New Scalarane Sesterterpenoids from the Formosan Sponge Ircinia felix
Abstract
:1. Introduction
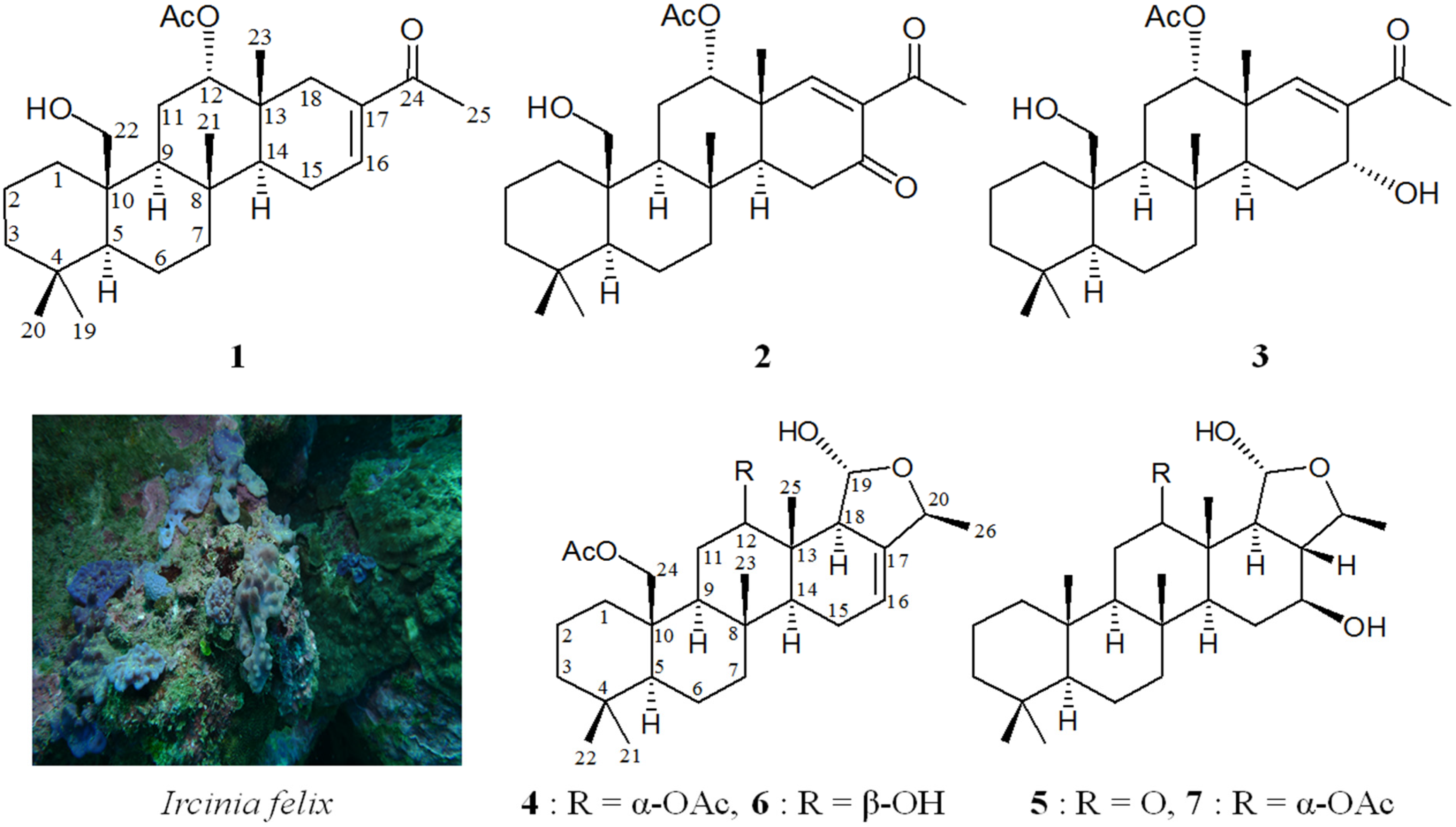
2. Results and Discussion
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 2.09 m; 0.52 ddd (12.8, 12.8, 3.2) | 34.4, CH2 | H2-2 | C-3, -10, -22 |
| 2 | 1.51 m; 1.39 m | 17.8, CH2 | H2-1, H2-3 | n. o. a |
| 3 | 1.43 ddd (12.8, 4.0, 4.0) | 41.7, CH2 | H2-2 | C-1, -4, -5, -19, -20 |
| 1.17 ddd (12.8, 12.8, 4.8) | ||||
| 4 | 33.0, C | |||
| 5 | 0.97 dd (12.0, 2.0) | 56.9, CH | H2-6 | C-3, -4, -6, -7, -9, -10, -20, -22 |
| 6 | 1.58 m; 1.45 m | 18.3, CH2 | H-5, H2-7 | C-5, -8 |
| 7 | 1.81 ddd (12, 8, 3.2, 3.2); 1.05 m | 41.9, CH2 | H2-6 | C-21 |
| 8 | 37.4, C | |||
| 9 | 1.35 br d (13.2) | 53.1, CH | H2-11 | C-5, -7, -8, -10, -11, -12, -14, -21, -22 |
| 10 | 41.8, C | |||
| 11 | 2.17 m; 1.96 m | 25.2, CH2 | H-9, H-12 | C-9, -13 |
| 12 | 4.72 dd (3.6, 2.0) | 77.1, CH | H2-11 | C-9, -14 |
| 13 | 35.8, C | |||
| 14 | 1.56 m | 48.0, CH | H2-15 | C-7, -8, -13, -15, -21, -23 |
| 15 | 2.34 m; 2.22 m | 24.0, CH2 | H-14, H-16 | C-16, -17 |
| 16 | 6.86 m | 139.4, CH | H2-15 | C-14, -24 |
| 17 | 137.7, C | |||
| 18 | 2.22 m; 1.92 m | 35.1, CH2 | C-13, -14, -16, -17, -23, -24 | |
| 19 | 0.87 s | 33.8, CH3 | C-3, -4, -5, -20 | |
| 20 | 0.77 s | 21.9, CH3 | C-3, -4, -5, -19 | |
| 21 | 1.10 s | 15.4, CH3 | C-7, -8, -9, -14 | |
| 22 | 4.03 d (11.6); 3.89 d (11.6) | 63.0, CH2 | C-1, -9, -10 | |
| 23 | 0.87 s | 19.6, CH3 | C-12, -13, -14 | |
| 24 | 199.1, C | |||
| 25 | 2.28 s | 25.2, CH3 | C-17, -24 | |
| 12-OAc | 170.2, C | |||
| 2.08 s | 21.5, CH3 | Acetate carbonyl |

| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 2.11, m; 0.53 ddd (13.2, 13.2, 4.0) | 34.2, CH2 | H2-2 | C-2, -3, -10, -22 |
| 2 | 1.58–1.42 m | 18.3, CH2 | H2-1, H2-3 | n. o. a |
| 3 | 1.42 m; 1.19 m | 41.6, CH2 | H2-2 | C-4, -20 |
| 4 | 33.0, C | |||
| 5 | 0.96 m | 57.0, CH | H2-6 | C-3, -4, -6, -7, -9, -10, -20, -22 |
| 6 | 1.54 m; 1.42 m | 17.7, CH2 | H-5, H2-7 | n. o. |
| 7 | 1.78 ddd (12.8, 3.2, 3.2); 1.05 m | 41.0, CH2 | H2-6 | C-21 |
| 8 | 37.2, C | |||
| 9 | 1.31 br d (13.2) | 53.1, CH | H2-11 | C-5, -7, -8, -10, -11, -21, -22 |
| 10 | 41.7, C | |||
| 11 | 2.29 ddd (13.6, 13.6, 2.4); 2.05 m | 24.9, CH2 | H-9, H-12 | n. o. |
| 12 | 4.97 dd (2.8, 2.8) | 76.3, CH | H2-11 | n. o. |
| 13 | 41.4, C | |||
| 14 | 2.11 m | 48.9, CH | H2-15 | C-8, -13, -21, -23 |
| 15 | 2.57–2.40 m | 35.0, CH2 | H-14 | C-13, -14, -16 |
| 16 | 197.7, C | |||
| 17 | 136.6, C | |||
| 18 | 7.30 s | 163.9, CH | C-12, -14, -17, -24 | |
| 19 | 0.87 s | 33.8, CH3 | C-3, -4, -5, -20 | |
| 20 | 0.76 s | 21.8, CH3 | C-3, -4, -5, -19 | |
| 21 | 1.12 s | 15.7, CH3 | C-7, -8, -9, -14 | |
| 22 | 4.04 d (12.0); 3.87 d (12.0) | 62.7, CH2 | C-1, -9, -10 | |
| 23 | 1.17 s | 18.4, CH3 | C-12, -13, -14, -18 | |
| 24 | 197.9, C | |||
| 25 | 2.42 s | 30.6, CH3 | C-17, -24 | |
| 12-OAc | 170.2, C | |||
| 2.05 s | 21.2, CH3 | Acetate carbonyl |

| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 2.08 m; 0.57 ddd (12.8, 12.8, 3.2) | 34.1, CH2 | H2-2 | n. o. a |
| 2 | 1.54 m; 1.39 m | 17.8, CH2 | H2-1, H2-3 | n. o. |
| 3 | 1.42 m; 1.16 m | 41.7, CH2 | H2-2 | C-20 |
| 4 | 33.0, C | |||
| 5 | 1.02 dd (12.8, 2.4) | 56.8, CH | H2-6 | C-4, -20, -22 |
| 6 | 1.54 m; 1.47 m | 18.4, CH2 | H-5, H2-7 | n. o. |
| 7 | 1.88 m; 1.11 m | 41.3, CH2 | H2-6 | C-8, -21 |
| 8 | 36.8, C | |||
| 9 | 1.45 m | 53.5, CH | H2-11 | C-10, -11, -21, -22 |
| 10 | 41.8, C | |||
| 11 | 1.96–1.81 m | 25.3, CH2 | H-9, H-12 | C-8, -10, -13 |
| 12 | 4.97 dd (2.8, 2.8) | 76.5, CH | H2-11 | n. o. |
| 13 | 41.4, C | |||
| 14 | 1.88 m | 44.0, CH | H2-15 | C-8, -13, -15, -16, -21, -23 |
| 15 | 1.88 m; 1.64 dd (14.0, 4.8) | 25.3, CH2 | H-14, H-16 | C-8, -13, -16, -17 |
| 16 | 4.55 d (3.6) | 63.3, CH | H2-15 | C-14, -17, -18 |
| 17 | 138.2, C | |||
| 18 | 6.59 s | 152.2, CH | C-12, -13, -14, -16, -24 | |
| 19 | 0.85 s | 33.8, CH3 | C-3, -4, -5, -20 | |
| 20 | 0.76 s | 21.8, CH3 | C-3, -4, -5, -19 | |
| 21 | 1.06 s | 16.4, CH3 | C-7, -8, -9, -14 | |
| 22 | 4.04 d (12.0); 3.90 d (12.0) | 62.8, CH2 | C-1, -9, -10 | |
| 23 | 1.06 s | 19.5, CH3 | C-12, -13, -14, -18 | |
| 24 | 201.4, C | |||
| 25 | 2.24 s | 25.4, CH3 | C-17, -24 | |
| 12-OAc | 170.9, C | |||
| 2.04 s | 21.4, CH3 | Acetate carbonyl |
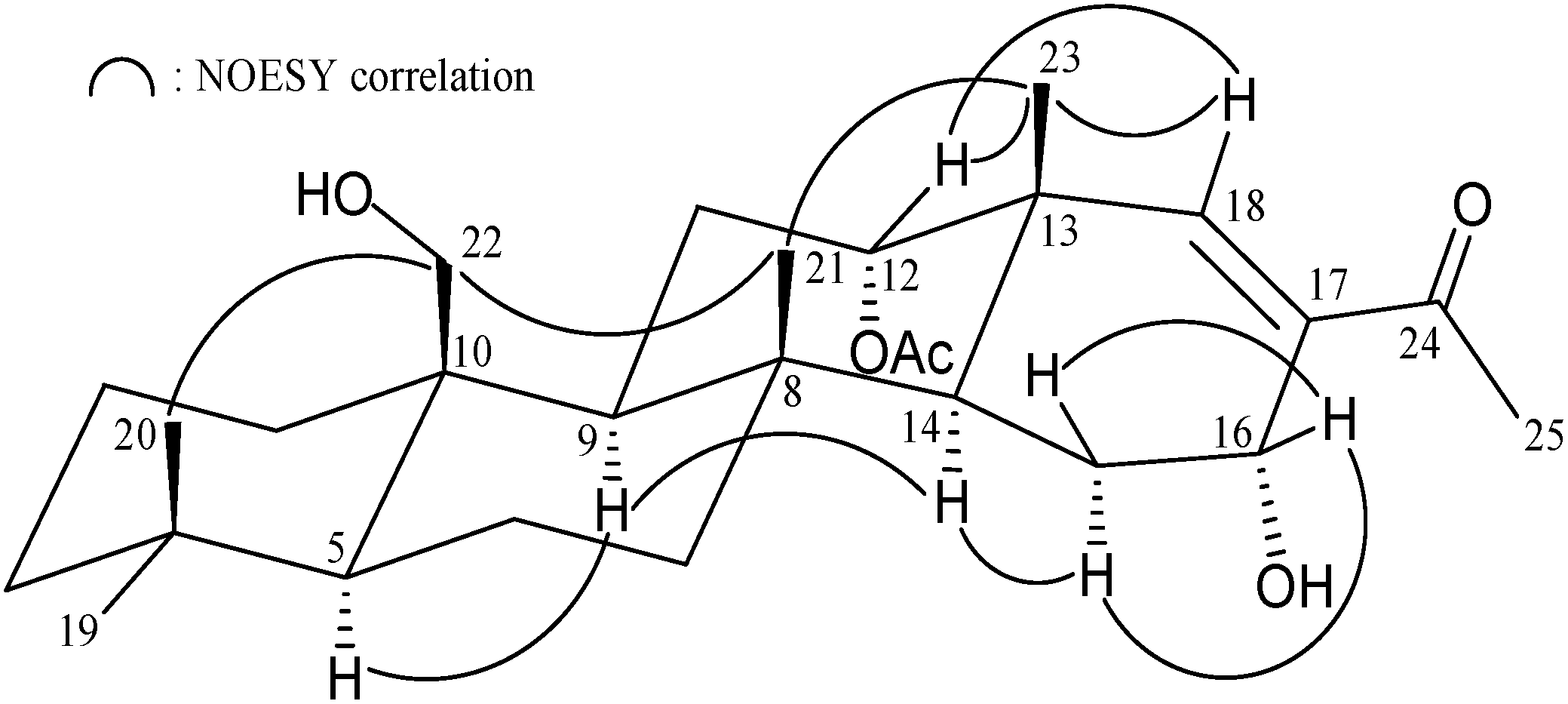
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 1.98 dd (13.2, 2.4) | 34.7, CH2 | H2-2 | C-5, -9, -24 |
| 0.57 ddd (13.2, 13.2, 2.4) | ||||
| 2 | 1.56 m; 1.43 m | 18.2, CH2 | H2-1, H2-3 | C-4 |
| 3 | 1.46 m; 1.15 m | 41.6, CH2 | H2-2 | C-2, -4, -21, -22 |
| 4 | 33.0, C | |||
| 5 | 1.04 dd (12.8, 2.0) | 56.8, CH | H2-6 | C-3, -4, -6, -7, -9, -10, -21, -22, -24 |
| 6 | 1.56 m; 1.38 dd (13.6, 3.2) | 17.9, CH2 | H-5, H2-7 | n. o. a |
| 7 | 1.79 ddd (12.8, 3.2, 3.2); 1.12 m | 41.7, CH2 | H2-6 | C-5, -8, -9, -14, -23 |
| 8 | 37.7, C | |||
| 9 | 1.46 m | 52.9, CH | H2-11 | C-1, -5, -8, -10, -11, -12, -14, -23, -24 |
| 10 | 40.1, C | |||
| 11 | 2.05–1.89 m | 25.1, CH2 | H-9, H-12 | C-8 |
| 12 | 4.91 dd (3.2, 2.4) | 74.6, CH | H2-11 | C-9, -14, acetate carbonyl |
| 13 | 36.9, C | |||
| 14 | 1.64 dd (11.2, 6.4) | 50.4, CH | H2-15 | C-7, -8, -9, -13, -15, -18, -25 |
| 15 | 2.16 m; 1.97 m | 22.9, CH2 | H-14, H-16 | C-8 |
| 16 | 5.35 br s | 114.4, CH | H2-15 | n. o. |
| 17 | 141.2, C | |||
| 18 | 2.82 br s | 54.7, CH | H-19 | n. o. |
| 19 | 5.21 d (3.2) | 96.7, CH | H-18 | C-20 |
| 20 | 4.62 m | 74.0, CH | H3-26 | n. o. |
| 21 | 0.89 s | 33.7, CH3 | C-3, -4, -5, -22 | |
| 22 | 0.83 s | 21.9, CH3 | C-3, -5, -21 | |
| 23 | 0.98 s | 15.4, CH3 | C-7, -9, -14 | |
| 24 | 4.59 d (12.0); 4.16 d (12.0) | 64.9, CH2 | C-1, -9, -10, acetate carbonyl | |
| 25 | 0.78 s | 14.7, CH3 | C-12, -14, -18 | |
| 26 | 1.26 d (6.0) | 17.6, CH3 | H-20 | C-17, -20 |
| 12-OAc | 170.9, C | |||
| 2.10 s | 21.5, CH3 | Acetate carbonyl | ||
| 23-OAc | 171.0, C | |||
| 2.05 s | 21.2, CH3 | Acetate carbonyl |
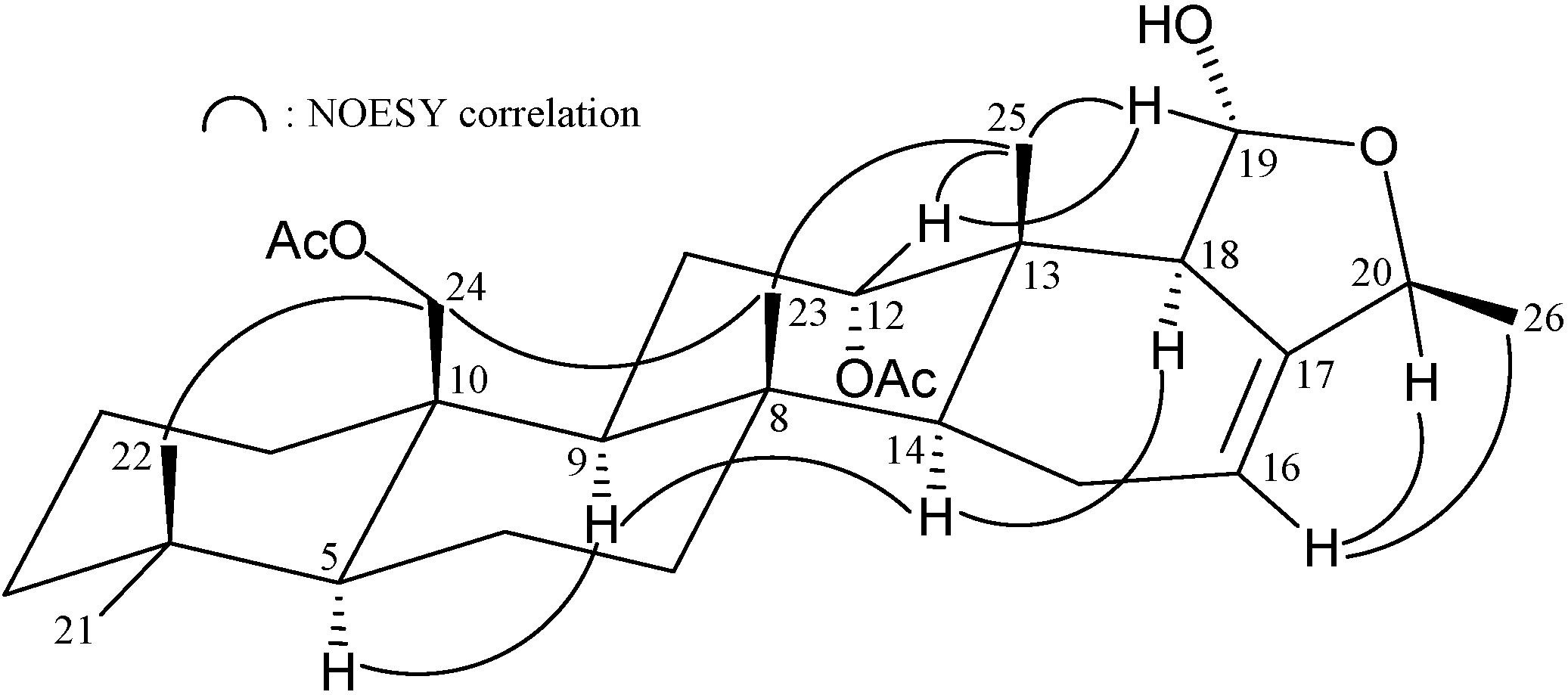
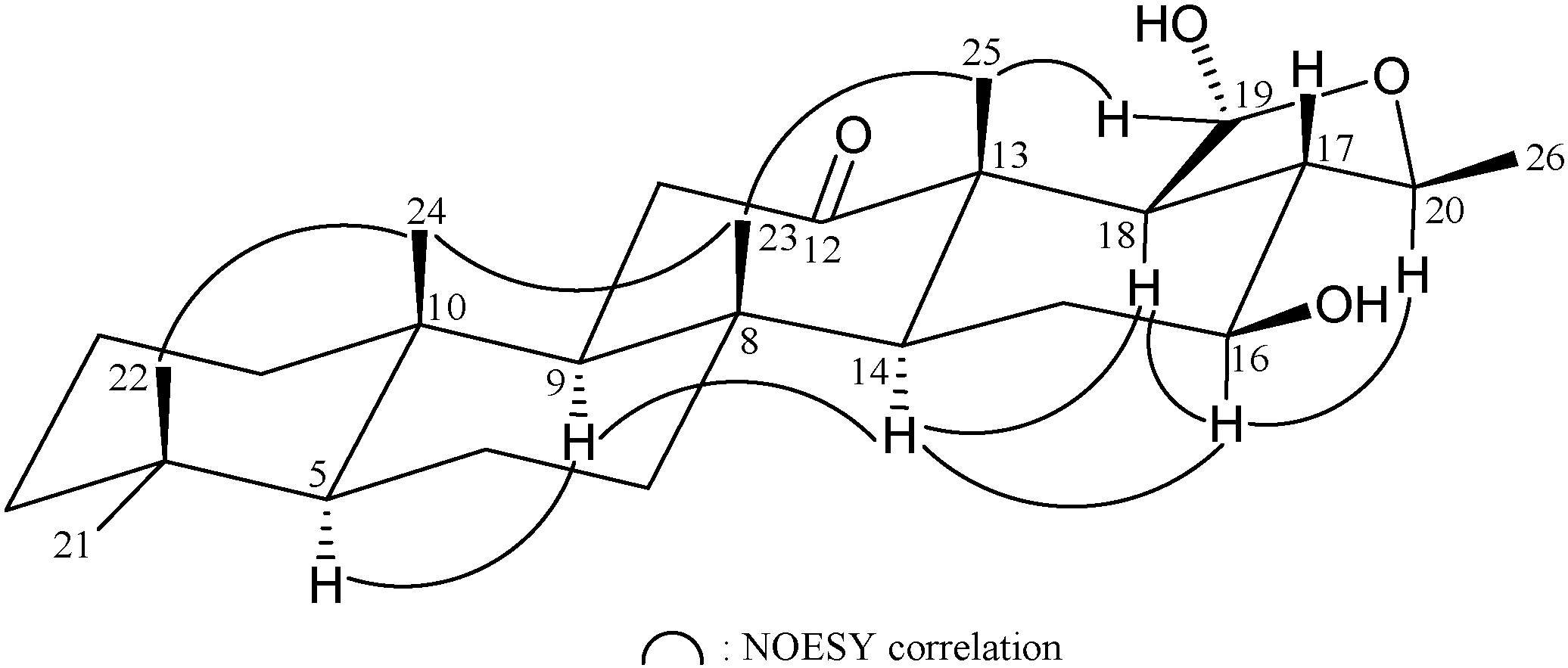
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 1.56 m; 0.76 m | 39.3 CH2 | H2-2 | C-5 |
| 2 | 1.64–1.34 m | 18.3, CH2 | H2-1, H2-3 | n. o. a |
| 3 | 1.80 m; 1.38 m | 41.6, CH2 | H2-2 | n. o. |
| 4 | 33.3, C | |||
| 5 | 0.94 m | 56.5, CH | H2-6 | C-4 |
| 6 | 1.64–1.34 m | 18.1, CH2 | H-5, H2-7 | n. o. |
| 7 | 1.81 m; 0.94 dd (13.2, 4.0) | 41.7, CH2 | H2-6 | C-5 |
| 8 | 37.8, C | |||
| 9 | 1.19 m | 61.4, CH | H2-11 | C-8, -12, -14, -23 |
| 10 | 38.2, C | |||
| 11 | 2.70 dd (14.0, 13.2); 2.32 dd (13.2, 2.4) | 35.3, CH2 | H-9 | C-8, -9, -12 |
| 12 | 219.0, C | |||
| 13 | 51.2, C | |||
| 14 | 1.21 m | 59.2, CH | H2-15 | C-12, -18 |
| 15 | 1.95 ddd (12.8, 4.4, 2.4); 1.41 m | 30.8, CH2 | H-14, H-16 | C-13 |
| 16 | 3.55 ddd (10.4, 10.4, 4.8) | 72.0, CH | H2-15, H-17 | n. o. |
| 17 | 1.62 m | 53.0, CH | H-16, H-18, H-20 | n. o. |
| 18 | 1.86 m | 59.2, CH | H-17, H-19 | C-13, -16, -19, -25 |
| 19 | 5.31 d (6.0) | 97.1, CH | H-18 | C-18, -20 |
| 20 | 4.10 qd (6.0, 3.2) | 78.1, CH | H-17, H3-26 | n. o. |
| 21 | 0.85 s | 33.2, CH3 | C-3, -4, -5, -22 | |
| 22 | 0.82 s | 21.3, CH3 | C-4, -21 | |
| 23 | 1.06 s | 16.9, CH3 | C-7, -8, -9, -14 | |
| 24 | 0.87 s | 15.6, CH3 | C-10 | |
| 25 | 1.24 s | 15.3, CH3 | C-12, -13, -14, -18 | |
| 26 | 1.38 d (6.0) | 20.5, CH3 | H-20 | C-17, -20 |
| Compounds | Cell Lines IC50 (μM) | |||||
|---|---|---|---|---|---|---|
| MOLT-4 | SUP-T1 | DLD-1 | LNCaP | T-47D | MCF7 | |
| 1 | NA b | NA | 10.9 | 24.3 | NA | NA |
| 2 | 14.9 | 27.1 | 8.5 | NA | 32.2 | 23.0 |
| 3 | 18.5 | NA | 15.0 | NA | NA | NA |
| 4 | 12.8 | 31.6 | 7.9 | 21.5 | 20.2 | NA |
| 5 | 14.0 | 31.1 | 7.2 | NA | 22.7 | 24.3 |
| Doxorubicin a | 0.02 | 0.09 | 0.64 | 0.02 | 0.09 | 0.79 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
3.4. MTT Antiproliferative Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Martinez, A.; Duque, C.; Sato, N.; Tanaka, R.; Fujimoto, Y. (18R)-Variabilin from the sponge Ircinia felix. Nat. Prod. Lett. 1995, 6, 1–6. [Google Scholar] [CrossRef]
- Martinez, A.; Duque, C.; Hara, N.; Fujimoto, Y. Variabilin 11-methyloctadecanoate, a branched-chain acid ester of furanosesterterpene tetronic acid, from the sponge Ircinia felix. Nat. Prod. Lett. 1995, 6, 281–284. [Google Scholar] [CrossRef]
- Martinez, A.; Duque, C.; Sato, N.; Fujimoto, Y. (8Z,13Z,20Z)-Strobilinin and (7Z,13Z,20Z)-felixinin: New furanosesterterpene tetronic acids from marine sponges of the genus Ircinia. Chem. Pharm. Bull. 1997, 45, 181–184. [Google Scholar] [CrossRef]
- Martínez, A.; Duque, C.; Fujimoto, Y. Novel fatty acid esters of (7E, 12E, 18R, 20Z)-variabilin from the marine sponge Ircinia felix. Lipids 1997, 32, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Granato, A.C.; de Oliveira, J.H.H.L.; Seleghim, M.H.R.; Berlinck, R.G.S.; Macedo, M.L.; Ferreira, A.G.; da Rocha, R.M.; Hajdu, E.; Peixinho, S.; Pessoa, C.O.; et al. Produtos naturais da ascídia Botrylloides giganteum, das esponjas Verongula gigantea, Ircinia felix, Cliona delitrix e do nudibrânquio Tambja eliora, da costa do Brasil. Quim. Nova 2005, 28, 192–198. [Google Scholar] [CrossRef]
- Waddell, B.; Pawlik, J.R. Defenses of Caribbean sponges against invertebrate predators. II. Assays with sea stars. Mar. Ecol. Prog. Ser. 2000, 195, 133–144. [Google Scholar] [CrossRef]
- Duque, C.; Bonilla, A.; Bautista, E.; Zea, S. Exudation of low molecular weight compounds (thiobismethane, methyl isocyanide, and methyl isothiocyanate) as a possible chemical defense mechanism in the marine sponge Ircinia felix. Biochem. Syst. Ecol. 2001, 29, 459–467. [Google Scholar] [CrossRef]
- Pawlik, J.R.; McFall, G.; Zea, S. Does the odor from sponges of the genus Ircinia protect them from fish predators? J. Chem. Ecol. 2002, 28, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Freeman, C.J.; Gleason, D.F. Chemical defenses, nutritional quality, and structural components in three sponges: Ircinia felix, I. campana, and Aplysina fulva. Mar. Biol. 2010, 157, 1083–1093. [Google Scholar] [CrossRef]
- Freeman, C.J.; Gleason, D.F. Does concentrating chemical defenses within specific regions of marine sponges results in enhanced protection from predators? Hydrobiologia 2012, 687, 289–297. [Google Scholar] [CrossRef]
- Gómez-Guiñán, Y.; Hidalgo, J.; Jiménez, M.; Salcedo, J. Actividad antibacteriana de extractos orgánicos de Penicillium sp. (Moniliales) aislados de la esponja Ircinia felix (Demospongiae). Rev. Biol. Trop. 2003, 51, 141–147. [Google Scholar] [PubMed]
- Sepčić, K.; Kauferstein, S.; Mebs, D.; Turk, T. Biological activities of aqueous and organic extracts from tropical marine sponges. Mar. Drugs 2010, 8, 1550–1566. [Google Scholar] [CrossRef] [PubMed]
- González, M.A. Scalarane sesterterpenoids. Curr. Bioact. Comp. 2010, 6, 178–206. [Google Scholar] [CrossRef]
- Fontana, A.; Mollo, E.; Ortea, J.; Gavagnin, M.; Cimino, G. Scalarane and homoscalarane compounds from the nudibranchs Glossodoris sedan and Glossodoris dalli: Chemical and biological properties. J. Nat. Prod. 2000, 63, 527–530. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.C.; Tanaka, J.; de Voogd, N.; Higa, T. New scalarane class sesterterpenes from an Indonesian sponge, Phyllospongia sp. J. Nat. Prod. 2002, 65, 1838–1842. [Google Scholar] [CrossRef] [PubMed]
- Pronzato, R.; Malva, R.; Manconi, R. The taxonomic status of Ircinia fasciculata, Ircinia felix, and Ircinia variabilis (Dictyoceratida, Irciniidae). Boll. Mus. Ist. Biol. Univ. Genova 2004, 68, 553–563. [Google Scholar]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar] [PubMed]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar] [PubMed]
- Lu, M.-C.; Hwang, S.-L.; Chang, F.-R.; Chen, Y.-H.; Chang, T.-T.; Hung, C.-S.; Wang, C.-L.; Chu, Y.-H.; Pan, S.-H.; Wu, Y.-C. Immunostimulatory effect of Antrodia camphorata extract on functional maturation of dendritic cells. Food Chem. 2009, 113, 1049–1057. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, Y.-Y.; Lu, M.-C.; Wang, L.-H.; Chen, J.-J.; Fang, L.-S.; Wu, Y.-C.; Sung, P.-J. New Scalarane Sesterterpenoids from the Formosan Sponge Ircinia felix. Mar. Drugs 2015, 13, 4296-4309. https://doi.org/10.3390/md13074296
Lai Y-Y, Lu M-C, Wang L-H, Chen J-J, Fang L-S, Wu Y-C, Sung P-J. New Scalarane Sesterterpenoids from the Formosan Sponge Ircinia felix. Marine Drugs. 2015; 13(7):4296-4309. https://doi.org/10.3390/md13074296
Chicago/Turabian StyleLai, Ya-Yuan, Mei-Chin Lu, Li-Hsueh Wang, Jih-Jung Chen, Lee-Shing Fang, Yang-Chang Wu, and Ping-Jyun Sung. 2015. "New Scalarane Sesterterpenoids from the Formosan Sponge Ircinia felix" Marine Drugs 13, no. 7: 4296-4309. https://doi.org/10.3390/md13074296
APA StyleLai, Y.-Y., Lu, M.-C., Wang, L.-H., Chen, J.-J., Fang, L.-S., Wu, Y.-C., & Sung, P.-J. (2015). New Scalarane Sesterterpenoids from the Formosan Sponge Ircinia felix. Marine Drugs, 13(7), 4296-4309. https://doi.org/10.3390/md13074296









