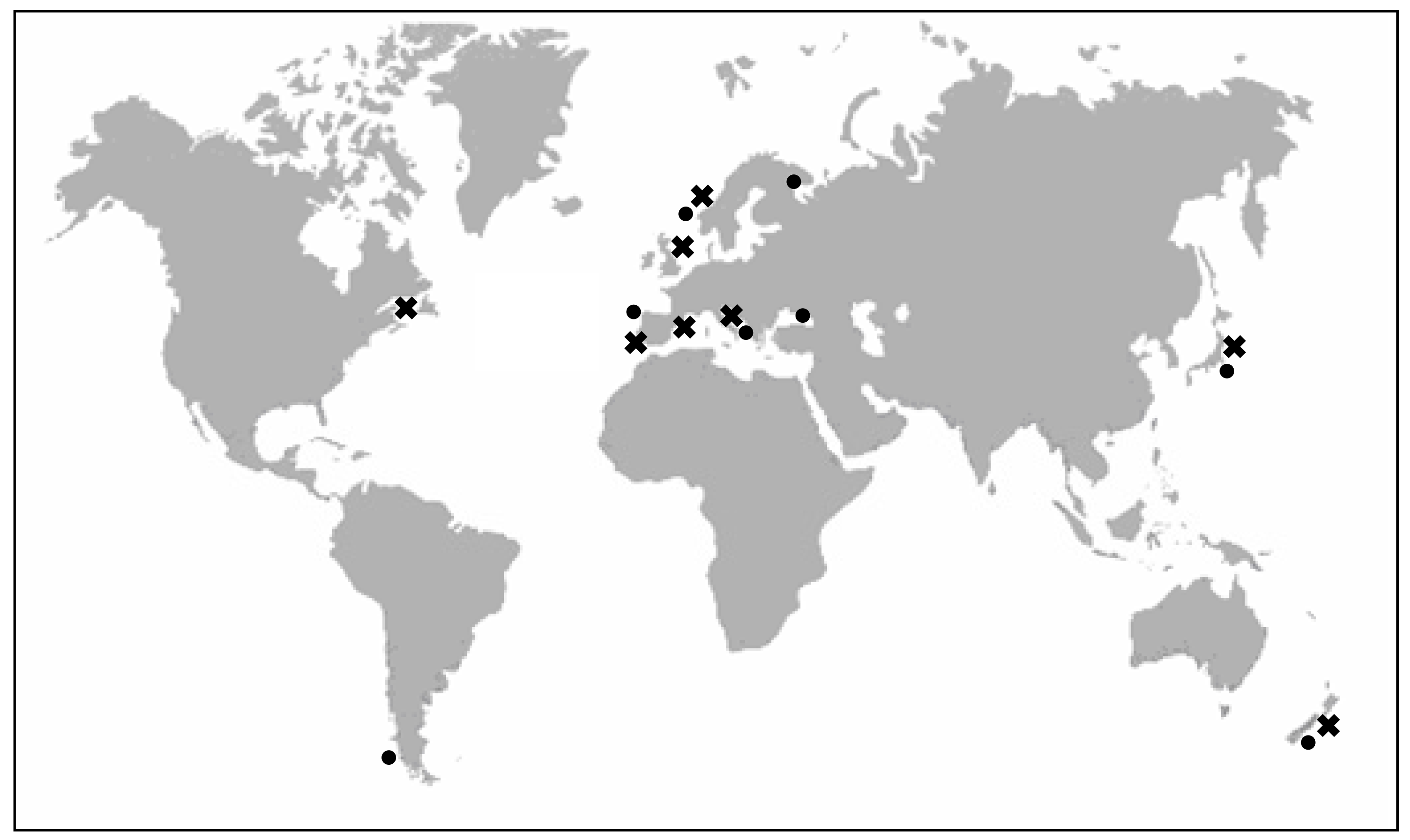
2. Producer organisms and vectors of YTXs
2.1. Characteristics of YTX-producer dinoflagellates
a) Protoceratium reticulatum (Claparède and Lachmann) Bütschli1885 (=Gonyaulax grindleyi Reinecke 1967)
b) Lingulodinium polyedrum (Stein) Dodge 1989 (=Gonyaulax polyedra Stein 1883)
c) Gonyaulax spinifera (Claparède and Lachmann) Diesing 1866
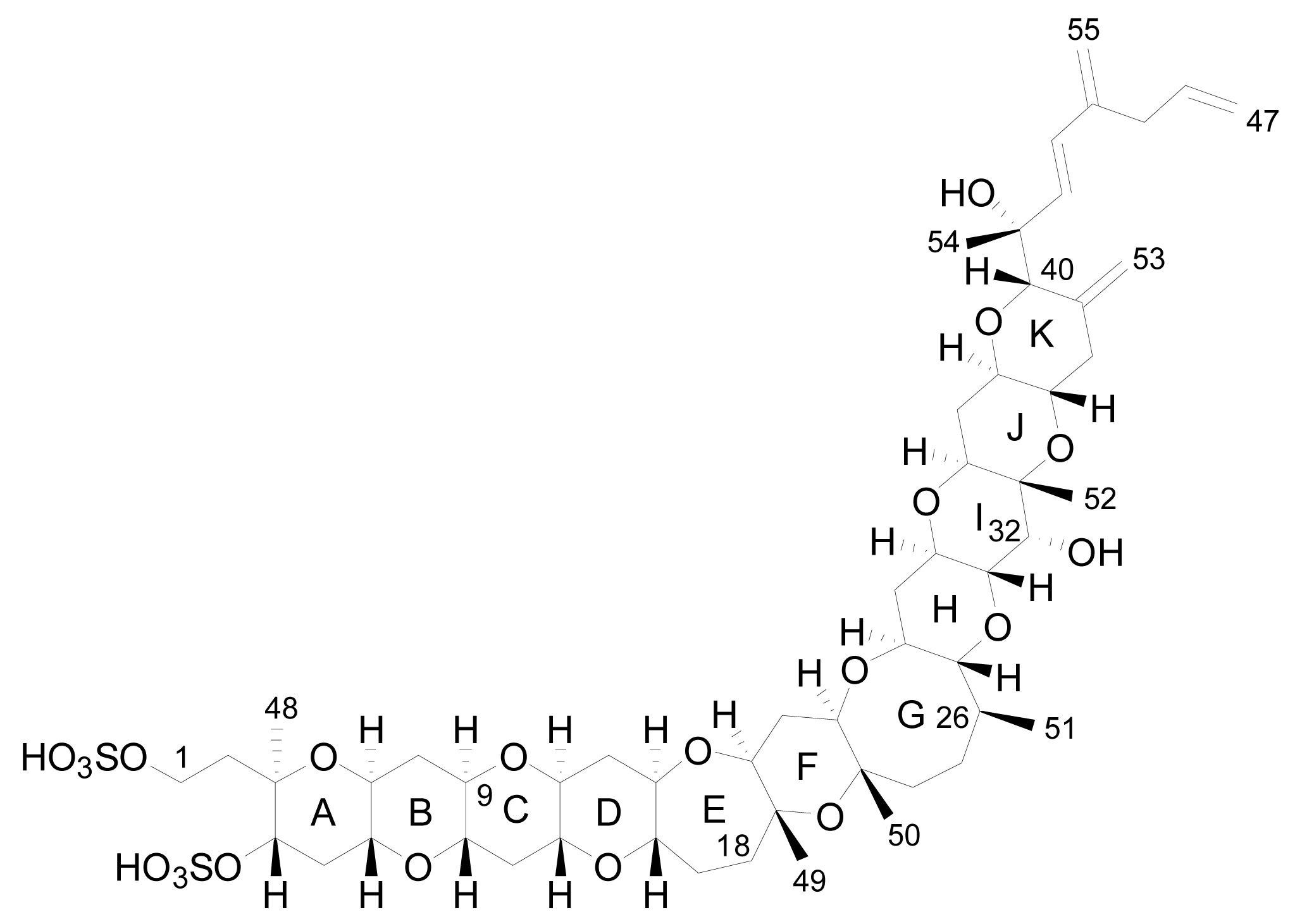
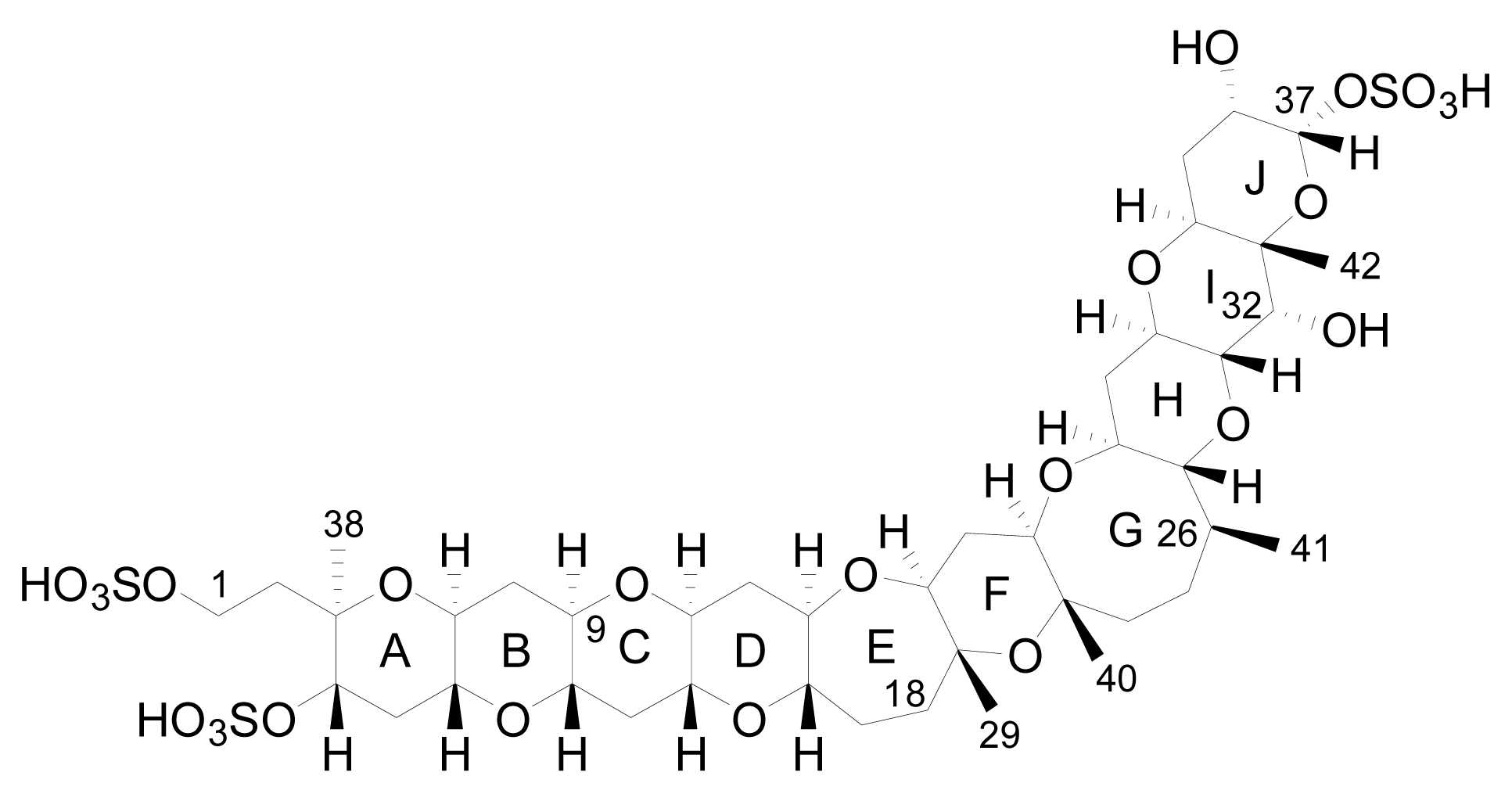
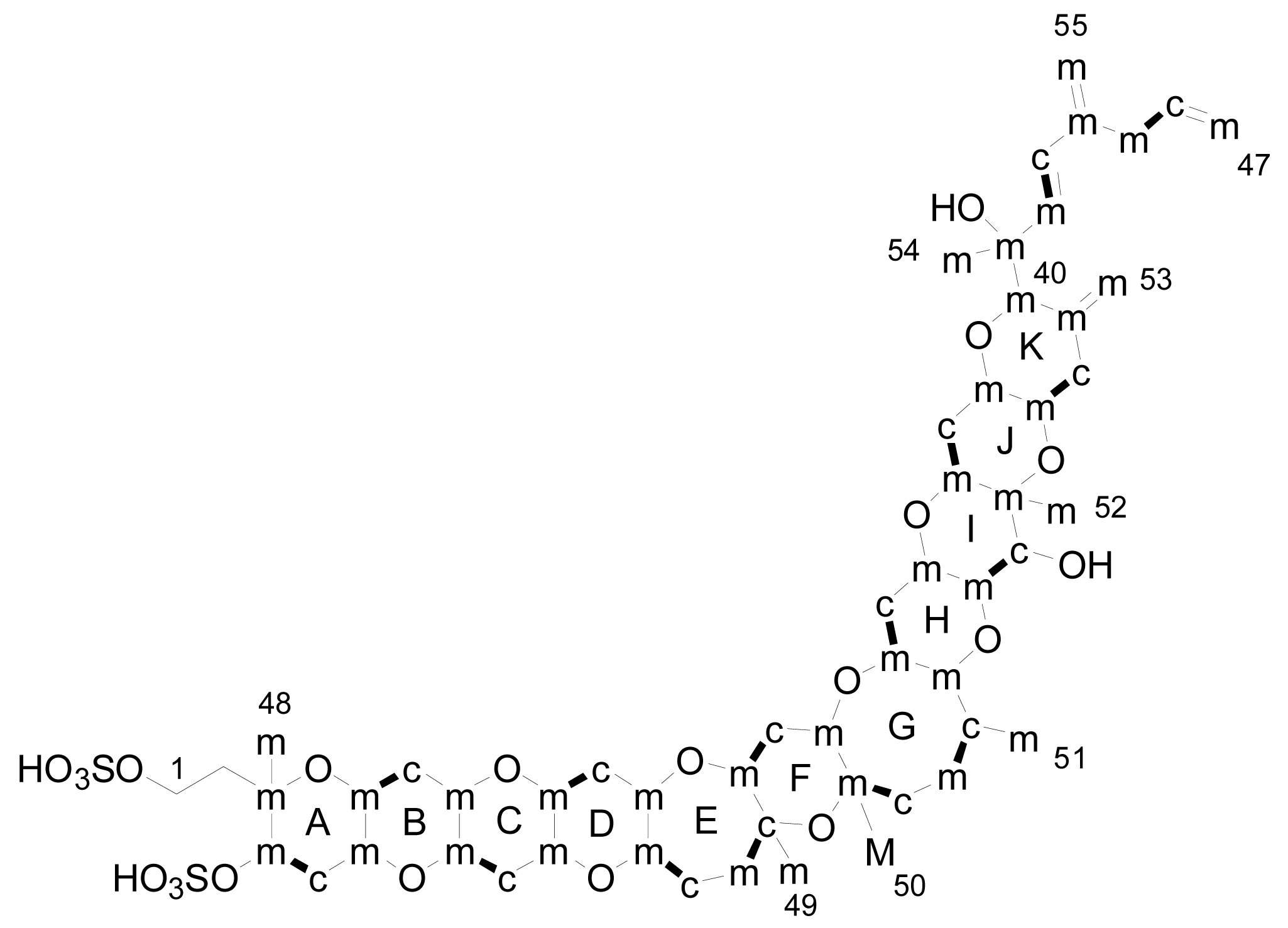
3. Structures of yessotoxin and its analogues
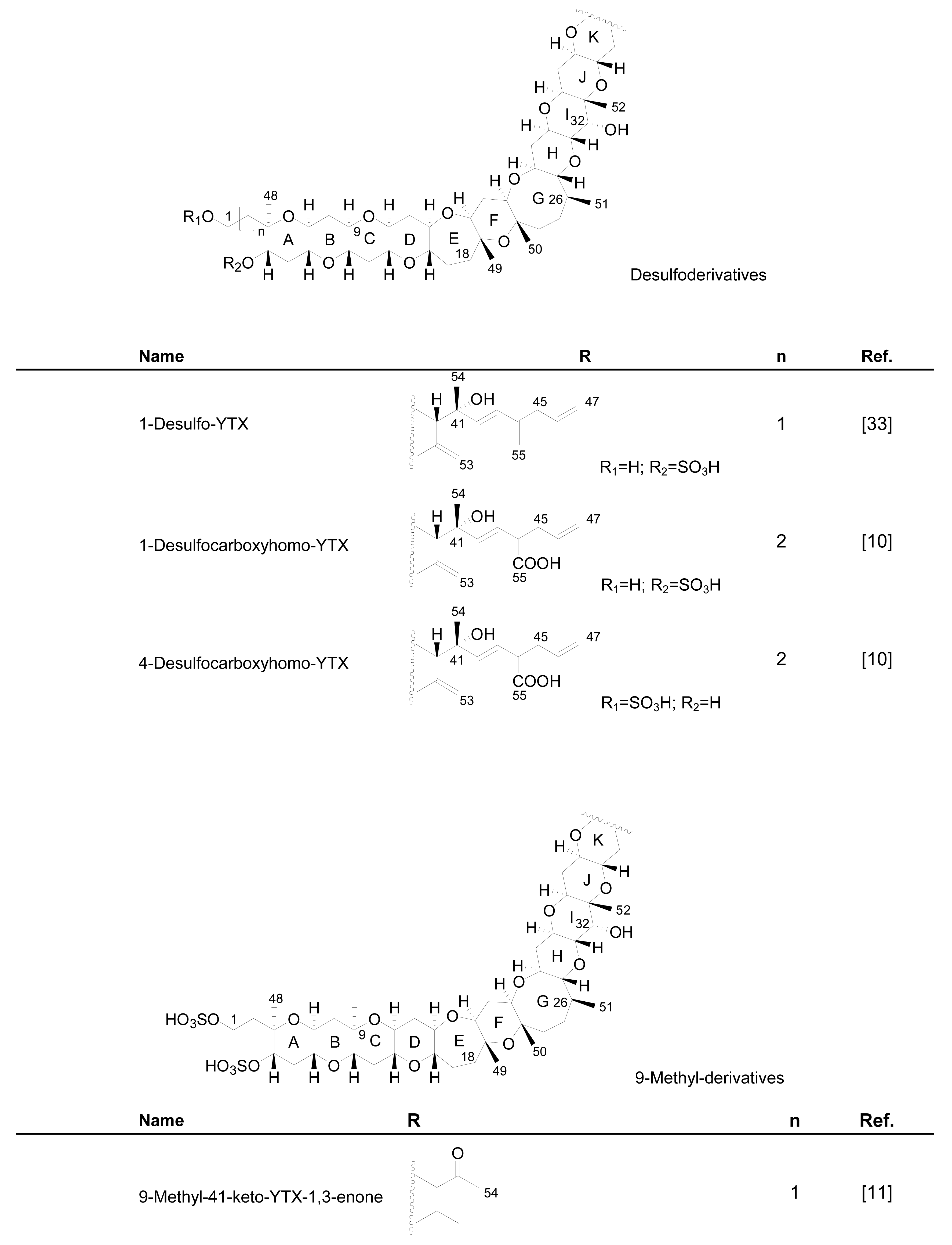
3.1. Analogues detected in dinoflagellates
3.2. Analogues detected in shellfish
4. Biosynthetic origin of yessotoxin
5. Toxicity and mode action of yessotoxins
5.1. Intraperitoneal toxicity
5.2. Oral toxicity
5.3. Mode of action of yessotoxins
6. Detection and quantification methods for yessotoxin and its analogues
6.1. In vivo assays
6.2. In vitro assays
- Functional assays. These assays depend on a characteristic response linked to the mode of action of the toxin in cultured cells; therefore they correlate well with real toxicity. The disadvantages are that: viable cell-strains are necessary, they are sensitive to interferences and that expertise is required to perform the assays. In addition, it is not possible to distinguish between different YTX analogues using them. A series of functional assays for YTXs have been recently developed:
- - Cytotoxicity assays: Initially designed to detect DSP toxins, these assays are also able to detect YTXs [101]. These assays are based on the microscopic examination of the morphological changes observed in freshly prepared rat hepatocytes after toxin exposure. With these assays it is possible to differentiate between OA, DTX1, PTX1 and YTXs as OA and DTX1 induce irregular-shaped cells with surface blebs, while PTX1 produces dose dependent vacuolisation or YTXs produce tiny blebs on the surface of cells but do not induce changes in the shape of the cells. However, this information is only qualitative, as cell changes produced by the toxins are not dose dependent.
- - Accumulation of fragmented E-cadherin [102]: In this assay the accumulation of a 100kDa fragment of E-cadherin induced by YTX in the epithelial MCF-7 breast cancer cells is measured. The increase of the fragmentation of this fragment of E-cadherin is concentration-dependent and is detected by Immunoblotting using anti-E-cadherin antibodies. It is a very sensitive and specific method (detection limit around 100 μg YTX equivalents/Kg) [103].
- - Intracellular decrease of adenosine 3′,5′-cyclic monophosphate (cAMP) [82, 104]: YTX in the presence of calcium activate PDEs activity with the consequent decrease in cAMP levels. This phenomenon is concentration-dependent and it is detected using a fluorescent derivative of cAMP, anthranyloyl-cAMP. To be exact, it is detected by a decrease in fluorescence, due to the improved hydrolytic effect of PDEs in anthranyloyl-cAMP [104]. The rate of cAMP hydrolysis is linearly correlated with different concentrations of YTX between 0.5 and 10 μM.
- Structural assays (immunoassays) based on measurements of the recognition of this toxin by specific antibodies. However, these assays not necessarily related to the biological activity of the toxin, therefore any correlation with real toxicity is not always as good as it is for functional assays.
- - ELISA (enzyme-linked immunosorbent assay): a specific ELISA to detect YTXs in shellfish, microalgae and/or seawater samples has been recently developed by Briggs et al. [105], and is now commercially available as a kit for rapid screening of YTXs [106]. It is a competitive, indirect immunoassay based on the use of polyclonal antibodies against YTX. The antibodies used, were obtained using conjugated YTX-protein (cBSA), and have a broad specificity for YTXs, because the YTXs have been conjugated in the non-sulphated end of the molecule. The working range is 70–1300 pgYTX/mL. The disadvantages of this method are due to the cross-reactivity of the antibody, and therefore it is not possible to distinguish among analogues. Moreover, the cross-reactivity of the antibodies is limited to compounds with specific compatible moieties, therefore it is not possible to detect all the analogues with the significant risk of obtaining false negatives. This assay has the advantage of being sensitive, rapid, relatively cheap, suitable for HTS screening large number of samples; it is affordable for most labs and operators require minimal training.
6.3. Chemical methods
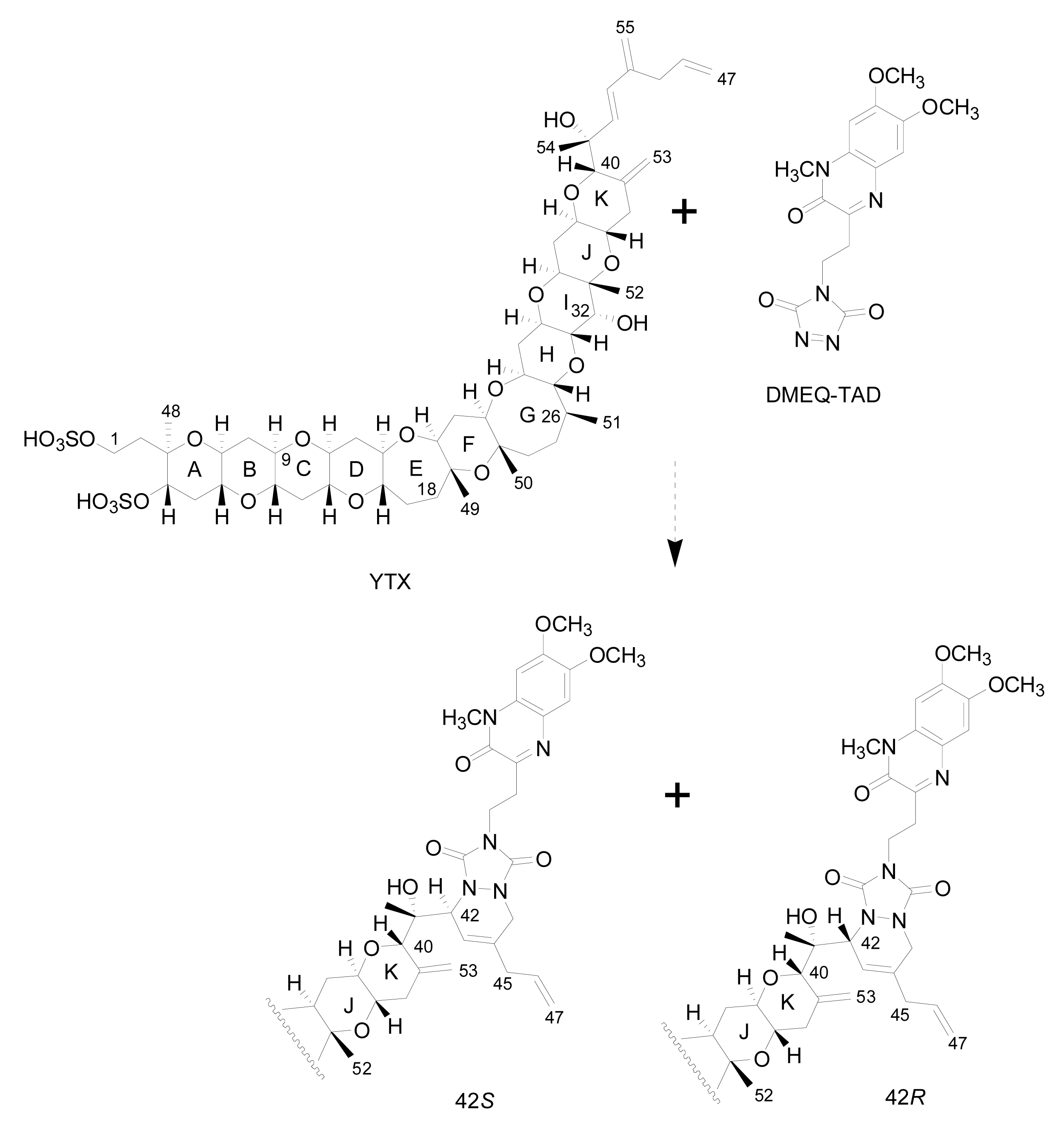
- Liquid chromatography with fluorescence detection (HPLC-FLD) [20]: This method is extensively used for the qualitative and quantitative analysis of YTXs both in molluscs and microalgae [107]. It is based on the determination of a fluorescent derivative of the toxin obtained by precolum derivatization with the dienophile reagent 4-(2-(6,7-dimethoxy-4-methyl-3-oxo-3,4dihydroquinoxalimylethyl)-1,2,4-triazoline-3,5-dione (DMEQ-TAD) (Figure 8).Previous to the analysis, a clean-up of the sample using solid phase extraction (SPE) cartridges is required to eliminate interferences. The fluorogenic reagent reacts with the conjugated diene in the side-chain and, as a consequence, this method can only detect YTX, 45-OH-YTX, trinor-YTX [20], desulfo-YTX [33], homo-YTX, 45-OHhomo-YTX and G-YTXs [108]. Other analogues such as carboxy-YTX, diOH-YTX, and noroxo-YTX or adriatoxin without the diene moiety cannot be detected by HPLC-FLD. The fluorescent adduct gives two diasteroisomeric peaks due to the formation of both C-42 epimers in a 3:1 ratio for YTX (Figure 8) [20]. YTX analogues elute at different retention times, therefore the use of toxin standards is necessary for toxin identification.Weaknesses of this method are that the detection of new analogues are limited to the presence of a conjugated diene, and that it is difficult to distinguish between analogues with similar retention times, such as YTX and homo-YTX [15]. On the other hand, advantages are that it is a very sensitive method with a linear response (0.4–6 mg/Kg), requires relatively low-cost equipment, it is affordable to many labs and it is faster than the mouse bioassay.
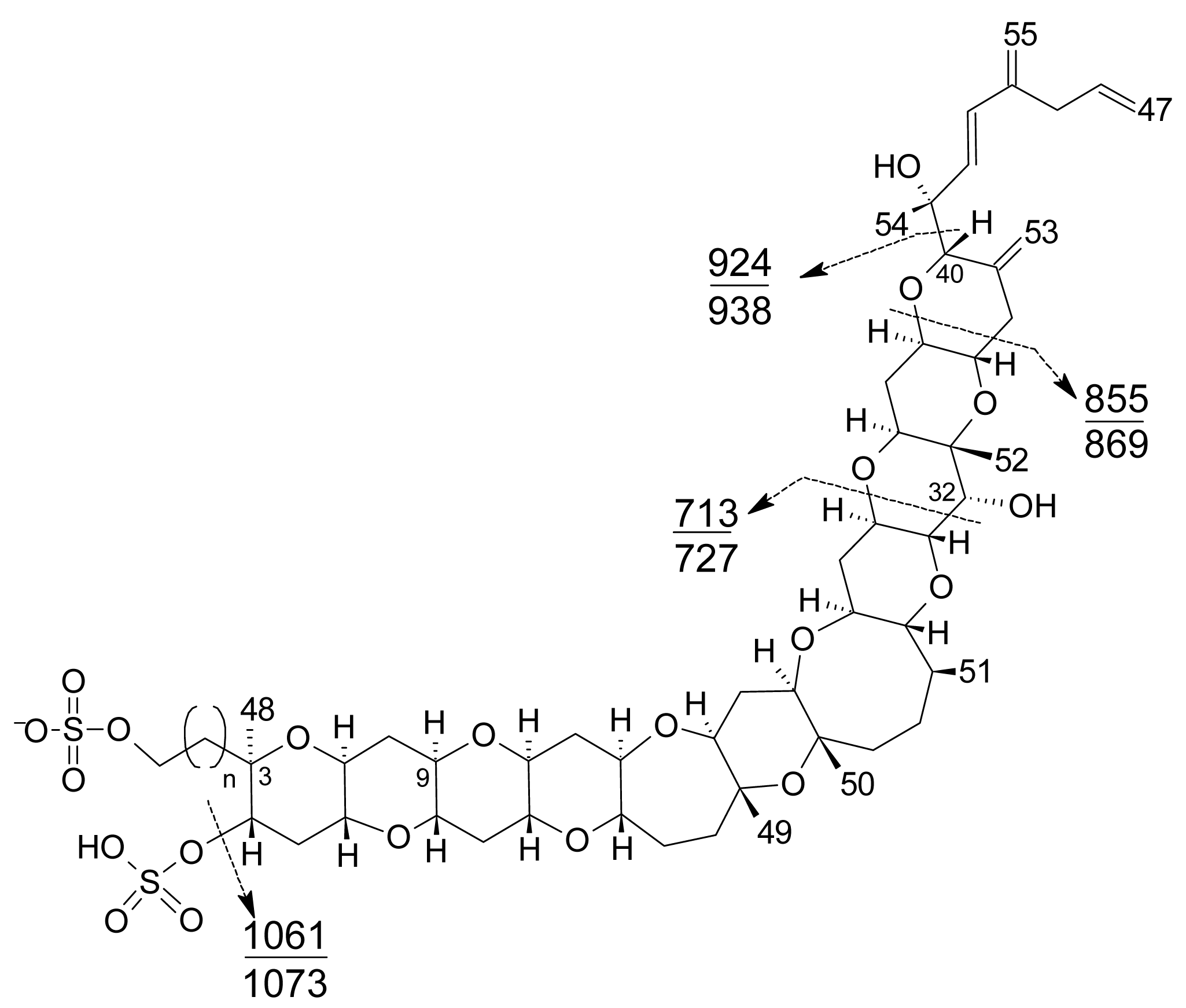
- Liquid chromatography coupled with mass spectrometry (LC-MS): currently this is the most powerful analytical tool used to identify multiple toxins. This technique has several advantages, such as its high sensitivity and selectivity. In addition, collision experiments (MS/MS) can provide valuable structural information in the confirmation of toxin identities, as well as in the identification of new toxins. It does not require complex derivatization and purification steps needed for HPLC-FLD methods. It is possible to analyze all the YTXs, even those derivatized with DMEQ-TAD [108], and also different algal toxins. In fact, several multi-toxin detection methods have been developed [109]. However, as is the case for other chemical methods, calibration standards are required for method development and toxin quantification. LC-MS methods can provide relevant information about the presence of compounds related to a known structure, even if the toxin standard is available only for one relevant toxin of the group. The main disadvantage of this technique is that the equipment is expensive.Several specific LC-MS methods have been developed for YTX detection, which differ basically in the mobile or stationary phase used or the electrospray mode selected (positive or negative) [25, 110–115]. Most methods use negative ion mode detection because YTXs easily lose a proton from the sulfate group. The fragmentation of YTX is achieved easily even when low collision energies (C.E.) are used and the loss of the sulfate group and the subsequent fragmentation of the unsaturated side chain are characteristic (Figure 9) [111]. Recently an ultra-performance liquid chromatography mass spectrometry (UPLC) method has been developed [116].
- Capillary electrophoresis with UV/MS detection: This is an analytical method designed as an alternative to HPLC-FLD for the determination of YTXs. It shows high resolution and small amounts of sample are required. Coupled with MS instrumentation it allows confirmation of the presence of YTXs in samples [117].
7. Current regulations for yessotoxins
8. Conclusions
Acknowledgements
References and Notes
- Moestrup, Ø; Codd, GA; Elbrächter, M; Faust, MA; Fraga, S; Fukuyo, Y; Cronberg, G; Halim, Y; Taylor, FJR; Zingone, A. IOC Taxonomic Reference List of Toxic Algae, UNESCO. 2004. http://ioc.unesco.org/hab/data.htm.
- Aune, T; Sorby, R; Yasumoto, T; Ramstad, H; Landsverk, T. Comparison of oral and intraperitoneal toxicity of yessotoxin towards mice. Toxicon 2002, 40, 77–82. [Google Scholar]
- Tubaro, A; Sosa, S; Carbonatto, M; Altinier, G; Vita, F; Melato, M; Satake, M; Yasumoto, T. Oral and intraperitoneal acute toxicity studies of yessotoxin and homoyessotoxins in mice. Toxicon 2003, 41, 783–792. [Google Scholar]
- Tubaro, A; Sidari, L; Della-Loggia, R; Yasumoto, T. Reguera, B, Blanco, J, Fenández, ML, Wyatt, T, Eds.; Occurrence of yessotoxin-like toxins in phytoplankton and mussels from northern Adriatic Sea. In Harmful Algae; Xunta de Galicia and IOC of UNESCO, 1998; pp. 470–472. [Google Scholar]
- Ogino, H; Kumagai, M; Yasumoto, T. Toxicologic evaluation of yessotoxin. Nat Toxins 1997, 5, 255–259. [Google Scholar]
- Konishi, M; Yang, X; Li, B; Fairchild, CR; Shimizu, Y. Highly cytotoxic metabolites from the culture supernatant of the temperate dinoflagellate Protoceratium cf. reticulatum. J Nat Prod 2004, 67, 1309–1313. [Google Scholar]
- Bianchi, C; Fato, R; Angelin, A; Trombetti, F; Ventrella, V; Borgatti, AR; Fattorusso, E; Ciminiello, P; Bernardi, P; Lenaz, G; Parenti, CG. Yessotoxin, a shellfish biotoxin, is a potent inducer of the permeability transition in isolated mitochondria and intact cells. Biochim Biophys Acta 2004, 1656, 139–147. [Google Scholar]
- Pérez-Gómez, A; Novelli, A; Ferrero-Gutiérrez, A; Franco, JM; Paz, B; Fernández-Sánchez, MT. Potent neurotoxic action of the shellfish biotoxin yessotoxin on cultured cerebellar neurons. Toxicol Sci 2006, 90, 168–177. [Google Scholar]
- EC. Regulation Nº 853/2004 of the European Parlamient and of the Council of 29 April 2004, laying down specific hygiene rules for food of animal origin. 2004, L 226, p. 22.
- Ciminiello, P; Dell-Aversano, C; Fattorusso, E; Forino, M; Grauso, L; Magno, S; Poletti, R; Tartaglione, L. Desulfoyessotoxins from Adriatic Mussels: a new problem for seafood safety control. Chem Res Toxicol 2007, 20, 95–98. [Google Scholar]
- Miles, CO; Wilkins, AL; Allan, DH; Selwood, AI; Jensen, DJ; Cooney, JM; Beuzenberg, V; MacKenzie, AL. Identification of 45-hydroxy-46,47-dinoryessotoxin, 44-oxo-45,46,47-trinoryessotoxin, and 9-methyl-42,43,44,45,46,47,55-heptanor-38-en-41-oxoyessotoxin, and partial characterization of some minor yessotoxins, from Protoceratium reticulatum. Toxicon 2006, 47, 229–240. [Google Scholar]
- Eiki, K; Satake, M; Koike, K; Ogata, T; Mitsuya, T; Oshima, Y. Confirmation of yessotoxin production by the dinoflagellate Protoceratium reticulatum in Mutsu Bay. Fish Sci 2005, 71, 633–638. [Google Scholar]
- Ciminiello, P; Dell-Aversano, C; Fattorusso, E; Forino, M; Magno, S; Guerrini, F; Pistocchi, R; Boni, L. Complex yessotoxins profile in Protoceratium reticulatum from northwestern Adriatic sea revealed by LC–MS analysis. Toxicon 2003, 42, 7–14. [Google Scholar]
- Samdal, IA; Olseng, CD; Sandvik, M; Miles, CO; Briggs, L; Torgersen, T; Jensen, DJ; Cooney, JM. Henshilwood, K, Deegan, B, McMahon, T, Cusack, C, Keaveney, S, Silke, J, O’Cinneide, M, Lyons, D, Hess, P, Eds.; Profile of yessotoxin analogues in a Norwegian strain of Protoceratium reticulatum; Galway: Ireland, 2006; p. 118. [Google Scholar]
- Paz, B; Riobó, P; Ramilo, I; Franco, JM. Yessotoxins profile in strains of Protoceratium reticulatum from Spain and USA. Toxicon 2007, 50, 1–17. [Google Scholar]
- Suzuki, T; Satake, M; Yoshimatsu, S; Oshima, Y; Horie, Y; Koike, K; Iwataki, M. Yessotoxin analogues in several strains of Protoceratium reticulatum in Japan determined by liquid chromatography-hybrid triple quadrupole/linear ion trap mass spectrometry. J ChromatogrA 2007, 1142, 172–177. [Google Scholar]
- Murata, M; Masanori, K; Lee, J-S; Yasumoto, T. Isolation and structure of Yessotoxin, a novel polyether compound implicated in diarrhetic shellfish poisoning. Tetrahedron Lett 1987, 28, 5869–5872. [Google Scholar]
- Lee, J-S; Tangen, K; Dahl, E; Hovgaard, P; Yasumoto, T. Diarrhetic shellfish toxins in Norwegian mussels. Nippon Suisan Gakkaishi/Bull Jap Soc Sci Fish 1988, 54, 1953–1957. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Magno, S; Poletti, R; Satake, M; Viviani, R; Yasumoto, T. Yessotoxin in mussels of the northern Adriatic Sea. Toxicon 1997, 35, 177–183. [Google Scholar]
- Yasumoto, T; Takizawa, A. Fluorometric measurement of yessotoxins in shellfish by high-pressure liquid chromatography. Biosci Biotechnol Biochem 1997, 61, 1775–1777. [Google Scholar]
- Arévalo, F; Pazos, Y; Correa, J; Salgado, C; Moroño, A; Paz, B; Franco, JM. Henshilwood, K, Deegan, B, McMahon, T, Cusack, C, Keaveney, S, Silke, J, O’Cinneide, M, Lyons, D, Hess, P, Eds.; First report of yessotoxins in mussels of Galician Rías during a bloom of Lingulodinium polyedra. In Stein (Dodge); Galway: Ireland, 2006; pp. 184–189. [Google Scholar]
- Vershinin, A; Moruchkov, A; Morton, SL; Leighfield, TA; Quilliam, MA; Ramsdell, JS. Phytoplankton composition of the Kandalaksha Gulf, Russian White Sea: Dinophysis and lipophilic toxins in the blue mussel (Mytilus edulis). Harmful Algae 2006, 5, 558–564. [Google Scholar]
- Morton, SL; Vershinin, A; Leighfield, TA; Smith, L; Quilliam, M. Identification of yessotoxin in mussels from the Caucasian Black Sea Coast of the Russian Federation. Toxicon 2007, 50, 581–584. [Google Scholar]
- Satake, M; Ichimura, T; Sekiguchi, K; Yoshimatsu, S; Oshima, Y. Confirmation of yessotoxin and 45,46,47-trinoryessotoxin production by Protoceratium reticulatum collected in Japan. Nat Toxins 1999, 7, 147–150. [Google Scholar]
- Stobo, LA; Lewis, J; Quilliam, MA; Hardstaff, WR; Gallacher, S; Webster, L; Smith, E; McKenzie, M. Bates, S, Ed.; Detection of yessotoxin in UK and Canadian isolates of phytoplankton and optimization and validation of LC-MS methods; Gulf Fisheries Centre: Moncton, New Brunswick, Canada, 2003; pp. 8–14. [Google Scholar]
- Samdal, IA; Naustvoll, LJ; Olseng, CD; Briggs, LR; Miles, CO. Use of ELISA to identify Protoceratium reticulatum as a source of yessotoxin in Norway. Toxicon 2004, 44, 75–82. [Google Scholar]
- Paz, B; Riobó, P; Fernández, ML; Fraga, S; Franco, JM. Production and release of yessotoxins by the dinoflagellates Protoceratium reticulatum and Lingulodinium polyedrum in culture. Toxicon 2004, 44, 251–258. [Google Scholar]
- Draisci, R; Ferretti, E; Palleschi, L; Marchiafava, C; Poletti, R; Milandri, A; Ceredi, A; Pompei, M. High levels of yessotoxin in mussels and presence of yessotoxin and homoyessotoxin in dinoflagellates of the Adriatic Sea. Toxicon 1999, 37, 1187–1193. [Google Scholar]
- Boni, L; Ceredi, A; Guerrini, F; Milandri, A; Pistocchi, R; Poletti, R; Pompei, M. Hallegraeff, GM, Blackburn, SI, Bolch, CJ, Lewis, RJ, Eds.; Toxic Protoceratium reticulatum. (Peridiniales, dinophyta) in the North-Western Adriatic Sea (Italy). In Harmful Algal Blooms 2000; IOC of UNESCO, 2001; pp. 137–140. [Google Scholar]
- Ramstad, H; Hovgaard, P; Yasumoto, T; Larsen, S; Aune, T. Monthly variations in diarrhetic toxins and yessotoxin in shellfish from coast to the inner part of the Sognefjord, Norway. Toxicon 2001, 39, 1035–1043. [Google Scholar]
- Armstrong, M; Kudela, R. Evaluation of California isolates of Lingulodinium poliedrum for the production of yessotoxin. Afr J Mar Sci 2006, 25, 399–401. [Google Scholar]
- Holmes, MJ; Lewis, RJ; Jones, A; Wong Hoy, AW. Cooliatoxin, the first toxin from Coolia monotis (Dinophyceae). Nat Toxins 1995, 3, 355–362. [Google Scholar]
- Daiguji, M; Satake, M; Ramstad, H; Aune, T; Naoki, H; Yasumoto, T. Structure and fluorometric HPLC determination of 1-desulfoyessotoxin, a new yessotoxin analog isolated from mussels from Norway. Nat Toxins 1998, 6, 235–239. [Google Scholar]
- Rhodes, L; McNabb, P; de Salas, M; Briggs, L; Beuzenberg, V; Gladstone, M. Yessotoxin production by Gonyaulax spinifera. Harmful Algae 2006, 5, 148–155. [Google Scholar]
- Hansen, G; Moestrup, Ø; Roberts, KR. Light and Electron Microscopical observations on Protoceratium reticulatum (Dinophyceae). Arch Protistenkd 1996–97, 147, 381–391. [Google Scholar]
- Mackenzie, L; Suzuki, T; Adamson, J. Hallegraeff, GM, Blackburn, SI, Bolch, CJ, Lewis, RJ, Eds.; Elimination and differential transformation of yessotoxin by the greenshell mussel Perna canaliculus and the blue mussel Mytilus gallopovincialis. In Harmful Algal Blooms 2000; IOC of UNESCO, 2001; pp. 371–374. [Google Scholar]
- Franchini, A; Milandri, A; Poletti, R; Ottaviani, E. Inmunolocalization of yessotoxins in the mussel Mytilus galloprovincialis. Toxicon 2003, 41, 967–970. [Google Scholar]
- Poupin, J; Cussatlegras, A-S; Geistdoerfer, P. Plancton Marin Bioluminescent. Inventaire documenté des espèces et bilan des formes les plus communes de la mer d’Iroise; Laboratoire d’Océanographie de l’École Navale, LOEN Lanvéoc-Poulmic: France, 1999; pp. 1–83. [Google Scholar]
- Rodríguez, JJG; Mirón, AS; Belarbi, EH; García, MCC; Camacho, FG; Grima, EM. New culture approaches for yessotoxin production from the dinoflagellate Protoceratium reticulatum. Biotechnol Prog 2007, 23, 339–350. [Google Scholar]
- Lewis, J; Hallet, R. Lingulodinium polyedrum (Gonyaulax polyedra) a blooming dinoflagellate. Oceanogr Mar Biol, Annu Rev 1997, 35, 97–161. [Google Scholar]
- Latz, MI; Rohr, J. Luminiscent response of the red tide dinoflagellates Lingulodinium polyedrum to laminar and turbulent flow. Limnology and Oceanography 1999, 44, 1423–1435. [Google Scholar]
- Lewis, J; Burton, P. A study of newly excysted cells of Gonyaulax poyedra (Dinophyceae) by electron microscopy. Br phycol J 1988, 23, 49–60. [Google Scholar]
- Figueroa, RI; Bravo, I. Sexual reproduction and two different encystment strategies of Lingulodinium polyedrum (Dinophyceae) in culture. Journal of Phycology 2005, 41, 370–379. [Google Scholar]
- Hansen, G; Moestrup, O; Roberts, KR. Fine structural observations on Gonyaulax spinifera (Dinophyceae), with special emphasis on the flagellar apparatus. Phycologia 1996, 354–366. [Google Scholar]
- Steidinger, K; Tangen, K. Dinoflagellates; San Diego; Academic Press, 1996; pp. 387–598. [Google Scholar]
- Wall, D; Dale, B. Modern dinoflagellate cysts and evolution of the Peridiniales. Micropaleontology 1968, 14, 265–304. [Google Scholar]
- Dale, B. Fryxell, GA, Ed.; Dinoflagellate resting cysts: benthic plankton. In Survival strategies of the algae; Cambridge; Cambridge Univ. Press, 1983; pp. 69–136. [Google Scholar]
- Guerrini, F; Ciminiello, P; Dell-Aversano, C; Tartaglione, L; Fattorusso, E; Boni, L; Pistocchi, R. Influence of temperature, salinity and nutrient limitation on yessotoxin production and release by the dinoflagellate Protocetatium reticulatum in batch-cultures. Harmful Algae 2007, 6, 707–717. [Google Scholar]
- Miles, CO; Samdal, IA; Aasen, JAG; Jensen, DJ; Quilliam, MA; Petersen, D; Briggs, LR; Wilkins, AL; Rise, F; Cooney, JM; MacKenzie, L. Evidence for numerous analogs of Yessotoxin in Protoceratium reticulatum. Harmful Algae 2005, 4, 1075–1091. [Google Scholar]
- Satake, M; Terasawa, K; Kadowaki, Y; Yasumoto, T. Relative configuration of YTX and isolation of two new analogs from toxic scallops. Tetrahedron Lett 1996, 37, 5955–5958. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Poletti, R; Viviani, R. A new analogue of yessotoxin, carboxyyessotoxin, isolated from Adriatic Sea mussels. Eur J Org Chem 2000, 291–295. [Google Scholar]
- Yasumoto, T; Murata, M; Lee, JS; Torigoe, K. Polyether toxins produced by dinoflagellates; Amsterdam; Elsevier, 1989; pp. 375–382. [Google Scholar]
- Satake, M; Eiki, K; Ichimura, T; Ota, S; Sekiguchi, K; Oshima, Y. Structure of 45,46,47-trinorhomoyessotoxin, a new yessotoxin analog, from Protoceratium reticulatum which represents the first detection of a homoyessotoxin analog in Japan. Harmful Algae 2006, 5, 731–735. [Google Scholar]
- Satake, M; Tubaro, A; Lee, JS; Yasumoto, T. Two new analogs of yessotoxin, homoyessotoxin and 45-hydroxyhomoyessotoxin, isolated from mussels of the Adriatic Sea. Nat Toxins 1997, 5, 107–110. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Poletti, R; Viviani, R. Structure determination of carboxyhomoyessotoxin, a new yessotoxin analogue isolated from adriatic mussels. Chem Res Toxicol 2000, 13, 770–774. [Google Scholar]
- Aasen, J; Samdal, IA; Miles, CO; Dahl, E; Briggs, LR; Aune, T. Yessotoxins in Norwegian blue mussels (Mytilus edulis): uptake from Protoceratium reticulatum, metabolism and depuration. Toxicon 2005, 45, 265–272. [Google Scholar]
- Ciminiello, P; Dell-Aversano, C; Fattorusso, E; Forino, M; Magno, S; Poletti, R. The detection and identification of 42,43,44,45,46,47,55-heptanor-41-oxoyessotoxin, a new marine toxin from adriatic shellfish, by liquid chromatography-mass spectrometry. Chem Res Toxicol 2002, 15, 979–984. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Poletti, R. 42,43,44,45,46,47,55-Heptanor-41-oxohomoyessotoxin, a new biotoxin from mussels of the northern Adriatic sea. Chem Res Toxicol 2001, 14, 596–599. [Google Scholar]
- Miles, CO; Wilkins, AL; Hawkes, AD; Selwood, A; Jensen, DJ; Aasen, J; Munday, R; Samdal, IA; Briggs, LR; Beuzenberg, V; MacKenzie, AL. Isolation of a 1,3-enone isomer of heptanor-41-oxoyessotoxin from Protoceratium reticulatum cultures. Toxicon 2004, 44, 325–336. [Google Scholar]
- Miles, CO; Wilkins, AL; Jensen, DJ; Cooney, JM; Quilliam, MA; Aasen, J; MacKenzie, AL. Isolation of 41a-Homoyessotoxin and the identification of 9-Methyl-41a-homoyessotoxin and Nor-ring-A-yessotoxin from Protoceratium reticulatum. Chem Res Toxicol 2004, 17, 1414–1422. [Google Scholar]
- Miles, CO; Wilkins, AL; Hawkes, AD; Selwood, A; Jensen, DJ; Munday, R; Cooney, JM; Beuzenberg, V. Polyhydroxilated amide analogs of yessotoxin from Protoceratium reticulatum. Toxicon 2005, 45, 61–71. [Google Scholar]
- Finch, SC; Wilkins, AL; Hawkes, AD; Jensen, DJ; MacKenzie, L; Beuzenberg, V; Quilliam, MA; Olseng, CD; Samdal, IA; Aasen, JAG; Selwood, AI; Cooney, JM; Sandvik, M; Miles, CO. Isolation and identification of (44-R,S)-44,55-dihydroxyyessotoxin from Protoceratium reticulatum, and its occurrence in extracts of shellfish from New Zealand, Norway and Canada. Toxicon 2005, 46, 160–170. [Google Scholar]
- Souto, ML; Fernandez, JJ; Franco, JM; Paz, B; Gil, LV; Norte, M. Glycoyessotoxin A, a new yessotoxin derivate from cultures of Protoceratium reticulatum. J Nat Prod 2005, 68, 420–422. [Google Scholar]
- Miles, CO; Wilkins, AL; Selwood, AI; Hawkes, AD; Jensen, DJ; Cooney, JM; Beuzenberg, V; MacKenzie, AL. Isolation of Yessotoxin 32-O-[β-l-arabinofuranosyl-(5’-1’’)-β-l-arabinofuranoside] from Protoceratium reticulatum. Toxicon 2006, 47, 510–516. [Google Scholar]
- Mitrovic, SM; Hamilton, B; McKenzie, L; Furey, A; James, KJ. Persistence of yessotoxin under light and dark conditions. Mar Environ Res 2005, 60, 397–401. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Magno, S; Poletti, R; Viviani, R. Isolation of Adriatoxin, a New Analogue of Yessotoxin from Mussels of the Adriatic Sea. Tetrahedron Lett 1998, 39, 8897–8900. [Google Scholar]
- MacKenzie, L; Holland, P; McNabb, P; Beuzenberg, V; Selwood, A; Suzuki, T. Complex toxin profiles in phytoplankton and Greenshell mussels (Perna canaliculus), revealed by LC-MS/MS analysis. Toxicon 2002, 40, 1321–1330. [Google Scholar]
- Samdal, IA; Aasen, JAG; Briggs, LR; Dahl, E; Miles, CO. Comparison of ELISA and LC-MS analyses for yessotoxins in blue mussels (Mytilus edulis). Toxicon 2005, 46, 7–15. [Google Scholar]
- Samdal, IA. Yessotoxins in algae and mussels -Studies on its sources, disposition, and levels; Oslo; Norwegian School of Veterinary Science, 2005; p. 53. [Google Scholar]
- Satake, M. Biosynthesis of the marine polyether toxin, yessotoxin. Tenner Yuki Kagobutsu Toronkay Koen Yoshishu 2000, 42, 259–264. [Google Scholar]
- Nakanishi, K. The chemistry of brevetoxins: A review. Toxicon 1985, 23, 473–479. [Google Scholar]
- Vilotijevic, I; Jamison, TF. Epoxide-Opening cascades promoted by water. Science 2007, 317, 1189–1192. [Google Scholar]
- Terao, K; Ito, E; Oarada, M; Murata, M; Yasumoto, T. Histopatological studies on experimental marine toxin poisoning-5. The effects in mice of yessotoxin isolated from Patinopecten yessoensis and of a desulfated derivate. Toxicon 1990, 28, 1095–1104. [Google Scholar]
- Satake, M; MacKenzie, L; Yasumoto, T. Identification of Protoceratium reticulatum as the biogenetic origin of yessotoxin. Nat Toxins 1997, 5, 164–167. [Google Scholar]
- Suárez-Korsnes, M; Hetland, DL; Espenes, A; Tranulis, MA; Aune, T. Apoptotic events induced by yessotoxin in myoblast cell lines from rat and mouse. Toxicol in Vitro 2006, 20, 1077–1087. [Google Scholar]
- Franchini, A; Marchesini, E; Poletti, R; Ottaviani, E. Acute toxic effect of the algal yessotoxin on Purkinje cells from the cerebellum of Swiss CD1 mice. Toxicon 2004, 43, 347–352. [Google Scholar]
- Franchini, A; Marchesini, E; Poletti, R; Ottaviani, E. Lethal and sub-lethal yessotoxin dose induced morpho-functional alterations in intraperitoneal injected Swiss CD1 mice. Toxicon 2004, 44, 83–90. [Google Scholar]
- Franchini, A; Marchesini, E; Poletti, R; Ottaviani, E. Swiss mice CD1 fed on mussels contaminated by okadaic acid and yessotoxins: effects on thymus and spleen. Eur J Histochem 2005, 49, 179–188. [Google Scholar]
- Tubaro, A; Sosa, S; Altinier, G; Soranzo, MR; Satake, M; Loggia, RD; Yasumoto, T. Short-term oral toxicity of homoyessotoxins, yessotoxin and okadaic acid in mice. Toxicon 2004, 43, 439–445. [Google Scholar]
- De la Rosa, LA; Alfonso, A; Vilariño, N; Vieytes, MR; Botana, LM. Modulation of cytosolic calcium levels of human lymphocytes by yessotoxin, a novel marine phycotoxin. Biochem Pharmacol 2001, 61, 827–833. [Google Scholar]
- De la Rosa, LA; Alfonso, A; Vilariño, N; Vieytes, MR; Yasumoto, T; Botana, LM. Maitotoxin-induced calcium entry in human lymphocytes modulation by yessotoxin, Ca2+ channel blockers and kinases. Cellular Signalling 2001, 12, 711–716. [Google Scholar]
- Alfonso, A; de la Rosa, LA; Vieytes, MR; Yasumoto, T; Botana, LM. Yessotoxin, a novel phycotoxin, activates phosphodiesterase activity. Effect of yessotoxin on cAMP levels in human lymphocytes. Biochem Pharmacol 2003, 65, 193–208. [Google Scholar]
- Pazos, MJ; Alfonso, A; Vieytes, MR; Yasumoto, T; Botana, LM. Kinetic Analysis of the Interaction between Yessotoxin and Analogues and Immobilized Phosphodiesterases Using a Resonant Mirror Optical Biosensor. Chem Res Toxicol 2005, 18, 1155–1160. [Google Scholar]
- Pazos, M-J; Alfonso, A; Vieytes, MR; Yasumoto, T; Botana, LM. Study of the Interaction between Different Phosphodiesterases and Yessotoxin Using a Resonant Mirror Biosensor. Chem Res Toxicol 2006, 19, 794–800. [Google Scholar]
- Leira, F; Alvarez, C; Vieites, JM; Vieytes, MR; Botana, LM. Characterization of distinct apoptotic changes induced by okadaic acid and yessotoxin in the BE(2)-M17 neuroblastoma cell line. Toxinol in Vitro 2002, 16, 23–31. [Google Scholar]
- Malaguti, C; Ciminiello, P; Fattorusso, E; Rossini, GP. Caspase activation and death induced by yessotoxin in HeLa cells. Toxicol in Vitro 2002, 16, 357–363. [Google Scholar]
- Suárez-Korsnes, M; Hetland, DL; Espenes, A; Aune, T. Induction of apoptosis by YTX in myoblast cell lines via mitochondrial signalling transduction pathway. Toxicol in Vitro 2006, 20, 1419–1426. [Google Scholar]
- Suárez-Korsnes, M; Hetland, DL; Espenes, A; Aune, T. Cleavage of tensin during cytoskeleton disruption in YTX-induced apoptosis. Toxicol in Vitro 2007, 21, 9–15. [Google Scholar]
- Ronzitti, G; Callegari, F; Malaguti, C; Rossini, GP. Selective disruption of the E-cadherin–catenin system by an algal toxin. Br J Cancer 2004, 90, 1100–1107. [Google Scholar]
- Malagoli, D; Ottaviani, E. Yessotoxin affects fMLP-induced cell shape changes in Mytilus galloprovincialis immunocytes. Cell Biol Int 2004, 28, 57–61. [Google Scholar]
- Malagoli, D; Casarini, L; Ottaviani, E. Algal toxin yessotoxin signalling pathways involve immunocyte mussel calcium channels. Cell Biol Int 2006, 30, 721–726. [Google Scholar]
- Malagoli, D; Marchesini, E; Ottaviani, E. Lysosomes as the target of yessotoxin in invertebrate and vertebrate cell lines. Toxicol Lett 2006, 167, 75–83. [Google Scholar]
- Ferrari, S; Ciminiello, P; Dell-Aversano, C; Forino, M; Malaguti, C; Tubaro, A; Poletti, R; Yasumoto, T; Fattorusso, E; Rossini, GP. Structure-Activity relationships of yessotoxins in cultured cells. Chem Res Toxicol 2004, 17, 1251–1257. [Google Scholar]
- Yasumoto, T; Oshima, Y; Yamaguchi, M. Occurrence of a new type of shellfish poisoning in the Tohoku district. Bull Jpn Soc Sci Fish 1978, 44, 1249–1255. [Google Scholar]
- Yasumoto, T; Murata, M; Oshima, Y; Matsumoto, GK; Clardy, J. Ragelis, EP, Ed.; Diarrhetic Shellfish Toxins. In Seafood toxins; Washington, D. C; American Chemical Society, 1984; pp. 207–214. [Google Scholar]
- EC. Commision Regulation Nº 2074/2005 of 5 December 2005, laying down implementing measures for certain products under Regulation (EC) Nº 853/2004 of the European Parliament and of the Council and for the organisation of official controls under Regulation (EC) Nº 854/2004 of the European Parliament and of the Council and Regulation (EC) Nº 882/2004 of the European Parliament and of the Council, derogating from Regulation (EC) Nº 852/2004 of the European Parliament and of the Council and amending Regulations (EC) Nº 853/2004 and (EC) Nº 854/2004. Official Journal of the European Union 2005, L 338, p. 27.
- Stabell, OB; Steffenak, I; Pedersen, K; Underdal, B. Diversity of shellfish toxins of "diarrhetic" type revealed by biological and chemical assays. J Toxicol Environ Health 1991, 33, 273–282. [Google Scholar]
- Suzuki, T; Jin, T; Shirota, Y; Mitsuya, T; Okomura, Y; Kamiyama, T. Quantification of lipophilic toxins associated with diarrhetic shellfish poisoning in Japanese bivalves by liquid chromatography–mass spectrometry and comparison with mouse bioassay. Fish Sci 2005, 71, 1370–1378. [Google Scholar]
- Hess, P; McMahon, T; Slattery, D; Swords, DDG; McCarron, M; Clarke, D; Gibbons, W; Silke, J; O’Cinneide, M. Villalba, A, Reguera, B, Romalde, JL, Beiras, R, Eds.; Use of LC-MS texting to identify lipophilic toxins, to establish local trends and interspecies differences and to test the comparability of LC-MS testing with the mouse bioassay: an example from the Irish biotoxin monitoring programme 2001. In Molluscan Shellfish Safety; Santiago de Compostela; Consellería de Pesca, Xunta de Galicia and IOC of UNESCO, 2003; pp. 57–66. [Google Scholar]
- Hess, P; Grune, B; Anderson, DB; Aune, T; Botana, LM; Caricato, P; Egmond, HPv; Halder, M; Hall, S; Lawrence, JF; Moffat, C; Poletti, R; Richmond, J; Rossini, GP; Seamer, C; Vilageliu, JS. Three Rs approaches in marine biotoxin testing. ATLA: Alternatives to Laboratory Animals 2006, 36, 193–224. [Google Scholar]
- Aune, T; Yasumoto, T; Engeland, E. Light and scanning electron microscopic studies on effects of marine algal toxins toward freshly prepared hepatocytes. J Toxicol Environ Health 1991, 34, 1–9. [Google Scholar]
- Pierotti, S; Malaguti, C; Milandri, A; Poletti, R; Rossini, GP. Functional assay to measure yessotoxins in contaminated mussel samples. Anal Biochem 2003, 312, 208–216. [Google Scholar]
- Pierotti, S; Albano, C; Milandri, A; Callegari, F; Poletti, R; Rossini, GP. A slot blot procedure for the measurement of yessotoxins by a functional assay. Toxicon 2007, 49, 36–45. [Google Scholar]
- Pazos, MJ; Alfonso, A; Vieytes, MR; Yasumoto, T; Vieites, JM; Botana, LM. Resonant mirror biosensor detection method based on yessotoxin–phosphodiesterase interactions. Anal Biochem 2004, 335, 112–118. [Google Scholar]
- Briggs, LR; Miles, CO; Fitzgerald, JM; Ross, KM; Garthwaite, I; Towers, NR. Enzyme-linked immunosorbent assay for the detection of yessotoxin and its analogues. J Agric Food Chem 2004, 52, 5836–5842. [Google Scholar]
- Kleivdal, H; Briggs, L; Miles, CO. Development and validation of YTX ELISA-a rapid assay for the determination of yessotoxin in shellfish and environmental samples; Baiona: Spain, 2005; p. 53. [Google Scholar]
- Ramstad, H; Larsen, S; Aune, T. Repeatability and validity of a fluorimetric HPLC method in the quantification of yessotoxin in blue mussels (Mytilus edulis) related to the mouse bioassay. Toxicon 2001, 39, 1393–1397. [Google Scholar]
- Paz, B; Riobó, P; Souto, ML; Gil, LV; Norte, M; Fernandez, JJ; Franco, JM. Detection and identification of glycoyessotoxin A in a culture of the dinoflagellate Protoceratium reticulatum. Toxicon 2006, 48, 611–619. [Google Scholar]
- Stobo, LA; Lacaze, JP; Scott, AC; Gallacher, S; Smith, EA; Quilliam, MA. Liquid chromatography with mass spectrometry-detection of lipophilic shellfish toxins. J AOAC Int 2005, 88, 1371–1382. [Google Scholar]
- Draisci, R; Giannetti, L; Lucentini, L; Ferretti, E; Palleschi, L; Marchiafava, C. Direct identification of yessotoxin in shellfish by liquid chromatography coupled with mass spectrometry and tandem mass spectrometry. Rapid Commun Mass Spectrom 1998, 12, 1291–1296. [Google Scholar]
- Ciminiello, P; Dell-Aversano, C; Fattorusso, E; Forino, M; Magno, S; Poletti, R. Direct detection of yessotoxin and its analogues by liquid chromatography coupled with electrospray ion trap mass spectrometry. J Chromatogr A 2002, 968, 61–69. [Google Scholar]
- Cooney, JM; Jensen, DJ; Miles, CO. Ion-trap LC-MS as a tool for structural characterisation of matrine algal toxins; Cawthron Institute, P. R: New Zealand, 2003; pp. 59–64. [Google Scholar]
- Draisci, R; Palleschi, L; Giannetti, L; Lucentini, L; James, KJ; Bishop, AG; Satake, M; Yasumoto, T. New approach to the direct detection of known and new diarrhoeic shellfish toxins in mussels and phytoplankton by liquid chromatography-mass spectrometry. J Chromatogr A 1999, 847, 213–221. [Google Scholar]
- Fernandez-Amandi, M; Furey, A; Lehane, M; Ramstad, H; James, KJ. Liquid chromatography with electrospray ion-trap mass spectrometry for the determination of yessotoxins in shellfish. J Chromatogr A 2002, 976, 329–334. [Google Scholar]
- Goto, H; Igarashi, T; Yamamoto, M; Yasuda, M; Sekiguchi, R; Watai, M; Tanno, K; Yasumoto, T. Quantitative determination of marine toxins associated with diarrhetic shellfish poisoning by liquid chromatography coupled with mass spectrometry. J Chromatogr A 2001, 907, 181–189. [Google Scholar]
- Fux, E; McMillan, D; Bire, R; Hess, P. Development of an ultra-performance liquid chromatography.mass spectrometry method for the detection of lipophilic marine toxins. J Chromatogr A 2007, 1157, 273–280. [Google Scholar]
- De la Iglesia, P; Gago-Martinez, A; Yasumoto, T. Advanced studies for the application of high performance capillary electrophoresis for the analysis of yessotoxin and 45-hydroxyyessotoxin. J Chromatogr A 2007, 1156, 160–166. [Google Scholar]
- EC. Commission Decision 2002/225/EC of 15 March 2002 laying down detailed rules for the implementation of Council Directive 91/492/EEC as regards the maximum levels and the methods of analysis of certain marine biotoxins in bivalve molluscs, echinoderms, tunicates and marine gastropods. Official Journal of the European Communities 2002, p. 62.




| P. reticulatum Strain | Location | Analysis Technique | YTXs (pg/cell) | Reference |
|---|---|---|---|---|
| Yamada Bay | Japan | LC-FLD | 14 | [24] |
| New Zealand | New Zealand | LC-FLD | 3.0 | [24] |
| Mutsu Bay | Japan | LC-FLD | 0.9–11 | [12] |
| Emilia-Romagna | Italy | LC-FLD | 15.7 | [29] |
| VGO758 | Spain | LC-FLD | 28.7 | [15] |
| VGO764* | Spain | LC-FLD | 20.5 * | [15] |
| GG1AM | Spain | LC-FLD | 2.9 | [15] |
| UW351 | UK | LC-MS | 0.3 | [25] |
| UW409 | Canada | LC-MS | 5 | [25] |
| Adriatic | Italy | LC-MS | 11.4 | [13] |
| CAWD40 | New Zealand | LC-MS | 10–15 | [65] |
| OM6-NP31* | Japan | LC-MS | 71.7 * | [16] |
| 10628-OK-PR-C | Japan | LC-MS | 59.8 | [16] |
| 020717-OK-PR-7 | Japan | LC-MS | 1.0 | [16] |
| CAWD40 | New Zealand | ELISA | 30 | [26] |
| AP2 | Norway | ELISA | 19-22 | [26] |
| Sognfj03 | Norway | ELISA | 19-34 | [26] |
| CAWD40 | New Zealand | ELISA | 8.3 | [34] |
Share and Cite
Paz, B.; Daranas, A.H.; Norte, M.; Riobó, P.; Franco, J.M.; Fernández, J.J. Yessotoxins, a Group of Marine Polyether Toxins: an Overview. Mar. Drugs 2008, 6, 73-102. https://doi.org/10.3390/md6020073
Paz B, Daranas AH, Norte M, Riobó P, Franco JM, Fernández JJ. Yessotoxins, a Group of Marine Polyether Toxins: an Overview. Marine Drugs. 2008; 6(2):73-102. https://doi.org/10.3390/md6020073
Chicago/Turabian StylePaz, Beatriz, Antonio H. Daranas, Manuel Norte, Pilar Riobó, José M. Franco, and José J. Fernández. 2008. "Yessotoxins, a Group of Marine Polyether Toxins: an Overview" Marine Drugs 6, no. 2: 73-102. https://doi.org/10.3390/md6020073
APA StylePaz, B., Daranas, A. H., Norte, M., Riobó, P., Franco, J. M., & Fernández, J. J. (2008). Yessotoxins, a Group of Marine Polyether Toxins: an Overview. Marine Drugs, 6(2), 73-102. https://doi.org/10.3390/md6020073





