Cytoglobosins H and I, New Antiproliferative Cytochalasans from Deep-Sea-Derived Fungus Chaetomium globosum
Abstract
:1. Introduction
2. Results and Discussion
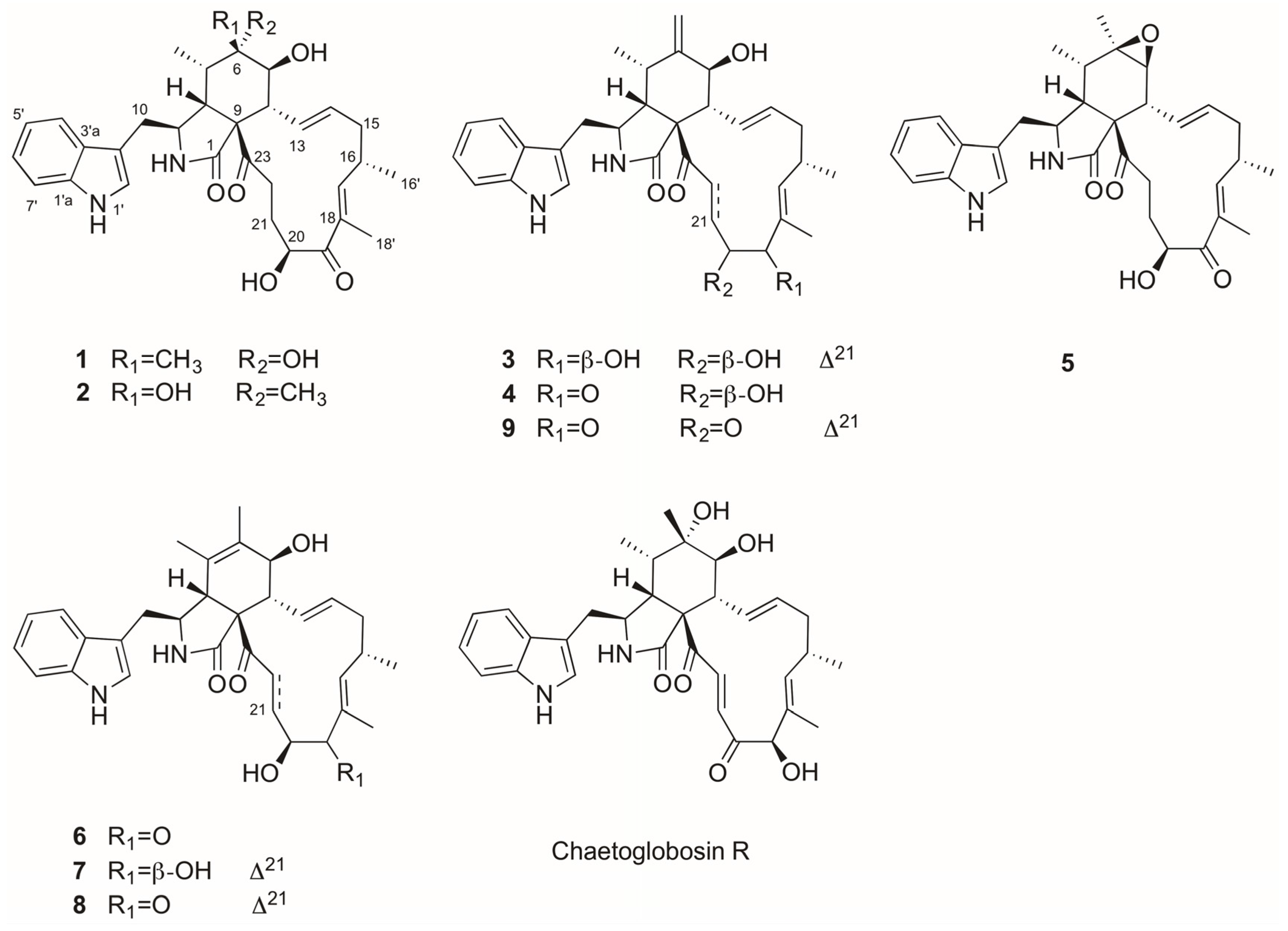
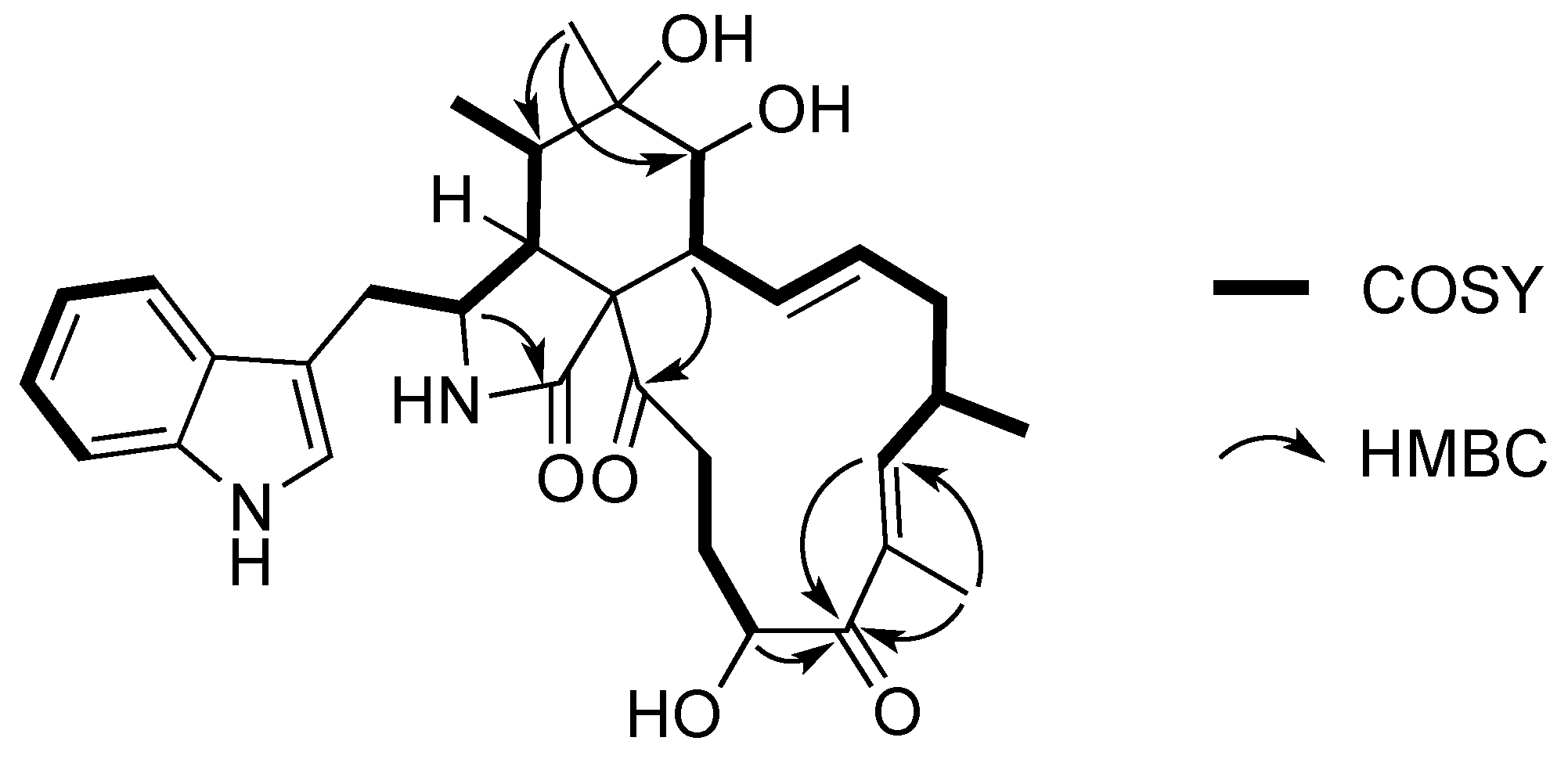
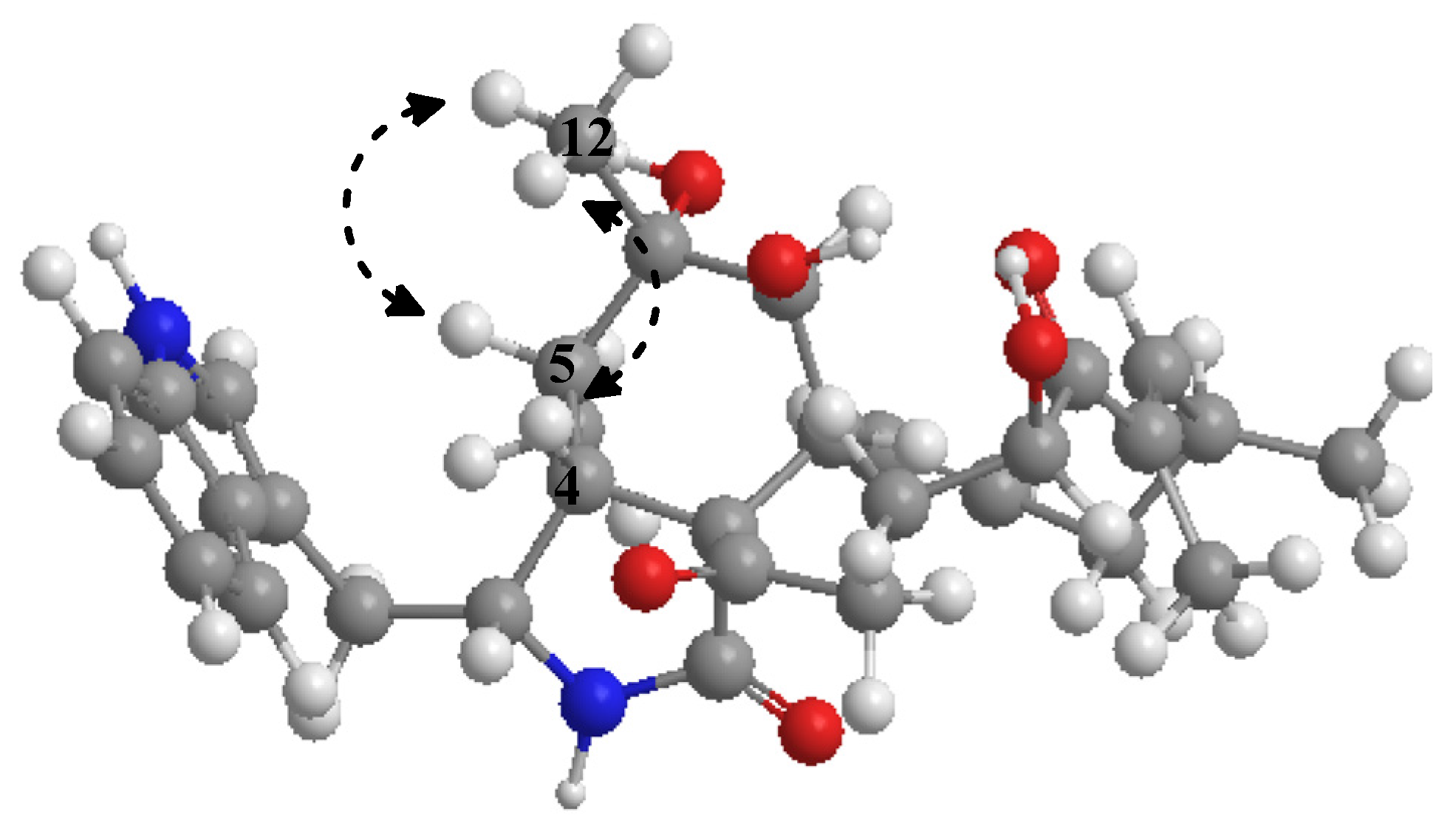
2.1. Purification and Structure Elucidation of Compounds 1–9
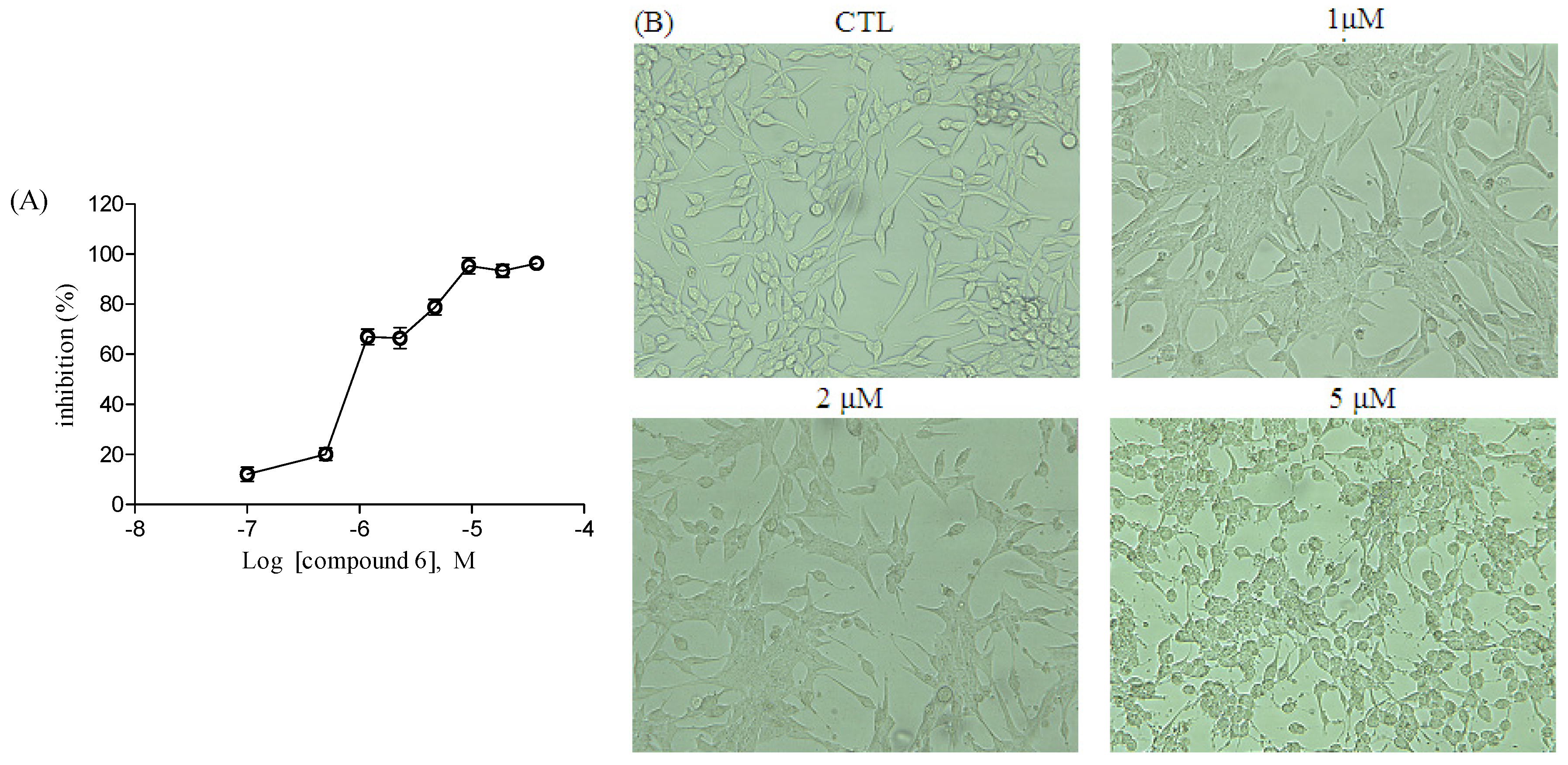
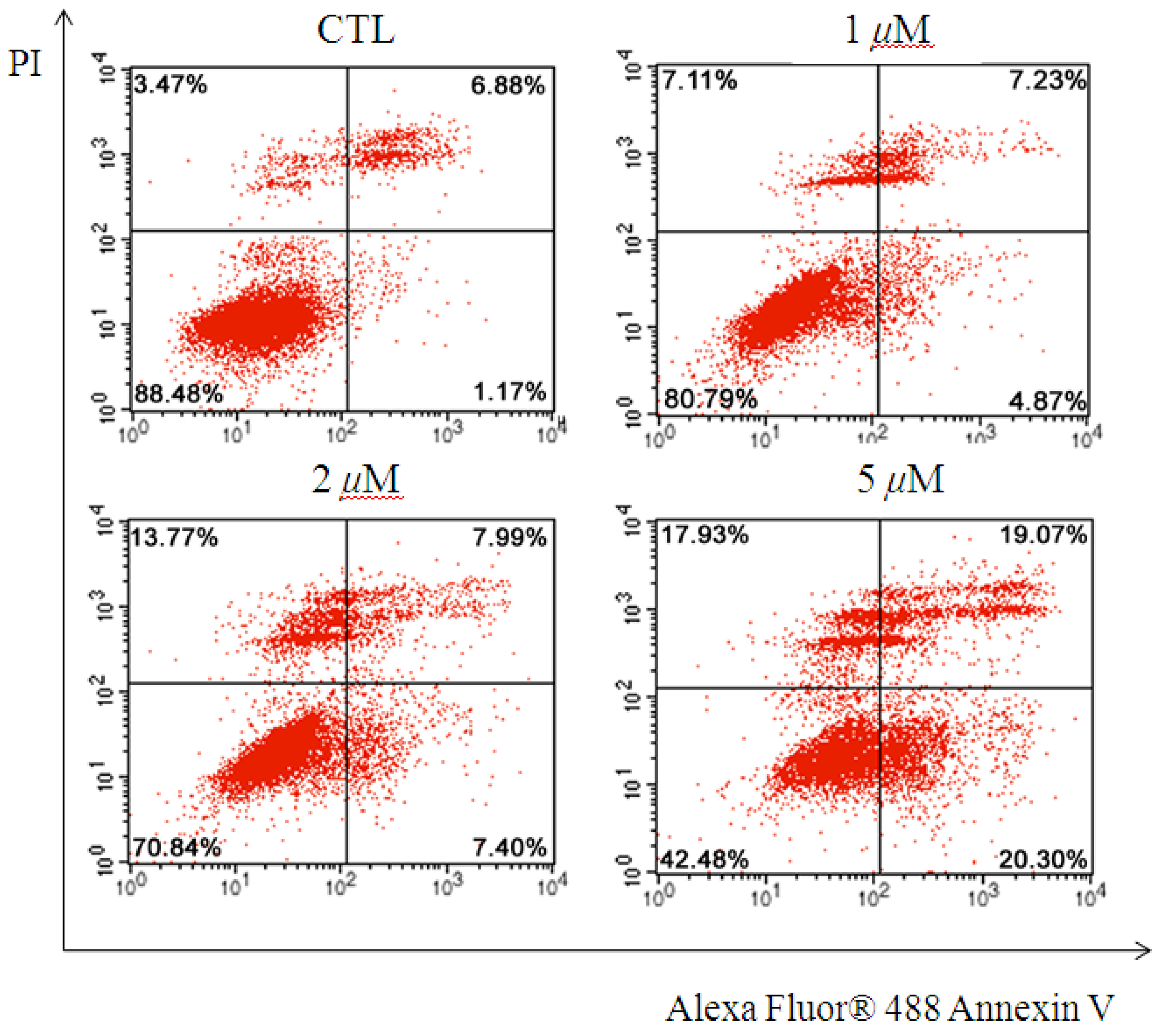
2.2. Antiproliferative Assay of Compounds 1–9
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Materials
3.3. Fermentation and Isolation
3.4. Antiproliferative Activity of Compounds 1–9
3.5. Apoptosis Evaluation of Compound 6
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2016, 33, 382–431. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.T.; Xue, Y.R.; Liu, C.H. A brief review of bioactive metabolites derived from deep-sea fungi. Mar. Drugs 2015, 13, 4594–4616. [Google Scholar] [CrossRef] [PubMed]
- Imhoff, J. Natural products from marine fungi—Still an underrepresented resource. Mar. Drugs 2016, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.J.; Feng, Y.J.; Blunt, J.W.; Cole, A.L.J.; Munro, M.H.G. Chaetoglobosins Q, R, and T, three further new metabolites from Chaetomium globosum. J. Nat. Prod. 2004, 67, 1722–1725. [Google Scholar] [CrossRef] [PubMed]
- Bashyal, B.P.; Wijeratne, E.M.K.; Faeth, S.H.; Gunatilaka, A.A.L. Globosumones A–C, cytotoxic orsellinic acid esters from the Sonoran desert endophytic fungus Chaetomium globosum. J. Nat. Prod. 2005, 68, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Ding, G.; Song, Y.C.; Chen, J.R.; Xu, C.; Ge, H.M.; Wang, X.T.; Tan, R.X. Chaetoglobosin U, a cytochalaan alkaloid from endophytic Chaetomium globosum IFB-E019. J. Nat. Prod. 2006, 69, 302–304. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Muroga, Y.; Tanaka, R. New azaphilones, seco-chaetomugilins A and D, produced by a marine-fish-derived Chaetomium globosum. Mar. Drugs 2009, 7, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.M.; Li, X.M.; Li, C.S.; Proksch, P.; Wang, B.G. Cytoglobosins A-G, cytochalasans from a marine-derived endophytic fungus, Chaetomium globosum QEN-14. J. Nat. Prod. 2010, 73, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Schumann, J.; Hertweck, C. Molecular basis of cytochalasan biosynthesis in fungi: gene cluster analysis and evidence for the involvement of a PKS-NRPS hybrid synthase by RNA silencing. J. Am. Chem. Soc. 2007, 129, 9564–9565. [Google Scholar] [CrossRef] [PubMed]
- Haidle, A.M.; Myers, A.G. An enantioselective, modular, and general route to the cytochalasins: Synthesis of L-696,474 and cytochalasin B. Proc. Natl. Acad. Sci. USA 2004, 101, 12048–12053. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Bao, J.; Zhang, X.Y.; Tu, Z.T.; Shi, Y.M.; Qi, S.H. Asperterrestide A, a cytotoxic cyclic tetrapeptide from the marine-derived fungus Aspergillus terreus SCSGAF0162. J. Nat. Prod. 2013, 76, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Liu, Z.; Yang, J.; Fu, P.; Peng, J.; Zhu, W.M.; Qi, S.H. A novel antifouling alkaloid from halotolerant fungus Penicillium sp. OUCMDZ-776. Tetrahedron Lett. 2012, 53, 2280–2283. [Google Scholar] [CrossRef]
- He, F.; Han, Z.; Peng, J.; Qian, P.Y.; Qi, S.H. Antifouling indole alkaloids from two marine derived strains. Nat. Prod. Commun. 2013, 8, 329–332. [Google Scholar] [PubMed]
- He, F.; Sun, Y.L.; Liu, K.S.; Zhang, X.Y.; Qian, P.Y.; Wang, Y.F.; Qi, S.H. Indole alkaloids from marine derived fungi Aspergillus sp. SCSIO 00305. J. Antibiot. 2012, 65, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Dou, H.; Song, Y.X.; Liu, X.Q.; Gong, W.; Li, E.G.; Tan, R.X.; Hou, Y.Y. Chaetoglobosin Fex from the marine-derived endophytic fungus inhibits induction of inflammatory mediators via Toll-like receptor 4 signaling in macrophages. Biol. Pharm. Bull. 2011, 34, 1864–1873. [Google Scholar] [CrossRef] [PubMed]
- Sekita, S.; Yoshihira, K.; Natori, S.; Udagawa, S.; Sakabe, F.; Kurata, H.; Umeda, M. Chaetoglobosins, cytotoxic 10-(indol-3-yl)-[13] cytochalasans from Chaetomium spp. I. Production, isolation and some cytological effects of chaetoglobosins A–J. Chem. Pharm. Bull. 1982, 30, 1609–1617. [Google Scholar] [CrossRef] [PubMed]
- Sekita, S.; Yoshihira, K.; Natori, S.; Kuwano, H. Structures of chaetoglobosins C, D, E, and F, cytotoxic indol-3-yl-[13]cytochalasans from Chaetomium globosum. Tetrahedron Lett. 1976, 17, 1351–1354. [Google Scholar] [CrossRef]
- Sekita, S.; Yoshihira, K.; Natori, S.; Kuwano, H. Chaetoglobosins, cytotoxic 10-(indol-3-yl)-[13]cytochalasans from Chaetomium spp. III. Structures of chaetoglobosins C, E, F, G, and J. Chem. Pharm. Bull. 1982, 30, 1629–1638. [Google Scholar] [CrossRef]
| No. | 1 | 2 | ||
|---|---|---|---|---|
| δH (m, J in Hz) | δC, Type | δH (m, J in Hz) | δC, Type | |
| 1 | 176.4, C | 176.8, C | ||
| 3 | 4.37 (dd, 5.0, 9.8) | 54.9, CH | 3.85 (dd, 4.7, 9.1) | 54.4, CH |
| 4 | 2.59 (dd, 11.2, 11.2) | 47.8, CH | 2.54 (m) | 45.8, CH |
| 5 | 1.75 (m) | 39.0, CH | 2.05 (m) | 40.1, CH |
| 6 | 77.4, C | 74.1, C | ||
| 7 | 3.57 (d, 12.1) | 76.7, CH | 3.19 (d, 11.8) | 73.6, CH |
| 8 | 2.32 (m) | 47.0, CH | 2.67 (dd, 11.0, 11.0) | 47.3, CH |
| 9 | 66.7, C | 65.7, C | ||
| 10 | 3.10 (dd, 3.9, 14.8) | 33.3, CH2 | 2.84 (dd, 4.8, 14.7) | 33.4, CH2 |
| 2.83 (dd, 5.3, 14.8) | 3.00 (dd, 4.8, 14.7) | |||
| 11 | 1.17 (d, 6.9) | 12.5, CH3 | 1.05 (d, 7.4) | 13.5, CH3 |
| 12 | 1.17 (s) | 22.3, CH3 | 1.24 (s) | 24.8, CH3 |
| 13 | 5.81 (dd, 10.0, 15.0) | 129.9, CH | 5.98 (dd, 10.6, 15.7) | 129.5, CH |
| 14 | 5.15 (m) | 134.5, CH | 5.17 (m) | 135.0, CH |
| 15 | 2.03 (m) | 41.9, CH2 | 2.44 (m) | 42.0, CH2 |
| 2.43 (d, 11.8) | 2.00 (m) | |||
| 16 | 2.73 (m) | 34.5, CH | 2.78 (m) | 34.6, CH |
| 17 | 6.24 (d, 9.0) | 149.8, CH | 6.22 (d, 9.1) | 150.0, CH |
| 18 | 136.3, C | 136.4, C | ||
| 19 | 205.9, C | 205.7, C | ||
| 20 | 4.75 (dd, 6.2, 6.2) | 72.3, CH | 4.69 (dd, 5.5, 5.5) | 72.5, CH |
| 21 | 1.67 (m) | 32.1, CH2 | 1.40 (m) | 31.9, CH2 |
| 1.37 (m) | 1.64 (m) | |||
| 22 | 2.38 (m) | 37.9, CH2 | 2.54 (m) | 38.6, CH2 |
| 1.67 (m) | 1.86 (m) | |||
| 23 | 209.8, C | 210.0, C | ||
| 2′ | 7.11 (m) | 125.6, CH | 7.06 (m) | 125.7, CH |
| 3′ | 110.3, C | 109.9, C | ||
| 3′a | 129.4, C | 129.3, C | ||
| 4′ | 7.58 (d, 7.9) | 119.5, CH | 7.53 (d, 7.9) | 119.3, CH |
| 5′ | 7.04 (dd, 7.9, 7.9) | 120.1, CH | 7.06 (m) | 120.2, CH |
| 6′ | 7.11 (m) | 122.5, CH | 7.12 (dd, 7.4, 7.4) | 122.5, CH |
| 7′ | 7.36 (d, 8.2) | 112.4, CH | 7.36 (d, 9.5) | 112.6, CH |
| 1′a | 138.0, C | 138.0, C | ||
| 16-Me | 1.03 (d, 6.7) | 20.2, CH3 | 1.03 (d, 6.6) | 20.2, CH3 |
| 18-Me | 1.78 (s) | 13.3, CH3 | 1.79 (s) | 12.4, CH3 |
| Compounds | MDA-MB-231 | LNCap | B16F10 |
|---|---|---|---|
| 1 | >10 | 9.25 ± 0.80 | >10 |
| 2 | >10 | >10 | >10 |
| 3 | >10 | >10 | 7.15 ± 1.21 |
| 4 | >10 | 2.93 ± 0.61 | 3.74 ± 0.32 |
| 5 | >10 | 1.05 ± 0.24 | 2.10 ± 0.26 |
| 6 | >10 | 0.62 ± 0.05 | 2.78 ± 0.15 |
| 7 | >10 | 7.78 ± 1.80 | 4.67 ± 1.02 |
| 8 | >10 | 3.29 ± 0.61 | 4.84 ± 0.75 |
| 9 | >10 | 7.12 ± 2.02 | >10 |
| cis-platin | 2.48 ± 0.74 | 1.04 ± 0.06 | 2.80 ± 0.11 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Min, X.; Huang, J.; Zhong, Y.; Wu, Y.; Li, X.; Deng, Y.; Jiang, Z.; Shao, Z.; Zhang, L.; et al. Cytoglobosins H and I, New Antiproliferative Cytochalasans from Deep-Sea-Derived Fungus Chaetomium globosum. Mar. Drugs 2016, 14, 233. https://doi.org/10.3390/md14120233
Zhang Z, Min X, Huang J, Zhong Y, Wu Y, Li X, Deng Y, Jiang Z, Shao Z, Zhang L, et al. Cytoglobosins H and I, New Antiproliferative Cytochalasans from Deep-Sea-Derived Fungus Chaetomium globosum. Marine Drugs. 2016; 14(12):233. https://doi.org/10.3390/md14120233
Chicago/Turabian StyleZhang, Zhihan, Xitian Min, Junjun Huang, Yue Zhong, Yuehua Wu, Xiaoxia Li, Yinyue Deng, Zide Jiang, Zongze Shao, Lianhui Zhang, and et al. 2016. "Cytoglobosins H and I, New Antiproliferative Cytochalasans from Deep-Sea-Derived Fungus Chaetomium globosum" Marine Drugs 14, no. 12: 233. https://doi.org/10.3390/md14120233
APA StyleZhang, Z., Min, X., Huang, J., Zhong, Y., Wu, Y., Li, X., Deng, Y., Jiang, Z., Shao, Z., Zhang, L., & He, F. (2016). Cytoglobosins H and I, New Antiproliferative Cytochalasans from Deep-Sea-Derived Fungus Chaetomium globosum. Marine Drugs, 14(12), 233. https://doi.org/10.3390/md14120233





