Tetrodotoxin – Distribution and Accumulation in Aquatic Organisms, and Cases of Human Intoxication
Abstract
:1. Introduction
2. TTX distribution in aquatic organisms
2.1. Distribution of TTX in pufferfish species
2.2. Distribution of TTX in pufferfish bodies
2.3. Distribution of TTX in animals other than pufferfish
3. Mechanism of TTX accumulation in TTX-bearing organisms
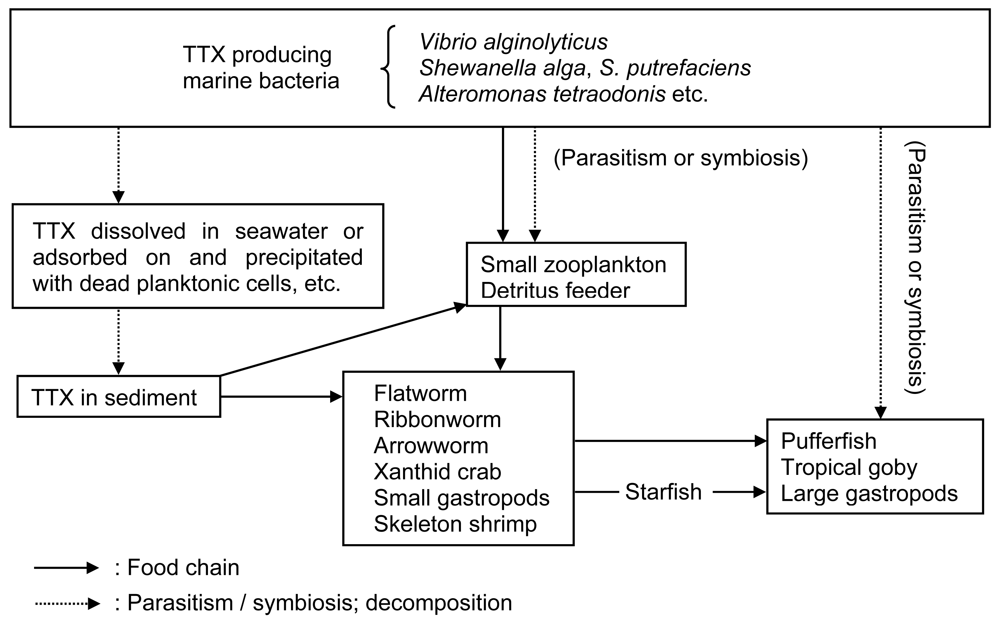
3.1. TTX producers
3.2. Mechanism of TTX accumulation in pufferfish
3.3. Mechanism of TTX accumulation in amphibians
3.4. Resistance of animals to TTX
4. Cases of human intoxication by TTX
4.1. Poisonings due to pufferfish
4.1.1. Cases in Japan
Case report of pufferfish poisoning [96]
4.1.2. Cases in Taiwan/China
Case report of poisoning cases in Taiwan [100]
4.1.3. Cases in other countries
4.2. Poisoning due to small gastropods
4.3. Poisoning due to other TTX-bearing animals
5. Conclusion
References
- Yokoo, A. Chemical studies on pufferfish toxin (3) – separation of spheroidine. Nippon Kagaku Zasshi 1950, 71, 590–592. [Google Scholar]
- Tsuda, K; Ikuma, S; Kawamura, M; Tachikawa, R; Sakai, K; Tamura, C; Amakasu, D. Tetrodotoxin. VII. On the structures of tetrodotoxin and its derivatives. Chem Pharm Bull 1964, 12, 1357–1374. [Google Scholar]
- Woodward, RB. The structure of tetrodotoxin. Pure Appl Chem 1964, 9, 49–74. [Google Scholar]
- Goto, T; Takahashi, S; Kishi, Y; Hirata, Y. Tetrodotoxin. Tetrahedron 1965, 21, 2059–2088. [Google Scholar]
- Narahashi, T. Pharmacology of tetrodotoxin. J Toxicol-Toxin Rev 2001, 20, 67–84. [Google Scholar]
- Noguchi, T; Ebesu, JSM. Puffer poisoning: epidemiology and treatment. J Toxicol-Toxin Rev 2001, 20, 1–10. [Google Scholar]
- Yotsu-Yamashita, M. Chemistry of puffer fish toxin. J Toxicol-Toxin Rev 2001, 20, 51–66. [Google Scholar]
- Jang, JH; Yotsu-Yamashita, M. 6,11-Dideoxytetrodotoxin from the puffer fish, Fugu pardalis. Toxicon 2007, 50, 947–951. [Google Scholar]
- Miyazawa, K; Noguchi, T. Distribution and origin of tetrodotoxin. J Toxicol-Toxin Rev 2001, 20, 11–33. [Google Scholar]
- Noguchi, T; Narita, H; Maruyama, J; Hashimoto, K. Tetrodotoxin in the starfish Astropecten polyacanthus, in association with toxification of a trumpet shell, “boshubora”, Charonia sauliae. Bull Japan Soc Sci Fish 1982, 48, 1173–1177. [Google Scholar]
- Noguchi, T; Jeon, JK; Arakawa, O; Sugita, H; Deguchi, Y; Shida, Y; Hashimoto, K. Occurrence of tetrodotoxin in Vibrio sp. isolated from intestines of xanthid crab, Atergatis floridus. J Biochem 1986, 99, 311–314. [Google Scholar]
- Matsui, T; Sato, H; Hamada, S; Shimizu, C. Comparison of toxicity of the cultured and wild puffer fish Fugu niphobles. Bull Japan Soc Sci Fish 1982, 48, 253. [Google Scholar]
- Saito, T; Maruyama, J; Kanoh, S; Jeon, JK; Noguchi, T; Harada, T; Murata, O; Hashimoto, K. Toxicity of the cultured pufferfish Fugu rubripes rubripes along with their resistibility against tetrodotoxin. Bull Japan Soc Sci Fish 1984, 50, 1573–1575. [Google Scholar]
- Noguchi, T; Arakawa, O; Takatani, T. Toxicity of pufferfish Takifugu rubripes cultured in netcages at sea or aquaria on land. Comp Biochem Physiol, Part D 2006, 1, 153–157. [Google Scholar]
- Matsui, T; Hamada, S; Konosu, S. Difference in accumulation of puffer fish toxin and crystalline tetrodotoxin in the puffer fish, Fugu rubripes rubripes. Bull Japan Soc Sci Fish 1981, 47, 535–537. [Google Scholar]
- Yamamori, K; Kono, M; Furukawa, K; Matsui, T. The toxification of juvenile cultured kusafugu Takifugu niphobles by oral administration of crystalline tetrodotoxin. J Food Fyg Soc Japan 2004, 45, 73–75. [Google Scholar]
- Honda, S; Arakawa, O; Takatani, T; Tachibana, K; Yagi, M; Tanigawa, A; Noguchi, T. Toxification of cultured puffer fish Takifugu rubripes by feeding on tetrodotoxin-containing diet. Nippon Suisan Gakkaishi 2005, 71, 815–820. [Google Scholar]
- Tani, I. Studies on Japanese Pufffish in Association with Poisoning due to Ingestion of Them; Teikoku Tosho: Tokyo, Japan, 1945; p. 103. [Google Scholar]
- Environmental Health Bureau, Ministry of Health and Welfare, Pufferfishes Available in Japan-An Illustrated Guide to Their Identification; Chuo-Hokishuppan: Tokyo, Japan, 1984; p. 79.
- Kanoh, S. Hashimoto, K, Ed.; Distribution of tetrodotoxin in vertebrates. In Recent Advances in Tetrodotoxin Research; Koseisha-Koseikaku: Tokyo, Japan, 1988; pp. 32–44. [Google Scholar]
- Fuchi, Y; Narimatsu, H; Nakama, S; Kotobuki, H; Hirakawa, H; Torishima, Y; Noguchi, T; Ohtomo, N. Tissue distribution of toxicity in a pufferfish, Arothron firmamentum (“hoshifugu”). J Food Hyg Soc Japan 1991, 32, 520–524. [Google Scholar]
- Khora, SS; Isa, J; Yasumoto, T. Toxicity of puffers from Okinawa, Japan. Nippon Suisan Gakkaishi 1991, 57, 163–167. [Google Scholar]
- Hwang, DF; Kao, CY; Yang, HC; Jeng, SS; Noguchi, T; Hashimoto, K. Toxicity of puffer in Taiwan. Nippon Suisan Gakkaishi 1992, 58, 1541–1547. [Google Scholar]
- Boylan, DB; Scheuer, PJ. Pahutoxin: a fish poison. Science 1967, 155, 52. [Google Scholar]
- Thomson, DA. Freudenthal, HD, Ed.; Drugs from the Sea; Marine Technology Society: Washington, D.C., USA, 1968. [Google Scholar]
- Taniyama, S. Toxicity and toxin profile of marine boxfish. In Studies on Parrotfish Poisoning and Similar Incidents Caused by Other Fishes; Ph.D. Thesis, Nagasaki University: Nagasaki, Japan, 2002; pp. 73–80. [Google Scholar]
- Mahmud, Y; Yamamori, K; Noguchi, T. Occurrence of TTX in a brackishwater puffer “midorifugu” Tetraodon nigroviridis, collected from Thailand. J Food Hyg Soc Japan 1999, 40, 363–367. [Google Scholar]
- Mahmud, Y; Yamamori, K; Noguchi, T. Toxicity and tetrodotoxin as the toxic principle of a brackishwater puffer Tetraodon steindachneri, collected from Thailand. J Food Hyg Soc Japan 1999, 40, 391–395. [Google Scholar]
- Kungsuwan, A; Arakawa, O; Promdet, M; Onoue, Y. Occurrence of paralytic shellfish poisons in Thai freshwater puffers. Toxicon 1997, 35, 1341–1346. [Google Scholar]
- Sato, S; Kodama, M; Ogata, T; Saitanu, K; Furuya, M; Hirayama, K; Kakinuma, K. Saxitoxin as toxic principle of a freshwater puffer, Tetraodon fangi, in Thailand. Toxicon 1997, 35, 137–140. [Google Scholar]
- Oliveira, JS; Fernandes, SCR; Schwartz, CA; Bloch, C, Jr; Melo, JAT; Pires, OR, Jr; Freitas, JC. Toxicity and toxin identification in Colomesus asellus, an Amazonian (Brazil) freshwater puffer fish. Toxicon 2006, 48, 55–63. [Google Scholar]
- Ngy, L; Tada, K; Yu, CF; Takatani, T; Arakawa, O. Occurrence of paralytic shellfish toxins in Cambodian Mekong pufferfish Tetraodon turgidus: Selective toxin accumulation in the skin. Toxicon 2008, 51, 280–288. [Google Scholar]
- Sato, S; Ogata, T; Borja, V; Gonzales, C; Fukuyo, Y; Kodama, M. Frequent occurrence of paralytic shellfish poisoning toxins as dominant toxins in marine puffer from tropical water. Toxicon 2000, 38, 1101–1109. [Google Scholar]
- Nakashima, K; Arakawa, O; Taniyama, S; Nonaka, M; Takatani, T; Yamamori, K; Fuchi, Y; Noguchi, T. Occurrence of saxitoxins as a major toxin in the ovary of a marine puffer Arothron firmamentum. Toxicon 2004, 43, 207–212. [Google Scholar]
- Taniyama, S; Mahmud, Y; Tanu, MB; Takatani, T; Arakawa, O; Noguchi, T. Delayed haemolytic activity by the freshwater puffer Tetraodon sp. toxin. Toxicon 2001, 39, 725–727. [Google Scholar]
- Mahmud, Y; Tanu, MB; Takatani, T; Asayama, E; Arakawa, O; Noguchi, T. Chelonodon patoca, a highly toxic marine puffer in Japan. J Nat Toxins 2001, 10, 69–74. [Google Scholar]
- Zaman, L; Arakawa, O; Shimosu, A; Onoue, Y. Occurrence of paralytic shellfish poison in Bangladeshi freshwater puffers. Toxicon 1997, 35, 423–431. [Google Scholar]
- Tanu, MB; Mahmud, Y; Takatani, T; Kawatsu, K; Hamano, Y; Arakawa, O; Noguchi, T. Localization of tetrodotoxin in the skin of a brackishwater puffer Tetraodon steindachneri on the basis of immunohistological study. Toxicon 2002, 40, 103–106. [Google Scholar]
- Mahmud, Y; Okada, K; Takatani, T; Kawatsu, K; Hamano, Y; Arakawa, O; Noguchi, T. Intra-tissue distribution of tetrodotoxin in two marine puffers Takifugu vermicularis and Chelonodon patoca. Toxicon 2003, 41, 13–18. [Google Scholar]
- Mahmud, Y; Arakawa, O; Ichinose, A; Tanu, MB; Takatani, T; Tsuruda, K; Kawatsu, K; Hamano, Y; Noguchi, T. Intracellular visualization of tetrodotoxin (TTX) in the skin of a puffer Tetraodon nigroviridis by immunoenzymatic technique. Toxicon 2003, 41, 605–611. [Google Scholar]
- Kodama, M; Ogata, T; Sato, S. External secretion of tetrodotoxin from puffer fishes stimulated by electric shock. Marine Biol 1985, 87, 199–202. [Google Scholar]
- Saito, T; Noguchi, T; Harada, T; Murata, O; Hashimoto, K. Tetrodotoxin as a biological defense agent for puffers. Bull Japan Soc Sci Fish 1985, 51, 1175–1180. [Google Scholar]
- Mosher, HS; Fuhrman, GJ; Fuhrman, FA; Fischer, HG. Tarichatoxin-tetrodotoxin, a potent neurotoxin. Science 1965, 144, 1100–1110. [Google Scholar]
- Miyazawa, K; Jeon, JK; Maruyama, J; Noguchi, T; Ito, K; Hashimot, K. Occurrence of tetrodotoxin in the flatworm Planocera multitentaculata. Toxicon 1986, 24, 645–650. [Google Scholar]
- Miyazawa, K; Higashiyama, M; Ito, K; Noguchi, T; Arakawa, O; Shida, Y; Hashimoto, K. Tetrodotoxin in two species of ribbon worm (Nemertini), Lineus fuscoviridis and Tubulanus punctatus. Toxicon 1988, 26, 867–874. [Google Scholar]
- Ali, AE; Arakawa, O; Noguchi, T; Miyazawa, K; Shida, Y; Hashimoto, K. Tetrodotoxin and related substances in a ribbon worm Cephalothrix linearis (Nemertean). Toxicon 1990, 28, 1083–1093. [Google Scholar]
- Narita, H; Noguchi, T; Maruyama, J; Ueda, Y; Hashimoto, K; Watanabe, Y; Hida, K. Occurrence of tetrodotoxin in a trumpet shell, “boshubora” Charonia sauliae. Bull Japan Soc Sci Fish 1981, 47, 935–941. [Google Scholar]
- Noguchi, T; Maruyama, J; Ueda, Y; Hashimoto, K; Harada, T. Occurrence of tetrodotoxin in the Japanese ivory shell Babylonia japonica. Bull Japan Soc Sci Fish 1981, 47, 901–913. [Google Scholar]
- Noguchi, T; Maruyama, J; Narita, H; Hashimoto, K. Occurrence of tetrodotoxin in the gastropod mollusk Tutufa lissostoma (frog shell). Toxicon 1984, 22, 219–226. [Google Scholar]
- Narita, H; Noguchi, T; Maruyama, J; Hashimoto, K. Occurrence of a tetrodotoxin-associated substance in a gastropod, “hanamushirogai” Zeuxis siquijorensis. Bull Japan Soc Sci Fish 1984, 50, 85–88. [Google Scholar]
- Jeon, JK; Narita, H; Nara, M; Noguchi, T; Maruyama, J; Hashimoto, K. Occurrence of tetrodotoxin in a gastropod mollusk, “araregai” Niotha clathrata. Bull Japan Soc Sci Fish 1984, 50, 2099–2102. [Google Scholar]
- Hwang, DF; Chueh, CH; Jeng, SS. Occurrence of tetrodotoxin in the gastropod mollusk Natica lineata (lined moon shell). Toxicon 1990, 28, 21–27. [Google Scholar]
- Narita, H. Tetrodotoxin, with special reference to its origin and the mechanism of toxification. Seikatsu Eisei 1991, 35, 2–15. [Google Scholar]
- Sheumack, DD; Howden, MEH. Maculotoxin: a neurotoxin from the venom glands of the octopus Hapalochlaena maculosa identified as tetrodotoxin. Science 1978, 199, 188–189. [Google Scholar]
- Yotsu-Yamashita, M; Mebs, D; Flachsenberger, W. Distribution of tetrodotoxin in the body of the blue-ringed octopus (Hapalochlaena maculosa). Toxicon 2007, 49, 410–412. [Google Scholar]
- Yasumoto, T; Yotsu, M; Endo, A; Murata, M; Kao, CY. Interspecies distribution and biogenetic origin of tetrodotoxin and its derivatives. Pure Appl Chem 1989, 61, 505–508. [Google Scholar]
- Noguchi, T; Uzu, A; Koyama, K; Hashimoto, K. Occurrence of tetrodotoxin as the major toxin in xanthid crab Atergatis floridus. Bull Japan Soc Sci Fish 1983, 49, 1887–1892. [Google Scholar]
- Yasumura, D; Oshima, Y; Yasumoto, T; Alcala, AC; Alcala, LC. Tetrodotoxin and paralytic shellfish toxins in Philippine crabs. Agric Biol Chem 1986, 50, 593–598. [Google Scholar]
- Kungsuwan, A; Nagashima, Y; Noguchi, T; Shida, Y; Suvapeepan, S; Suwansakornkul, P; Hashimoto, K. Tetrodotoxin in the horseshoe crab Carcinoscorpius rotundicauda inhabiting Thailand. Nippon Suisan Gakkaishi 1987, 53, 261–266. [Google Scholar]
- Thuesen, EV; Kogure, K; Hashimoto, K; Nemoto, T. Poison arrowworms: a tetrodotoxin venom in the marine phylum Chaetonnatha. J Exp Mar Biol Ecol 1988, 116, 249–256. [Google Scholar]
- Maruyama, J; Noguchi, T; Jeon, JK; Harada, T; Hashimoto, K. Occurrence of tetrodotoxin in the starfish Astropecten latespinosus. Experientia 1984, 40, 1395–1396. [Google Scholar]
- Maruyama, J; Noguchi, T; Narita, H; Jeon, JK; Otsuka, M; Hashimoto, K. Occurrence of tetrodotoxin in a starfish, Astropecten scoparius. Agric Biol Chem 1985, 49, 3069–3070. [Google Scholar]
- Noguchi, T; Hashimoto, Y. Isolation of tetrodotoxin from a goby Gobius criniger. Toxicon 1973, 11, 305–307. [Google Scholar]
- Yotsu, M; Iorizzi, M; Yasumoto, T. Distribution of tetrodotoxin, 6-epitetrodotoxin, and 11-deoxytetrodotoxin in newts. Toxicon 1990, 28, 238–241. [Google Scholar]
- Yotsu-Yamashita, M; Mebs, D. The levels of tetrodotoxin and its analogue 6-epitetrodotoxin in the red-spotted newt Notophthalmus viridescens. Toxicon 2001, 39, 1261–1263. [Google Scholar]
- Yasumoto, T; Yotsu, M; Murata, M. New tetrodotoxin analogues from the newt Cynops ensicauda. J Am Chem Soc 1988, 110, 2344–2345. [Google Scholar]
- Yotsu-Yamashita, M; Mebs, D; Kwet, A; Schneider, M. Tetrodotoxin and its analogue 6-epitetrodotoxin in newts (Triturus spp.; Urodela, Salamandridae) from southern Germany. Toxicon 2007, 50, 306–309. [Google Scholar]
- Kim, YH; Brown, GB; Mosher, HS. Tetrodotoxin: occurrence in atelopid frogs of Costa Rica. Science 1975, 189, 151–152. [Google Scholar]
- Daly, JW; Gusovsky, F; Myers, CW; Yotsu-Yamashita, M; Yasumoto, T. First occurrence of tetrodotoxin in a dendrobatid frog (Colostethus inguinalis), with further reports for the bufonid genus Atelopus. Toxicon 1994, 32, 279–285. [Google Scholar]
- Tanu, MB; Mahmud, Y; Tsuruda, K; Arakawa, O; Noguchi, T. Occurrence of tetrodotoxin in the skin of a rhacophoridid frog Polypedates sp. from Bangladesh. Toxicon 2001, 39, 937–941. [Google Scholar]
- Pires, OR, Jr; Sebben, A; Schwartz, EF; Largura, SWR; Bloch, C, Jr; Morales, RAV; Schwartz, CA. Occurrence of tetrodotoxin (TTX) and its analogues in the Brazilian frog Brachycephalus ephippium (Anura: Brachycephalidae). Toxicon 2002, 40, 761–766. [Google Scholar]
- Pires, OR, Jr; Sebben, A; Schwartz, EF; Morales, RAV; Bloch, C, Jr; Schwartz, CA. Further report of the occurrence of tetrodotoxin and new analogues in the Anuran family Brachycephalidae. Toxicon 2005, 45, 73–79. [Google Scholar]
- Narita, H; Matsubara, S; Miwa, N; Akahane, S; Murakami, M; Goto, T; Nara, M; Noguchi, T; Saito, T; Shida, Y; Hashimoto, K. Vibrio alginolyticus, a TTX-producing bacterium isolated from the starfish Astropecten polyacanthus. Bull Japan Soc Sci Fish 1987, 47, 935–941. [Google Scholar]
- Hashimoto, K; Noguchi, T; Watabe, S. Pohland, AE, Ed.; New aspect of tetrodotoxin. In Microbial Toxins in Foods and Feeds; Plenum Press: New York, 1990; pp. 159–172. [Google Scholar]
- Suenaga, K; Kotoku, S. Detection of tetrodotoxin in autopsy material by gas chromatography. Arch Toxicol 1980, 44, 291–297. [Google Scholar]
- Yasumoto, T; Yasumura, D; Yotsu, M; Michishita, T; Endo, A; Kotaki, Y. Bacterial production of tetrodotoxin and anhydrotetrodotoxin. Agric Biol Chem 1986, 50, 793–795. [Google Scholar]
- Matsui, T; Taketsugu, S; Sato, H; Yamamori, K; Kodama, K; Ishi, A; Hirose, H; Shimizu, C. Toxification of cultured puffer fish by the administration of tetrodotoxin producing bacteria. Nippon Suisan Gakkaishi 1990, 56, 705. [Google Scholar]
- Simidu, U; Noguchi, T; Hwang, DF; Shida, Y; Hashimoto, K. Marine bacteria which produce tetrodotoxin. Appl Environ Microbiol 1987, 53, 1714–1715. [Google Scholar]
- Noguchi, T. Hashimoto, K, Ed.; Food chain-associated toxification of tetrodotoxin-bearing animals. In Recent Advances in Tetrodotoxin Research; Koseisha-Koseikaku: Tokyo, 1988; pp. 85–93. [Google Scholar]
- Shimizu, Y; Kobayashi, M. Apparent lack of tetrodotoxin biosynthesis in captured Taricha torosa and Taricha granulosa. Chem Pharm Bull 1983, 31, 3625–3631. [Google Scholar]
- Hanifin, CT; Brodie, ED, III; Brodie, ED, Jr. Tetrodotoxin levels of the rough-skin newt, Taricha granulosa, increase in long-term captivity. Toxicon 2002, 40, 1149–1153. [Google Scholar]
- Cardall, BL; Brodie, ED, Jr; Brodie, ED, III; Hanifin, CT. Secretion and regeneration of tetrodotoxin in the rough-skin newt Taricha granulosa. Toxicon 2004, 44, 933–938. [Google Scholar]
- Tsuruda, K; Arakawa, O; Noguchi, T. Toxicity and toxin profiles of the newt, Cynops pyrrhogaster from western Japan. J Nat Toxins 2001, 10, 79–89. [Google Scholar]
- Arakawa, K. Resistibility against TTX and PSP. In Studies on the Toxicity of a Japanese Newt Cynops pyrrhogaster; Doctoral thesis, Nagasaki University: Nagasaki, 2001; pp. 50–53. [Google Scholar]
- Daly, JW; Padgett, WL; Saunders, RL; Cover, JF, Jr. Absence of tetrodotoxin in a captive-raised riparian frog, Atelopus varius. Toxicon 1997, 35, 705–709. [Google Scholar]
- Saito, T; Noguchi, T; Harada, T; Murata, O; Abe, T; Hashimoto, K. Resistibility of toxic and nontoxic pufferfish against tetrodotoxin. Bull Japan Soc Sci Fish 1985, 51, 1371. [Google Scholar]
- Koyama, K; Noguchi, T; Uzu, A; Hashimoto, K. Resistibility of toxic and nontoxic crabs against paralytic shellfish poison and tetrodotoxin. Bull Japan Soc Sci Fish 1983, 49, 485–489. [Google Scholar]
- Kaneko, Y; Matsumoto, G; Hanyu, Y. TTX resistivity of Na+ channel in newt retinal neuron. Biochem Biophys Res Commun 1997, 240, 651–656. [Google Scholar]
- Yotsu-Yamashita, M; Nishimori, K; Nitanai, Y; Isemura, M; Sugimoto, A; Yasumoto, T. Binding properties of 3H-PbTx-3 and 3H-saxitoxin to brain membranes and to skeletal muscle membranes of puffer fish Fugu pardalis and the primary structure of a voltage-gated Na+ channel α-subunit (fMNa1) from skeletal muscle of F pardalis. Biochem Biophys Res Commun 2000, 267, 403–412. [Google Scholar]
- Venkatesh, B; Lu, SQ; Dandona, N; See, SL; Benner, S; Soong, TW. Genetic basis of tetrodotoxin resistance in pufferfishes. Current Biol 2005, 15, 2069–2072. [Google Scholar]
- Maruta, S; Yamaoka, K; Yotsu-Yamashita, M. Two critical residues in p-loop regions of puffer fish Na+ channels on TTX-sensitivity. Toxicon 2008, 51, 381–387. [Google Scholar]
- Geffeney, SL; Fujimoto, E; Brodie, ED, III; Brodie, ED, Jr; Ruben, PC. Evolutionary diversification of TTX-resistant sodium channels in a predator-prey interaction. Nature 2005, 434, 759–763. [Google Scholar]
- Bricejl, VM; Connell, L; Konoki, K; MacQuarrie, SP; Scheuer, T; Catterall, WA; Trainer, VL. Sodium channel mutation leading to saxitoxin resistance in clams increases risk of PSP. Nature 2005, 434, 763–767. [Google Scholar]
- Shiomi, K; Yamaguchi, S; Kikuchi, T; Yamamori, K; Matsui, T. Occurrence of tetrodotxin-binding high molecular weight substances in the body fluid of shore crab (Hemigrapsus sanguineus). Toxicon 1992, 30, 1529–1537. [Google Scholar]
- Nagashima, Y; Yamamoto, K; Shimakura, K; Shiomi, K. A tetrodotoxin-binding protein in the hemolymph of shore crab Hemigrapsus sanguineus: purification and properties.
- Noguchi, T; Akaeda, H. Pufferfish poisoning. Jpn J Toxicol 1998, 11, 339–345. [Google Scholar]
- Du, SS; Fu, YM; Shih, YC; Chang, PC; Chou, SS; Lue, YH; Hwang, DF. First report on suspected food poisoning with ingestion of dried seasoned fish fillet. J Food Drug Anal 1999, 7, 163–167. [Google Scholar]
- Hsieh, YW; Hwang, PA; Pan, HH; Chen, JB; Hwang, DF. Identification of tetrodotoxin and fish species in an adulterated dried mullet roe implicated in food poisoning. J Food Sci 2003, 68, 142–146. [Google Scholar]
- Hwang, DF; Hwang, PA; Tsai, YH; Lu, YH. Identification of tetrodotoxin and fish species in dried dressed fish fillets implicated in food poisoning. J Food Prot 2002, 65, 389–392. [Google Scholar]
- Hwang, DF; Noguchi, T. Tetrodotoxin poisoning. Adv Food Nutr Res 2007, 52, 142–236. [Google Scholar]
- Hsieh, YW; Shiu, YC; Cheng, CA; Chen, SK; Hwang, DF. Identification of toxin and fish species in cooked fish liver implicated in food poisoning. J Food Sci 2002, 67, 948–952. [Google Scholar]
- Ebesu, JSM; Hokama, Y; Noguchi, T. Hui, YH, Kitts, D, Stanfield, PS, Eds.; Tetrodotoxin. In Foodborne Disease Handbook; Marcel Dekker, Inc.: New York, USA, 2000; Volume 2, pp. 274–276. [Google Scholar]
- Helfrich, P. Fish poisoning in Hawaii. Hawaii Med J 1963, 22, 361. [Google Scholar]
- Sims, JK; Ostman, DC. Emergency diagnosis and management of mild human tetrodotoxin. Ann Emerg Med 1986, 15, 1094. [Google Scholar]
- Mahmud, Y; Tanu, MB; Noguchi, T. First occurrence of a food poisoning incident due to ingestion of Takifugu oblongus, along with a toxicological report on three marine puffer species in Bangladesh. J Food Hyg Soc Japan 1999, 40, 473–480. [Google Scholar]
- Laobhripatr, S; Limpakarnjanarat, K; Sangwonloy, O; Sudhasaneya, S; Anuchatvorakul, B; Leelasitor, S; Saitanu, K. Food poisoning due to consumption of the freshwater puffer Tetraodon fungi in Thailand. Toxicon 1990, 28, 1372–1375. [Google Scholar]
- Mahmud, Y; Arakawa, O; Noguchi, T. An epidemic survey on freshwater puffer poisoning in Bangladesh. J Nat Toxins 2000, 9, 319–326. [Google Scholar]
- Hwang, DF; Cheng, CA; Tsai, YH; Shih, DYC; Ko, HC; Yang, RZ; Jeng, SS. Identification of tetrodotoxin and paralytic shellfish toxins in marine gastropods implicated in food poisoning. Fish Sci 1995, 61, 675–679. [Google Scholar]
- Hwang, DF; Shiu, YC; Hwang, PA; Lu, YH. Tetrodotoxin in gastropods snails implicated in food poisoning in northern Taiwan. J Food Sci 2002, 65, 1341–1344. [Google Scholar]
- Hwang, PA; Tsai, YH; Lu, YH; Hwang, DF. Paralytic toxins in three new gastropod Olividae species implicated in food poisoning in southern Taiwan. Toxicon 2003, 41, 529–533. [Google Scholar]
- Hwang, PA; Tsai, YH; Deng, JF; Cheng, CA; Ho, PH; Hwang, DF. Identification of tetrodotoxin in a marine gastropod Nassarius glans responsible for human morbidity and mortality in Taiwan. J Food Prot 2005, 68, 1696–1701. [Google Scholar]
- Shiu, YC; Lu, YH; Tsai, YH; Hwang, DF. Occurrence of tetrodotoxin in the causative gastropod Polinices didyma and another gastropod Natica lineata collected from western Taiwan. J Food Drug Anal 2003, 11, 159–163. [Google Scholar]
- Sui, LM; Chen, K; Hwang, PA; Hwang, DF. Identification of tetrodotoxin in marine gastropod implicated in food poisoning. J Nat Toxins 2002, 11, 213–220. [Google Scholar]
- Shui, LM; Chen, K; Wang, JY; Mei, HZ; Wang, AZ; Lu, YH; Hwang, DF. Paralytic snail poisoning in Zhoushan: A 25-year retrospective analysis. J Food Prot 2003, 66, 110–114. [Google Scholar]
- Maruyama, J; Noguchi, T; Jeon, JK; Yamazaki, K; Hashimoto, K. Another occurrence of tetrodotoxin in a trumpet shell Charonia sauliae. J Food Hyg Soc Japan 1983, 24, 465–468. [Google Scholar]
- Kanchanapongkul, J; Krittayapoositpot, P. An epidemic of tetrodotoxin poisoning following ingestion of the horseshoe crab Carcinoscorpius rotundicauda. Southeast Asian J Trop Med Public Health 1995, 26, 364–367. [Google Scholar]
- Tanu, MB; Noguchi, T. Tetrodotoxin as a toxin principle in the horseshoe crab Carcinoscorpius rotundicauda collected from Bangladesh. J Food Hyg Soc Japan 1999, 40, 426–431. [Google Scholar]
- Ngy, L; Yu, CF; Takatani, T; Arakawa, O. Toxicity assessment for the horseshoe crab Carcinoscorpius rotundicauda collected from Cambodia. Toxicon 2007, 49, 843–847. [Google Scholar]
- Fusetani, N; Endo, H; Hashimoto, K; Kodama, M. Occurrence and properties of toxins in the horseshoe crab Carcinoscorpius rotundicauda. Toxicon 1983, 21, 165–168. [Google Scholar]
- Tanu, MB; Mahmud, Y; Arakawa, O; Takatani, T; Kajihara, H; Kawatsu, K; Hamano, Y; Asakawa, M; Miyazawa, K; Noguchi, T. Immunoenzymatic visualization of tetrodotoxin (TTX) in Cephalothrix species (Nemertea: Anopla: Palaeonemertea: Cephalotrichidae) and Planocera reticulata (Platyhelminthes: Turbellaria: Polycladida: Planoceridae). Toxicon 2004, 44, 515–520. [Google Scholar]
- Tsuruda, K; Arakawa, O; Kawatsu, K; Hamano, Y; Takatani, T; Noguchi, T. Secretory glands of tetrodotoxin in the skin of a Japanese newt Cynops pyrrhogaster. Toxicon 2002, 40, 131–136. [Google Scholar]
- Honda, S; Ichibu, T; Arakawa, O; Takatani, T; Tachibana, K; Yagi, M; Tanigawa, A; Noguchi, T. Antibody productivity against sheep red blood cells and splenocyte proliferation reaction of tiger puffer (Takifugu rubripes) fed with tetrodotoxin-containing diets. Aquaculture Sci 2005, 53, 205–210. [Google Scholar]
- Nagashima, Y; Toyoda, M; Hasobe, M; Shimakura, K; Shiomi, K. In vitro accumulation of tetrodotoxin in pufferfish liver tissue slices. Toxicon 2003, 41, 569–547. [Google Scholar]
- Matsumoto, T; Nagashima, Y; Kusuhara, H; Sugiyama, Y; Ishizaki, S; Shimakura, K; Shiomi, K. Involvement of carrier-mediated transport system in uptake of tetrodotoxin into liver tissue slices of puffer fish Takifugu rubripes. Toxicon 2007, 50, 173–179. [Google Scholar]
- Matsui, T; Yamamori, K; Furukawa, K; Kono, M. Purification and some properties of a tetrodotoxin binding protein from the blood plasma of kusafugu, Takifugu niphobles. Toxicon 2000, 38, 463–468. [Google Scholar]
- Yotsu-Yamashita, M; Sugimoto, A; Terakawa, T; Shoji, Y; Miyazawa, T; Yasumoto, T. Purification, characterization and cDNA cloning of a novel soluble saxitoxin and tetrodotoxin binding protein from plasma of the puffer fish, Fugu pardalis. Eur J Biochem 2001, 268, 5937–5946. [Google Scholar]
- Lee, JH; Kondo, H; Sato, S; Akimoto, S; Saito, T; Kodama, M; Watabe, S. Identification of novel genes related to tetrodotoxin intoxication in pufferfish. Toxicon 2007, 49, 939–953. [Google Scholar]
- Kawatsu, K; Hamano, Y; Yoda, T; Terano, Y; Shibata, T. Rapid and highly sensitive enzyme immunoassay for quantitative determination of tetrodotoxin. Jpn J Med Sci Biol 1997, 50, 133–150. [Google Scholar]

| Maximal toxicity* | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Family | Habitat | Species | Ovary | Testis | Liver | Skin | Intestine | Muscle | Blood |
| Tetraodontidae | Marine | Japanese pufferfish | |||||||
| Takifugu niphobles | ● | ○ | ● | ◎ | ● | ○ | — | ||
| T. poecilonotus | ● | ◎ | ● | ◎ | ◎ | ○ | — | ||
| T. pardalis | ● | ○ | ● | ◎ | ◎ | × | × | ||
| T. snyderi | ● | × | ● | ◎ | ◎ | ○ | — | ||
| T. porphyreus | ● | × | ● | ◎ | ◎ | × | — | ||
| T. chinensis | ● | — | ● | — | — | — | — | ||
| T. obscurus | ● | × | ◎ | ◎ | ◎ | × | — | ||
| T. exascurus | ● | × | ◎ | ◎ | — | × | — | ||
| T. pseudommus | ● | × | ○ | ○ | ○ | × | — | ||
| T. chrysops | ◎ | × | ◎ | ◎ | ○ | × | × | ||
| T. vermicularis | ◎ | × | ◎ | ◎ | ○ | × | — | ||
| T. rubripes | ◎ | × | ◎ | × | ○ | × | × | ||
| T. xanthopterus | ◎ | × | ◎ | × | ○ | × | — | ||
| T. stictonotus | ◎ | × | ◎ | ○ | × | × | — | ||
| Tetraodon alboreticulatus | ● | — | ○ | ○ | ◎ | ○ | — | ||
| Pleuranacanthus sceleratus | ● | — | ○ | ○ | ◎ | ○ | — | ||
| Chelonodon patoca** | ◎ | ◎ | ◎ | ● | — | ◎ | — | ||
| Arothron firmamentum | ◎ | × | × | ○ | × | × | — | ||
| Canthigaster rivulata | × | — | ○ | ◎ | ○ | × | — | ||
| Lagocephalus lunaris | × | × | × | ◎ | × | ● | — | ||
| L. inermis | × | × | ◎ | × | × | × | — | ||
| L. wheeleri | × | × | × | × | × | × | — | ||
| L. gloveri | × | × | × | × | × | × | — | ||
| Sphoeroides pachygaster | × | × | × | × | × | × | — | ||
| Marine | Chinese pufferfish | ||||||||
| Takifugu flavidus | ● | ◎ | ● | ◎ | ◎ | ○ | — | ||
| Brackish | Thai pufferfish | ||||||||
| Tetraodon nigroviridis | — | — | × | ◎ | ○ | ○ | — | ||
| T. steindachneri | — | — | × | ◎ | × | × | — | ||
| Diodontidae | Marine | Japanese pufferfish | |||||||
| Diodon holocanthus | × | — | × | × | × | × | — | ||
| Chilomycterus affinis | × | — | × | × | × | × | — | ||
| Ostraciidae | Marine | Japanese pufferfish | |||||||
| Ostracion immaculatum | × | × | × | × | × | × | — | ||
| Lactoria diaphana | × | × | × | × | × | × | — | ||
| Aracana aculeata | × | × | × | × | × | × | — | ||
| Animals | Toxic parts | Maximal toxicity* | Ref | |
|---|---|---|---|---|
| Platyhelminthes | ||||
| Turbellaria | ||||
| Flatworms | Planocera spp. | Whole body | ● | [44] |
| Nemertinea | ||||
| Ribbonworms | Lineus fuscoviridis | Whole body | ● | [45] |
| Tubulanus punctatus | Whole body | ◎ | [45] | |
| Cephalothrix linearis | Whole body | ● | [46] | |
| Mollusca | ||||
| Gastropoda | Charonia sauliae | Digestive gland | ● | [47] |
| Babylonia japonica | Digestive gland | ○ | [48] | |
| Tutufa lissostoma | Digestive gland | ◎ | [49] | |
| Zeuxis siquijorensis | Whole body | ● | [50] | |
| Niotha clathrata | Whole body | ● | [51] | |
| Niotha lineata | Whole body | ● | [52] | |
| Cymatium echo | Digestive gland | ○ | [53] | |
| Pugilina ternotoma | Digestive gland | ○ | [53] | |
| Cephalopoda | Hapalochlaena maculosa | Posterior salivary gland (adult) | ● | [54] |
| Whole body (semi-adult) | ○ | [55] | ||
| Annelida: | ||||
| Polychaeta | Pseudopolamilla occelata | Whole body | ○ | [56] |
| Arthropoda: | ||||
| Xanthidae crabs | Atergatis floridus | Whole body | ○ | [57] |
| Zosimus aeneus | Whole body | ○ | [58] | |
| Horseshoe crab | Carcinoscorpius rotundicauda | Egg | ○ | [59] |
| Chaetognatha: | ||||
| Arrowworms | Parasagitta spp. | Head | ▵ | [60] |
| Flaccisagitta spp. | Head | ▵ | [60] | |
| Echinodermata: | ||||
| Starfish | Astropecten spp. | Whole body | ◎ | [61,62] |
| Vertebrata: | ||||
| Pisces | ||||
| Goby | Yongeichthys criniger | Skin, viscera, gonad | ◎ | [63] |
| Amphibia | ||||
| Newts | Taricha spp. | Skin, egg, ovary, muscle, blood | ◎ | [43] |
| Notophthalmus spp. | Skin, egg ovary | ○ | [64,65] | |
| Cynopsis spp. | Skin, egg, ovary, muscle, blood | ○ | [66] | |
| Triturus spp. | Skin, egg, ovary, muscle, blood | ▵ | [64,67] | |
| Frogs | Atelopus spp. | Skin | ● | [68] |
| Colostethus sp. | Skin | ◎ | [69] | |
| Polypedates sp. | Skin | ◎ | [70] | |
| Brachycephalus spp. | Skin, liver | ◎ | [71,72] | |
| Place of culture | Year of collection | Age | Number of collection* | Toxicity (MU/g) |
|---|---|---|---|---|
| Nagasaki** | 2001 | 1–3 | 494 | < 2 |
| 2002 | 1–3 | 1021 | < 2 | |
| 2003 | 2 | 240 | < 2 | |
| Kumamoto | 2001 | 1–2 | 829 | < 2 |
| 2002 | 2 | 587 | < 2 | |
| 2003 | Unknown | 145 | < 2 | |
| Kagoshima | 1981–83 | 1–2 | 47 | < 10 |
| 1990–91 | 1–2 | 40 | < 5 | |
| 2002 | 2 | 46 | < 2 | |
| Ehime | 2001 | 2 | 379 | < 2 |
| 2002 | 2 | 140 | <2 | |
| Fukui | 1982–83 | 1–2 | 25 | < 10 |
| Wakayama | 1983 | 1 | 12 | < 10 |
| 2002 | 2 | 81 | < 2 | |
| Shizuoka | 2003 | 2 | 70 | <2 |
| Unknown | 2001 | 2 | 101 | <2 |
| Total | 4257 | |||
| Place of culture | Year of collection | Age | Number of collection* | Toxicity (MU/g) |
|---|---|---|---|---|
| Yobuko, Saga | 2001 | 2 | 114 | < 2 |
| 2002 | 2 | 228 | < 2 | |
| 2003 | 2 | 358 | < 2 | |
| 2004 | 2 | 349 | < 2 | |
| 2005 | 2 | 100 | < 2 | |
| 2006 | 2 | 250 | < 2 | |
| 2007 | 2 | 266 | < 4–8 | |
| 2 | 60 | < 2–4 | ||
| 1725 | ||||
| Species | MLD (MU/20 g)* | Ref | |
|---|---|---|---|
| TTX-bearing organisms | |||
| Xanthid crab | Atergatis floridus | 1000 | [87] |
| Tropical goby | Yongeichthys criniger | > 300 | [63] |
| Japanese newt | Cynops pyrrhogaster | > 10000 | [84] |
| Pufferfish | [86] | ||
| Toxic | Takifugu niphobles | 700–750 | |
| T. pardalis | 500–550 | ||
| T. rubripes (culture) | 300–500 | ||
| Generally non-toxic or rarely toxic | Lagocephalus wheeleri | 15–18 | |
| L. gloveri | 19–20 | ||
| Liosaccus cutaneous | 13–15 | ||
| Non-toxic | Ostracion immaculatum | 0.9–1.3 | |
| TTX-free vertebrates | [86] | ||
| Teleosts | Oplegnathus punctatus | 0.8–0.9 | |
| O. fasciatus | 0.8–1.8 | ||
| Girella punctata | 0.3–0.5 | ||
| Land mammal | |||
| Mouse | Mus musculus | 1 | |
| Family | Species | Muscle | Edible part Skin | Male gonad |
|---|---|---|---|---|
| Tetraodontidae | “Kusafugu” Takifugu niphobles | ○ | — | — |
| “Komonfugu” T. poecilonotus | ○ | — | — | |
| “Higanfugu” T. pardalis | ○ | — | — | |
| “Shousaifugu” T. snyderi | ○ | — | ○ | |
| “Nashifugu” T. vermicularisa* | ○ | — | — | |
| “Mafugu” T. porphyreus | ○ | — | ○ | |
| “Mefugu” T. obscurus | ○ | — | ○ | |
| “Akamefugu” T. chrysops | ○ | — | ○ | |
| “Torafugu” T. rubripes | ○ | ○ | ○ | |
| “Karasu” T. chinensis | ○ | ○ | ○ | |
| “Shimafugu” T. xanthopterus | ○ | ○ | ○ | |
| “Gomafugu” T. stictonotus | ○ | — | ○ | |
| “Kanafugu” Lagocephalus inermis | ○ | ○ | ○ | |
| “Shirosabafugu” L. wheeleri | ○ | ○ | ○ | |
| “Kurosabafugu” L. gloveri | ○ | ○ | ○ | |
| “Yoritofugu” Sphoeroides pachygaster | ○ | ○ | ○ | |
| “Sansaifugu” T. flavidus | ○ | — | — | |
| Diodontidae | “Ishigakifugu” Chilomycterus reticulatus | ○ | ○ | ○ |
| “Harisenbon” Diodon holocanthus | ○ | ○ | ○ | |
| “Hitozuraharisenbon” D. liturosus | ○ | ○ | ○ | |
| “Nezumifugu” D. hystrix | ○ | ○ | ○ | |
| Ostracidae | “Hakofugu” Ostraction immaculatum | ○ | — | ○ |
| Year | Number of incidents | Number of patients | Number of deaths | Mortality (%) |
|---|---|---|---|---|
| 1965 | 106 | 152 | 88 | 57.9 |
| 1970 | 46 | 73 | 33 | 45.2 |
| 1975 | 52 | 75 | 30 | 40.0 |
| 1980 | 46 | 90 | 15 | 16.7 |
| 1985 | 30 | 41 | 9 | 22.0 |
| 1990 | 33 | 55 | 1 | 1.8 |
| 1995 | 30 | 42 | 2 | 4.8 |
| 1996 | 21 | 34 | 3 | 8.8 |
| 1997 | 28 | 44 | 6 | 13.6 |
| 1998 | 27 | 39 | 4 | 10.3 |
| 1999 | 20 | 34 | 2 | 5.9 |
| 2000 | 29 | 40 | 0 | 0.0 |
| 2001 | 31 | 52 | 3 | 5.8 |
| 2002 | 32 | 49 | 5 | 10.2 |
| 2003 | 28 | 35 | 3 | 8.6 |
| 2004 | 43 | 58 | 2 | 3.4 |
| 2005 | 40 | 49 | 2 | 4.1 |
| 2006 | 25 | 32 | 1 | 3.1 |
| 2007 | 24 | 38 | 2 | 5.3 |
Share and Cite
Noguch, T.; Arakawa, O. Tetrodotoxin – Distribution and Accumulation in Aquatic Organisms, and Cases of Human Intoxication. Mar. Drugs 2008, 6, 220-242. https://doi.org/10.3390/md6020220
Noguch T, Arakawa O. Tetrodotoxin – Distribution and Accumulation in Aquatic Organisms, and Cases of Human Intoxication. Marine Drugs. 2008; 6(2):220-242. https://doi.org/10.3390/md6020220
Chicago/Turabian StyleNoguch, Tamao, and Osamu Arakawa. 2008. "Tetrodotoxin – Distribution and Accumulation in Aquatic Organisms, and Cases of Human Intoxication" Marine Drugs 6, no. 2: 220-242. https://doi.org/10.3390/md6020220
APA StyleNoguch, T., & Arakawa, O. (2008). Tetrodotoxin – Distribution and Accumulation in Aquatic Organisms, and Cases of Human Intoxication. Marine Drugs, 6(2), 220-242. https://doi.org/10.3390/md6020220




