Abstract
The presence and impact of toxins have been detected in various regions worldwide ever since the discovery of azaspiracids (AZAs) in 1995. These toxins have had detrimental effects on marine resource utilization, marine environmental protection, and fishery production. Over the course of more than two decades of research and development, scientists from all over the world have conducted comprehensive studies on the in vivo metabolism, in vitro synthesis methods, pathogenic mechanisms, and toxicology of these toxins. This paper aims to provide a systematic introduction to the discovery, distribution, pathogenic mechanism, in vivo biosynthesis, and in vitro artificial synthesis of AZA toxins. Additionally, it will summarize various detection methods employed over the past 20 years, along with their advantages and disadvantages. This effort will contribute to the future development of rapid detection technologies and the invention of detection devices for AZAs in marine environmental samples.
1. Introduction
Since the 20th century, human beings have progressively explored and exploited marine resources. The high nutritional value of shellfish, along with their abundance of unsaturated fatty acids, has been a significant factor in attracting human consumption. However, shellfish possess self-protective mechanisms that often lead to the production of toxins and other harmful substances for humans. Additionally, as apex predators in the planktonic food chain, shellfish can accumulate toxins produced by algae [1].
Azaspiracids (AZAs) are a type of polyether toxin (Figure 1) that was initially discovered in an episode of food poisoning in Ireland in 1995 [2]. Several individuals in Ireland exhibited symptoms of diarrhea after consuming mussels, leading to the isolation and identification of these toxins [3]. AZAs were originally named Killary Toxin or KT-3 [4], and their name was later changed to Azaspiracids to better reflect the chemical structural formula. Azaspiracids are known to be produced by the Protists Azadinium [5,6] and Amphidoma [7,8], which belong to the order Lumbar Flagellate. Furthermore, these toxins have been reported and detected in various shellfish species, such as oysters, scallops, and clams [9,10,11].
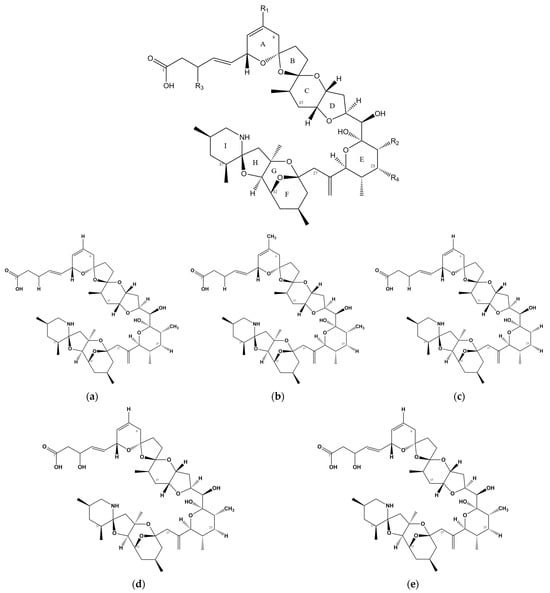
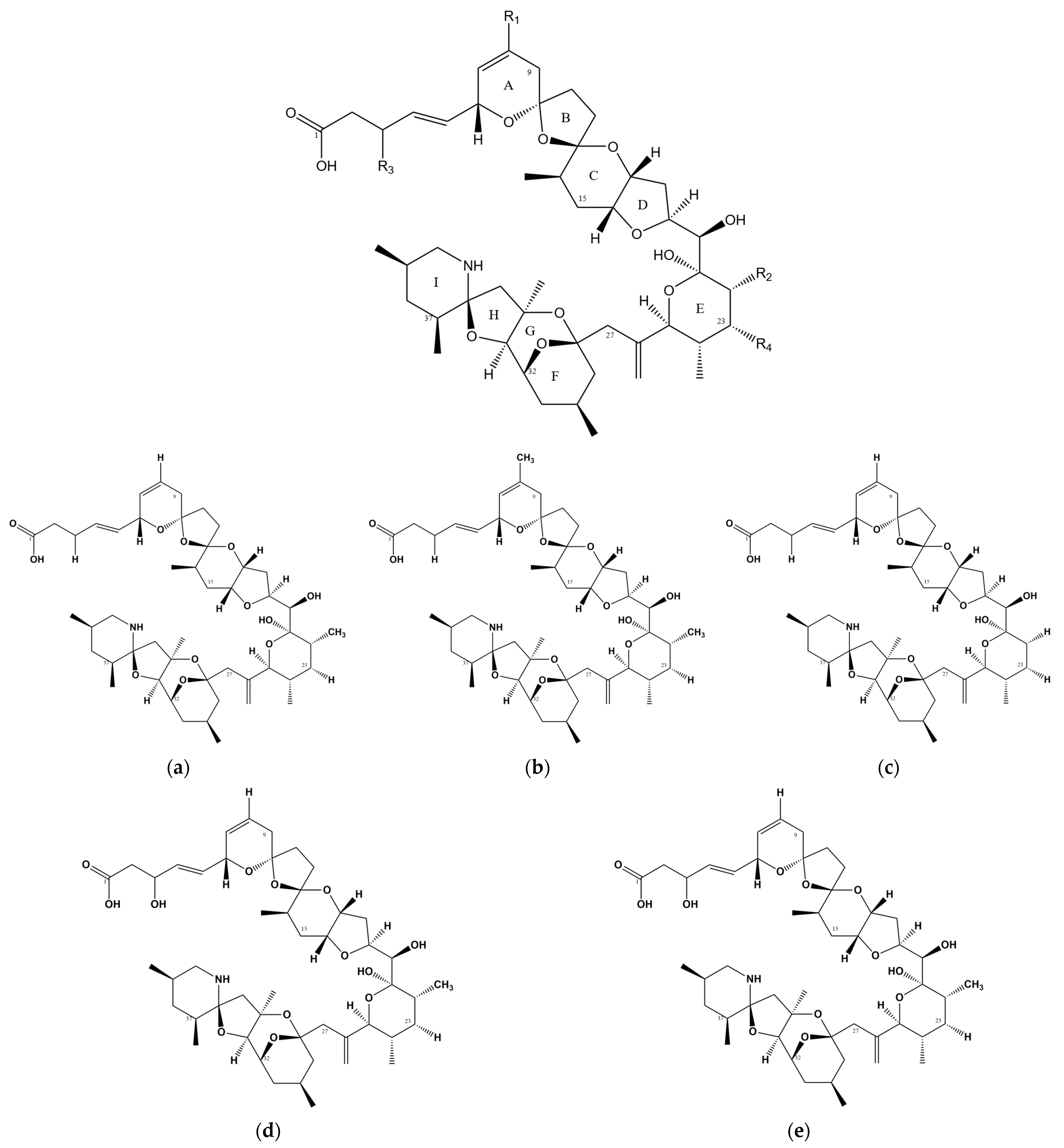
Figure 1.
Chemical structure of AZAs. (a) Chemical structure of AZA-1, (b) chemical structure of AZA-2, (c) chemical structure of AZA-3, (d) chemical structure of AZA-6, and (e) chemical structure of AZA-7.
Since the identification and isolation of AZA-2 and AZA-3 in 1997 [12], numerous analogs of AZAs have been discovered and purified (Table 1). Among these, Azadinium dexteroporum is currently known to produce the highest number of AZA variants, with up to six types reported [13]. The total number of named AZA toxins and their analogs currently stands at AZA-68 [14]. (Table 2).

Table 1.
Chemical structure of some AZA analogs.

Table 2.
Classification of AZA homologous analogs.
2. Toxin Distribution
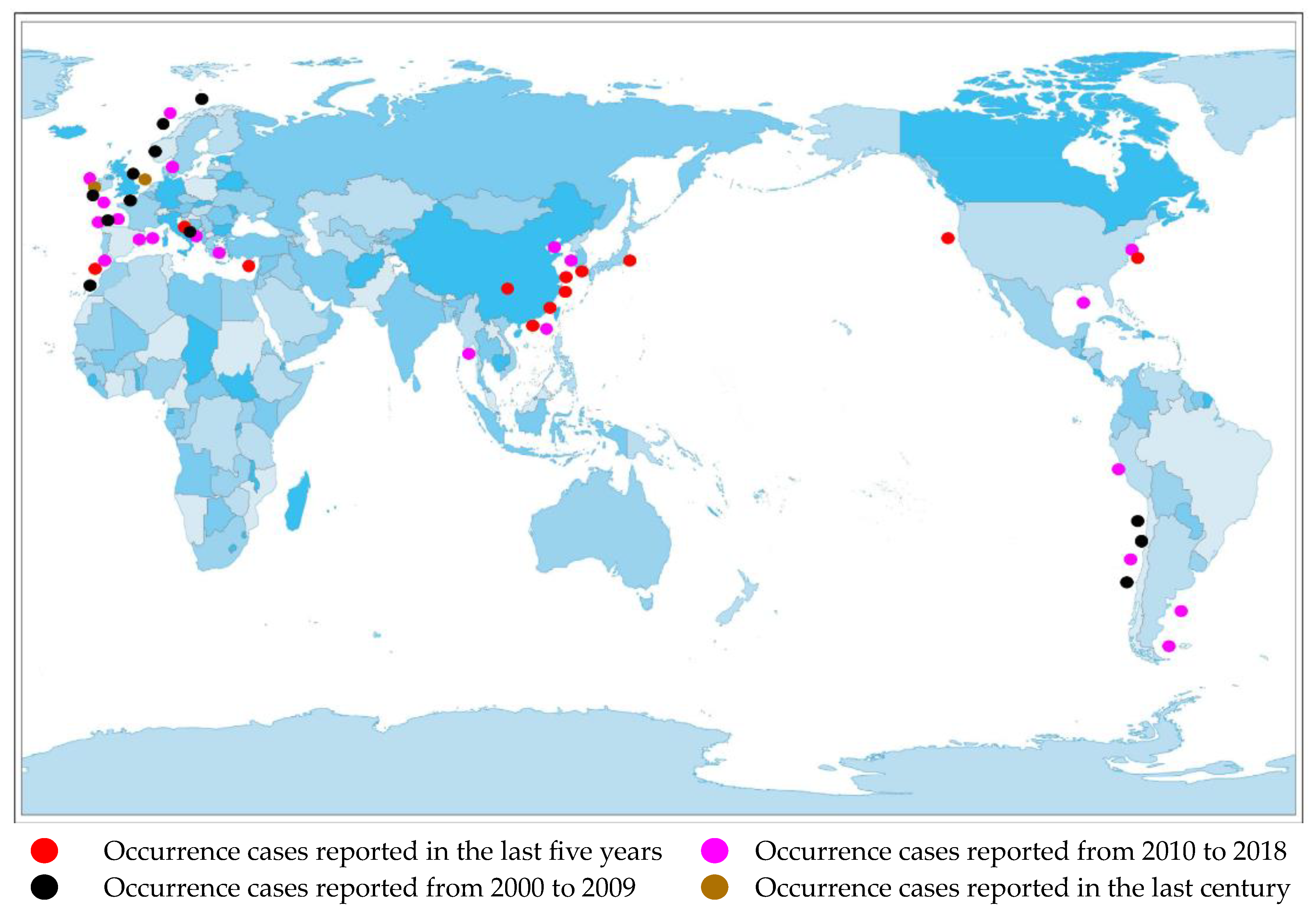
Currently, there are reports of cases of AZA poisoning on many continents, including North America [15], South America [16], Africa [17], Europe [18], and Asia [19].(Figure 2).
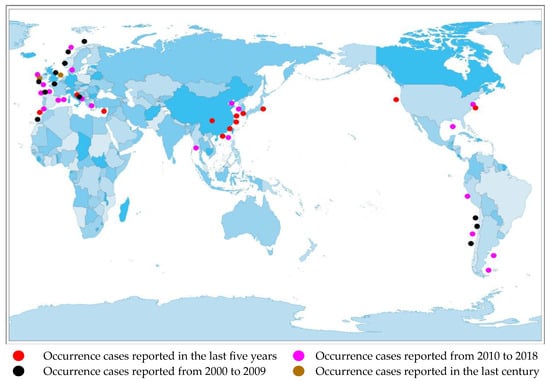
Figure 2.
Distribution of AZA separation or occurrence.
3. Toxicology and Pathology
The acute lethal efficacy LD50 values of oral AZA-1, AZA-2, and AZA-3 in mice were determined to be 443 μg/kg, 626 μg/kg, and 875 μg/kg [20], respectively. The final symptoms of oral administration of AZAs in mice include immobility, sternal lateral immobility, tremors, abdominal breathing, hypothermia, and cyanosis. Although diarrhea is the main toxic sign of human ingestion of seafood contaminated with AZAs, oral exposure from AZA-1 to AZA-3 in mice did not cause significant diarrhea. The lethal dose of intraperitoneal injection in mice targeting AZA-1, AZA-2, and AZA-3 is 200 μg/kg, 110 μg/kg, and 140 μg/kg [12]. The signs and symptoms observed after intraperitoneal injection of purified AZAs in mice include progressive paralysis of the limbs, difficulty breathing, and pre-death convulsions [21]. Regarding the determination of 8 AZA-1 analogs and 12 fragments from the synthesis process of AZA-1, it was found that they have very low or almost no toxic effects compared to AZA-1 itself, indicating that the entire AZA-1 molecule and its stereo-orientation are necessary for exerting toxic effects. Animals subjected to AZA treatment exhibit organ swelling, along with the presence of fat droplets and vacuoles in liver cells [22]. The villi of the small intestine become blunt, accompanied by a reduction in the thickness of the brush-like edge. Additionally, there is a mild to moderate increase in apoptotic cells and infiltrating multi-nucleated cells in the mucosal lamina propria. Depletion of white medullary lymph nodes and lymphocyte necrosis are observed in the spleen. Hepatocellular necrosis is evident in liver cells. Prolonged exposure to small amounts of AZA toxins can lead to a decrease in digestive epithelial cells, increased lipid consumption, and an accumulation of lipofuscin [23]. The data on the oral toxicity of toxins in combination with other toxins indicate that neither the combination with OA (okadaic acid) nor YTX (yessotoxin) showed an increase in toxicity, and no overlapping or synergistic effects were found, only gastrointestinal symptoms were observed [24,25].
There are currently no reports on the long-term effects of AZA toxins on humans, but according to the European Food Safety Authority (EFSA), the lowest observed adverse effect level for individuals with a body weight of 60 kg is 1.9 μg AZA-1 equivalents/kg, and based on this, the most acute reference dose is calculated to be 0.2 μg AZA-1 equivalents/kg body weight [26].
4. Toxic Mechanism
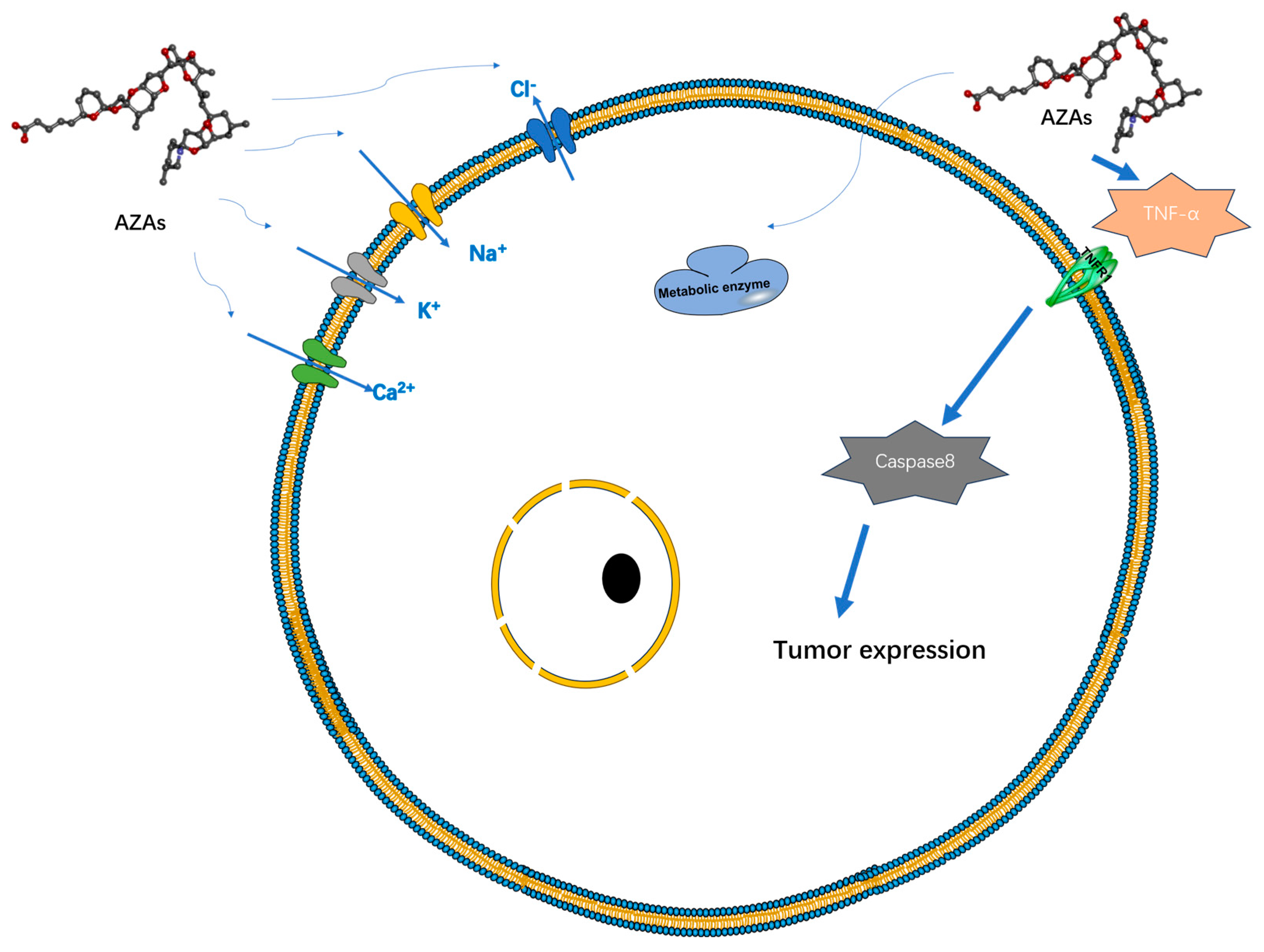
Currently, there is a lack of definitive experimental research regarding the therapeutic mechanism of AZAs that would indicate its specific target-blocking properties. However, several studies have demonstrated its ability to influence cell electrical activity by affecting potassium ion channels [27], sodium ion channels [28], chloride ion channels [29], and calcium ion channels [30] (Figure 3). However, the specific mode of action, whether through interactions or intermediates, remains to be further explored. Specifically, its effect on the sodium ion channel is limited to the modulation of a fast sodium channel flow rate, without inducing channel inactivation. However, under the action of high-concentration toxins (200 nM), the proportion of channel inhibition can reach 60%, seriously affecting the process of cell depolarization [28]. AZAs can also inhibit sodium current through Nav 1.6 channels in the presence of glutamic acid [31], indicating that AZA poisoning may be due to its synergistic effect on some metabolites. Regarding the mechanism of potassium ion channels, it has been reported that AZAs can inhibit hERG channels (hERG channels play a very important role in myocardial repolarization) [27], and experimental data have shown that it can affect the quantity of hERG in the membrane in rats [32]. Rats treated with different doses of toxins (11 or 55 μg/kg) experienced partial PR interval prolongation and heart rate changes due to potassium ion channel blockade [32]. Inhibition of Ca2+ channels primarily affects the storage channels responsible for intracellular calcium ions, thereby giving rise to neurological symptoms [30].
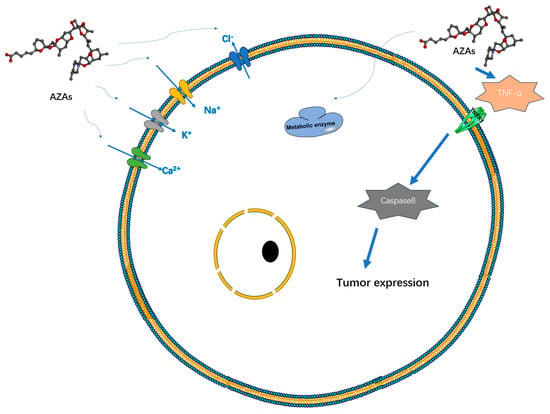
Figure 3.
Possible targets and modes of action of AZAs currently reported.
Additionally, experimental data suggest that AZA-1 may act as a tumor initiator [33]. The induction mechanism here may be related to the abundant production of TNF-α and can induce the expression of early response genes jun B, jun D, c-fos, c-jun, fos B, and fra-1 to achieve tumor induction and occurrence. Repeated administration of AZA in mice led to a significant increase in lung tumors, as well as inducing lymph necrosis in tissues such as the small intestine, spleen, and thymus [34]. Prolonged exposure to AZAs in the environment can result in alterations in cellular cytoskeletons and reduced metabolic activity in human cells [28]. Certain experiments have demonstrated an upregulation of mRNA expression of genes associated with cholesterol synthesis and glycolysis following treatment with AZA toxins, suggesting a potential mechanism for AZAs in modulating cellular metabolism [35].
The gastrointestinal symptoms associated with AZA exposure may arise from alterations in the human intestinal glial system which can impact the integrity of the intestinal barrier. These changes include, but are not limited to, induced neuronal alterations, oxidative stress, disruption of the cell cycle, and an increase in specific enteric glial cell (EGC) markers [36]. Furthermore, the synergistic effects of multiple toxins present in the natural marine environment can enhance the virulence of AZAs [37]. Additionally, AZA-1 has been found to have a partial promoting effect on cell apoptosis and induce an increase in genetic toxicity. In terms of the blood system, AZAs can affect the damage to immune system cell lysosomes, consequently impacting phagocytic function [23].
Heart cells subjected to AZA-1 treatment exhibited heightened levels of apoptotic markers, including caspase-3 and -8, cleavage of PARP, and upregulation of Fas ligands [38]. These molecular changes are reflected at the tissue level, resulting in alterations in arterial blood pressure and deposition of cardiac collagen. Long-term experimental studies have demonstrated that AZAs can induce structural changes in the heart that contribute to heart failure [39] and provoke arrhythmias by modulating ion channels [32] (Figure 3).
In summary, although the activity of toxins as inhibitors of PP [40], kinases, and GPCR or as inhibitors of actin polymerization/depolymerization has been experimentally overturned [41], other experiments have shown their cytotoxicity, affecting cytoskeleton arrangement, promoting tumors, and potentially affecting the activity of multiple ion channels. However, at this point in time, there seems to be no scientific consensus on a specific target or mechanism of AZAs that can jointly explain the various effects observed in experiments and the gastrointestinal symptoms observed in exposed individuals, so further exploration of treatment is needed in the future.
5. AZA Analogs from Different Sources
The initial characterization of AZA and its analogs was conducted in 1998 using mass spectrometry (MS) and nuclear magnetic resonance (NMR) techniques [3]. Over 60 types of AZA analogs have been identified, with the majority being produced through metabolic processes in mussels.
AZA-38 and 39 are primarily produced by Amphidoma languida, a small dinoflagellate species belonging to the Amphidomataceae family. Recently, these toxins have undergone structural modifications [42]. The toxins produced by different ribosomal subtypes of Azadinium spinosum exhibit variations, with subtype A mainly producing AZA-1 and AZA-2, while subtype B primarily produces AZA-11 and AZA-51 [43]. In the case of ribosomal subtype A of Azadinium pomorum, there is either no production or only a minimal amount of AZAs, whereas ribosomal subtype C can generate AZA-40 and AZA-2 [44]. Different strains of Azadinium pomorum produce the hydroxylation product AZA-42 from AZA-41 and the dehydrogenation product AZA-62 from AZA-11. The former strain is isolated from the South China Sea, while the latter strain is isolated from the northern coast of Chile [45]. Furthermore, AZA-59 is the sole AZA toxin produced by Azadinium pomorum strains isolated from the Pacific northwest coast of the United States.
6. Synthesis In Vivo
To date, a majority of studies have pointed to the blue mussel, Mytilus edulis, as the primary vector for AZAs. However, other organisms such as mollusks, arthropods, and echinoderms have also been reported as potential vectors [46]. In mussels, several AZAs undergo acyl ester formation, with some studies suggesting that these esters exhibit higher toxicity than the free toxins themselves [47]. The average distribution of AZAs in mussels is as follows: hepatopancreas (60.6%), gills (12.0%), and adductor muscle (27.4%) [48].
AZA has been detected in various bivalve mollusks, including oysters (Crassostrea gigas), scallops (Pecten maximus), clams (Tapes filipinarium), and cockles (Cardium rule), as well as in numerous phytoplankton species [10]. In the experiment involving feeding blue mussels (Mytilus edulis) with toxins, it was found that AZA-17 and AZA-19 were mainly fast metabolites feeding AZA-1 and AZA-2 [49], indicating that carboxylation of methyl groups at the C22 position is a dominant metabolic pathway, while hydroxylation and decarboxylation are secondary degradation pathways. Notably, AZA-65 and AZA-66 have been identified as intermediate products in the conversion of AZA-1 and AZA-2 to AZA-17 and AZA-19. In mussels, the expression of AZA-1-3 can lead to the production of AZA-8, AZA-12, and AZA-5 through C-23 α hydroxylation. Additionally, the double hydroxylation of AZA-67 and AZA-68 can generate AZA-1 and AZA-2 as secondary metabolites [14].
Experimental evidence has demonstrated a correlation between toxin production in algae and temperature, with the highest toxin concentrations observed at 26 °C [50].
AZA-1 treated with rat liver microsomal extract undergoes oxidation at the F ring and can bind with glucuronic acid at C1 to generate glucuronides [51].
7. Synthesis In Vitro
In the synthesis of AZAs, the FGHI ring exhibits relatively high stability in the region spanning from C-26 to C-40, whereas the variability is primarily observed in the C-1 to C-22 [52] region. As early as 2008, the structure of the AZA-1 compound was fragmented and partially synthesized, encompassing the synthesis of the E ring, HI ring, CD ring, and FG ring. The conversion of furan into the ABCD ring can be achieved through a single oxygen-initiated one-pot process [53]. Subsequently, the combination of the EFGHI ring and the ABCD ring enables the complete synthesis of AZA-1 [54]. The artificial in vitro synthesis of fragments from C-22 to C-40 has been enhanced and refined [55]. Currently, through the full synthesis of AZA-1 and continuous modification and optimization of reaction conditions, an artificial stereoisomeric composite closely resembling the structure of natural AZA-3 has been achieved [56].
8. Detection of Toxins
Many countries, including those in the European Union (EU), have established regulatory limits for AZA content in shellfish intended for human consumption. In the EU, this limit is set at 160 µg of AZA equivalents per kg of shellfish meat (whole body or any edible part) [57]. Currently, the European official method for detecting AZAs is the mouse bioassay [58]. Evaluating the suitability of chromatographic conditions, multi-experimental groups also provided experimental evidence for the standardized authentication of AZA concentrations and a new CRM from the NRC Certified Reference Materials Program (Halifax, NS, Canada) [59,60,61] (Table 3).

Table 3.
LOD and LOQ of different detection methods and sources of samples.
8.1. LC-MS
LC-MS, or liquid chromatography–mass spectrometry, is a powerful analytical technique that combines the physical separation capabilities of liquid chromatography (LC) with the mass analysis capabilities of mass spectrometry (MS). When combined, LC separates the chemical components of a mixture, and then, those individual components are funneled into the mass spectrometer. The mass spectrometer further separates the chemicals based on their mass-to-charge ratio, and then identifies and quantifies them. In the field of toxin detection, LC-MS has been intensively employed to generate high-quality data, demonstrating its pivotal role in advancing our understanding of various toxins.
In 1999 [12], an LC/MS method was first used that offered sensitive and specific determination of AZA and its two analogs. This method achieved a detection limit of 50 pg for AZAs, demonstrating a sensitivity approximately 8 × 104 times greater than that of the mouse bioassay. Later, a variety of optimized LC-MS (n) methods were developed successively. A highly sensitive LC-MS method achieved a detection limit of 4 pg and allowed for the simultaneous analysis of multiple types of AZAs [9,80]. Additionally, an on-board LC-MS-MS system was developed for near real-time analysis of phycotoxins in plankton [81]. A variety of AZAs have since been found, including its isomers [13,52,82,83]. Since the first discovery of AZA and its two analogs in 1999, this type of toxin has been detected in various marine coasts and marine products worldwide [16,62,84,85,86,87,88,89,90,91,92,93,94,95,96] (Table 4).

Table 4.
Reported cases of azaspiracid poisoning (AZP) mentioned in the article.
Matrix effects can result in either signal enhancement (increase in the apparent concentration of the analyte) or signal suppression (decrease in the apparent concentration of the analyte). It is a significant concern in LC-MS analysis that greatly influences the accuracy and reliability of toxin detection. After an in-depth investigation of matrix effects, it was found that AZA-1 significantly inhibited the signal in the presence of matrix effects. Subsequently, this issue was greatly improved through the use of a rapid LC-MS method that separated major toxins based on the MS ionization mode [97,98]. Furthermore, LC-MS has facilitated rapid simultaneous separation, monitoring, and quantification of toxins [67,79,99].
8.1.1. UHPLC/HPLC-MS
The utilization of ultrahigh-performance liquid chromatography (UHPLC) coupled with advanced mass spectrometry techniques has transformed the detection and quantification of these toxins. Several research groups have combined this method with other techniques such as ESI [71,100,101] and SPE [70,102,103] and optimized it to achieve the simultaneous detection of multiple toxins [76,77], improve detection limits, reduce matrix effects [75], and enhance the effect of chromatographic gradients [104].
8.1.2. SPE/MSPE+LC-MS
The development and refinement of extraction techniques, particularly SPE [64] and MSPE [75], coupled with LC-MS also have significantly advanced our ability to detect and quantify toxins [66,75,102,103,105]. After validation, optimization can significantly reduce matrix effects [106].
8.1.3. LC-ESI-MS
Sensitive LC–electrospray ionization–mass spectrometry (LC-ESI-MS(n)) methods, including ion trap mass spectrometry [84,86,105,107] and Orbitrap technology [51,71,78,101,108,109], have been developed for the determination of major AZAs and their hydroxyl analogs, enabling the separation of multiple AZAs in a short period of time. The combination of LC-MS with ESI technology allows for the determination of MS dissociation pathways and the differences between CID spectra to be obtained [63,65,68,84,110,111,112]. Subsequently, through a series of practical tests [69,113], the utility of this approach was demonstrated, including its ability to reduce matrix effects [114,115].
8.1.4. LC-HRMS
The use of HRMS was described in combination with passive sampling as a progressive approach to marine algal toxin surveys [116]. This method can also be used in conjunction with the Orbitrap exactive HCD mass spectrometer [78], solid–liquid ultrasound-assisted extraction, and solid–phase extraction [103], and has been implemented in the detection process [47,117].
8.1.5. LC-MS+NMR
The combination of NMR and LC-MS is primarily used for the discovery of new AZAs and the detection of the structure and concentration of the toxins [13,118,119,120,121].
8.1.6. LC-MS + Others
Other methods used in conjunction with LC-MS detection technology include live microscopy, quantitative polymerase chain reaction (qPCR) [94], micro-liquid chromatography–tandem mass spectrometry (micro-LC-MS-MS) [63], selected ion monitoring (SIM), multi-reaction monitoring (MRM) [122], and the use of a gel to selectively capture and release AZAs [123].
8.2. MBA and CBA
The detection and quantification of toxins, particularly azaspiracids (AZAs), have been significantly advanced by the development and refinement of bioassay techniques, including mouse bioassay (MBA) [124,125,126,127] and cell-based assay (CBA) [127,128]. Due to cost and ethical considerations, these two methods are currently not mainstream detection techniques. They are typically used in conjunction with other technologies to validate the detection results, although such cases are actually quite rare.
8.3. Biosensor
At present, the quantification and identification of AZAs are possible only for those compounds that have available certified standards. The development of biosensor assays for detecting AZAs remains a challenge, with no such assay currently available.
Recently, significant progress has been made in the development of AZA-specific antibodies that have shown binding affinity for several AZA analogs, including AZA-1, 2, 3, and 6 [129,130]. These antibodies have been utilized to design competition and sandwich enzyme-linked immunosorbent assays (ELISAs) with a synthetic fragment of the AZA molecule that is conserved for many analogs.
These developments mark a significant step forward in the detection of AZAs. The hope is that in the near future, these new antibodies will be applied in the development of electrochemical or optical immunosensors for the detection of AZAs.
8.4. ELISA
In 2015, Samdal [72] demonstrated an ELISA with a working range of 0.45–8.6 ng/mL and a limit of quantitation for total AZAs in whole shellfish of 57 μg/kg. He also produced a new plate coater, OVA-cdiAZA-1, resulting in an ELISA with a working range of 0.30–4.1 ng/mL and a limit of quantification of 37 μg/kg for AZA-1 in shellfish, in 2019 [74]. ELISA can also be employed in conjunction with gel methods [123]. This method is usually combined with bioassay techniques; two research teams have already implemented this technique [125,131].
8.5. Other Immunoassays
As part of the advancements in immune technology, microsphere-based immunoassays [132], magnetic bead (MB)-based direct immunoassays [73], and immunoaffinity chromatography (IAC) columns [133], specifically designed for the purification and concentration of AZAs, have been reported. An immunoassay kit was discovered that provided a more sensitive, specific, and swift approach to determining toxins in total shellfish extracts compared to LC-MS [134].
Due to the extensive research and the establishment of regulatory measures concerning AZAs, most studies focused on detection and quantification are primarily concerned with the safety of samples. At the same time, there are also some studies that utilize reference standards to explore new analogs [3,52,82,83]. Since AZA-1 to 3 were the earliest discovered in this group and constitute a significant portion of the entire toxin group (especially AZA-1 and AZA-2 as precursor compounds), with established regulations in terms of food safety, therefore, in the majority of detection efforts, significant attention is placed on these three toxins or the entire AZA toxin family as the target of analysis [92].
We compared the detection results of our investigation group and found that in contrast to other lipophilic toxins, the levels of AZAs were generally low in the samples. In the majority of the tests, AZAs accounted for a minimal proportion and were far below the relevant regulations for food safety. This also implies that the standardization of this toxin is relatively challenging to extract, and it requires a high sensitivity of the detection techniques in practical applications. The AZP toxin group encompasses a wide range of analogs, making it possible for misidentification of toxin types in certain experiments. Therefore, a more precise differentiation of toxin analogs may be a prospective research direction for future studies.
To date, more than 60 analogs of AZAs have been identified, exhibiting complex diversity and similar structures. Therefore, in order to distinguish them more effectively, gaining a clear understanding of their structure and chemical composition through the utilization of MS and NMR spectroscopy, as well as other identification techniques, has become a focal point in the research of these toxins. Gaining a more detailed and in-depth understanding of toxins and developing faster and more efficient detection techniques can contribute to the early development of commercialized detection kits and standardized testing procedures.
Currently, there are relatively few explicitly regulated AZAs. However, in certain detections, the proportion of unregulated AZAs may be higher than that of the established regulated types—potentially even by an order of magnitude. This fact highlights the underestimation of the total AZA load by current regulatory strategies.
9. Conclusions
In summary, AZAs are an important part of toxin research. Currently, many countries and institutions have developed monitoring plans and regulatory measures to ensure the safety of seafood products and to safeguard public health. Continuous in-depth research on monitoring and detection methods is crucial for effectively monitoring AZA pollution and protecting consumers from potential harm. Although progress has been made in understanding the toxicology and occurrence of AZAs, there are still some knowledge gaps. Further research is needed to investigate the long-term health effects and potential risks associated with long-term exposure to AZAs. In addition, more research is needed to elucidate the mechanism of action of AZAs and determine the exact relationship between AZAs and their potential carcinogenicity.
Although the probability of large-scale outbreaks of shellfish toxins is low, considering the toxicity, unpredictability, and significant differences in the abundance of AZA toxins in recent years, as well as the threat to consumers and marine professionals, we still need to establish a complete monitoring and protection system. The design of stable and efficient methods and equipment for detecting toxins is also conducive to promoting the development and utilization of marine resources, promoting the safety of marine fisheries, and promoting human health.
Author Contributions
Investigation, resources, writing—original draft preparation, writing—review and editing, data curation, J.Y., W.S.; visualization, Y.C.; supervision, M.S.; project administration, L.W.; funding acquisition, L.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Nation Natural Science Foundation of China (No. 82173732).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Huss, H. Control of indigenous pathogenic bacteria in seafood. Food Control 1997, 18, 91–98. [Google Scholar] [CrossRef]
- McMahon, T. Winter toxicity of unknown aetiology in mussels. Harmful Algae News 1996, 14, 2. [Google Scholar]
- Satake, M.; Ofuji, K.; Naoki, H.; James, K.J.; Furey, A.; McMahon, T.; Silke, J.; Yasumoto, T. Azaspiracid, a new marine toxin having unique spiro ring assemblies, isolated from Irish mussels, Mytilus edulis. J. Am. Chem. Soc. 1998, 120, 9967–9968. [Google Scholar] [CrossRef]
- Satake, M.; Ofuji, K.; James, K.J.; Furey, A.; Yasumoto, T. New toxic event caused by Irish mussels. In Proceedings of the Harmful Algae, Proceedings of the VIII International Conference on Harmful Algae, Vigo, Spain, 25–29 June 1999; pp. 468–469. [Google Scholar]
- Krock, B.; Tillmann, U.; John, U.; Cembella, A.D. Characterization of azaspiracids in plankton size-fractions and isolation of an azaspiracid-producing dinoflagellate from the North Sea. Harmful Algae 2009, 8, 254–263. [Google Scholar] [CrossRef]
- Tillmann, U.; Elbrächter, M.; Krock, B.; John, U.; Cembella, A. Azadinium spinosum gen. et sp. nov (Dinophyceae) identified as a primary producer of azaspiracid toxins. Eur. J. Phycol. 2009, 44, 63–79. [Google Scholar] [CrossRef]
- Tillmann, U.; Salas, R.; Gottschling, M.; Krock, B.; O’Driscoll, D.; Elbrächter, M. Amphidoma languida sp. Nov (Dinophyceae) reveals a close relationship between Amphidoma and Azadinium. Protist 2012, 163, 701–719. [Google Scholar] [CrossRef]
- Tillmann, U.; Salas, R.; Jauffrais, T.; Hess, P.; Silke, J. AZA: The producing organisms–biology and trophic transfer. In Seafood and Freshwater Toxins; Botana, L.M., Ed.; CRC Press: Boca Raton, FL, USA, 2014; pp. 773–798. [Google Scholar]
- Furey, A.; Braña-Magdalena, A.; Lehane, M.; Moroney, C.; James, K.J.; Satake, M.; Yasumoto, T. Determination of azaspiracids in shellfish using liquid chromatography/tandem electrospray mass spectrometry. Rapid Commun. Mass Spectrom. RCM 2002, 16, 238–242. [Google Scholar] [CrossRef]
- Furey, A.; Moroney, C.; Braña-Magdalena, A.; Saez, M.J.; Lehane, M.; James, K.J. Geographical, temporal, and species variation of the polyether toxins, azaspiracids, in shellfish. Environ. Sci. Technol. 2003, 37, 3078–3084. [Google Scholar] [CrossRef]
- Magdalena, A.B.; Lehane, M.; Moroney, C.; Furey, A.; James, K.J. Food safety implications of the distribution of azaspiracids in the tissue compartments of scallops (Pecten maximus). Food Addit. Contam. 2003, 20, 154–160. [Google Scholar] [CrossRef]
- Ofuji, K.; Satake, M.; McMahon, T.; Silke, J.; James, K.J.; Naoki, H.; Oshima, Y.; Yasumoto, T. Two analogs of azaspiracid isolated from mussels, Mytilus edulis, involved in human intoxication in Ireland. Nat. Toxins 1999, 7, 99–102. [Google Scholar] [CrossRef]
- Rossi, R.; Dell’Aversano, C.; Krock, B.; Ciminiello, P.; Percopo, I.; Tillmann, U.; Soprano, V.; Zingone, A. Mediterranean Azadinium dexteroporum (Dinophyceae) produces six novel azaspiracids and azaspiracid-35: A structural study by a multi-platform mass spectrometry approach. Anal. Bioanal. Chem. 2017, 409, 1121–1134. [Google Scholar] [CrossRef]
- Sandvik, M.; Miles, C.O.; Løvberg, K.L.E.; Kryuchkov, F.; Wright, E.J.; Mudge, E.M.; Kilcoyne, J.; Samdal, I.A. In Vitro Metabolism of Azaspiracids 1-3 with a Hepatopancreatic Fraction from Blue Mussels (Mytilus edulis). J. Agric. Food Chem. 2021, 69, 11322–11335. [Google Scholar] [CrossRef]
- Klontz, K.C.; Abraham, A.; Plakas, S.M.; Dickey, R.W. Mussel-associated azaspiracid intoxication in the United States. Ann. Intern. Med. 2009, 150, 361. [Google Scholar] [CrossRef]
- Alvarez, G.; Uribe, E.; Avalos, P.; Mariño, C.; Blanco, J. First identification of azaspiracid and spirolides in Mesodesma donacium and Mulinia edulis from Northern Chile. Toxicon Off. J. Int. Soc. Toxinol. 2010, 55, 638–641. [Google Scholar] [CrossRef]
- Taleb HVale, P.; Amanhir, R.; Benhadouch, A.; Sagou, R.; Chafik, A. First detection of azaspiraicds in mussels in north west Africa. J. Shellfish Res. 2006, 25, 1067–1070. [Google Scholar]
- James, K.J.; Furey, A.; Satake, M.; Yasumoto, T. Azaspiracid poisoning (AZP): A new shellfish toxic syndrome in Europe. In Proceedings of the Ninth International Conference on Harmful Algal Bloom, Tasmania, Australia, 7–11 February 2000; pp. 250–253. [Google Scholar]
- Wang, J.; Li, R.; Liu, B.; Zhang, Q.; Wang, X.; Zhu, Y.; Zhang, Y. Occurrence and distribution of lipophilic marine algal toxins in the coastal seawater of Southeast China and the South China Sea. Mar. Pollut. Bull. 2023, 187, 114584. [Google Scholar] [CrossRef]
- Pelin, M.; Kilcoyne, J.; Nulty, C.; Crain, S.; Hess, P.; Tubaro, A.; Sosa, S. Toxic equivalency factors (TEFs) after acute oral exposure of azaspiracid 1, -2 and -3 in mice. Toxicol. Lett. 2018, 282, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Furey, A.; O’Doherty, S.; O’Callaghan, K.; Lehane, M.; James, K.J. Azaspiracid poisoning (AZP) toxins in shellfish: Toxicological and health considerations. Toxicon Off. J. Int. Soc. Toxinol. 2010, 56, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Pelin, M.; Kilcoyne, J.; Florio, C.; Hess, P.; Tubaro, A.; Sosa, S. Azaspiracids Increase Mitochondrial Dehydrogenases Activity in Hepatocytes: Involvement of Potassium and Chloride Ions. Mar. Drugs 2019, 17, 276. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, M.E.; Accoroni, S.; Mezzelani, M.; Lugarini, F.; Bacchiocchi, S.; Siracusa, M.; Tavoloni, T.; Piersanti, A.; Totti, C.; Regoli, F.; et al. Biological Effects of the Azaspiracid-Producing Dinoflagellate Azadinium dexteroporum in Mytilus galloprovincialis from the Mediterranean Sea. Mar. Drugs 2019, 17, 595. [Google Scholar] [CrossRef]
- Aasen, J.A.; Espenes, A.; Miles, C.O.; Samdal, I.A.; Hess, P.; Aune, T. Combined oral toxicity of azaspiracid-1 and yessotoxin in female NMRI mice. Toxicon Off. J. Int. Soc. Toxinol. 2011, 57, 909–917. [Google Scholar] [CrossRef]
- Aune, T.; Espenes, A.; Aasen, J.A.; Quilliam, M.A.; Hess, P.; Larsen, S. Study of possible combined toxic effects of azaspiracid-1 and okadaic acid in mice via the oral route. Toxicon Off. J. Int. Soc. Toxinol. 2012, 60, 895–906. [Google Scholar] [CrossRef]
- EFSA, Marine biotoxins in shellfish–azaspiracid group, scientific opinion of the panel on contaminants in the food chain. EFSA J. 2008, 723, 1–52.
- Twiner, M.J.; Doucette, G.J.; Rasky, A.; Huang, X.P.; Roth, B.L.; Sanguinetti, M.C. Marine algal toxin azaspiracid is an open-state blocker of hERG potassium channels. Chem. Res. Toxicol. 2012, 25, 1975–1984. [Google Scholar] [CrossRef] [PubMed]
- Boente-Juncal, A.; Raposo-García, S.; Costas, C.; Louzao, M.C.; Vale, C.; Botana, L.M. Partial blockade of human voltage-dependent sodium channels by the marine toxins Azaspiracids. Chem. Res. Toxicol. 2020, 33, 2593–2604. [Google Scholar] [CrossRef] [PubMed]
- Boente-Juncal, A.; Raposo-García, S.; Louzao, M.C.; Vale, C.; Botana, L.M. Targeting chloride ion channels: New insights into the mechanism of action of the marine toxin azaspiracid. Chem. Res. Toxicol. 2021, 34, 865–879. [Google Scholar] [CrossRef]
- Alfonso, A.; Román, Y.; Vieytes, M.R.; Ofuji, K.; Satake, M.; Yasumoto, T.; Botana, L.M. Azaspiracid-4 inhibits Ca2+ entry by stored operated channels in human T lymphocytes. Biochem. Pharmacol. 2005, 69, 1627–1636. [Google Scholar] [CrossRef] [PubMed]
- Chevallier, O.P.; Graham, S.F.; Alonso, E.; Duffy, C.; Silke, J.; Campbell, K.; Botana, L.M.; Elliott, C.T. New insights into the causes of human illness due to consumption of azaspiracid contaminated shellfish. Sci. Rep. 2015, 5, 9818. [Google Scholar] [CrossRef]
- Ferreiro, S.F.; Vilariño, N.; Carrera, C.; Louzao, M.C.; Santamarina, G.; Cantalapiedra, A.G.; Rodríguez, L.P.; Cifuentes, J.M.; Vieira, A.C.; Nicolaou, K.C.; et al. In vivo arrhythmogenicity of the marine biotoxin azaspiracid-2 in rats. Arch. Toxicol. 2014, 88, 425–434. [Google Scholar] [CrossRef]
- Ito, E. Toxicology of Azaspiracid-1: Acute and chronic poisoning, tumourigenicity and chemical structure relationship to toxicity in a mouse bioassay. In Seafood and Freshwater Toxins: Pharmacology, Physiology and Detection; CRC Press: Boca Raton, FL, USA, 2008; pp. 775–784. [Google Scholar]
- Ito, E.; Satake, M.; Ofuji, K.; Higashi, M.; Harigaya, K.; McMahon, T.; Yasumoto, T. Chronic effects in mice caused by oral administration of sublethal doses of azaspiracid, a new marine toxin isolated from mussels. Toxicon Off. J. Int. Soc. Toxinol. 2002, 40, 193–203. [Google Scholar] [CrossRef]
- Bodero, M.; Hoogenboom, R.; Bovee, T.F.H.; Portier, L.; de Haan, L.; Peijnenburg, A.; Hendriksen, P.J.M. Whole genome mRNA transcriptomics analysis reveals different modes of action of the diarrheic shellfish poisons okadaic acid and dinophysis toxin-1 versus azaspiracid-1 in Caco-2 cells. Toxicol. Vitr. Int. J. Public Assoc. BIBRA 2018, 46, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Reale, O.; Huguet, A.; Fessard, V. Novel Insights on the Toxicity of Phycotoxins on the Gut through the Targeting of Enteric Glial Cells. Mar. Drugs 2019, 17, 429. [Google Scholar] [CrossRef] [PubMed]
- Ferron, P.J.; Dumazeau, K.; Beaulieu, J.F.; Le Hégarat, L.; Fessard, V. Combined effects of lipophilic phycotoxins (okadaic acid, azapsiracid-1 and yessotoxin) on human intestinal cells models. Toxins 2016, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, S.F.; Vilariño, N.; Carrera, C.; Louzao, M.C.; Santamarina, G.; Cantalapiedra, A.G.; Cifuentes, J.M.; Crespo, A.; Botana, L.M. In vivo cardiomyocyte response to YTX- and AZA-1-induced damage: Autophagy versus apoptosis. Arch. Toxicol. 2017, 91, 1859–1870. [Google Scholar] [CrossRef]
- Ferreiro, S.F.; Vilariño, N.; Carrera, C.; Louzao, M.C.; Cantalapiedra, A.G.; Santamarina, G.; Cifuentes, J.M.; Vieira, A.C.; Botana, L.M. Subacute cardiovascular toxicity of the marine phycotoxin azaspiracid-1 in rats. Toxicol. Sci. Off. J. Soc. Toxicol. 2016, 151, 104–114. [Google Scholar] [CrossRef]
- Twiner, M.J.; Hess, P.; Dechraoui, M.Y.; McMahon, T.; Samons, M.S.; Satake, M.; Yasumoto, T.; Ramsdell, J.S.; Doucette, G.J. Cytotoxic and cytoskeletal effects of azaspiracid-1 on mammalian cell lines. Toxicon Off. J. Int. Soc. Toxinol. 2005, 45, 891–900. [Google Scholar] [CrossRef]
- Twiner, M.J.; Hess, P.; Doucette, G.J. Azaspiracids: Toxicology, Pharmacology, and Risk Assessment, Seafood and Freshwater Toxins; CRC Press: Boca Raton, FL, USA, 2014; pp. 823–856. [Google Scholar]
- Salas, R.; Murphy, E.; Doohan, R.; Tillmann, U.; Thomas, O.P. Production of the dinoflagellate Amphidoma languida in a large scale photobioreactor and structure elucidation of its main metabolite AZA-39. Harmful Algae 2023, 127, 102471. [Google Scholar] [CrossRef]
- Tillmann, U.; Wietkamp, S.; Gu, H.; Krock, B.; Salas, R.; Clarke, D. Multiple new strains of amphidomataceae (Dinophyceae) from the north atlantic revealed a high toxin profile variability of Azadinium spinosum and a new non-toxigenic az. cf. spinosum. Microorganisms 2021, 9, 134. [Google Scholar] [CrossRef]
- Luo, Z.; Krock, B.; Giannakourou, A.; Venetsanopoulou, A.; Pagou, K.; Tillmann, U.; Gu, H. Sympatric occurrence of two Azadinium poporum ribotypes in the Eastern Mediterranean Sea. Harmful Algae 2018, 78, 75–85. [Google Scholar] [CrossRef]
- Krock, B.; Tillmann, U.; Tebben, J.; Trefault, N.; Gu, H. Two novel azaspiracids from Azadinium poporum, and a comprehensive compilation of azaspiracids produced by Amphidomataceae, (Dinophyceae). Harmful Algae 2019, 82, 1–8. [Google Scholar] [CrossRef]
- Silva, M.; Rodríguez, I.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. Lipophilic toxins occurrence in non-traditional invertebrate vectors from North Atlantic Waters (Azores, Madeira, and Morocco): Update on geographical tendencies and new challenges for monitoring routines. Mar. Pollut. Bull. 2020, 161 Pt B, 111725. [Google Scholar] [CrossRef]
- Mudge, E.M.; Miles, C.O.; Hardstaff, W.R.; McCarron, P. Fatty acid esters of azaspiracids identified in mussels (Mytilus edulis) using liquid chromatography-high resolution mass spectrometry. Toxicon X 2020, 8, 100059. [Google Scholar] [CrossRef]
- O’Driscoll, D.; Škrabáková, Z.; James, K.J. Confirmation of extensive natural distribution of azaspiracids in the tissue compartments of mussels (Mytilus edulis). Toxicon Off. J. Int. Soc. Toxinol. 2014, 92, 123–128. [Google Scholar] [CrossRef]
- Jauffrais, T.; Marcaillou, C.; Herrenknecht, C.; Truquet, P.; Séchet, V.; Nicolau, E.; Tillmann, U.; Hess, P. Azaspiracid accumulation, detoxification and biotransformation in blue mussels (Mytilus edulis) experimentally fed Azadinium spinosum. Toxicon Off. J. Int. Soc. Toxinol. 2012, 60, 582–595. [Google Scholar] [CrossRef]
- Li, X.; Yan, M.; Gu, J.; Lam, V.T.T.; Wai, T.C.; Baker, D.M.; Thompson, P.D.; Yiu, S.K.F.; Lam, P.K.S.; Leung, P.T.Y. The effect of temperature on physiology, toxicity and toxin content of the benthic dinoflagellate Coolia malayensis from a seasonal tropical region. Water Res. 2020, 185, 116264. [Google Scholar] [CrossRef]
- Kittler, K.; Preiss-Weigert, A.; These, A. Identification strategy using combined mass spectrometric techniques for elucidation of phase I and phase II in vitro metabolites of lipophilic marine biotoxins. Anal. Chem. 2010, 82, 9329–9335. [Google Scholar] [CrossRef]
- Kilcoyne, J.; McCarron, P.; Twiner, M.J.; Nulty, C.; Crain, S.; Quilliam, M.A.; Rise, F.; Wilkins, A.L.; Miles, C.O. Epimers of azaspiracids: Isolation, structural elucidation, relative LC-MS response, and in vitro toxicity of 37-epi-azaspiracid-1. Chem. Res. Toxicol. 2014, 27, 587–600. [Google Scholar] [CrossRef]
- Triantafyllakis, M.; Tofi, M.; Montagnon, T.; Kouridaki, A.; Vassilikogiannakis, G. Singlet oxygen-mediated synthesis of bis-spiroketals found in azaspiracids. Org. Lett. 2014, 16, 3150–3153. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.A.; Kvaernø, L.; Dunn, T.B.; Beauchemin, A.; Raymer, B.; Mulder, J.A.; Olhava, E.J.; Juhl, M.; Kagechika, K.; Favor, D.A. Total synthesis of (+)-azaspiracid-1. An exhibition of the intricacies of complex molecule synthesis. J. Am. Chem. Soc. 2008, 130, 16295–16309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, Y.; Adu-Ampratwum, D.; Okumu, A.A.; Kenton, N.T.; Forsyth, C.J. Synthesis of the C22-C40 Domain of the Azaspiracids. Org. Lett. 2016, 18, 1824–1827. [Google Scholar] [CrossRef] [PubMed]
- Kenton, N.T.; Adu-Ampratwum, D.; Okumu, A.A.; McCarron, P.; Kilcoyne, J.; Rise, F.; Wilkins, A.L.; Miles, C.O.; Forsyth, C.J. Stereochemical Definition of the Natural Product (6R,10R,13R, 14R,16R,17R,19S,20S,21R,24S,25S,28S,30S,32R,33R,34R,36S,37S,39R)-Azaspiracid-3 by Total Synthesis and Comparative Analyses. Angew. Chem. (Int. Ed. Engl.) 2018, 57, 810–813. [Google Scholar] [CrossRef] [PubMed]
- Byrne, B.; Dunne, G.; Lyng, J.; Bolton, D.J. The development of a ‘clean sheep policy’ in compliance with the new Hygiene Regulation (EC) 853/2004 (Hygiene 2). Food Microbiol. 2007, 24, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Ben-Gigirey, B.; Rodríguez-Velasco, M.L.; Otero, A.; Vieites, J.M.; Cabado, A.G. A comparative study for PSP toxins quantification by using MBA and HPLC official methods in shellfish. Toxicon Off. J. Int. Soc. Toxinol. 2012, 60, 864–873. [Google Scholar] [CrossRef] [PubMed]
- García-Altares, M.; Diogène, J.; de la Iglesia, P. The implementation of liquid chromatography tandem mass spectrometry for the official control of lipophilic toxins in seafood: Single-laboratory validation under four chromatographic conditions. J. Chromatogr. A 2013, 1275, 48–60. [Google Scholar] [CrossRef] [PubMed]
- McCarron, P.; Giddings, S.D.; Reeves, K.L.; Hess, P.; Quilliam, M.A. A mussel (Mytilus edulis) tissue certified reference material for the marine biotoxins azaspiracids. Anal. Bioanal. Chem. 2015, 407, 2985–2996. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.J.; McCarron, P. A mussel tissue certified reference material for multiple phycotoxins. Part 5: Profiling by liquid chromatography-high-resolution mass spectrometry. Anal. Bioanal. Chem. 2021, 413, 2055–2069. [Google Scholar] [CrossRef]
- Ofuji, K.; Satake, M.; Oshima, Y.; McMahon, T.; James, K.J.; Yasumoto, T. A sensitive and specific determination method for azaspiracids by liquid chromatography mass spectrometry. Nat. Toxins 1999, 7, 247–250. [Google Scholar] [CrossRef]
- Draisci, R.; Palleschi, L.; Ferretti, E.; Furey, A.; James, K.J.; Satake, M.; Yasumoto, T. Development of a method for the identification of azaspiracid in shellfish by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2000, 871, 13–21. [Google Scholar] [CrossRef]
- Lehane, M.; Braña-Magdalena, A.; Moroney, C.; Furey, A.; James, K.J. Liquid chromatography with electrospray ion trap mass spectrometry for the determination of five azaspiracids in shellfish. J. Chromatogr. A 2002, 950, 139–147. [Google Scholar] [CrossRef]
- Lehane, M.; Fidalgo Sáez, M.J.; Magdalena, A.B.; Ruppén Cañás, I.; Díaz Sierra, M.; Hamilton, B.; Furey, A.; James, K.J. Liquid chromatography--multiple tandem mass spectrometry for the determination of ten azaspiracids, including hydroxyl analogues in shellfish. J. Chromatogr. A 2004, 1024, 63–70. [Google Scholar] [CrossRef]
- Skrabáková, Z.; O’Halloran, J.; van Pelt, F.N.; James, K.J. Food contaminant analysis at ultra-high mass resolution: Application of hybrid linear ion trap-orbitrap mass spectrometry for the determination of the polyether toxins, azaspiracids, in shellfish. Rapid Commun. Mass Spectrom. RCM 2010, 24, 2966–2974. [Google Scholar] [CrossRef]
- Yao, J.; Tan, Z.; Zhou, D.; Guo, M.; Xing, L.; Yang, S. Determination of azaspiracid-1 in shellfishes by liquid chromatography with tandem mass spectrometry. Chin. J. Chromatogr. 2010, 28, 363–367. [Google Scholar] [CrossRef]
- Blay, P.; Hui, J.P.; Chang, J.; Melanson, J.E. Screening for multiple classes of marine biotoxins by liquid chromatography-high-resolution mass spectrometry. Anal. Bioanal. Chem. 2011, 400, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Guo, M.; Tan, Z.; Cheng, H.; Li, Z.; Zhai, Y. Liquid chromatography quadrupole linear ion trap mass spectrometry for multiclass screening and identification of lipophilic marine biotoxins in bivalve mollusks. J. Chromatogr. A 2014, 1358, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Yao, X.; Wang, L.; Li, J. Solid-phase extraction-based ultra-sensitive detection of four lipophilic marine biotoxins in bivalves by high-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. Sci. 2015, 53, 373–379. [Google Scholar] [CrossRef]
- Orellana, G.; Van Meulebroek, L.; Van Vooren, S.; De Rijcke, M.; Vandegehuchte, M.; Janssen, C.R.; Vanhaecke, L. Quantification and profiling of lipophilic marine toxins in microalgae by UHPLC coupled to high-resolution orbitrap mass spectrometry. Anal. Bioanal. Chem. 2015, 407, 6345–6356. [Google Scholar] [CrossRef] [PubMed]
- Samdal, I.A.; Løvberg, K.E.; Briggs, L.R.; Kilcoyne, J.; Xu, J.; Forsyth, C.J.; Miles, C.O. Development of an ELISA for the detection of Azaspiracids. J. Agric. Food Chem. 2015, 63, 7855–7861. [Google Scholar] [CrossRef]
- Leonardo, S.; Rambla-Alegre, M.; Samdal, I.A.; Miles, C.O.; Kilcoyne, J.; Diogène, J.; O’Sullivan, C.K.; Campàs, M. Immunorecognition magnetic supports for the development of an electrochemical immunoassay for azaspiracid detection in mussels. Biosens. Bioelectron. 2017, 92, 200–206. [Google Scholar] [CrossRef]
- Samdal, I.A.; Løvberg, K.E.; Kristoffersen, A.B.; Briggs, L.R.; Kilcoyne, J.; Forsyth, C.J.; Miles, C.O. A Practical ELISA for Azaspiracids in Shellfish via Development of a New Plate-Coating Antigen. J. Agric. Food Chem. 2019, 67, 2369–2376. [Google Scholar] [CrossRef]
- Xu, F.; Liu, F.; Wang, C.; Wei, Y. Reversed-phase/weak anion exchange magnetic mesoporous microspheres for removal of matrix effects in lipophilic marine biotoxins analysis by ultrahigh-performance liquid chromatography coupled to tandem mass spectrometry. Food Chem. 2019, 294, 104–111. [Google Scholar] [CrossRef]
- Onofrio, M.D.; Mallet, C.R.; Place, A.R.; Smith, J.L. A screening tool for the direct analysis of marine and freshwater phycotoxins in organic SPATT extracts from the chesapeake bay. Toxins 2020, 12, 322. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Wang, P.; Liu, Y.; Gu, J.; Lü, M.; Wang, J. Determination of three azaspiracids in edible shellfishes by QuEChERS method combined with ultra high performance liquid chromatography-tandem mass spectrometry. Chin. J. Chromatogr. 2013, 31, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Domènech, A.; Cortés-Francisco, N.; Palacios, O.; Franco, J.M.; Riobó, P.; Llerena, J.J.; Vichi, S.; Caixach, J. Determination of lipophilic marine toxins in mussels. Quantification and confirmation criteria using high resolution mass spectrometry. J. Chromatogr. A 2014, 1328, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Singh, A.; Lankford, S.K.; Stuart, J.; Rice, D.; Wu, W.H.; Hungerford, J.M. A rapid method for the detection of diarrhetic shellfish toxins and Azaspiracid shellfish toxins in Washington State shellfish by liquid chromatography tandem mass spectrometry. J. AOAC Int. 2020, 103, 792–799. [Google Scholar] [CrossRef]
- Stobo, L.A.; Lacaze, J.P.; Scott, A.C.; Gallacher, S.; Smith, E.A.; Quilliam, M.A. Liquid chromatography with mass spectrometry--detection of lipophilic shellfish toxins. J. AOAC Int. 2005, 88, 1371–1382. [Google Scholar] [CrossRef] [PubMed]
- Krock, B.; Tillmann, U.; John, U.; Cembella, A. LC-MS-MS aboard ship: Tandem mass spectrometry in the search for phycotoxins and novel toxigenic plankton from the North Sea. Anal. Bioanal. Chem. 2008, 392, 797–803. [Google Scholar] [CrossRef]
- James, K.J.; Sierra, M.D.; Lehane, M.; Braña Magdalena, A.; Furey, A. Detection of five new hydroxyl analogues of azaspiracids in shellfish using multiple tandem mass spectrometry. Toxicon Off. J. Int. Soc. Toxinol. 2003, 41, 277–283. [Google Scholar] [CrossRef]
- Kim, J.H.; Tillmann, U.; Adams, N.G.; Krock, B.; Stutts, W.L.; Deeds, J.R.; Han, M.S.; Trainer, V.L. Identification of Azadinium species and a new azaspiracid from Azadinium poporum in Puget Sound, Washington State, USA. Harmful Algae 2017, 68, 152–167. [Google Scholar] [CrossRef]
- James, K.J.; Furey, A.; Lehane, M.; Ramstad, H.; Aune, T.; Hovgaard, P.; Morris, S.; Higman, W.; Satake, M.; Yasumoto, T. First evidence of an extensive northern European distribution of azaspiracid poisoning (AZP) toxins in shellfish. Toxicon Off. J. Int. Soc. Toxinol. 2002, 40, 909–915. [Google Scholar] [CrossRef]
- Villar-González, A.; Rodríguez-Velasco, M.L.; Ben-Gigirey, B.; Botana, L.M. Lipophilic toxin profile in Galicia (Spain): 2005 toxic episode. Toxicon Off. J. Int. Soc. Toxinol. 2007, 49, 1129–1134. [Google Scholar] [CrossRef]
- Torgersen, T.; Bremnes, N.B.; Rundberget, T.; Aune, T. Structural confirmation and occurrence of azaspiracids in Scandinavian brown crabs (Cancer pagurus). Toxicon Off. J. Int. Soc. Toxinol. 2008, 51, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Amzil, Z.; Sibat, M.; Royer, F.; Savar, V. First report on azaspiracid and yessotoxin groups detection in French shellfish. Toxicon Off. J. Int. Soc. Toxinol. 2008, 52, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Stobo, L.A.; Lacaze, J.P.; Scott, A.C.; Petrie, J.; Turrell, E.A. Surveillance of algal toxins in shellfish from Scottish waters. Toxicon Off. J. Int. Soc. Toxinol. 2008, 51, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Liang, Y.; Wu, X.; Xu, D.; Liu, Y.; Liu, L. First report on the detection of pectenotoxin groups in Chinese shellfish by LC-MS/MS. Toxicon Off. J. Int. Soc. Toxinol. 2011, 57, 1000–1007. [Google Scholar] [CrossRef]
- Wu, H.; Yao, J.; Guo, M.; Tan, Z.; Zhou, D.; Zhai, Y. Distribution of marine lipophilic toxins in shellfish products collected from the chinese market. Mar. Drugs 2015, 13, 4281–4295. [Google Scholar] [CrossRef]
- Tillmann, U.; Borel, C.M.; Barrera, F.; Lara, R.; Krock, B.; Almandoz, G.O.; Witt, M.; Trefault, N. Azadinium poporum from the Argentine Continental Shelf, Southwestern Atlantic, produces azaspiracid-2 and azaspiracid-2 phosphate. Harmful Algae 2016, 51, 40–55. [Google Scholar] [CrossRef]
- Tillmann, U.; Jaén, D.; Fernández, L.; Gottschling, M.; Witt, M.; Blanco, J.; Krock, B. Amphidoma languida (Amphidomatacea, Dinophyceae) with a novel azaspiracid toxin profile identified as the cause of molluscan contamination at the Atlantic coast of southern Spain. Harmful Algae 2017, 62, 113–126. [Google Scholar] [CrossRef]
- Schirone, M.; Berti, M.; Visciano, P.; Chiumiento, F.; Migliorati, G.; Tofalo, R.; Suzzi, G.; Di Giacinto, F.; Ferri, N. Determination of lipophilic marine biotoxins in mussels harvested from the adriatic sea by LC-MS/MS. Front. Microbiol. 2018, 9, 152. [Google Scholar] [CrossRef] [PubMed]
- Wietkamp, S.; Krock, B.; Gu, H.; Voß, D.; Klemm, K.; Tillmann, U. Occurrence and distribution of Amphidomataceae (Dinophyceae) in Danish coastal waters of the North Sea, the Limfjord and the Kattegat/Belt area. Harmful Algae 2019, 88, 101637. [Google Scholar] [CrossRef] [PubMed]
- Otero, P.; Miguéns, N.; Rodríguez, I.; Botana, L.M. LC-MS/MS analysis of the emerging toxin Pinnatoxin-G and high levels of esterified OA group toxins in galician commercial mussels. Toxins 2019, 11, 394. [Google Scholar] [CrossRef]
- Vale, P.; Bire, R.; Hess, P. Confirmation by LC-MS/MS of azaspiracids in shellfish from the Portuguese north-western coast. Toxicon Off. J. Int. Soc. Toxinol. 2008, 51, 1449–1456. [Google Scholar] [CrossRef]
- Fux, E.; Rode, D.; Bire, R.; Hess, P. Approaches to the evaluation of matrix effects in the liquid chromatography-mass spectrometry (LC-MS) analysis of three regulated lipophilic toxin groups in mussel matrix (Mytilus edulis). Food Addit. Contam. Part A Chem. Anal. Control Exp. Risk Assess. 2008, 25, 1024–1032. [Google Scholar] [CrossRef]
- McCarron, P.; Giddings, S.D.; Quilliam, M.A. A mussel tissue certified reference material for multiple phycotoxins. Part 2: Liquid chromatography-mass spectrometry, sample extraction and quantitation procedures. Anal. Bioanal. Chem. 2011, 400, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Gerssen, A.; Mulder, P.P.; McElhinney, M.A.; de Boer, J. Liquid chromatography-tandem mass spectrometry method for the detection of marine lipophilic toxins under alkaline conditions. J. Chromatogr. A 2009, 1216, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, R.C.; Kong, F.Z.; Li, C.; Dai, L.; Chen, Z.F.; Zhou, M.J. Lipophilic marine toxins discovered in the Bohai Sea using high performance liquid chromatography coupled with tandem mass spectrometry. Chemosphere 2017, 183, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Rúbies, A.; Muñoz, E.; Gibert, D.; Cortés-Francisco, N.; Granados, M.; Caixach, J.; Centrich, F. New method for the analysis of lipophilic marine biotoxins in fresh and canned bivalves by liquid chromatography coupled to high resolution mass spectrometry: A quick, easy, cheap, efficient, rugged, safe approach. J. Chromatogr. A 2015, 1386, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Alfonso, C.; Alfonso, A.; Otero, P.; Rodríguez, P.; Vieytes, M.R.; Elliot, C.; Higgins, C.; Botana, L.M. Purification of five azaspiracids from mussel samples contaminated with DSP toxins and azaspiracids. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 865, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Bosch-Orea, C.; Sanchís, J.; Farré, M.; Barceló, D. Analysis of lipophilic marine biotoxins by liquid chromatography coupled with high-resolution mass spectrometry in seawater from the Catalan Coast. Anal. Bioanal. Chem. 2017, 409, 5451–5462. [Google Scholar] [CrossRef]
- D’Amore, T.; Lo Magro, S.; Vita, V.; Di Taranto, A. Optimization and validation of a high throughput UHPLC-MS/MS method for determination of the EU regulated lipophilic marine toxins and occurrence in fresh and processed shellfish. Mar. Drugs 2022, 20, 173. [Google Scholar] [CrossRef] [PubMed]
- Moroney, C.; Lehane, M.; Braña-Magdalena, A.; Furey, A.; James, K.J. Comparison of solid-phase extraction methods for the determination of azaspiracids in shellfish by liquid chromatography-electrospray mass spectrometry. J. Chromatogr. A 2002, 963, 353–361. [Google Scholar] [CrossRef]
- Gerssen, A.; McElhinney, M.A.; Mulder, P.P.; Bire, R.; Hess, P.; de Boer, J. Solid phase extraction for removal of matrix effects in lipophilic marine toxin analysis by liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2009, 394, 1213–1226. [Google Scholar] [CrossRef] [PubMed]
- Magdalena, A.B.; Lehane, M.; Krys, S.; Fernández, M.L.; Furey, A.; James, K.J. The first identification of azaspiracids in shellfish from France and Spain. Toxicon Off. J. Int. Soc. Toxinol. 2003, 42, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Orellana, G.; Vanden Bussche, J.; Van Meulebroek, L.; Vandegehuchte, M.; Janssen, C.; Vanhaecke, L. Validation of a confirmatory method for lipophilic marine toxins in shellfish using UHPLC-HR-Orbitrap MS. Anal. Bioanal. Chem. 2014, 406, 5303–5312. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.Q.V.; Afonso, I.I.; Lage, S.; Costa, P.R.; Canário, A.V.M.; Da Silva, J.P. Quantitation overcoming matrix effects of lipophilic toxins in Mytilus galloprovincialis by liquid chromatography-full scan high resolution mass spectrometry analysis (LC-HR-MS). Mar. Drugs 2022, 20, 143. [Google Scholar] [CrossRef] [PubMed]
- Brombacher, S.; Edmonds, S.; Volmer, D.A. Studies on azaspiracid biotoxins. II. Mass spectral behavior and structural elucidation of azaspiracid analogs. Rapid Commun. Mass Spectrom. RCM 2002, 16, 2306–2316. [Google Scholar] [CrossRef] [PubMed]
- Díaz Sierra, M.; Furey, A.; Hamilton, B.; Lehane, M.; James, K.J. Elucidation of the fragmentation pathways of azaspiracids, using electrospray ionisation, hydrogen/deuterium exchange, and multiple-stage mass spectrometry. J. Mass Spectrom. 2003, 38, 1178–1186. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Huang, B.; Zhao, Q.; Wang, Z.; Liu, W.; Zhang, J.; Zhou, Y.; Sun, Q.; Huang, H.; Huang, X.; et al. Shellfish contamination with lipophilic toxins and dietary exposure assessments from consumption of shellfish products in Shenzhen, China. Ecotoxicol. Environ. Saf. 2021, 221, 112446. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Uchida, H.; Watanabe, R.; Matsushima, R.; Oikawa, H.; Takahashi, K.; Iwataki, M.; Suzuki, T. Complex profiles of azaspiracid analogues in two culture strains of Azadinium poporum (Amphidomataceae, Dinophyceae) isolated from Japanese coastal waters determined by LC-MS/MS. Toxicon Off. J. Int. Soc. Toxinol. 2021, 199, 145–155. [Google Scholar] [CrossRef]
- Kilcoyne, J.; Fux, E. Strategies for the elimination of matrix effects in the liquid chromatography tandem mass spectrometry analysis of the lipophilic toxins okadaic acid and azaspiracid-1 in molluscan shellfish. J. Chromatogr. A 2010, 1217, 7123–7130. [Google Scholar] [CrossRef]
- McCarron, P.; Wright, E.; Quilliam, M.A. Liquid chromatography/mass spectrometry of domoic acid and lipophilic shellfish toxins with selected reaction monitoring and optional confirmation by library searching of product ion spectra. J. AOAC Int. 2014, 97, 316–324. [Google Scholar] [CrossRef]
- Zendong, Z.; McCarron, P.; Herrenknecht, C.; Sibat, M.; Amzil, Z.; Cole, R.B.; Hess, P. High resolution mass spectrometry for quantitative analysis and untargeted screening of algal toxins in mussels and passive samplers. J. Chromatogr. A 2015, 1416, 10–21. [Google Scholar] [CrossRef]
- Dom, I.; Biré, R.; Hort, V.; Lavison-Bompard, G.; Nicolas, M.; Guérin, T. Extended targeted and non-targeted strategies for the analysis of marine toxins in mussels and oysters by (LC-HRMS). Toxins 2018, 10, 375. [Google Scholar] [CrossRef]
- Kilcoyne, J.; Nulty, C.; Jauffrais, T.; McCarron, P.; Herve, F.; Foley, B.; Rise, F.; Crain, S.; Wilkins, A.L.; Twiner, M.J.; et al. Isolation, structure elucidation, relative LC-MS response, and in vitro toxicity of azaspiracids from the dinoflagellate Azadinium spinosum. J. Mol. Evol. 2014, 77, 2465–2474. [Google Scholar] [CrossRef]
- Kilcoyne, J.; Twiner, M.J.; McCarron, P.; Crain, S.; Giddings, S.D.; Foley, B.; Rise, F.; Hess, P.; Wilkins, A.L.; Miles, C.O. Structure Elucidation, Relative LC-MS Response and In Vitro Toxicity of Azaspiracids 7-10 Isolated from Mussels (Mytilus edulis). J. Agric. Food Chem. 2015, 63, 5083–5091. [Google Scholar] [CrossRef]
- Perez, R.A.; Rehmann, N.; Crain, S.; LeBlanc, P.; Craft, C.; MacKinnon, S.; Reeves, K.; Burton, I.W.; Walter, J.A.; Hess, P.; et al. The preparation of certified calibration solutions for azaspiracid-1, -2, and -3, potent marine biotoxins found in shellfish. Anal. Bioanal. Chem. 2010, 398, 2243–2252. [Google Scholar] [CrossRef]
- Kilcoyne, J.; McCarron, P.; Twiner, M.J.; Rise, F.; Hess, P.; Wilkins, A.L.; Miles, C.O. Identification of 21,22-dehydroazaspiracids in mussels (Mytilus edulis) and in vitro toxicity of Azaspiracid-26. J. Mol. Evol. 2018, 81, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Ciminiello, P.; Dell’Aversano, C.; Fattorusso, E.; Forino, M.; Tartaglione, L.; Boschetti, L.; Rubini, S.; Cangini, M.; Pigozzi, S.; Poletti, R. Complex toxin profile of Mytilus galloprovincialis from the Adriatic sea revealed by LC-MS. Toxicon Off. J. Int. Soc. Toxinol. 2010, 55, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Kilcoyne, J.; McCarron, P.; Giddings, S.D.; Waaler, T.; Rundberget, T.; Samdal, I.A.; Løvberg, K.E. Selective extraction and purification of Azaspiracids from blue mussels (Mytilus edulis) using boric acid gel. J. Agric. Food Chem. 2018, 66, 2962–2969. [Google Scholar] [CrossRef] [PubMed]
- Ciminiello, P.; Dell’Aversano, C.; Fattorusso, E.; Forino, M.; Magno, S.; Santelia, F.; Tsoukatou, M. Investigation of the toxin profile of Greek mussels Mytilus galloprovincialis by liquid chromatography-mass spectrometry. Toxicon Off. J. Int. Soc. Toxinol. 2006, 47, 174–181. [Google Scholar] [CrossRef] [PubMed]
- James, K.J.; Lehane, M.; Moroney, C.; Fernandez-Puente, P.; Satake, M.; Yasumoto, T.; Furey, A. Azaspiracid shellfish poisoning: Unusual toxin dynamics in shellfish and the increased risk of acute human intoxications. Food Addit. Contam. 2002, 19, 555–561. [Google Scholar] [CrossRef] [PubMed]
- García-Mendoza, E.; Sánchez-Bravo, Y.A.; Turner, A.; Blanco, J.; O’Neil, A.; Mancera-Flores, J.; Pérez-Brunius, P.; Rivas, D.; Almazán-Becerril, A.; Peña-Manjarrez, J.L. Lipophilic toxins in cultivated mussels (Mytilus galloprovincialis) from Baja California, Mexico. Toxicon Off. J. Int. Soc. Toxinol. 2014, 90, 111–123. [Google Scholar] [CrossRef]
- Ledreux, A.; Sérandour, A.L.; Morin, B.; Derick, S.; Lanceleur, R.; Hamlaoui, S.; Furger, C.; Biré, R.; Krys, S.; Fessard, V.; et al. Collaborative study for the detection of toxic compounds in shellfish extracts using cell-based assays. Part II: Application to shellfish extracts spiked with lipophilic marine toxins. Anal. Bioanal. Chem. 2012, 403, 1995–2007. [Google Scholar] [CrossRef]
- Flanagan, A.F.; Callanan, K.R.; Donlon, J.; Palmer, R.; Forde, A.; Kane, M. A cytotoxicity assay for the detection and differentiation of two families of shellfish toxins. Toxicon Off. J. Int. Soc. Toxinol. 2001, 39, 1021–1027. [Google Scholar] [CrossRef]
- Forsyth, C.J.; Xu, J.; Nguyen, S.T.; Samdal, I.A.; Briggs, L.R.; Rundberget, T.; Sandvik, M.; Miles, C.O. Antibodies with broad specificity to azaspiracids by use of synthetic haptens. J. Am. Chem. Soc. 2006, 128, 15114–15116. [Google Scholar] [CrossRef] [PubMed]
- Frederick, M.O.; De Lamo Marin, S.; Janda, K.D.; Nicolaou, K.C.; Dickerson, T.J. Monoclonal antibodies with orthogonal azaspiracid epitopes. Chembiochem 2009, 10, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
- Tebben, J.; Zurhelle, C.; Tubaro, A.; Samdal, I.A.; Krock, B.; Kilcoyne, J.; Sosa, S.; Trainer, V.L.; Deeds, J.R.; Tillmann, U. Structure and toxicity of AZA-59, an azaspiracid shellfish poisoning toxin produced by Azadinium poporum (Dinophyceae). Harmful Algae 2023, 124, 102388. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, L.P.; Vilariño, N.; Louzao, M.C.; Dickerson, T.J.; Nicolaou, K.C.; Frederick, M.O.; Botana, L.M. Microsphere-based immunoassay for the detection of azaspiracids. Anal. Biochem. 2014, 447, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Samdal, I.A.; Sandvik, M.; Vu, J.; Sukenthirarasa, M.S.; Kanesamurthy, S.; Løvberg, K.L.E.; Kilcoyne, J.; Forsyth, C.J.; Wright, E.J.; Miles, C.O. Preparation and characterization of an immunoaffinity column for the selective extraction of azaspiracids. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2022, 1207, 123360. [Google Scholar] [CrossRef] [PubMed]
- Elgarch, A.; Vale, P.; Rifai, S.; Fassouane, A. Detection of diarrheic shellfish poisoning and azaspiracid toxins in Moroccan mussels: Comparison of the LC-MS method with the commercial immunoassay kit. Mar. Drugs 2008, 6, 587–594. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).