Microalgae n-3 PUFAs Production and Use in Food and Feed Industries
Abstract
1. Algae, a Source of Bioactive Compounds
2. Polyunsaturated Fatty Acids and Human Health
2.1. n-3 PUFAs and Cardiovascular Diseases
2.2. n-3 PUFAs Against Obesity and Diabetes
2.3. Anti-Inflammatory Properties of n-3 PUFAs in the Context of the COVID-19 Pandemic
2.4. EPA and DHA Roles in Gut Microbiota
2.5. Daily Recommendation and Requirement of n-3 PUFAs
2.6. Role of n-3 PUFAs on Brain Development, Tumors and Cancer
2.7. N-3 PUFAs and Retinopathies
3. Increasing Demand for n-3 Polyunsaturated Fatty Acids
4. Microalgae as n-3 Polyunsaturated Fatty Acids Producers
5. Synthesis and Production of n-3 Polyunsaturated Fatty Acids
5.1. Nomenclature
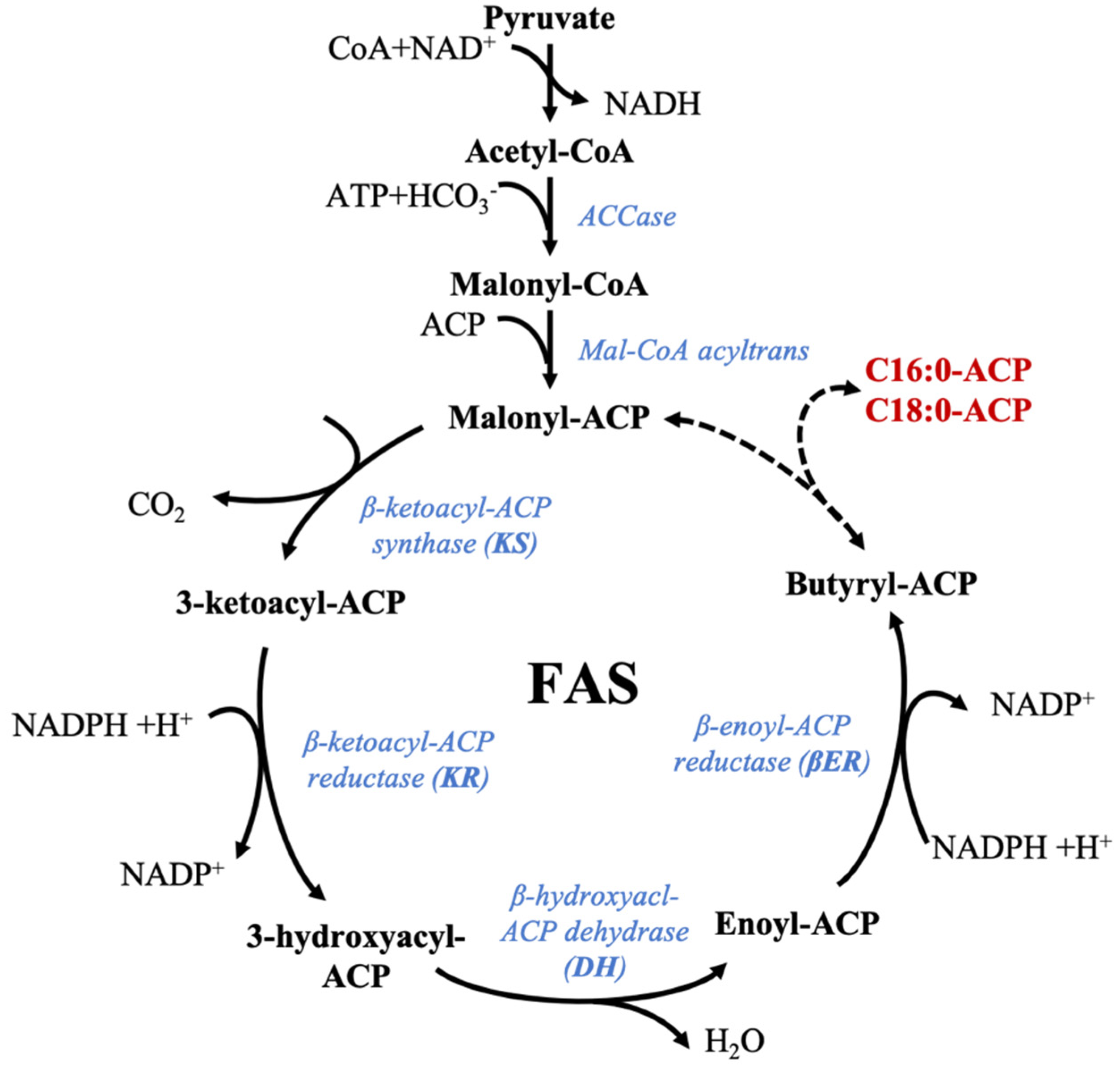
5.2. Fatty Acid Synthase (FAS): Synthesis of Saturated Fatty Acids
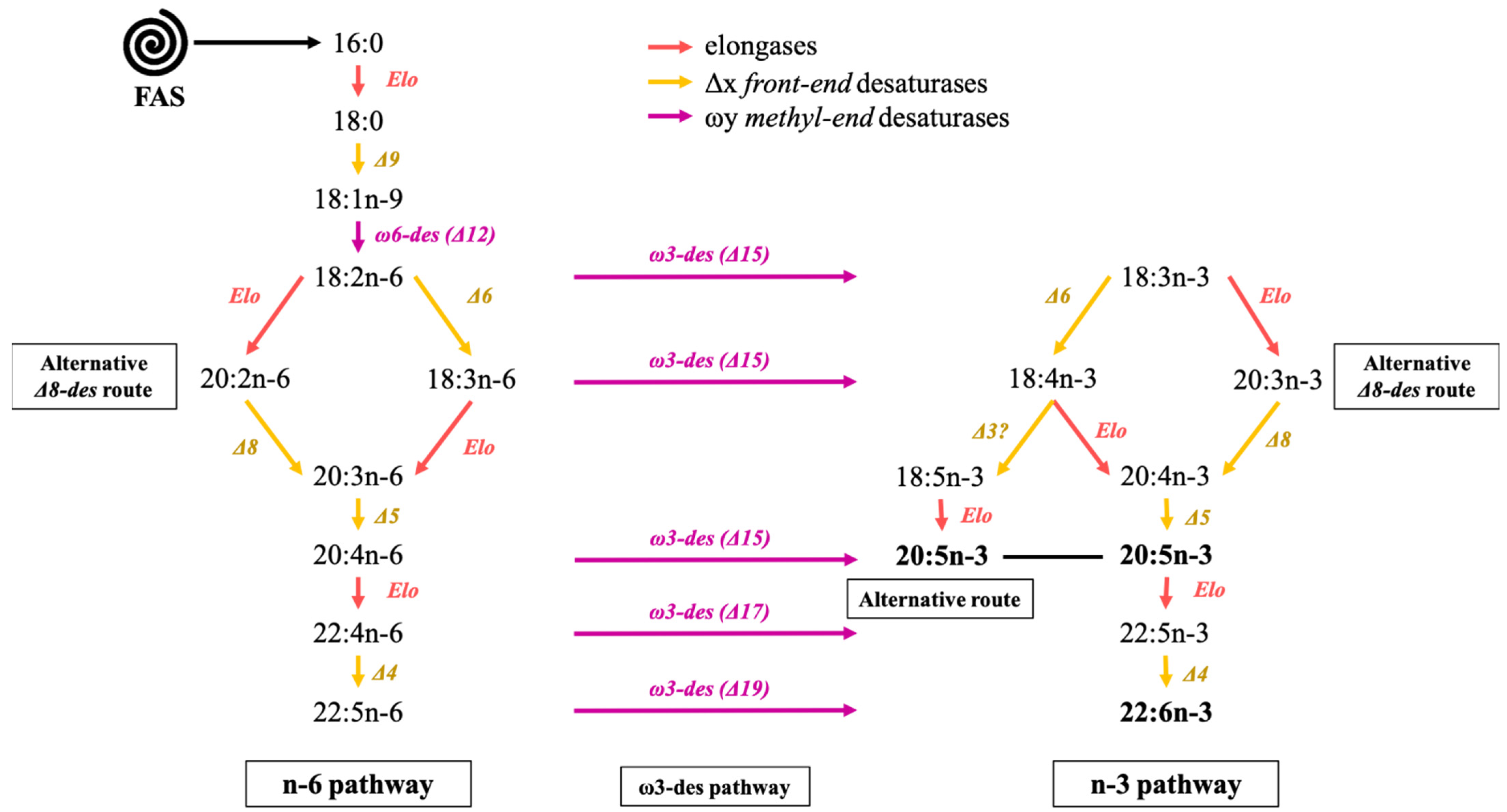
5.3. Elongation and Desaturation Steps of the n-3 and n-6 Pathways
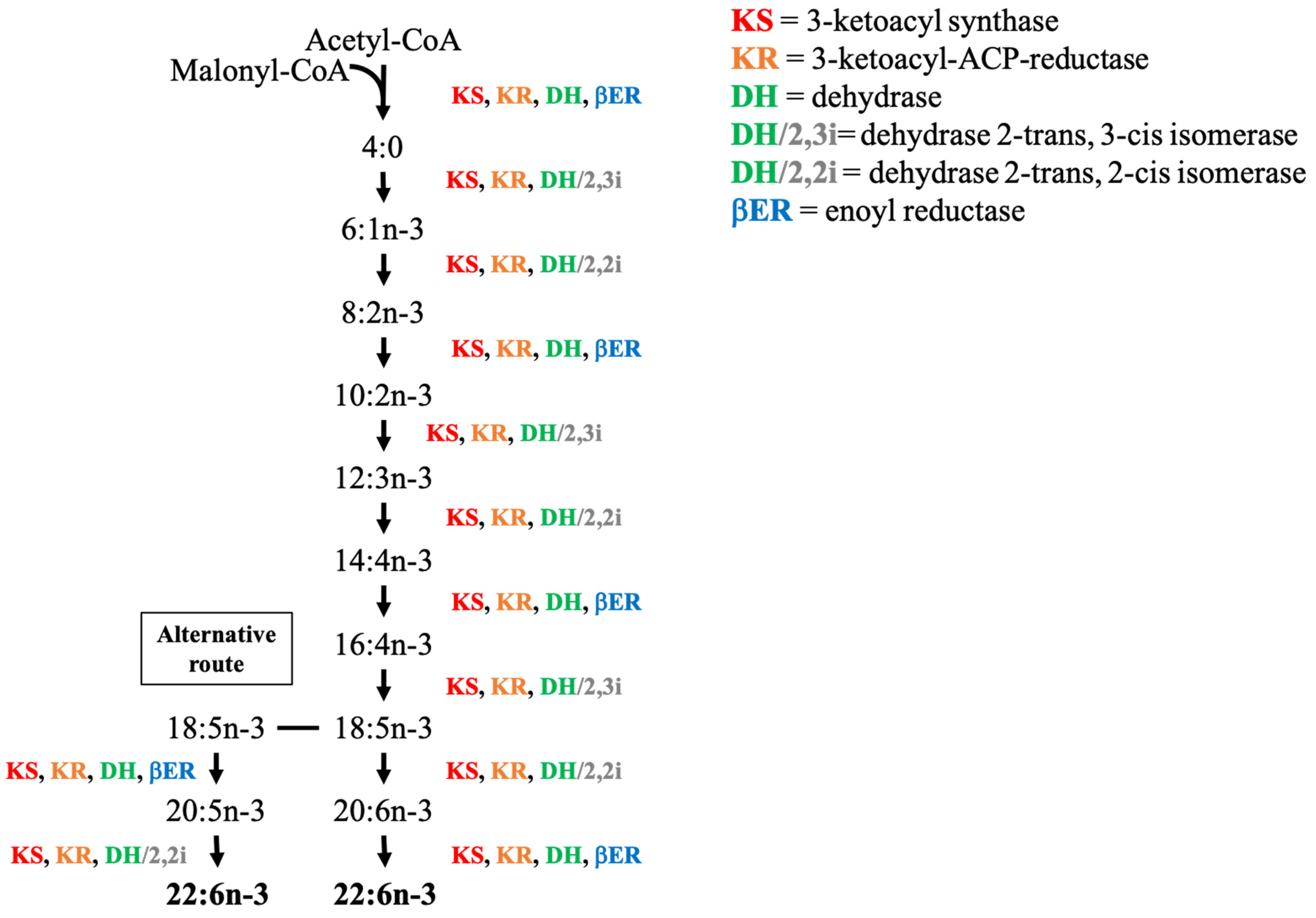
5.4. Polyketide Synthase Pathway (PKS)
5.5. Elucidation of Microalgae Synthesis Pathways
6. Use of n-3 Polyunsaturated Fatty Acids in Food and Feed Industries
6.1. Use and Proprieties of n-3 Polyunsaturated Fatty Acids in the Food Industry
6.2. Example of Microalgae Valuable in Food Industries
6.3. Use of n-3 Polyunsaturated Fatty Acids in the Feed Industry and Aquaculture
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Patras, D.; Moraru, C.V.; Socaciu, C. Bioactive Ingredients from Microalgae: Food and Feed Applications. BUASVMCN-FST 2019, 76, 1–9. [Google Scholar] [CrossRef]
- Levring, T.; Hoppe, H.A.; Schmid, O.J. Marine Algae: A Survey of Research and Utilization; Walter de Gruyter GmbH & Co KG: Hamburg, Germany, 2019; ISBN 978-3-11-143168-0. [Google Scholar]
- Pinckney, J.L. A Mini-Review of the Contribution of Benthic Microalgae to the Ecology of the Continental Shelf in the South Atlantic Bight. Chesap. Sci. 2018, 41, 2070–2078. [Google Scholar] [CrossRef]
- García, J.L.; De Vicente, M.; Galán, B. Microalgae, old sustainable food and fashion nutraceuticals. Microb. Biotechnol. 2017, 10, 1017–1024. [Google Scholar] [CrossRef]
- Chacón-Lee, T.; González-Mariño, G. Microalgae for “Healthy” Foods-Possibilities and Challenges. Compr. Rev. Food Sci. Food Saf. 2010, 9, 655–675. [Google Scholar] [CrossRef] [PubMed]
- Barka, A.; Blecker, C. Microalgae as a Potential Source of Single-Cell Proteins. A Review. Biotechnol. Agron. Société Environ. 2016, 10. [Google Scholar] [CrossRef]
- Spier, M.R.; Peron-Schlosser, B.; Paludo, L.C.; Gallo-García, L.A.; Zanette, C.M. Microalgae as enzymes biofactories. In Handbook of Microalgae-Based Processes and Products; Elsevier: Amsterdam, The Netherlands, 2020; pp. 687–706. [Google Scholar]
- Raghukumar, S. Thraustochytrid Marine Protists: Production of PUFAs and Other Emerging Technologies. Mar. Biotechnol. 2008, 10, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Leyland, B.; Leu, S.; Boussiba, S. Are Thraustochytrids algae? Fungal Biol. 2017, 121, 835–840. [Google Scholar] [CrossRef]
- Gouveia, L.; Batista, A.P.; Sousa, I.; Raymundo, A.; Bandarra, N. Microalgae in novel food products. In Food Chemistry Research Developments; Papadopoulos, K.N., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2008; pp. 2–37. ISBN 978-1-60456-262-0. [Google Scholar]
- Nova, P.; Martins, A.P.; Teixeira, C.; Abreu, H.; Silva, J.G.; Silva, A.M.; Freitas, A.C.; Gomes, A.M. Foods with microalgae and seaweeds fostering consumers health: A review on scientific and market innovations. Environ. Boil. Fishes 2020, 32, 1789–1802. [Google Scholar] [CrossRef]
- Bernaerts, T.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 37, 107419. [Google Scholar] [CrossRef]
- Sathasivam, R.; Radhakrishnan, R.; Hashem, A.; Allah, E.F.A. Microalgae metabolites: A rich source for food and medicine. Saudi J. Biol. Sci. 2019, 26, 709–722. [Google Scholar] [CrossRef]
- Hamed, I.; Özogul, F.; Özogul, Y.; Regenstein, J.M. Marine Bioactive Compounds and Their Health Benefits: A Review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 446–465. [Google Scholar] [CrossRef]
- Li, D.; Wahlqvist, M.L.; Sinclair, A.J. Advances in n-3 polyunsaturated fatty acid nutrition. Asia Pac. J. Clin. Nutr. 2019, 28, 1–5. [Google Scholar] [PubMed]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Wu, J.H.Y. (n-3) Fatty Acids and Cardiovascular Health: Are Effects of EPA and DHA Shared or Complementary? J. Nutr. 2012, 142, 614S–625S. [Google Scholar] [CrossRef] [PubMed]
- Gladyshev, M.I.; Sushchik, N.N.; Makhutova, O.N. Production of EPA and DHA in aquatic ecosystems and their transfer to the land. Prostaglandins Other Lipid Mediat. 2013, 107, 117–126. [Google Scholar] [CrossRef]
- Rimm, E.B.; Appel, L.J.; Chiuve, S.E.; Djoussé, L.; Engler, M.B.; Kris-Etherton, P.M.; Mozaffarian, D.; Siscovick, D.S.; Lichtenstein, A.H. A Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and Cardiovascular Disease: A Science Advisory From the American Heart Association. Circulation 2018, 138, e35–e47. [Google Scholar] [CrossRef]
- Colussi, G.; Catena, C.; Novello, M.; Bertin, N.; Sechi, L.A. Impact of omega-3 polyunsaturated fatty acids on vascular function and blood pressure: Relevance for cardiovascular outcomes. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Casula, M.; Olmastroni, E.; Gazzotti, M.; Galimberti, F.; Zambon, A.; Catapano, A.L. Omega-3 polyunsaturated fatty acids supplementation and cardiovascular outcomes: Do formulation, dosage, and baseline cardiovascular risk matter? An updated meta-analysis of randomized controlled trials. Pharmacol. Res. 2020, 160, 105060. [Google Scholar] [CrossRef]
- Itsiopoulos, C.; Marx, W.; Mayr, H.; Tatucu-Babet, O.; Dash, S.; George, E.; Trakman, G.; Kelly, J.; Thomas, C.; Brazionis, L. The role of omega-3 polyunsaturated fatty acid supplementation in the management of type 2 diabetes mellitus: A narrative review. J. Nutr. Intermed. Metab. 2018, 14, 42–51. [Google Scholar] [CrossRef]
- Madingou, N.; Gilbert, K.; Tomaro, L.; Touchette, C.P.; Trudeau, F.; Fortin, S.; Rousseau, G. Comparison of the effects of EPA and DHA alone or in combination in a murine model of myocardial infarction. Prostaglandins Leukot. Essent. Fat. Acids 2016, 111, 11–16. [Google Scholar] [CrossRef]
- Kalstad, A.A.; Myhre, P.L.; Laake, K.; Tveit, S.H.; Schmidt, E.B.; Smith, P.; Nilsen, D.W.T.; Tveit, A.; Fagerland, M.W.; Solheim, S.; et al. Effects of n-3 Fatty Acid Supplements in Elderly Patients after Myocardial Infarction: A Randomized Controlled Trial. Circulation 2020. [Google Scholar] [CrossRef]
- Sakamoto, A.; Saotome, M.; Iguchi, K.; Maekawa, Y. Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure: Current Understanding for Basic to Clinical Relevance. Int. J. Mol. Sci. 2019, 20, 4025. [Google Scholar] [CrossRef]
- Uljević, M.V.; Starčević, K.; Mašek, T.; Bočina, I.; Restović, I.; Kević, N.; Racetin, A.; Kretzschmar, G.; Grobe, M.; Vukojević, K.; et al. Dietary DHA/EPA Supplementation Ameliorates Diabetic Nephropathy by Protecting from Distal Tubular Cell Damage. Cell Tissue Res. 2019, 378, 301–317. [Google Scholar] [CrossRef] [PubMed]
- Farsi, P.F.; Djazayery, A.; Eshraghian, M.R.; Koohdani, F.; Akbar, A.; Derakhshanian, H.; Zarei, M.; Javanbakht, M.H.; Djalali, M. Effects of Supplementation with Omega-3 on Insulin Sensitivity and Non-Esterified Free Fatty Acid (NEFA) in Type 2 Di-abetic Patients. Arq. Bras. Endocrinol. Metabol. 2014, 58, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Avallone, L.; Shaikh, A.; Hassan, A.; Tajuddin, N. Prescription omega-3 fatty acid products: Considerations for patients with diabetes mellitus. Diabetes Metab. Syndr. Obesity Targets Ther. 2016, 9, 109–118. [Google Scholar] [CrossRef]
- Reis, C.E.G.; Landim, K.C.; Nunes, A.C.S.; Dullius, J. Safety in the hypertriglyceridemia treatment with N-3 polyunsaturated fatty acids on glucose metabolism in subjects with type 2 diabetes mellitus. Nutr. Hosp. 2014, 31, 570–576. [Google Scholar] [PubMed]
- Calder, P.C. n−3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [CrossRef]
- Chang, J.P.-C.; Pariante, C.M.; Su, K.-P. Omega-3 fatty acids in the psychological and physiological resilience against COVID-Prostaglandins Leukot. Essent. Fat. Acids 2020, 161, 102177. [Google Scholar] [CrossRef] [PubMed]
- Torrinhas, R.S.; Calder, P.C.; Lemos, G.O.; Waitzberg, D.L. Parenteral fish oil: An adjuvant pharmacotherapy for coronavirus disease 2019? Nutrition 2021, 81, 110900. [Google Scholar] [CrossRef]
- Fung, T.C.; Olson, C.A.; Hsiao, E.Y. Interactions between the microbiota, immune and nervous systems in health and disease. Nat. Neurosci. 2017, 20, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Ding, Y.; Saedi, N.; Choi, M.; Sridharan, G.V.; Sherr, D.H.; Yarmush, M.L.; Alaniz, R.C.; Jayaraman, A.; Lee, K. Gut Microbiota-Derived Tryptophan Metabolites Modulate Inflammatory Response in Hepatocytes and Macrophages. Cell Rep. 2018, 23, 1099–1111. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganism 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef]
- Vetrani, C.; Maukonen, J.; Bozzetto, L.; Della Pepa, G.; Vitale, M.; Costabile, G.; Riccardi, G.; Rivellese, A.A.; Saarela, M.; Annuzzi, G. Diets Naturally Rich in Polyphenols and/or Long-Chain n-3 Polyunsaturated Fatty Acids Differently Affect Mi-crobiota Composition in High-Cardiometabolic-Risk Individuals. Acta Diabetol. 2020, 8, 853–860. [Google Scholar] [CrossRef]
- Bentley-Hewitt, K.L.; Narbad, A.; Majsak-Newman, G.; Philo, M.R.; Lund, E.K. Lactobacilli survival and adhesion to colonic epithelial cell lines is dependent on long chain fatty acid exposure. Eur. J. Lipid Sci. Technol. 2017, 119, 1700062. [Google Scholar] [CrossRef]
- Duru, M. Trends in agri-food choices for health since the 1960s: The case of fatty acids. Oilseeds fats Crop. Lipids 2019, 26, 44. [Google Scholar] [CrossRef]
- Tocher, D.R.; Betancor, M.B.; Sprague, M.; Olsen, R.E.; Napier, J.A. Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand. Nutrients 2019, 11, 89. [Google Scholar] [CrossRef]
- Richter, C.; Skulas-Ray, A.; Kris-Etherton, P. Recommended intake of fish and fish oils worldwide. In Fish and Fish Oil in Health and Disease Prevention; Elsevier: London, UK, 2016; pp. 27–48. ISBN 978-0-12-802844-5. [Google Scholar]
- ANSES. Actualisation des Repères du PNNS: Révision des Repères de Consommation Alimentaires; Agence Nationale de Sécurité Sanitaire de l’Alimentation, de l’environnement et du Travail: CEDEX 94701, Maison-Alfort, France, 2016; p. 280.
- Bowen, K.J.; Harris, W.S.; Kris-Etherton, P.M. Omega-3 Fatty Acids and Cardiovascular Disease: Are There Benefits? Curr. Treat. Options Cardiovasc. Med. 2016, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Garg, P.; Pejaver, R.K.; Sukhija, M.; Ahuja, A. Role of DHA, ARA, & phospholipids in brain development: An Indian perspective. Clin. Epidemiol. Glob. Heal. 2017, 5, 155–162. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H. The Importance of Marine Omega-3s for Brain Development and the Prevention and Treatment of Behavior, Mood, and Other Brain Disorders. Nutrients 2020, 12, 2333. [Google Scholar] [CrossRef] [PubMed]
- Sugasini, D.; Thomas, R.; Yalagala, P.C.R.; Tai, L.M.; Subbaiah, P.V. Dietary Docosahexaenoic Acid (DHA) as Lysophospha-tidylcholine, but Not as Free Acid, Enriches Brain DHA and Improves Memory in Adult Mice. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Liu, J.-H.; Wang, Q.; You, Q.-L.; Li, Z.-L.; Hu, N.-Y.; Wang, Y.; Jin, Z.-L.; Li, S.-J.; Li, X.-W.; Yang, J.-M.; et al. Acute EPA-induced learning and memory impairment in mice is prevented by DHA. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Jandacek, R.; Tso, P.; Blom, T.J.; Welge, J.A.; Strawn, J.R.; Adler, C.M.; Strakowski, S.M.; DelBello, M.P. Adolescents with or at ultra-high risk for bipolar disorder exhibit erythrocyte docosahexaenoic acid and eicosapentaenoic acid deficits: A candidate prodromal risk biomarker. Early Interv. Psychiatry 2015, 10, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Unay, B.; Sarıcı, S.Ü.; Ulaş, Ü.H.; Akın, R.; Alpay, F.; Gökçay, E. Nutritional effects on auditory brainstem maturation in healthy term infants. Arch. Dis. Child. Fetal Neonatal Ed. 2004, 89, F177–F179. [Google Scholar] [CrossRef]
- Haubner, L.Y.; Stockard, J.E.; Saste, M.D.; Benford, V.J.; Phelps, C.P.; Chen, L.T.; Barness, L.; Wiener, D.; Carver, J.D. Maternal dietary docosahexanoic acid content affects the rat pup auditory system. Brain Res. Bull. 2002, 58, 1–5. [Google Scholar] [CrossRef]
- Le Belle, J.; Sperry, J.; Ludwig, K.; Harris, N.; Caldwell, M.; Kornblum, H. Docosahexaenoic Acid Has Stem Cell-Specific Effects in the SVZ and Restores Olfactory Neurogenesis and Function in the Aging Brain. Neuroscience 2020. [Google Scholar] [CrossRef]
- Khoury, S.; Soubeyre, V.; Cabaret, S.; Merle, L.; Grégoire, S.; Deprêtre, N.; Jarriault, D.; Grosmaitre, X.; Bretillon, L.; Berdeaux, O.; et al. Perinatal exposure to diets with different n-6:n-3 fatty acid ratios affects olfactory tissue fatty acid composition. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.-T.; Xu, J.; Wang, Y.-M.; Xue, C.-H. Health benefits of dietary marine DHA/EPA-enriched glycerophospholipids. Prog. Lipid Res. 2019, 75, 100997. [Google Scholar] [CrossRef]
- Mohajeri, M.H.; Troesch, B.; Weber, P. Inadequate supply of vitamins and DHA in the elderly: Implications for brain aging and Alzheimer-type dementia. Nutrition 2015, 31, 261–275. [Google Scholar] [CrossRef]
- Sun, G.Y.; Simonyi, A.; Fritsche, K.L.; Chuang, D.Y.; Hannink, M.; Gu, Z.; Greenlief, C.M.; Yao, J.K.; Lee, J.C.-M.; Beversdorf, D.Q. Docosahexaenoic acid (DHA): An essential nutrient and a nutraceutical for brain health and diseases. Prostaglandins Leukot. Essent. Fat. Acids 2018, 136, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Mei, Y.; Han, H.; Liu, F.; Yang, X.M.; Shao, Y.; Xie, B.; Long, B. Effects of Omega-3 in the treatment of violent schizophrenia patients. Schizophr. Res. 2018, 195, 283–285. [Google Scholar] [CrossRef]
- Goupille, C.; Vibet, S.; Frank, P.G.; Mahéo, K. EPA and DHA Fatty Acids Induce a Remodeling of Tumor Vasculature and Potentiate Docetaxel Activity. Int. J. Mol. Sci. 2020, 21, 4965. [Google Scholar] [CrossRef]
- Elmasry, K.; Ibrahim, A.S.; Abdulmoneim, S.; Al-Shabrawey, M. Bioactive lipids and pathological retinal angiogenesis. Br. J. Pharmacol. 2019, 176, 93–109. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, J.; Li, Q.; Cao, B. The Effect of Omega-3 Polyunsaturated Fatty Acid Supplementations on anti-Tumor Drugs in Triple Negative Breast Cancer. Nutr. Cancer 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Sun, S.; Li, J. Docosahexaenoic acid (DHA) induces apoptosis in human hepatocellular carcinoma cells. HPB 2016, 18, e166. [Google Scholar] [CrossRef][Green Version]
- Xue, M.; Ge, Y.; Yu, C.; Zheng, Z.; He, X.; Zhao, J. Apoptosis is induced by docosahexaenoic acid in breast cancer cells via death receptor and mitochondria-mediated pathways. Mol. Med. Rep. 2017, 16, 978–982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, Y.; Wang, F.; Yang, W.; Zou, Z. MAG-DHA Induces Apoptosis and Autophagy in Breast Cancer Cells via Lipid Peroxidation-Mediated Endoplasmic Reticulum Stress. Preprint 2020. [Google Scholar] [CrossRef]
- Wu, J.; Cho, E.; Giovannucci, E.L.; Rosner, B.A.; Sastry, S.M.; Willett, W.C.; Schaumberg, D.A. Dietary Intakes of Eicosapentaenoic Acid and Docosahexaenoic Acid and Risk of Age-Related Macular Degeneration. Ophthalmology 2017, 124, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Lofqvist, C.A.; Shao, Z.; Sun, Y.; Joyal, J.-S.; Hurst, C.G.; Cui, R.Z.; Evans, L.P.; Tian, K.; SanGiovanni, J.P.; et al. Dietary ω-3 polyunsaturated fatty acids decrease retinal neovascularization by adipose–endoplasmic reticulum stress reduction to increase adiponectin. Am. J. Clin. Nutr. 2015, 101, 879–888. [Google Scholar] [CrossRef]
- Simón, M.V.; Agnolazza, D.L.; German, O.L.; Garelli, A.; Politi, L.E.; Agbaga, M.; Anderson, R.E.; Rotstein, N.P. Synthesis of docosahexaenoic acid from eicosapentaenoic acid in retina neurons protects photoreceptors from oxidative stress. J. Neurochem. 2015, 136, 931–946. [Google Scholar] [CrossRef]
- Prokopiou, E.; Kolovos, P.; Kalogerou, M.; Neokleous, A.; Papagregoriou, G.; Deltas, C.; Malas, S.; Georgiou, T. Therapeutic potential of omega-3 fatty acids supplementation in a mouse model of dry macular degeneration. BMJ Open Ophthalmol. 2017, 1, e000056. [Google Scholar] [CrossRef]
- Behl, T.; Kotwani, A. Omega-3 fatty acids in prevention of diabetic retinopathy. J. Pharm. Pharmacol. 2017, 69, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Suzumura, A.; Terao, R.; Kaneko, H. Protective Effects and Molecular Signaling of n-3 Fatty Acids on Oxidative Stress and Inflammation in Retinal Diseases. Antioxidants 2020, 9, 920. [Google Scholar] [CrossRef] [PubMed]
- FAO. Meeting the Sustainable Development Goals; The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2018; ISBN 978-92-5-130562-1. [Google Scholar]
- Caddy, J.F.; Garibaldi, L. Apparent changes in the trophic composition of world marine harvests: The perspective from the FAO capture database. Ocean Coast. Manag. 2000, 43, 615–655. [Google Scholar] [CrossRef]
- Pauly, D.; Christensen, V.; Dalsgaard, J.; Froese, R.; Torres, F. Fishing Down Marine Food Webs. Science 1998, 279, 860–863. [Google Scholar] [CrossRef]
- Tocher, D. Issues surrounding fish as a source of omega-3 long-chain polyunsaturated fatty acids. Lipid Technol. 2009, 21, 13–16. [Google Scholar] [CrossRef]
- Guschina, I.A.; Harwood, J.L. Algal lipids and effect of the environment on their biochemistry. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 1–24. ISBN 978-0-387-88607-7. [Google Scholar]
- Hixson, S.M.; Arts, M.T. Climate warming is predicted to reduce omega-3, long-chain, polyunsaturated fatty acid production in phytoplankton. Glob. Chang. Biol. 2016, 22, 2744–2755. [Google Scholar] [CrossRef]
- Winter, R.; Dzwolak, W. Exploring the temperature-pressure configurational landscape of biomolecules: From lipid membranes to proteins. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2004, 363, 537–563. [Google Scholar] [CrossRef] [PubMed]
- Renaud, S.M.; Thinh, L.-V.; Lambrinidis, G.; Parry, D.L. Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 2002, 211, 195–214. [Google Scholar] [CrossRef]
- Rousch, J.M.; Bingham, S.E.; Sommerfeld, M.R. Changes in fatty acid profiles of thermo-intolerant and thermo-tolerant marine diatoms during temperature stress. J. Exp. Mar. Biol. Ecol. 2003, 295, 145–156. [Google Scholar] [CrossRef]
- Adarme-Vega, T.C.; Thomas-Hall, S.R.; Schenk, P.M. Towards sustainable sources for omega-3 fatty acids production. Curr. Opin. Biotechnol. 2014, 26, 14–18. [Google Scholar] [CrossRef] [PubMed]
- FAO. The State of World Fisheries and Aquaculture 2020: Sustainability in Action; The State of World Fisheries and Aquaculture (SOFIA); FAO: Rome, Italy, 2020; ISBN 978-92-5-132692-3. [Google Scholar]
- Domergue, F.; Abbadi, A.; Heinz, E. Relief for fish stocks: Oceanic fatty acids in transgenic oilseeds. Trends Plant Sci. 2005, 10, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Robert, S.S. Production of Eicosapentaenoic and Docosahexaenoic Acid-Containing Oils in Transgenic Land Plants for Human and Aquaculture Nutrition. Mar. Biotechnol. 2006, 8, 103–109. [Google Scholar] [CrossRef]
- Khachatourians, G.C.; Hui, Y.H.; Scorza, R.; Nip, W.-K. Transgenic Plants and Crops; CRC Press: Boca Raton, FL, USA, 2002; ISBN 978-0-203-91097-9. [Google Scholar]
- Abbadi, A.; Domergue, F.; Bauer, J.; Napier, J.A.; Welti, R.; Zähringer, U.; Cirpus, P.; Heinza, E. Biosynthesis of Very-Long-Chain Polyunsaturated Fatty Acids in Transgenic Oilseeds: Constraints on Their Accumulation. Plant Cell 2004, 16, 2734–2748. [Google Scholar] [CrossRef]
- Qi, B.; Fraser, T.; Mugford, S.T.; Dobson, G.; Sayanova, O.; Butler, J.; Napier, J.A.; Stobart, A.K.; Lazarus, C.M. Production of very long chain polyunsaturated omega-3 and omega-6 fatty acids in plants. Nat. Biotechnol. 2004, 22, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Truksa, M.; Datla, N.; Vrinten, P.; Bauer, J.; Zank, T.; Cirpus, P.; Heinz, E.; Qiu, X. Stepwise engineering to produce high yields of very long-chain polyunsaturated fatty acids in plants. Nat. Biotechnol. 2005, 23, 1013–1017. [Google Scholar] [CrossRef]
- Ruiz-Lopez, N.; Haslam, R.P.; Napier, J.A.; Sayanova, O. Successful high-level accumulation of fish oil omega-3 long-chain polyunsaturated fatty acids in a transgenic oilseed crop. Plant J. 2014, 77, 198–208. [Google Scholar] [CrossRef]
- Betancor, M.; Sprague, M.; Usher, S.; Sayanova, O.; Campbell, P.; Napier, J.A.; Tocher, D.R. A nutritionally-enhanced oil from transgenic Camelina sativa effectively replaces fish oil as a source of eicosapentaenoic acid for fish. Sci. Rep. 2015, 5, 8104. [Google Scholar] [CrossRef] [PubMed]
- Napier, J.A.; Olsen, R.; Tocher, D.R. Update on GM canola crops as novel sources of omega-3 fish oils. Plant Biotechnol. J. 2019, 17, 703–705. [Google Scholar] [CrossRef]
- Ribeiro, T.G.; Barone, B.; Behrens, J.H. Genetically modified foods and their social representation. Food Res. Int. 2016, 84, 120–127. [Google Scholar] [CrossRef]
- Volkman, J.; Jeffrey, S.; Nichols, P.; Rogers, G.; Garland, C. Fatty acid and lipid composition of 10 species of microalgae used in mariculture. J. Exp. Mar. Biol. Ecol. 1989, 128, 219–240. [Google Scholar] [CrossRef]
- Ötleş, S.; Pire, R. Fatty Acid Composition of Chlorella and Spirulina Microalgae Species. J. Assoc. Off. Agric. Chem. Int. 2001, 84, 1708–1714. [Google Scholar] [CrossRef]
- Leblond, J.D.; Evans, T.J.; Chapman, P.J. The biochemistry of dinoflagellate lipids, with particular reference to the fatty acid and sterol composition of a Karenia brevis bloom. Phycologia 2003, 42, 324–331. [Google Scholar] [CrossRef]
- Pernet, F.; Tremblay, R.; Demers, E.; Roussy, M. Variation of lipid class and fatty acid composition of Chaetoceros muelleri and Isochrysis sp. grown in a semicontinuous system. Aquaculture 2003, 221, 393–406. [Google Scholar] [CrossRef]
- Patil, V.; Källqvist, T.; Olsen, E.; Vogt, G.; Gislerød, H.R. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquac. Int. 2006, 15, 1–9. [Google Scholar] [CrossRef]
- Niccolai, A.; Zittelli, G.C.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae of interest as food source: Biochemical composition and digestibility. Algal Res. 2019, 42, 101617. [Google Scholar] [CrossRef]
- Gladyshev, M.I.; Sushchik, N.N. Long-chain Omega-3 Polyunsaturated Fatty Acids in Natural Ecosystems and the Human Diet: Assumptions and Challenges. Biomolecules 2019, 9, 485. [Google Scholar] [CrossRef] [PubMed]
- Taipale, S.; Peltomaa, E.; Salmi, P. Variation in ω-3 and ω-6 Polyunsaturated Fatty Acids Produced by Different Phytoplankton Taxa at Early and Late Growth Phase. Biomolecules 2020, 10, 559. [Google Scholar] [CrossRef] [PubMed]
- Arts, M.T.; Ackman, R.G.; Holub, B.J. “Essential fatty acids” in aquatic ecosystems: A crucial link between diet and human health and evolution. Can. J. Fish. Aquat. Sci. 2001, 58, 122–137. [Google Scholar] [CrossRef]
- Mendes, A.; Reis, A.; Vasconcelos, R.; Guerra, P.; Da Silva, T.L. Crypthecodinium cohnii with emphasis on DHA production: A review. Environ. Boil. Fishes 2009, 21, 199–214. [Google Scholar] [CrossRef]
- Fan, K.-W.; Jiang, Y.; Faan, Y.-W.; Chen, F. Lipid Characterization of Mangrove Thraustochytrid—Schizochytrium mangrovei. J. Agric. Food Chem. 2007, 55, 2906–2910. [Google Scholar] [CrossRef]
- Leblond, J.D.; Chapman, P.J. Lipid class distribution of highly unsaturated long chain fatty acids in marine dinoflagellates. J. Phycol. 2000, 36, 1103–1108. [Google Scholar] [CrossRef]
- Usup, G.; Hamid, S.Z.; Chiet, P.K.; Wah, C.K.; Ahmad, A. Marked Differences in Fatty Acid Profiles of Some Planktonic and Benthic Marine Dinoflagellates from Malaysian Waters. Phycologia 2008, 47, 105–111. [Google Scholar] [CrossRef]
- Da Costa, F.; Le Grand, F.; Quéré, C.; Bougaran, G.; Cadoret, J.P.; Robert, R.; Soudant, P. Effects of growth phase and nitrogen limitation on biochemical composition of two strains of Tisochrysis lutea. Algal Res. 2017, 27, 177–189. [Google Scholar] [CrossRef]
- Chang, K.J.L.; Nichols, C.M.; Blackburn, S.I.; Dunstan, G.A.; Koutoulis, A.; Nichols, P.D. Comparison of Thraustochytrids Aurantiochytrium sp., Schizochytrium sp., Thraustochytrium sp., and Ulkenia sp. for Production of Biodiesel, Long-Chain Omega-3 Oils, and Exopolysaccharide. Mar. Biotechnol. 2014, 16, 396–411. [Google Scholar] [CrossRef] [PubMed]
- Moreno, V.J.; De Moreno, J.E.A.; Brenner, R.R. Biosynthesis of unsaturated fatty acids in the Diatom Phaeodactylum tricornutum. Lipids 1979, 14, 15–19. [Google Scholar] [CrossRef]
- Gillan, F.; McFadden, G.; Wetherbee, R.; Johns, R. Sterols and fatty acids of an antarctic sea ice diatom, Stauroneis amphioxys. Phytochemistry 1981, 20, 1935–1937. [Google Scholar] [CrossRef]
- Arao, T.; Sakaki, T.; Yamada, M. Biosynthesis of polyunsaturated lipids in the diatom, Phaeodactylum tricornutum. Phytochemistry 1994, 36, 629–635. [Google Scholar] [CrossRef]
- Tonon, T.; Harvey, D.; Larson, T.R.; Graham, I.A. Long chain polyunsaturated fatty acid production and partitioning to triacylglycerols in four microalgae. Phytochemistry 2002, 61, 15–24. [Google Scholar] [CrossRef]
- Liang, Y.; Maeda, Y.; Yoshino, T.; Matsumoto, M.; Tanaka, T. Profiling of fatty acid methyl esters from the oleaginous diatom Fistulifera sp. strain JPCC DA0580 under nutrition-sufficient and -deficient conditions. Environ. Boil. Fishes 2014, 26, 2295–2302. [Google Scholar] [CrossRef]
- Yao, L.; Gerde, J.A.; Lee, S.-L.; Wang, T.; Harrata, K.A. Microalgae Lipid Characterization. J. Agric. Food Chem. 2015, 63, 1773–1787. [Google Scholar] [CrossRef] [PubMed]
- Zulu, N.N.; Zienkiewicz, K.; Vollheyde, K.; Feussner, I. Current trends to comprehend lipid metabolism in diatoms. Prog. Lipid Res. 2018, 70, 1–16. [Google Scholar] [CrossRef]
- Adams, C.; Godfrey, V.; Wahlen, B.; Seefeldt, L.; Bugbee, B. Understanding precision nitrogen stress to optimize the growth and lipid content tradeoff in oleaginous green microalgae. Bioresour. Technol. 2013, 131, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.K.; McHugh, E.; Hayes, J.; Moane, S.; Walsh, D.; Murray, P. Effect of various stress-regulatory factors on biomass and lipid production in microalga Haematococcus pluvialis. Bioresour. Technol. 2013, 128, 118–124. [Google Scholar] [CrossRef]
- Parrish, C.C. Lipids in Marine Ecosystems. Int. Sch. Res. Not. Oceanogr. 2013, 2013, 604045. [Google Scholar] [CrossRef]
- Watkins, S.M.; German, B.J. Unsaturated fatty acids. In Food Lipid: Chemistry, Nutrition, and Biotechnology; Food Science and Technology; CRC Press/Taylor & Francis Group: Boca Raton, CA, USA, 2008; pp. 513–537. [Google Scholar]
- Khozin-Goldberg, I. Lipid metabolism in microalgae. In The Physiology of Microalgae; Borowitzka, M.A., Beardall, J., Raven, J.A., Eds.; Developments in Applied Phycology; Springer International Publishing: Cham, Switzerland, 2016; pp. 413–484. ISBN 978-3-319-24945-2. [Google Scholar]
- Harwood, J.L. Fatty acid biosynthesis. In Plant Lipids: Biology, Utilisation and Manipulation; John Wiley & Sons: Cardiff, Wales, UK, 2009; pp. 27–66. ISBN 978-1-4051-4812-2. [Google Scholar]
- Bell, M.V.; Tocher, D.R. Biosynthesis of polyunsaturated fatty acids in aquatic ecosystems: General pathways and new direc-tions. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 211–236. ISBN 978-0-387-88607-7. [Google Scholar]
- Vieler, A.; Wu, G.; Tsai, C.-H.; Bullard, B.; Cornish, A.J.; Harvey, C.; Reca, I.-B.; Thornburg, C.; Achawanantakun, R.; Buehl, C.J.; et al. Genome, Functional Gene Annotation, and Nuclear Transformation of the Heterokont Oleaginous Alga Nannochloropsis oceanica CCMP1779. PLoS Genet. 2012, 8, e1003064. [Google Scholar] [CrossRef]
- Hoffmeister, M.; Piotrowski, M.; Nowitzki, U.; Martin, W. Mitochondrial trans-2-Enoyl-CoA Reductase of Wax Ester Fermentation from Euglena gracilis Defines a New Family of Enzymes Involved in Lipid Synthesis. J. Biol. Chem. 2005, 280, 4329–4338. [Google Scholar] [CrossRef] [PubMed]
- Los, D.A.; Murata, N. Structure and expression of fatty acid desaturases. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1998, 1394, 3–15. [Google Scholar] [CrossRef]
- Li-Beisson, Y.; Beisson, F.; Riekhof, W. Metabolism of acyl-lipids in Chlamydomonas reinhardtii. Plant J. 2015, 82, 504–522. [Google Scholar] [CrossRef]
- Meesapyodsuk, D.; Qiu, X. The Front-end Desaturase: Structure, Function, Evolution and Biotechnological Use. Lipids 2012, 47, 227–237. [Google Scholar] [CrossRef]
- Remize, M.; Planchon, F.; Loh, A.N.; Le Grand, F.; Bideau, A.; Le Goic, N.; Fleury, E.; Miner, P.; Corvaisier, R.; Volety, A.; et al. Study of Synthesis Pathways of the Essential Polyunsaturated Fatty Acid 20:5n-3 in the Diatom Chaetoceros Muelleri Using 13C-Isotope Labeling. Biomolecules 2020, 10, 797. [Google Scholar] [CrossRef]
- Arao, T.; Yamada, M. Biosynthesis of polyunsaturated fatty acids in the marine diatom, Phaeodactylum tricornutum. Phytochemistry 1994, 35, 1177–1181. [Google Scholar] [CrossRef]
- Domergue, F.; Lerchl, J.; Zähringer, U.; Heinz, E. Cloning and functional characterization of Phaeodactylum tricornutum front-end desaturases involved in eicosapentaenoic acid biosynthesis. Eur. J. Biochem. 2002, 269, 4105–4113. [Google Scholar] [CrossRef] [PubMed]
- Domergue, F.; Spiekermann, P.; Lerchl, J.; Beckmann, C.; Kilian, O.; Kroth, P.G.; Boland, W.; Zähringer, U.; Heinz, E. New Insight into Phaeodactylum tricornutum Fatty Acid Metabolism. Cloning and Functional Characterization of Plastidial and Microsomal Δ12-Fatty Acid Desaturases. Plant Physiol. 2003, 131, 1648–1660. [Google Scholar] [CrossRef]
- Gurr, M.I.; Harwood, J.L. Fatty acid structure and metabolism. In Lipid Biochemistry: An Introduction; Gurr, M.I., Harwood, J.L., Eds.; Springer: Boston, MA, USA, 1991; pp. 23–118. ISBN 978-1-4615-3862-2. [Google Scholar]
- Buist, P.H. Fatty acid desaturases: Selecting the dehydrogenation channel. Nat. Prod. Rep. 2004, 21, 249–262. [Google Scholar] [CrossRef]
- Ratledge, C. Fatty acid biosynthesis in microorganisms being used for Single Cell Oil production. Biochimie 2004, 86, 807–815. [Google Scholar] [CrossRef]
- Armenta, R.E.; Valentine, M.C. Single-Cell Oils as a Source of Omega-3 Fatty Acids: An Overview of Recent Advances. J. Am. Oil Chem. Soc. 2012, 90, 167–182. [Google Scholar] [CrossRef]
- Wallis, J.G.; Watts, J.L.; Browse, J. Polyunsaturated fatty acid synthesis: What will they think of next? Trends Biochem. Sci 2002, 27, 467–473. [Google Scholar] [CrossRef]
- Metz, J.G.; Roessler, P.; Facciotti, D.; Levering, C.; Dittrich, F.; Lassner, M.; Valentine, R.; Lardizabal, K.; Domergue, F.; Yamada, A.; et al. Production of Polyunsaturated Fatty Acids by Polyketide Synthases in Both Prokaryotes and Eukaryotes. Science 2001, 293, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X. Biosynthesis of docosahexaenoic acid (DHA, 22:6-4, 7,10,13,16,19): Two distinct pathways. Prostaglandins Leukot. Essent. Fat. Acids 2003, 68, 181–186. [Google Scholar] [CrossRef]
- Hauvermale, A.; Kuner, J.; Rosenzweig, B.; Guerra, D.; Diltz, S.; Metz, J.G. Fatty acid production in Schizochytrium sp.: Involvement of a polyunsaturated fatty acid synthase and a type I fatty acid synthase. Lipids 2006, 41, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Uttaro, A.D. Biosynthesis of polyunsaturated fatty acids in lower eukaryotes. Int. Union Biochem. Mol. Biol. Life 2006, 58, 563–571. [Google Scholar] [CrossRef]
- Murray, S.A.; Garby, T.; Hoppenrath, M.; Neilan, B.A. Genetic Diversity, Morphological Uniformity and Polyketide Production in Dinoflagellates (Amphidinium, Dinoflagellata). PLoS ONE 2012, 7, e38253. [Google Scholar] [CrossRef]
- Kimura, K.; Okuda, S.; Nakayama, K.; Shikata, T.; Takahashi, F.; Yamaguchi, H.; Skamoto, S.; Yamaguchi, M.; Tomaru, Y. RNA Sequencing Revealed Numerous Polyketide Synthase Genes in the Harmful Dinoflagellate Karenia mikimotoi. PLoS ONE 2015, 10, e0142731. [Google Scholar] [CrossRef] [PubMed]
- Remize, M.; Planchon, F.; Loh, A.N.; Le Grand, F.; Lambert, C.; Bideau, A.; Bidault, A.; Corvaisier, R.; Volety, A.; Soudant, P. Identification of Polyunsaturated Fatty Acids Synthesis Pathways in the Toxic Dinophyte Alexandrium minutum Using 13C-Labelling. Biomolecules 2020, 10, 1428. [Google Scholar] [CrossRef] [PubMed]
- John, U.; Beszteri, B.; Derelle, E.; Van De Peer, Y.; Read, B.; Moreau, H.; Cembella, A. Novel Insights into Evolution of Protistan Polyketide Synthases through Phylogenomic Analysis. Protist 2008, 159, 21–30. [Google Scholar] [CrossRef]
- John, U.; Beszteri, S.; Glöckner, G.; Singh, R.; Medlin, L.; Cembella, A.D. Genomic characterisation of the ichthyotoxic prymnesiophyte Chrysochromulina polylepis,and the expression of polyketide synthase genes in synchronized cultures. Eur. J. Phycol. 2010, 45, 215–229. [Google Scholar] [CrossRef]
- Shen, B. Polyketide biosynthesis beyond the type I, II and III polyketide synthase paradigms. Curr. Opin. Chem. Biol. 2003, 7, 285–295. [Google Scholar] [CrossRef]
- Freitag, M.; Beszteri, S.; Vogel, H.; John, U. Effects of physiological shock treatments on toxicity and polyketide synthase gene expression in Prymnesium parvum (Prymnesiophyceae). Eur. J. Phycol. 2011, 46, 193–201. [Google Scholar] [CrossRef]
- Eichholz, K.; Beszteri, B.; John, U. Putative Monofunctional Type I Polyketide Synthase Units: A Dinoflagellate-Specific Feature? PLoS ONE 2012, 7, e48624. [Google Scholar] [CrossRef]
- Kohli, G.S.; John, U.; Van Dolah, F.M.; Murray, S.A. Evolutionary distinctiveness of fatty acid and polyketide synthesis in eukaryotes. ISME J. 2016, 10, 1877–1890. [Google Scholar] [CrossRef]
- Monroe, E.A.; van Dolah, F.M. The Toxic Dinoflagellate Karenia brevis Encodes Novel Type I-like Polyketide Synthases Con-taining Discrete Catalytic Domains. Protist 2008, 159, 471–482. [Google Scholar] [CrossRef]
- Jenke-Kodama, H.; Sandmann, A.; Müller, R.; Dittmann, E. Evolutionary Implications of Bacterial Polyketide Synthases. Mol. Biol. Evol. 2005, 22, 2027–2039. [Google Scholar] [CrossRef] [PubMed]
- Cock, J.M.; Sterck, L.; Rouzé, P.; Scornet, D.; Allen, A.E.; Amoutzias, G.D.; Anthouard, V.; Artiguenave, F.; Aury, J.-M.; Badger, J.H.; et al. The Ectocarpus genome and the independent evolution of multicellularity in brown algae. Nat. Cell Biol. 2010, 465, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Qiao, W.; Yu, X.; Ji, X.; Huang, H.; Collier, J.L.; Liu, L. Reconstruction and analysis of the genome-scale metabolic model of schizochytrium limacinum SR21 for docosahexaenoic acid production. BMC Genom. 2015, 16, 799. [Google Scholar] [CrossRef]
- Zhang, K.; Li, H.; Chen, W.; Zhao, M.; Cui, H.; Min, Q.; Wang, H.; Chen, S.; Li, D. Regulation of the Docosapentaenoic Acid/Docosahexaenoic Acid Ratio (DPA/DHA Ratio) in Schizochytrium limacinum B4D1. Appl. Biochem. Biotechnol. 2017, 182, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Leblond, J.D.; Lasiter, A.D. Mono- and digalactosyldiacylglycerol composition of dinoflagellates. II.Lepidodinium chlorophorum, Karenia brevis, and Kryptoperidinium foliaceum, three dinoflagellates with aberrant plastids. Eur. J. Phycol. 2009, 44, 199–205. [Google Scholar] [CrossRef][Green Version]
- Courchesne, N.M.D.; Parisien, A.; Wang, B.; Lan, C.Q. Enhancement of lipid production using biochemical, genetic and transcription factor engineering approaches. J. Biotechnol. 2009, 141, 31–41. [Google Scholar] [CrossRef]
- Gong, Y.; Wan, X.; Jiang, M.; Hu, C.; Hu, H.; Huang, F. Metabolic engineering of microorganisms to produce omega-3 very long-chain polyunsaturated fatty acids. Prog. Lipid Res. 2014, 56, 19–35. [Google Scholar] [CrossRef]
- Mühlroth, A.; Li, K.; Røkke, G.; Winge, P.; Olsen, Y.; Hohmann-Marriott, M.F.; Vadstein, O.; Bones, A.M. Pathways of Lipid Metabolism in Marine Algae, Co-Expression Network, Bottlenecks and Candidate Genes for Enhanced Production of EPA and DHA in Species of Chromista. Mar. Drugs 2013, 11, 4662–4697. [Google Scholar] [CrossRef]
- Cui, J.; Diao, J.; Sun, T.; Shi, M.; Liu, L.; Wang, F.; Chen, L.; Zhang, W. 13C Metabolic Flux Analysis of Enhanced Lipid Accumulation Modulated by Ethanolamine in Crypthecodinium cohnii. Front. Microbiol. 2018, 9, 956. [Google Scholar] [CrossRef]
- Menzel, R.; Ngosong, C.; Ruess, L. Isotopologue profiling enables insights into dietary routing and metabolism of trophic biomarker fatty acids. Chemoecology 2017, 27, 101–114. [Google Scholar] [CrossRef]
- Wei, X.; Shi, B.; Koo, I.; Yin, X.; Lorkiewicz, P.; Suhail, H.; Rattan, R.; Giri, S.; McClain, C.J.; Zhang, X. Analysis of stable isotope assisted metabolomics data acquired by GC-MS. Anal. Chim. Acta 2017, 980, 25–32. [Google Scholar] [CrossRef]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Levasseur, W.; Perré, P.; Pozzobon, V. A review of high value-added molecules production by microalgae in light of the classification. Biotechnol. Adv. 2020, 41, 107545. [Google Scholar] [CrossRef]
- Pan-Utai, W.; Iamtham, S. Extraction, purification and antioxidant activity of phycobiliprotein from Arthrospira platensis. Process. Biochem. 2019, 82, 189–198. [Google Scholar] [CrossRef]
- Kulkarni, S.; Nikolov, Z. Process for selective extraction of pigments and functional proteins from Chlorella vulgaris. Algal Res. 2018, 35, 185–193. [Google Scholar] [CrossRef]
- Castejón, N.; Señoráns, F.J. Simultaneous extraction and fractionation of omega-3 acylglycerols and glycolipids from wet microalgal biomass of Nannochloropsis gaditana using pressurized liquids. Algal Res. 2019, 37, 74–82. [Google Scholar] [CrossRef]
- Lu, K.; Zhao, X.; Ho, S.-H.; Ma, R.; Xie, Y.; Chen, J. Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa. Mar. Drugs 2019, 17, 454. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Goswami, S. Microalgae—A green multi-product biorefinery for future industrial prospects. Biocatal. Agric. Biotechnol. 2020, 25, 101580. [Google Scholar] [CrossRef]
- Tang, D.Y.Y.; Yew, G.Y.; Koyande, A.K.; Chew, K.W.; Vo, D.-V.N.; Show, P.L. Green technology for the industrial production of biofuels and bioproducts from microalgae: A review. Environ. Chem. Lett. 2020, 18, 1967–1985. [Google Scholar] [CrossRef]
- Galasso, C.; Gentile, A.; Orefice, I.; Ianora, A.; Bruno, A.; Noonan, D.M.; Sansone, C.; Albini, A.; Brunet, C. Microalgal Derivatives as Potential Nutraceutical and Food Supplements for Human Health: A Focus on Cancer Prevention and Interception. Nutrients 2019, 11, 1226. [Google Scholar] [CrossRef]
- Allmicroalgae Natural Products Allma by Allmicroalgae. Available online: https://www.allmicroalgae.com/en/food/ (accessed on 7 December 2020).
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Krupanidhi, S.; Sanjeevi, C.B. Omega-3 Fatty Acids for Nutrition and Medicine: Considering Microalgae Oil as a Vegetarian Source of EPA and DHA. Curr. Diabetes Rev. 2007, 3, 198–203. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Algal physiology and large-scale outdoor cultures of microalgae. In The Physiology of Microalgae; Boro-witzka, M.A., Beardall, J., Raven, J.A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 601–652. ISBN 978-3-319-24943-8. [Google Scholar]
- Oren, A. A hundred years of Dunaliella research: 1905–2005. Saline Syst. 2005, 1, 2. [Google Scholar] [CrossRef]
- Beuzenberg, V.; Smith, K.; Packer, M. Isolation and characterisation of halo-tolerant Dunaliella strains from Lake Grassmere/Kapara Te Hau, New Zealand. N. Zealand J. Bot. 2014, 52, 136–152. [Google Scholar] [CrossRef]
- Babuskin, S.; Krishnan, K.R.; Saravana, P.A.; Sivarajan, M.; Sukumar, M. Functional Foods Enriched with Marine Microalga Nannochloropsis oculata as a Source of W-3 Fatty Acids. Food Technol. Biotechnol. 2014, 52, 292–299. [Google Scholar]
- Robertson, R.C.; Mateo, M.R.G.; O’Grady, M.N.; Guihéneuf, F.; Stengel, D.B.; Ross, R.P.; Fitzgerald, G.F.; Kerry, J.P.; Stanton, C. An assessment of the techno-functional and sensory properties of yoghurt fortified with a lipid extract from the microalga Pavlova lutheri. Innov. Food Sci. Emerg. Technol. 2016, 37, 237–246. [Google Scholar] [CrossRef]
- Lane, K.E.; Li, W.; Smith, C.A.; Derbyshire, E. The bioavailability of an omega-3-rich algal oil is improved by nanoemulsion technology using yogurt as a food vehicle. Int. J. Food Sci. Technol. 2014, 49, 1264–1271. [Google Scholar] [CrossRef]
- Geppert, J.; Kraft, V.; Demmelmair, H.; Koletzko, B. Docosahexaenoic acid supplementation in vegetarians effectively increases omega-3 index: A randomized trial. Lipids 2005, 40, 807–814. [Google Scholar] [CrossRef]
- Li, K.; Sinclair, A.J.; Zhao, F.; Li, D. Uncommon Fatty Acids and Cardiometabolic Health. Nutrients 2018, 10, 1559. [Google Scholar] [CrossRef]
- Wakimoto, T.; Kondo, H.; Nii, H.; Kimura, K.; Egami, Y.; Oka, Y.; Yoshida, M.; Kida, E.; Ye, Y.; Akahoshi, S.; et al. Furan fatty acid as an anti-inflammatory component from the green-lipped mussel Perna canaliculus. Proc. Natl. Acad. Sci. USA 2011, 108, 17533–17537. [Google Scholar] [CrossRef] [PubMed]
- Packer, M.A.; Harris, G.C.; Adams, S.L. Food and feed applications of algae. In Algae Biotechnology; Bux, F., Chisti, Y., Eds.; Green Energy and Technology; Springer International Publishing: Cham, Switzerland, 2016; pp. 217–247. ISBN 978-3-319-12333-2. [Google Scholar]
- Jordan, M.A.; Wilson, L. Microtubules as a Target for Anticancer Drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef]
- Ba, F.; Ursu, A.V.; Laroche, C.; Djelveh, G. Haematococcus pluvialis soluble proteins: Extraction, characterization, concentration/fractionation and emulsifying properties. Bioresour. Technol. 2016, 200, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Caporgno, M.P.; Haberkorn, I.; Böcker, L.; Mathys, A. Cultivation of Chlorella protothecoides under different growth modes and its utilisation in oil/water emulsions. Bioresour. Technol. 2019, 288, 121476. [Google Scholar] [CrossRef] [PubMed]
- Damodaran, S. Protein Stabilization of Emulsions and Foams. J. Food Sci. 2006, 70, R54–R66. [Google Scholar] [CrossRef]
- Law, S.Q.; Mettu, S.; AshokKumar, M.; Scales, P.J.; Martin, G.J. Emulsifying properties of ruptured microalgae cells: Barriers to lipid extraction or promising biosurfactants? Colloids Surfaces B Biointerfaces 2018, 170, 438–446. [Google Scholar] [CrossRef]
- Cabra, V.; Arreguín, R. Emulsifying Properties of Proteins. Boletín Soc. Química México 2008, 2, 80–89. [Google Scholar]
- Hasenhuettl, G.L. Analysis of food emulsifiers. In Food Emulsifiers and Their Applications; Hasenhuettl, G.L., Hartel, R.W., Eds.; Springer: New York, NY, USA, 2008; pp. 39–62. ISBN 978–0-387-75283-9. [Google Scholar]
- Mnif, I.; Ghribi, D. High Molecular Weight Bioemulsifiers, Main Properties and Potential Environmental and Biomedical Applications. World J. Microbiol. Biotechnol. 2015, 16, 691–706. [Google Scholar] [CrossRef]
- Lu, F.S.H.; Nielsen, N.S.; Baron, C.P.; Jacobsen, C. Marine phospholipids: The current understanding of their oxidation mechanisms and potential uses for food fortification. Crit. Rev. Food Sci. Nutr. 2017, 57, 2057–2070. [Google Scholar] [CrossRef]
- Pietrowski, B.N.; Tahergorabi, R.; Matak, K.E.; Tou, J.C.; Jaczynski, J. Chemical properties of surimi seafood nutrified with ω-3 rich oils. Food Chem. 2011, 129, 912–919. [Google Scholar] [CrossRef]
- Sedoski, H.D. Sensory Evaluation and Quality Indicators of Nutritionally Enhanced Egg Product with Omega-3 Rich Oils. Master’s Thesis, West Virginia University Libraries, Morgantown, WV, USA, 2011. [Google Scholar]
- Kassis, N.M.; Gigliotti, J.C.; Beamer, S.K.; Tou, J.C.; Jaczynski, J. Characterization of lipids and antioxidant capacity of novel nutraceutical egg products developed with omega-3-rich oils. J. Sci. Food Agric. 2011, 92, 66–73. [Google Scholar] [CrossRef] [PubMed]
- El-Baz, F.K.; Abdo, S.M.; Hussein, A.M.S. Microalgae Dunaliella salina for Use as Food Supplement to Improve Pasta Quality. Int. J. Pharm. Sci. Rev. Res. 2017, 46, 45–51. [Google Scholar]
- Pina-Pérez, M.C.; Rivas, A.; Martínez, A.; Rodrigo, D. Antimicrobial potential of macro and microalgae against pathogenic and spoilage microorganisms in food. Food Chem. 2017, 235, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Sidari, R.; Tofalo, R. A Comprehensive Overview on Microalgal-Fortified/Based Food and Beverages. Food Rev. Int. 2019, 35, 778–805. [Google Scholar] [CrossRef]
- Gouveia, L.; Coutinho, C.; Mendonça, E.; Batista, A.P.; Sousa, I.; Bandarra, N.M.; Raymundo, A. Functional biscuits with PUFA-ω3 from Isochrysis galbana. J. Sci. Food Agric. 2008, 88, 891–896. [Google Scholar] [CrossRef]
- Gouveia, L.; Batista, A.P.; Raymundo, A.; Bandarra, N. Spirulina maxima and Diacronema vlkianummicroalgae in vegetable gelled desserts. Nutr. Food Sci. 2008, 38, 492–501. [Google Scholar] [CrossRef]
- Kaur, P. Microalgae as nutraceutical for achieving sustainable food solution in future. In Microbial Biotechnology: Basic Research and Applications; Singh, J., Vyas, A., Wang, S., Prasad, R., Eds.; Environmental and Microbial Biotechnology; Springer: Singapore, 2020; pp. 91–125. ISBN 9789811528163. [Google Scholar]
- Raja, R.; Coelho, A.; Hemaiswarya, S.; Kumar, P.; Carvalho, I.S.; Alagarsamy, A. Applications of microalgal paste and powder as food and feed: An update using text mining tool. Beni Suef Univ. J. Basic Appl. Sci. 2018, 7, 740–747. [Google Scholar] [CrossRef]
- Griffiths, M.; Harrison, S.T.L.; Smit, M.; Maharajh, D. Major commercial products from micro and macroalgae. In Algae Biotechnology; Bux, F., Chisti, Y., Eds.; Green Energy and Technology; Springer International Publishing: Cham, Switzerland, 2016; pp. 269–300. ISBN 978-3-319-12333-2. [Google Scholar]
- Reboleira, J.; Freitas, R.; Pinteus, S.; Silva, J.; Alves, C.; Pedrosa, R.; Bernardino, S. Spirulina. In Nonvitamin and Nonmineral Nutritional Supplements; Elsevier: Amsterdam, The Netherlands, 2019; pp. 409–413. [Google Scholar]
- Matos, J.; Cardoso, C.; Bandarra, N.M.; Afonso, C. Microalgae as healthy ingredients for functional food: A review. Food Funct. 2017, 8, 2672–2685. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Reboleira, J.; Pedrosa, R.; Bernardino, S. Chlorella. In Nonvitamin and Nonmineral Nutritional Supplements; Elsevier: Amsterdam, The Netherlands, 2019; pp. 187–193. [Google Scholar]
- Rodriguez-Garcia, I.; Guil-Guerrero, J.L. Evaluation of the antioxidant activity of three microalgal species for use as dietary supplements and in the preservation of foods. Food Chem. 2008, 108, 1023–1026. [Google Scholar] [CrossRef]
- Vazhappilly, R.; Chen, F. Eicosapentaenoic acid and docosahexaenoic acid production potential of microalgae and their heterotrophic growth. J. Am. Oil Chem. Soc. 1998, 75, 393–397. [Google Scholar] [CrossRef]
- Seto, A.; Wang, H.L.; Hesseltine, C.W. Culture conditions affect eicosapentaenoic acid content of Chlorella minutissima. J. Am. Oil Chem. Soc. 1984, 61, 892–894. [Google Scholar] [CrossRef]
- European Parliament. European Council Commission Decision of 21 October 2009 Concerning the Extension of Uses of Algal Oil from the Microalgae Ulkenia Sp. as a Novel Food Ingredient under Regulation (EC) No 258/97 of the European Parliament and of the Council (Notified under Document C(2009) 7932). Off. J. Eur. Union 2009, L 278/54, 2. [Google Scholar]
- Fu, W.; Nelson, D.; Yi, Z.; Xu, M.; Khraiwesh, B.; Jijakli, K.; Chaiboonchoe, A.; Alzahmi, A.; Al-Khairy, D.; Brynjolfsson, S.; et al. Bioactive compounds from microalgae: Current development and prospects. In Studies in Natural Products Chemistry; Rahman, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 54, pp. 199–225. [Google Scholar]
- Torres-Tiji, Y.; Fields, F.J.; Mayfield, S.P. Microalgae as a future food source. Biotechnol. Adv. 2020, 41, 107536. [Google Scholar] [CrossRef]
- CEVA. Edible Seaweed and Microalgae—Regulatory Status in France and Europe—2019 Update; Centre d’Etude et de Valorisation des Algues: Saint Brieuc, France, 2020; p. 15. [Google Scholar]
- Nuzzo, D.; Contardi, M.; Kossyvaki, D.; Picone, P.; Cristaldi, L.; Galizzi, G.; Bosco, G.; Scoglio, S.; Athanassiou, A.; Carlo, M.D. Heat-Resistant Aphanizomenon Flos-Aquae (AFA) Extract (Klamin®) as a Functional Ingredient in Food Strategy for Preven-tion of Oxidative Stress. Oxidative Med. and Cell. Longev. 2019, 9481390. [Google Scholar]
- Niccolai, A.; Bažec, K.; Rodolfi, L.; Biondi, N.; Zlatić, E.; Jamnik, P.; Tredici, M.R. Lactic Acid Fermentation of Arthrospira platensis (Spirulina) in a Vegetal Soybean Drink for Developing New Functional Lactose-Free Beverages. Front. Microbiol. 2020, 11, 560684. [Google Scholar] [CrossRef] [PubMed]
- Darwish, R.; Gedi, M.A.; Akepach, P.; Assaye, H.; Zaky, A.S.; Gray, D.A. Chlamydomonas reinhardtii Is a Potential Food Supplement with the Capacity to Outperform Chlorella and Spirulina. Appl. Sci. 2020, 17, 6736. [Google Scholar] [CrossRef]
- Kawano, T.; Naito, J.; Nishioka, M.; Nishida, N.; Takahashi, M.; Kashiwagi, S.; Sugino, T.; Watanabe, Y. Effect of Food Con-taining Paramylon Derived from Euglena Gracilis EOD-1 on Fatigue in Healthy Adults: A Randomized, Double-Blind, Pla-cebo-Controlled, Parallel-Group Trial. Nutrients 2020, 15, 3098. [Google Scholar] [CrossRef]
- Ruiz-Domínguez, M.C.; Espinosa, C.; Paredes, A.; Palma, J.; Jaime, C.; Vílchez, C.; Cerezal, P. Determining the Potential of Haematococcus pluvialis Oleoresin as a Rich Source of Antioxidants. Molecules 2019, 24, 4073. [Google Scholar] [CrossRef]
- Xia, S.; Wang, K.; Wan, L.; Li, A.; Hu, Q.; Zhang, C. Production, Characterization, and Antioxidant Activity of Fucoxanthin from the Marine Diatom Odontella aurita. Mar. Drugs 2013, 11, 2667–2681. [Google Scholar] [CrossRef] [PubMed]
- Lafarga, T.; Acién-Fernández, F.G.; Castellari, M.; Villaró, S.; Bobo, G.; Aguiló-Aguayo, I. Effect of microalgae incorporation on the physicochemical, nutritional, and sensorial properties of an innovative broccoli soup. Food Sci. Technol. 2019, 111, 167–174. [Google Scholar] [CrossRef]
- Matos, Â.P.; Feller, R.; Moecke, E.H.S.; De Oliveira, J.V.; Junior, A.F.; Derner, R.B.; Sant’Anna, E.S. Chemical Characterization of Six Microalgae with Potential Utility for Food Application. J. Am. Oil Chem. Soc. 2016, 93, 963–972. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Raja, R.; Kumar, R.R.; Ganesan, V.; Anbazhagan, C. Microalgae: A sustainable feed source for aquaculture. World J. Microbiol. Biotechnol. 2010, 27, 1737–1746. [Google Scholar] [CrossRef]
- Camacho, F.; Macedo, A.; Malcata, F. Potential Industrial Applications and Commercialization of Microalgae in the Functional Food and Feed Industries: A Short Review. Mar. Drugs 2019, 17, 312. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.R.; Lutzu, G.A.; Alam, A.; Sarker, P.; Chowdhury, M.A.K.; Parsaeimehr, A.; Liang, Y.; Daroch, M. Microalgae in aquafeeds for a sustainable aquaculture industry. Environ. Boil. Fishes 2018, 30, 197–213. [Google Scholar] [CrossRef]
- Martínez-Fernández, E.; Southgate, P.C. Use of tropical microalgae as food for larvae of the black-lip pearl oyster Pinctada margaritifera. Aquaculture 2007, 263, 220–226. [Google Scholar] [CrossRef]
- Breteler, W.C.M.K.; Schogt, N.; Baas, M.; Schouten, S.; Kraay, G.W. Trophic upgrading of food quality by protozoans enhancing copepod growth: Role of essential lipids. Mar. Biol. 1999, 135, 191–198. [Google Scholar] [CrossRef]
- Bec, A.; Martin-Creuzburg, D.; Von Elert, E. Trophic upgrading of autotrophic picoplankton by the heterotrophic nanoflagellate Paraphysomonas sp. Limnol. Oceanogr. 2006, 51, 1699–1707. [Google Scholar] [CrossRef]
- Chu, F.; Lund, E.; Podbesek, J. Quantitative significance of n-3 essential fatty acid contribution by heterotrophic protists in marine pelagic food webs. Mar. Ecol. Prog. Ser. 2008, 354, 85–95. [Google Scholar] [CrossRef][Green Version]
- Ganuza, E.; Benítez-Santana, T.; Atalah, E.; Vega-Orellana, O.; Ganga, R.; Izquierdo, M. Crypthecodinium cohnii and Schizochytrium sp. as potential substitutes to fisheries-derived oils from seabream (Sparus aurata) microdiets. Aquaculture 2008, 277, 109–116. [Google Scholar] [CrossRef]
- Lubzens, E. Raising rotifers for use in aquaculture. Hydrobiologia 1987, 147, 245–255. [Google Scholar] [CrossRef]
- Ferreira, M.; Maseda, A.; Fabregas, J.; Otero, A. Enriching Rotifers with “Premium” Microalgae Isochrysis aff. galbana Clone T-ISO. Aquaculture 2008, 279, 126–130. [Google Scholar] [CrossRef]
- Ferreira, M.; Coutinho, P.; Seixas, P.; Fábregas, J.; Otero, A. Enriching Rotifers with “Premium” Microalgae. Nannochloropsis gaditana. Mar. Biotechnol. 2009, 11, 585–595. [Google Scholar] [CrossRef]
- Castillo, C.E.-D.; Gapasin, R.S.; Leaño, E.M. Enrichment potential of HUFA-rich thraustochytrid Schizochytrium mangrovei for the rotifer Brachionus plicatilis. Aquaculture 2009, 293, 57–61. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Kovač, D.J.; Simeunović, J.B.; Babić, O.B.; Mišan, A.Č.; Milovanović, I.L. Algae in Food and Feed. Food Feed Res. 2013, 11, 21–32. [Google Scholar]
- Alves, S.P.; Mendonça, S.H.; Silva, J.L.; Bessa, R.J.B. Nannochloropsis oceanica, a novel natural source of rumen-protected eicosapentaenoic acid (EPA) for ruminants. Sci. Rep. 2018, 8, 10269. [Google Scholar] [CrossRef] [PubMed]
- Beynen, A.C. Microalgae in Petfood. Creat. Companion 2019, 40, 7. [Google Scholar]
- Souza, C.M.M.; De Lima, D.C.; Bastos, T.S.; De Oliveira, S.G.; Beirão, B.C.B.; Félix, A.P. Microalgae Schizochytrium sp. as a source of docosahexaenoic acid (DHA): Effects on diet digestibility, oxidation and palatability and on immunity and inflammatory indices in dogs. Anim. Sci. J. 2019, 90, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Hadley, K.; Bauer, J.; Milgram, N. The oil-rich alga Schizochytrium sp. as a dietary source of docosahexaenoic acid improves shape discrimination learning associated with visual processing in a canine model of senescence. Prostaglandins Leukot. Essent. Fat. Acids 2017, 118, 10–18. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dahms, I.; Bailey-Hall, E.; Sylvester, E.; Parenteau, A.; Yu, S.; Karagiannis, A.; Roos, F.; Wilson, J. Safety of a novel feed ingredient, Algal Oil containing EPA and DHA, in a gestation-lactation-growth feeding study in Beagle dogs. PLoS ONE 2019, 14, e0217794. [Google Scholar] [CrossRef] [PubMed]
- Allmicroalgae Natural Products Allvitae by Allmicroalgae. Available online: https://www.allmicroalgae.com/en/feed-2/ (accessed on 7 December 2020).
- Ao, T.; Macalintal, L.M.; Paul, M.A.; Pescatore, A.J.; Cantor, A.H.; Ford, M.J.; Timmons, B.; Dawson, K.A. Effects of supplementing microalgae in laying hen diets on productive performance, fatty-acid profile, and oxidative stability of eggs. J. Appl. Poult. Res. 2015, 24, 394–400. [Google Scholar] [CrossRef]
- Corbion Inc. AlgaPrimeTM DHA Is the World’s Leading Source of Algae Omega-3 Feed Ingredient for Aquaculture, Production Animals and Companion Animals. Available online: https://algaprime.com (accessed on 7 December 2020).
| Scientific Name | FR [209] | EP [209] | USA [194,208] | Example of Application |
|---|---|---|---|---|
| Aphanizomenon flos aquae | DS | F | Functional ingredient with antioxidant properties for cookies [210] | |
| Arthrospira sp. | F | F | DS, F | Fermenting agent for lactose-free beverages [211] |
| A. major | DS | |||
| A. maxima | DS | |||
| A. platensis | DS | F | DS, F | |
| Auxenochlorella protothecoides | DS, F | Algal flour for baked goods [194] | ||
| Chlamydomonas reinhardtii | DS, F | Omega-3 fatty acids valuable in healthy food [212] | ||
| Chlorella sp. | F | Tablets and powder made with whole biomass for human food [167] | ||
| C. luteoviridis | F | |||
| C. pyrenoidosa | F | |||
| C. vulgaris | DS | F | DS, F | |
| Dunaliella salina | DS | DS, F | Biomass used in pasta [192] | |
| D. badarwil | DS, F | |||
| Euglena gracilis | DS, F | Produce paramylon acting against fatigue [213] | ||
| Haematoccocus pluvialis | DS | DS, E | Astaxanthin-rich oleoresin with antioxidant capacity for healthy food [214] | |
| Nannochloropsis oculata | DS | Ingredient in functional cookies [173] | ||
| Odontella aurita | DS | DS | Produces fucoxanthin with antioxidant activities [215] | |
| Schizochytrium sp. | DS | E | E | Used for DHA-rich oil [165] |
| Tetraselmis chuii | DS, E | Ingredient for broccoli soup [216] | ||
| Ulkenia sp. | DS | E | E | Used for DHA-rich oil [130] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Remize, M.; Brunel, Y.; Silva, J.L.; Berthon, J.-Y.; Filaire, E. Microalgae n-3 PUFAs Production and Use in Food and Feed Industries. Mar. Drugs 2021, 19, 113. https://doi.org/10.3390/md19020113
Remize M, Brunel Y, Silva JL, Berthon J-Y, Filaire E. Microalgae n-3 PUFAs Production and Use in Food and Feed Industries. Marine Drugs. 2021; 19(2):113. https://doi.org/10.3390/md19020113
Chicago/Turabian StyleRemize, Marine, Yves Brunel, Joana L. Silva, Jean-Yves Berthon, and Edith Filaire. 2021. "Microalgae n-3 PUFAs Production and Use in Food and Feed Industries" Marine Drugs 19, no. 2: 113. https://doi.org/10.3390/md19020113
APA StyleRemize, M., Brunel, Y., Silva, J. L., Berthon, J.-Y., & Filaire, E. (2021). Microalgae n-3 PUFAs Production and Use in Food and Feed Industries. Marine Drugs, 19(2), 113. https://doi.org/10.3390/md19020113







