SAR of Sponge-Inspired Hemibastadin Congeners Inhibiting Blue Mussel PhenolOxidase
Abstract
:1. Introduction
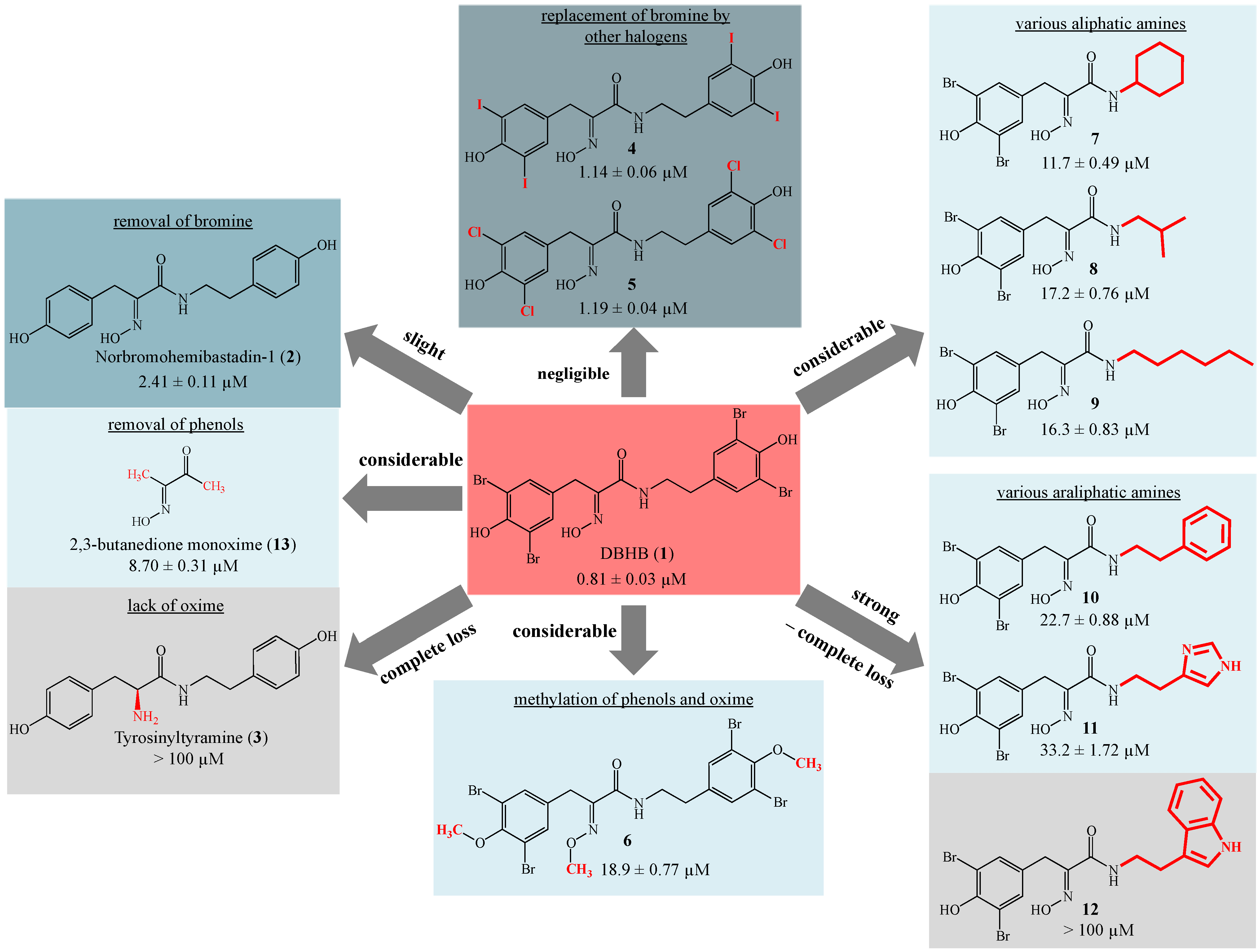
2. Results and Discussion

3. Experimental Section
3.1. General Experimental Procedures
3.2. Blue Mussel PO Inhibition Assay
3.3. Synthetic Procedures
3.3.1. l-Tyrosine-methyl Ester (15)
3.3.2. (E)-Methyl 2-(hydroxyimino)-3-(4-hydroxyphenyl)propanoate (16)
3.3.3. (E)-Methyl 3-(3,5-dibromo-4-hydroxyphenyl)-2-(hydroxyimino)propanoate (17)
3.3.4. 5,5′-Dibromohemibastadin-1 (1) and Norbromohemibastadin-1 (2)
3.3.5. Tyrosinyltyramine (3)
3.3.6. Tetraiodo-norbromohemibastadin-1 (4)
3.3.7. Tetrachloro-norbromohemibastadin-1 (5)
3.3.8. Tri-O-methyl-5,5′-dibromohemibastadin-1 (6)
3.3.9. Amides Resulting from Liquid Primary Amines (7–10)
3.3.10. Amides Resulting from Solid Primary Amines (11, 12)
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bao, V.W.W.; Leung, K.M.Y.; Qiu, J.-W.; Lam, M.H.W. Acute toxicities of five commonly used antifouling booster biocides to selected subtropical and cosmopolitan marine species. Mar. Pollut. Bull. 2011, 62, 1147–1151. [Google Scholar]
- Trepos, R.; Pinori, E.; Jonsson, P.R.; Berglin, M.; Svenson, J.; Coutinho, R.; Lausmaa, J.; Hellio, C. Innovative approaches for the development of new copper-free marine antifouling paints. J. Ocean Technol. 2015, in press. [Google Scholar]
- Hellio, C.; Bourgougnon, N.; Gal, Y.L. Phenoloxidase (E.C. 1.14.18.1) from the byssus gland of Mytilus edulis: Purification, partial characterization and application for screening products with potential antifouling activities. Biofouling 2000, 16, 235–244. [Google Scholar] [CrossRef]
- Numata, K.; Baker, P.J. Synthesis of adhesive peptides similar to those found in blue mussel (Mytilus edulis) using papain and tyrosinase. Biomacromolecules 2014, 15, 3206–3212. [Google Scholar] [CrossRef] [PubMed]
- Aladaileh, S.; Rodney, P.; Nair, S.V.; Raftos, D.A. Characterization of phenoloxidase activity in sydney rock oysters (Saccostrea glomerata). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2007, 148, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Silverman, H.G.; Roberto, F.F. Understanding marine mussel adhesion. Mar. Biotechnol. 2007, 9, 661–681. [Google Scholar] [CrossRef] [PubMed]
- Ortlepp, S.; Sjögren, M.; Dahlström, M.; Weber, H.; Ebel, R.; Edrada, R.; Thoms, C.; Schupp, P.; Bohlin, L.; Proksch, P. Antifouling activity of bromotyrosine-derived sponge metabolites and synthetic analogues. Mar. Biotechnol. 2007, 9, 776–785. [Google Scholar] [CrossRef]
- Bayer, M.; Hellio, C.; Maréchal, J.-P.; Frank, W.; Lin, W.; Weber, H.; Proksch, P. Antifouling bastadin congeners target mussel phenoloxidase and complex copper(II) ions. Mar. Biotechnol. 2011, 13, 1148–1158. [Google Scholar] [CrossRef] [PubMed]
- Ismaya, W.T.; Rozeboom, H.J.; Weijn, A.; Mes, J.J.; Fusetti, F.; Wichers, H.J.; Dijkstra, B.W. Crystal structure of Agaricus bisporus mushroom tyrosinase: Identity of the tetramer subunits and interaction with tropolone. Biochemistry 2011, 50, 5477–5486. [Google Scholar] [CrossRef] [PubMed]
- Boehlow, T.R.; Harburn, J.J.; Spilling, C.D. Approaches to the synthesis of some tyrosine-derived marine sponge metabolites: Synthesis of verongamine and purealidin N. J. Org. Chem. 2001, 66, 3111–3118. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Harayama, Y.; Kamimura, D.; Yoshida, M.; Shibata, T.; Fujiwara, K.; Morimoto, K.; Fujioka, H.; Kita, Y. The synthetic and biological studies of discorhabdins and related compounds. Org. Biomol. Chem. 2011, 9, 4959–4976. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niemann, H.; Hagenow, J.; Chung, M.-Y.; Hellio, C.; Weber, H.; Proksch, P. SAR of Sponge-Inspired Hemibastadin Congeners Inhibiting Blue Mussel PhenolOxidase. Mar. Drugs 2015, 13, 3061-3071. https://doi.org/10.3390/md13053061
Niemann H, Hagenow J, Chung M-Y, Hellio C, Weber H, Proksch P. SAR of Sponge-Inspired Hemibastadin Congeners Inhibiting Blue Mussel PhenolOxidase. Marine Drugs. 2015; 13(5):3061-3071. https://doi.org/10.3390/md13053061
Chicago/Turabian StyleNiemann, Hendrik, Jens Hagenow, Mi-Young Chung, Claire Hellio, Horst Weber, and Peter Proksch. 2015. "SAR of Sponge-Inspired Hemibastadin Congeners Inhibiting Blue Mussel PhenolOxidase" Marine Drugs 13, no. 5: 3061-3071. https://doi.org/10.3390/md13053061
APA StyleNiemann, H., Hagenow, J., Chung, M.-Y., Hellio, C., Weber, H., & Proksch, P. (2015). SAR of Sponge-Inspired Hemibastadin Congeners Inhibiting Blue Mussel PhenolOxidase. Marine Drugs, 13(5), 3061-3071. https://doi.org/10.3390/md13053061





