Dihydroisocoumarins from the Mangrove-Derived Fungus Penicillium citrinum
Abstract
:1. Introduction
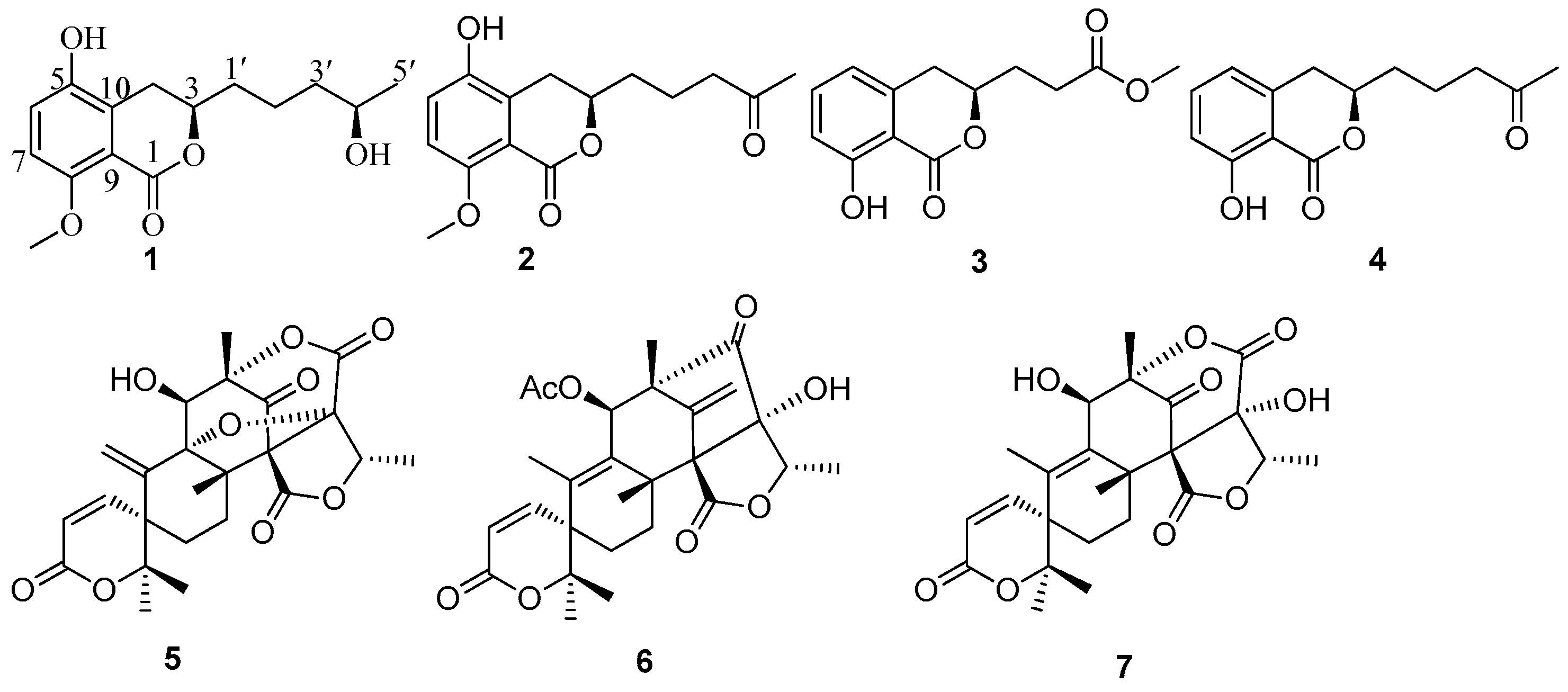
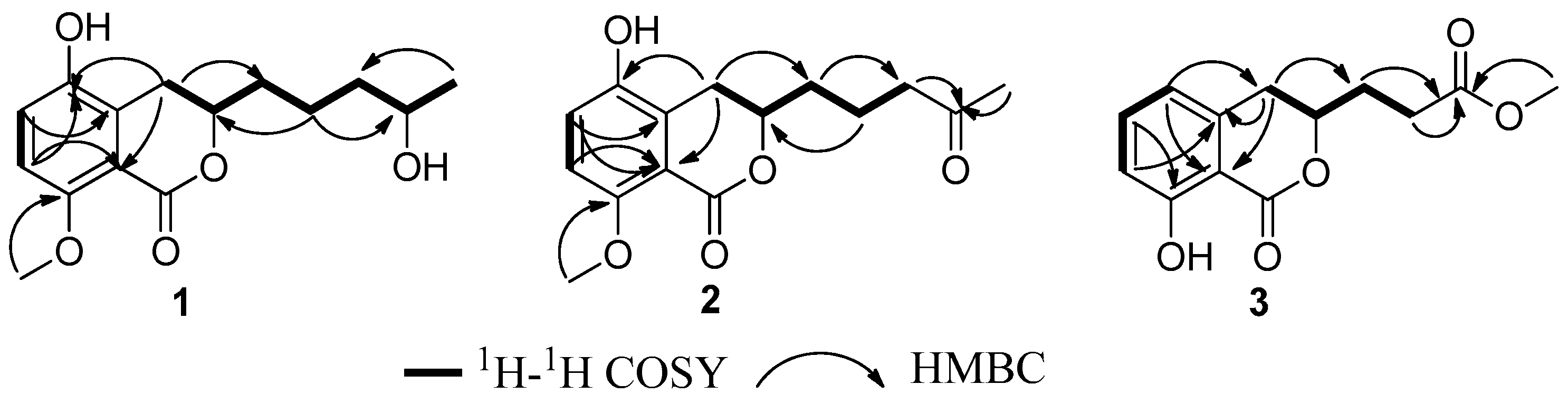
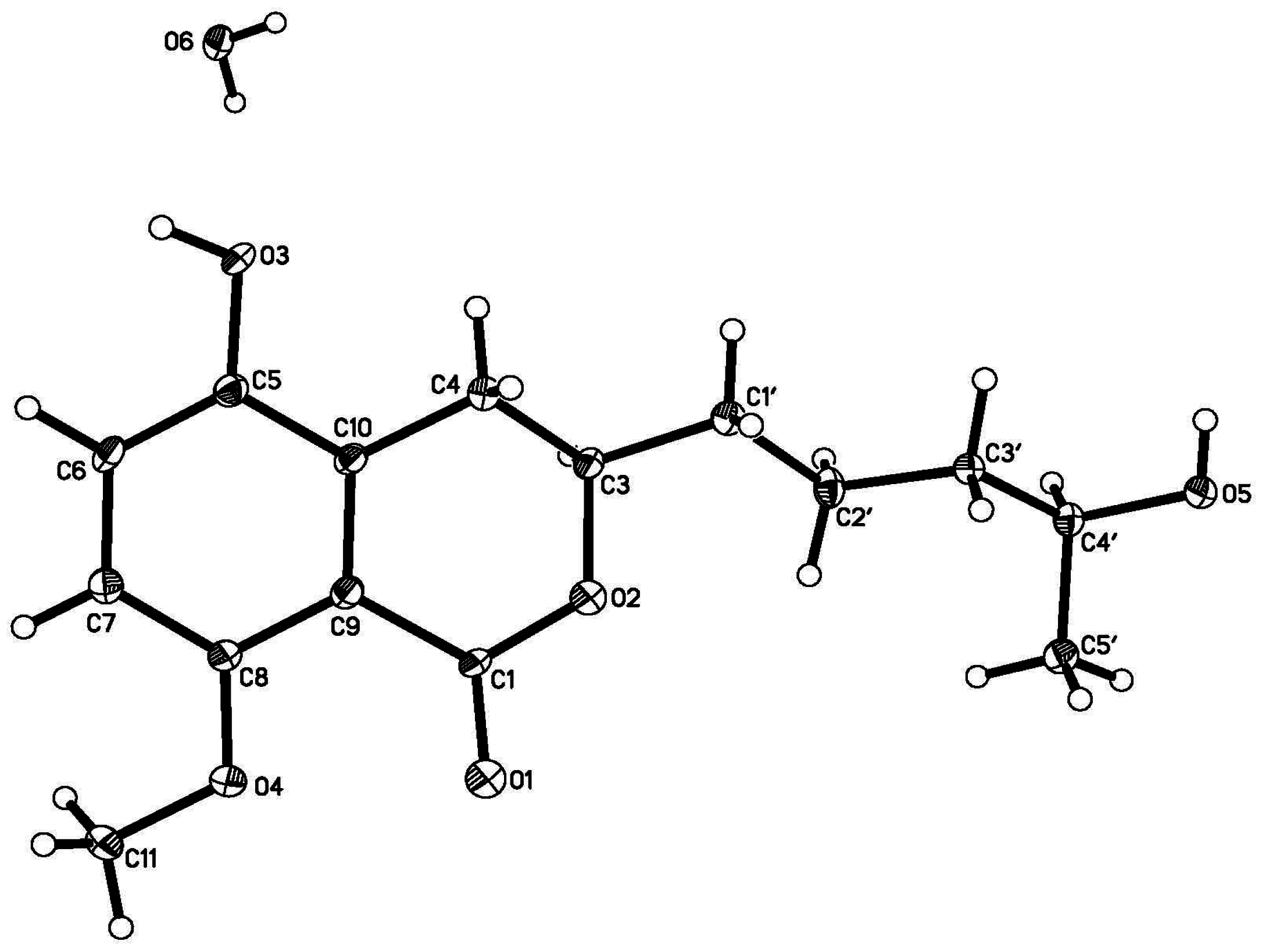
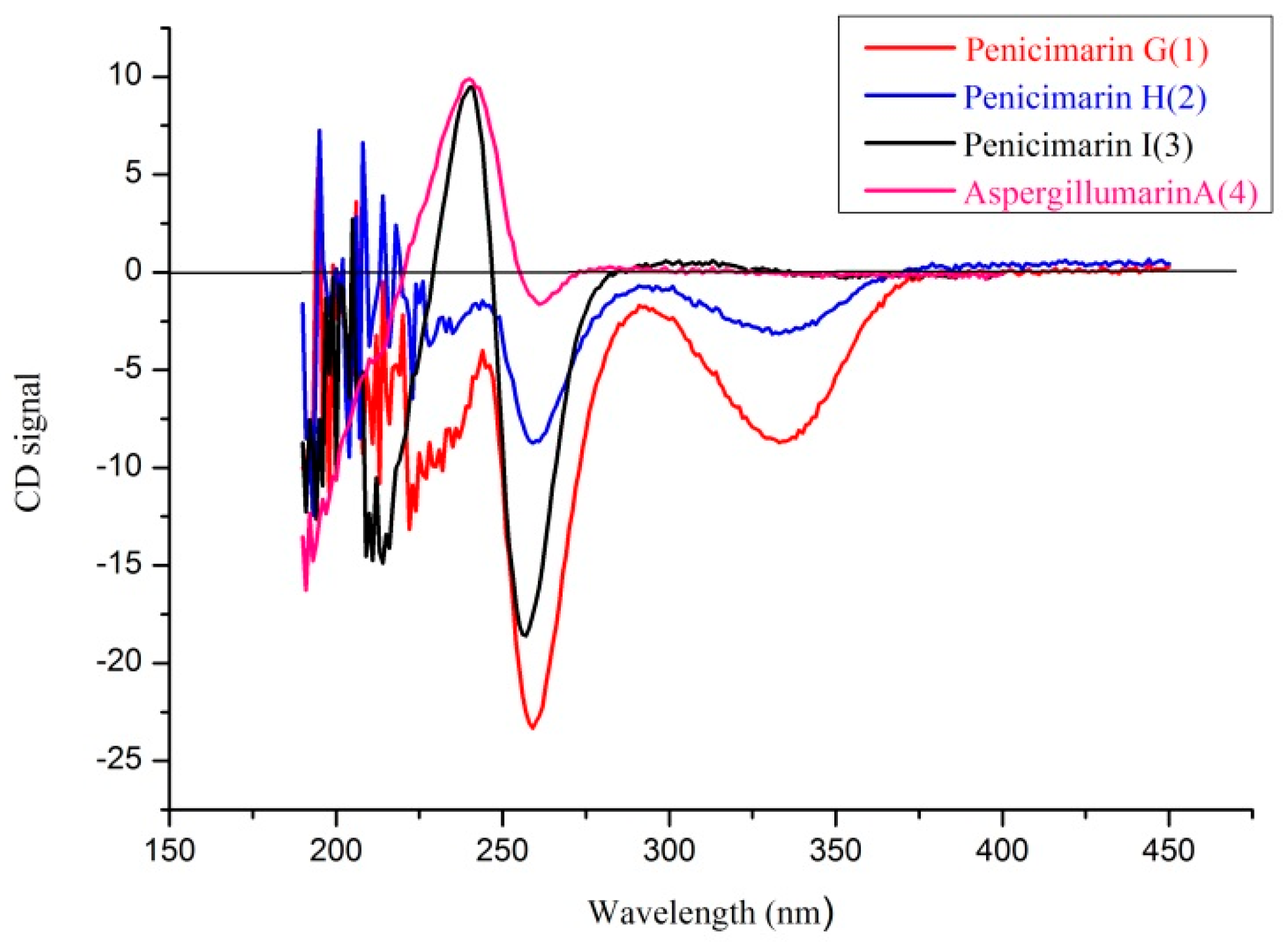
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Fungal Materials
3.3. Identification of Fungus
3.4. Extraction and Isolation
3.5. Biological Assays
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211. [Google Scholar] [CrossRef] [PubMed]
- Bao, J.; Sun, Y.L.; Zhang, X.Y.; Han, Z.; Gao, H.C.; He, F.; Qian, P.Y.; Qi, S.H. Antifouling and antibacterial polyketides from marine gorgonian coral-associated fungus Penicillium sp. SCSGAF 0023. J. Antibiot. 2013, 66, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.F.; Liu, P.P.; Wang, Y.; Wang, H.; Li, J.; Zhuang, Y.B.; Zhu, W.M. Antimicrobial aromatic polyketides from gorgonian-associated fungus, Penicillium commune 518. Chin. J. Chem. 2012, 30, 1236–1242. [Google Scholar] [CrossRef]
- Shao, C.L.; Wang, C.Y.; Gu, Y.C.; Wei, M.Y.; Pan, J.H.; Deng, D.S.; She, Z.G.; Lin, Y.C. Penicinoline, a new pyrrolyl 4-quinolinone alkaloid with an unprecedented ring system from an endophytic fungus Penicillium sp. Bioorg. Med. Chem. Lett. 2010, 20, 3284–3286. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Fang, Y.C.; Zhu, T.J.; Gu, Q.Q.; Zhu, W.M. Nine new gentisyl alcohol derivatives from marine-derived fungus Penicillium terrestre. J. Nat. Prod. 2008, 71, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Bringmann, G.; Lang, G.; Gulder, T.A.M.; Tsuruta, H.; Muhlbacher, J.; Maksimenka, K.; Steffens, S.; Schaumann, K.; Stohr, R.; Wiese, J.; et al. The first sorbicillinoid alkaloids, the antileukemic sorbicillactones A and B, from a sponge-derived Penicillium chrysogenum strain. Tetrahedron 2005, 61, 7252–7265. [Google Scholar] [CrossRef]
- Zhou, X.M.; Zheng, C.J.; Chen, G.Y.; Song, X.P.; Han, C.R.; Li, G.N.; Fu, Y.H.; Chen, W.H.; Niu, Z.G. Bioactive anthraquinone derivatives from the mangrove-derived fungus Stemphylium sp. 33231. J. Nat. Prod. 2014, 77, 2021–2028. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Shao, C.L.; Wu, L.Y.; Chen, M.; Wang, K.L.; Zhao, D.L.; Sun, X.P.; Chen, G.Y.; Wang, C.Y. Bioactive phenylalanine derivatives and cytochalasins from the soft coral-derived fungus Aspergillus elegans. Mar. Drugs 2013, 11, 2054–2068. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Wu, L.Y.; Li, X.B.; Song, X.M.; Niu, Z.G.; Song, X.P.; Chen, G.Y.; Wang, C.Y. Structure and absolute configuration of aspergilumamide A, a novel lumazine peptide from the mangrove-derived fungus Aspergillus sp. Helv. Chim. Acta 2015, 98, 368–373. [Google Scholar] [CrossRef]
- Zheng, C.J.; Huang, G.L.; Xu, Y.; Song, X.M.; Yao, J.; Liu, H.; Wang, R.P.; Sun, X.P. A new benzopyrans derivatives from a mangrove-derived fungus Penicillium citrinum from the South China Sea. Nat. Prod. Res. 2016, 30, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.M.; Zheng, C.J.; Chen, G.Y.; Song, X.P.; Han, C.R.; Tang, X.Z.; Liu, R.J.; Ren, L.L. Two new stemphol sulfates from the mangrove endophytic fungus Stemphylium sp. 33231. J. Antibiot. 2015, 68, 501–503. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Huang, G.L.; Tang, X.Z.; Wang, D.N.; Gong, X.L.; Zhang, Q.; Song, X.P.; Chen, G.Y. Secondary metabolites and antibacterial activities of a Bruguiera sexangula var. Rhynchopetala-derived fungus Penicillium sp. (J41221). Chin. J. Org. Chem. 2014, 34, 1172–1176. [Google Scholar] [CrossRef]
- Zhou, X.M.; Zheng, C.J.; Song, X.P.; Han, C.R.; Chen, W.H.; Chen, G.Y. Antibacterial α-pyrone derivatives from a mangrove-derived fungus Stemphylium sp. 33231 from the South China Sea. J. Antibiot. 2014, 67, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Li, S.D.; Wei, M.Y.; Chen, G.Y.; Lin, Y.C. Two new dihydroisocoumarins from the endophytic fungus Aspergillus sp. collected from the South China Sea. Chem. Nat. Compd. 2012, 48, 371–373. [Google Scholar] [CrossRef]
- Arunpanichlert, J.; Rukachaisirikul, V.; Phongpaichit, S.; Supaphon, O.; Sakayaroj, J. Meroterpenoid, isocoumarin, and phenol derivatives from the seagrass-derived fungus Pestalotiopsis sp. PSU-ES194. Tetrahedron 2015, 71, 882–888. [Google Scholar] [CrossRef]
- Hayashi, H.; Mukaihara, M.; Murao, S.; Arai, M.; Lee, A.Y.; Clardy, J. Acetoxydehydroaustin, a new bioactive compound, and related compound neoaustin from Penicillium sp. MG-11. Biosci. Biotechnol. Biochem. 1994, 58, 334–338. [Google Scholar] [CrossRef]
- Qi, J.; Shao, C.L.; Li, Z.Y.; Gan, L.S.; Fu, X.M.; Bian, W.T.; Zhao, Y.X.; Wang, C.Y. Isocoumarin derivatives and benzofurans from a sponge-derived Penicillium sp. fungus. J. Nat. Prod. 2013, 76, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Choukchou-Braham, N.; Asakawa, Y.; Lepoittevin, J.P. Isolation, structure determination and synthesis of new dihydroisocoumarins from Ginkgo biloba L. Tetrahedron Lett. 1994, 35, 3949–3952. [Google Scholar] [CrossRef]
- Pierce, C.G.; Uppuluri, P.; Teistan, A.R.; Wormley, F.L.J.; Mowat, E.; Ramage, G.; Lopez-ribot, J.L. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nat. Protoc. 2008, 3, 1494–1500. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar] [PubMed]
| Position | 1 a | 2 a | 3 b | |||
|---|---|---|---|---|---|---|
| δC, Type | δH (J in Hz) | δC, Type | δH (J in Hz) | δC, Type | δH (J in Hz) | |
| 1 | 165.8, C | 165.7, C | 169.5, C | |||
| 3 | 79.6, CH | 4.39 (m) | 79.4, CH | 4.39 (m) | 78.4, CH | 4.65 (m) |
| 4 | 28.7, CH2 | 3.16 (dd, 16.8, 3.0) 2.58 (dd, 16.8, 11.6) | 28.6, CH2 | 3.15 (dd, 16.8, 3.0) 2.59 (dd, 16.8, 12.0) | 32.9, CH2 | 2.92 (m) |
| 5 | 148.2, C | 148.2, C | 118.0, CH | 6.89 (d, 8.0) | ||
| 6 | 122.3, CH | 7.05 (d, 8.4) | 122.3, CH | 7.05 (d, 9.0) | 136.2, CH | 7.42 (dd, 8.0, 7.6) |
| 7 | 112.8, CH | 6.90 (d, 8.4) | 112.8, CH | 6.90 (d, 9.0) | 116.3, CH | 6.70 (d, 7.6) |
| 8 | 155.8, C | 155.8, C | 162.2, C | |||
| 9 | 114.8, C | 114.7, C | 139.0, C | |||
| 10 | 129.5, C | 129.4, C | 108.3, C | |||
| 1′ | 35.8, CH2 | 1.84 (m), 1.70 (m) | 35.1, CH2 | 1.79 (m) | 29.8, CH2 | 2.13 (m) |
| 2′ | 23.5, CH2 | 1.59 (m) | 20.3, CH2 | 1.72 (m) | 29.2, CH2 | 2.62 (dd, 14.4, 7.2) |
| 3′ | 39.8, CH2 | 1.55 (m) | 43.6, CH2 | 2.58 (m) | 173.2 | |
| 4′ | 68.3, CH | 3.75 (m) | 211.4, CH | 51.8, CH3 | 3.70 (s) | |
| 5′ | 22.3, CH3 | 1.17 (d, 6.2) | 29.8, CH3 | 2.15 (s) | ||
| 8-OMe | 56.7, CH3 | 3.82 (s) | 56.7, CH3 | 3.81 (s) | ||
| 8-OH | 10.94 (s) | |||||
| Compound | Minimum Inhibitory Concentration (MIC, μM) | ||||
|---|---|---|---|---|---|
| S. epidermidis | S. aureus | E. coli | B. cereus | V. alginolyticus | |
| 1 | 20 | 20 | 20 | 20 | 20 |
| 2 | 10 | 20 | >20 | 20 | 20 |
| 5 | 20 | >20 | >20 | >20 | >20 |
| 6 | 20 | >20 | >20 | >20 | >20 |
| 7 | 10 | >20 | >20 | 20 | >20 |
| Ciprofloxacin a | 0.30 | 0.30 | 0.60 | 1.20 | 1.25 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, G.-L.; Zhou, X.-M.; Bai, M.; Liu, Y.-X.; Zhao, Y.-L.; Luo, Y.-P.; Niu, Y.-Y.; Zheng, C.-J.; Chen, G.-Y. Dihydroisocoumarins from the Mangrove-Derived Fungus Penicillium citrinum. Mar. Drugs 2016, 14, 177. https://doi.org/10.3390/md14100177
Huang G-L, Zhou X-M, Bai M, Liu Y-X, Zhao Y-L, Luo Y-P, Niu Y-Y, Zheng C-J, Chen G-Y. Dihydroisocoumarins from the Mangrove-Derived Fungus Penicillium citrinum. Marine Drugs. 2016; 14(10):177. https://doi.org/10.3390/md14100177
Chicago/Turabian StyleHuang, Guo-Lei, Xue-Ming Zhou, Meng Bai, Yu-Xin Liu, Yan-Lei Zhao, You-Ping Luo, Yan-Yan Niu, Cai-Juan Zheng, and Guang-Ying Chen. 2016. "Dihydroisocoumarins from the Mangrove-Derived Fungus Penicillium citrinum" Marine Drugs 14, no. 10: 177. https://doi.org/10.3390/md14100177
APA StyleHuang, G.-L., Zhou, X.-M., Bai, M., Liu, Y.-X., Zhao, Y.-L., Luo, Y.-P., Niu, Y.-Y., Zheng, C.-J., & Chen, G.-Y. (2016). Dihydroisocoumarins from the Mangrove-Derived Fungus Penicillium citrinum. Marine Drugs, 14(10), 177. https://doi.org/10.3390/md14100177





