Ascophyllan Purified from Ascophyllum nodosum Induces Th1 and Tc1 Immune Responses by Promoting Dendritic Cell Maturation
Abstract
:1. Introduction
2. Results and Discussion
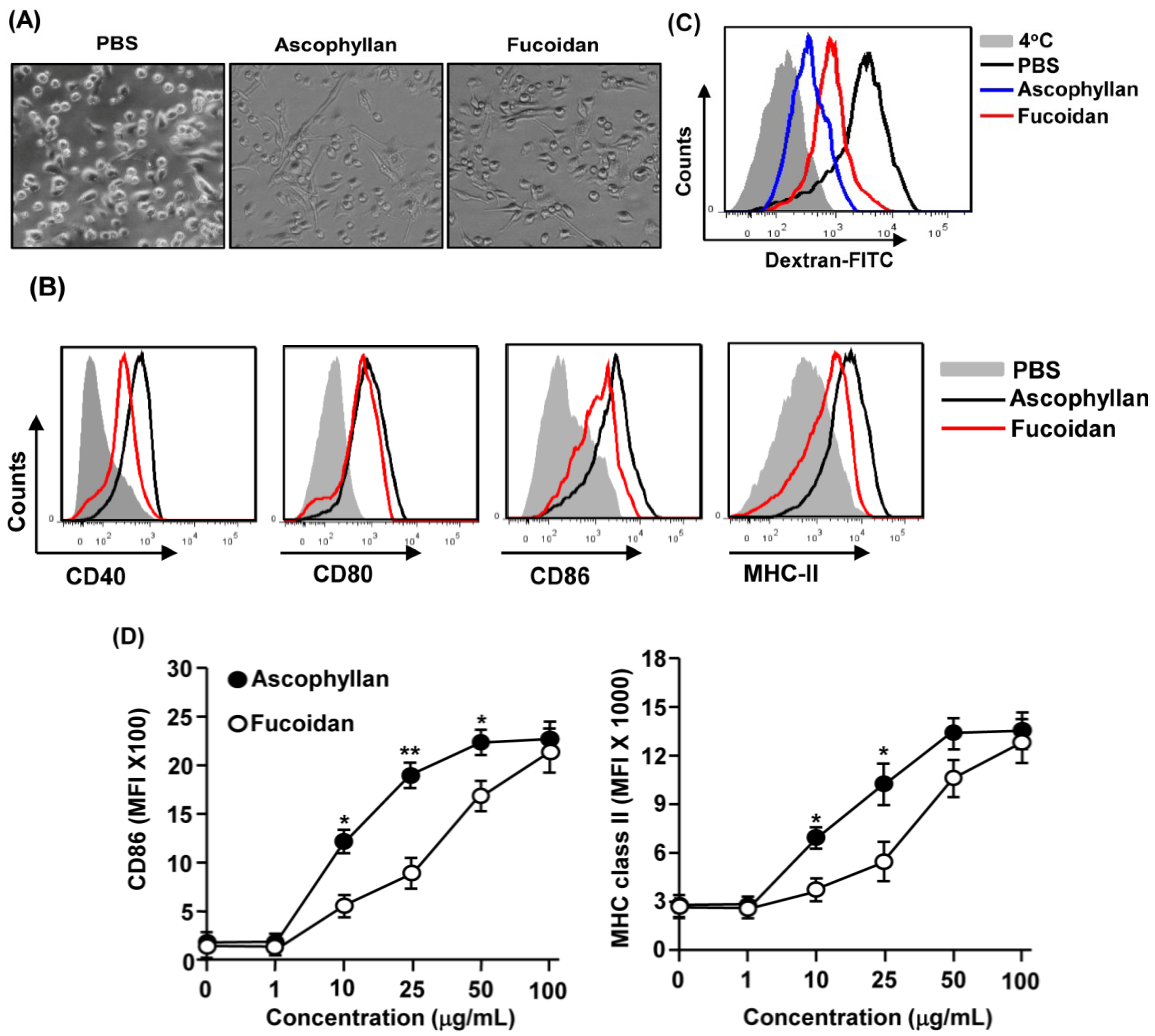
2.1. Ascophyllan Promotes the Activation of BMDCs
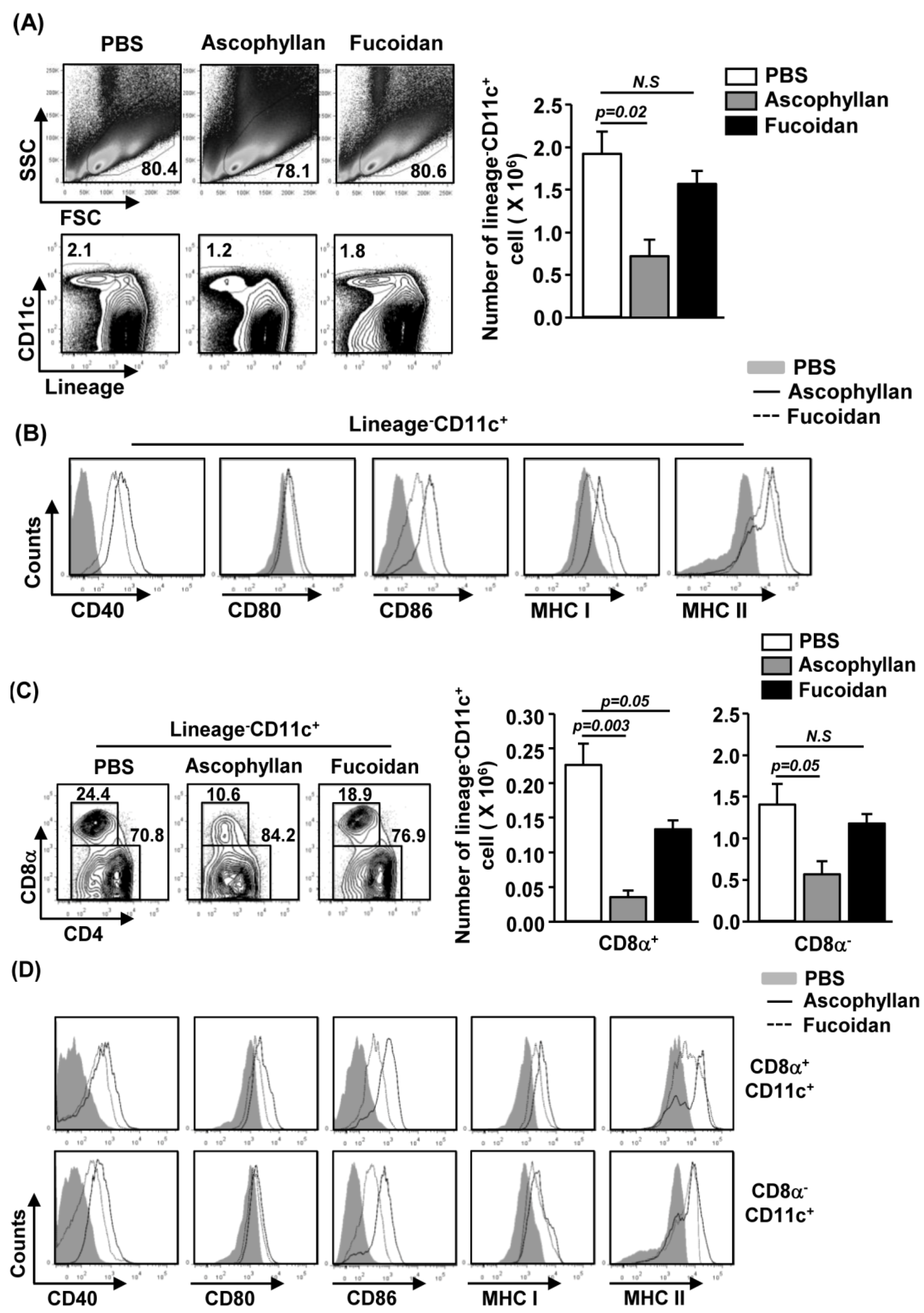
2.2. Ascophyllan Induces the Activation of Spleen DCs in Vivo
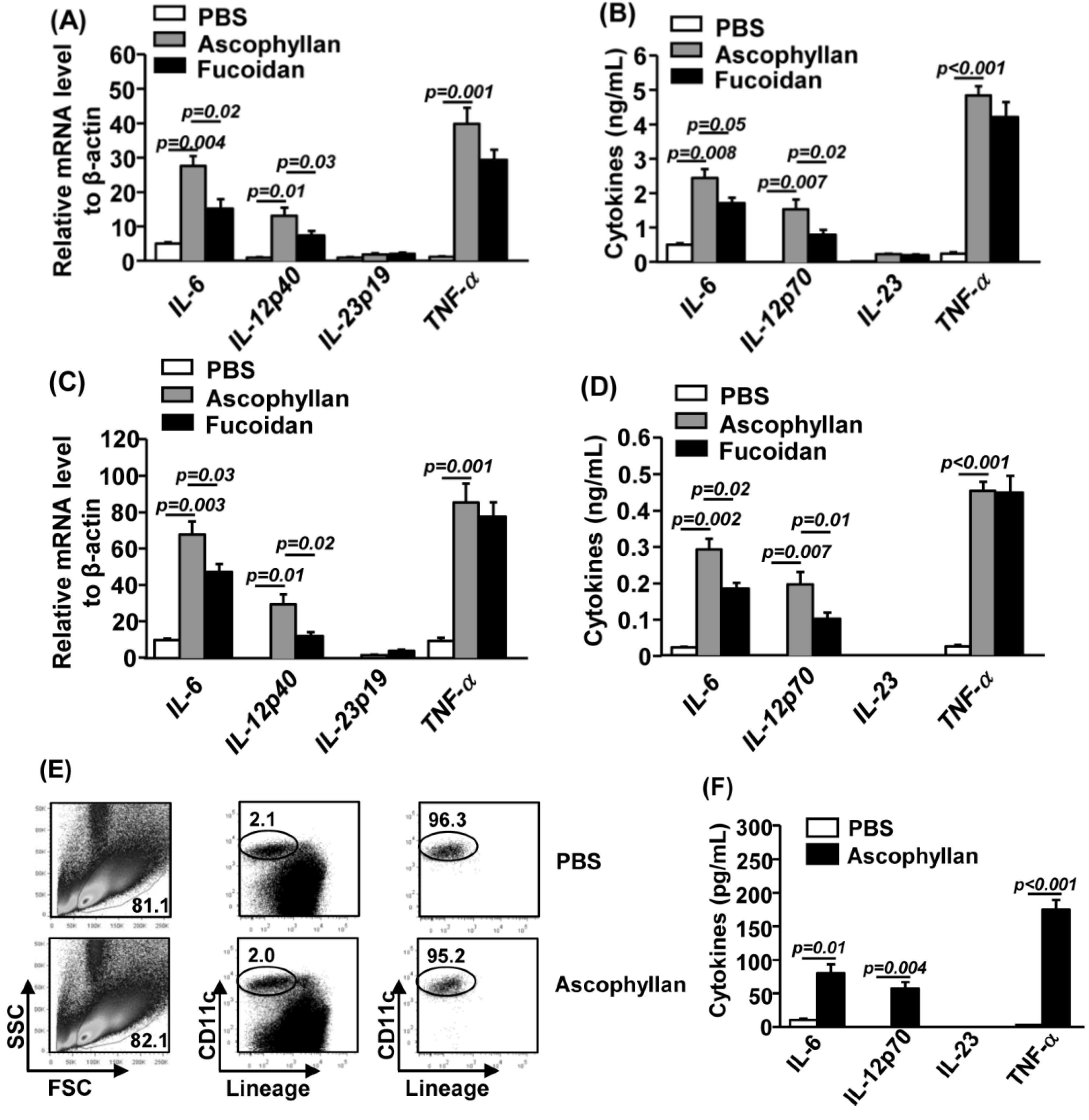
2.3. Ascophyllan Promotes Pro-Inflammatory Cytokine Production from DCs
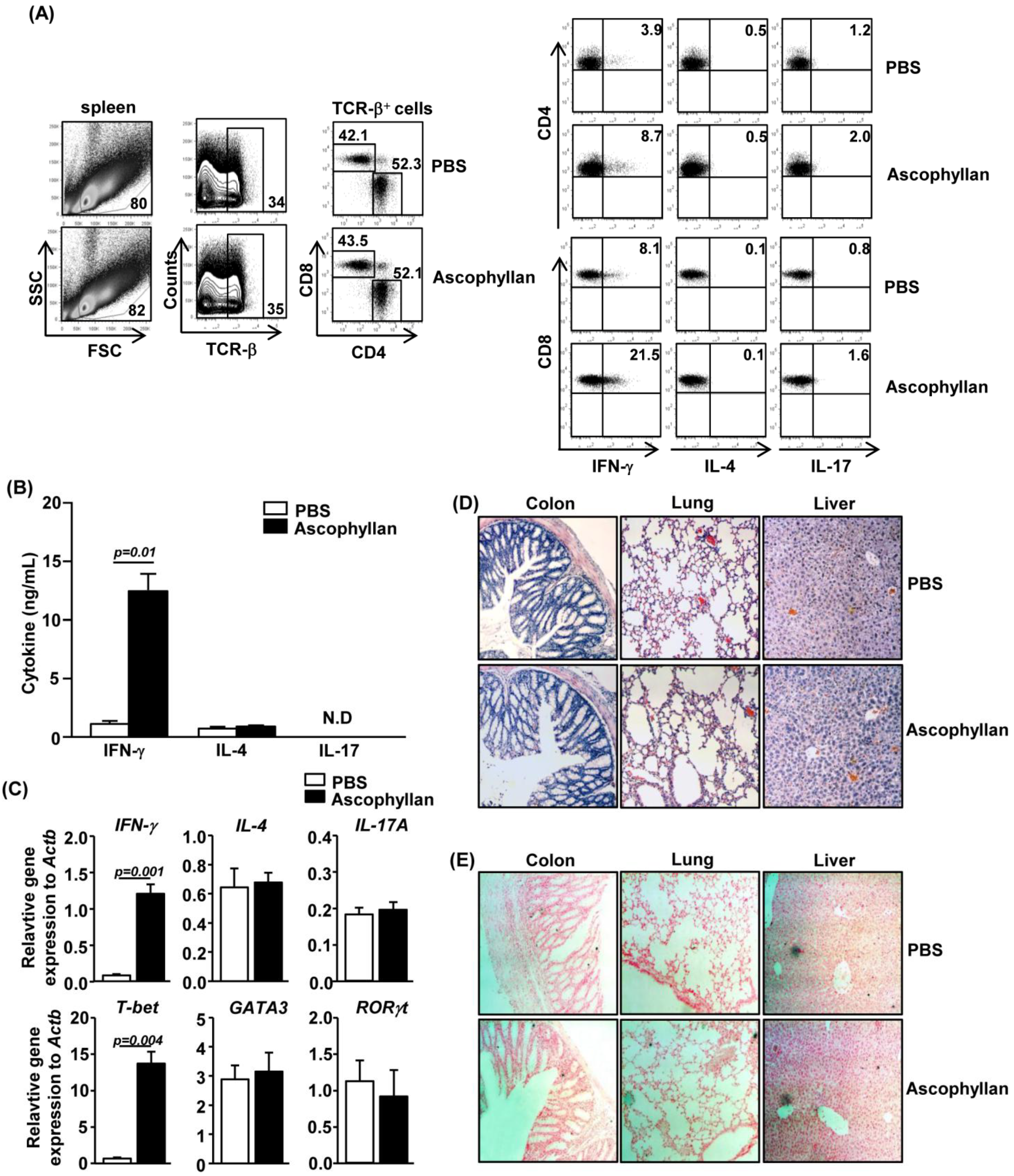
2.4. Ascophyllan Promotes Generation of Th1 and Tc1 Cells in Vivo
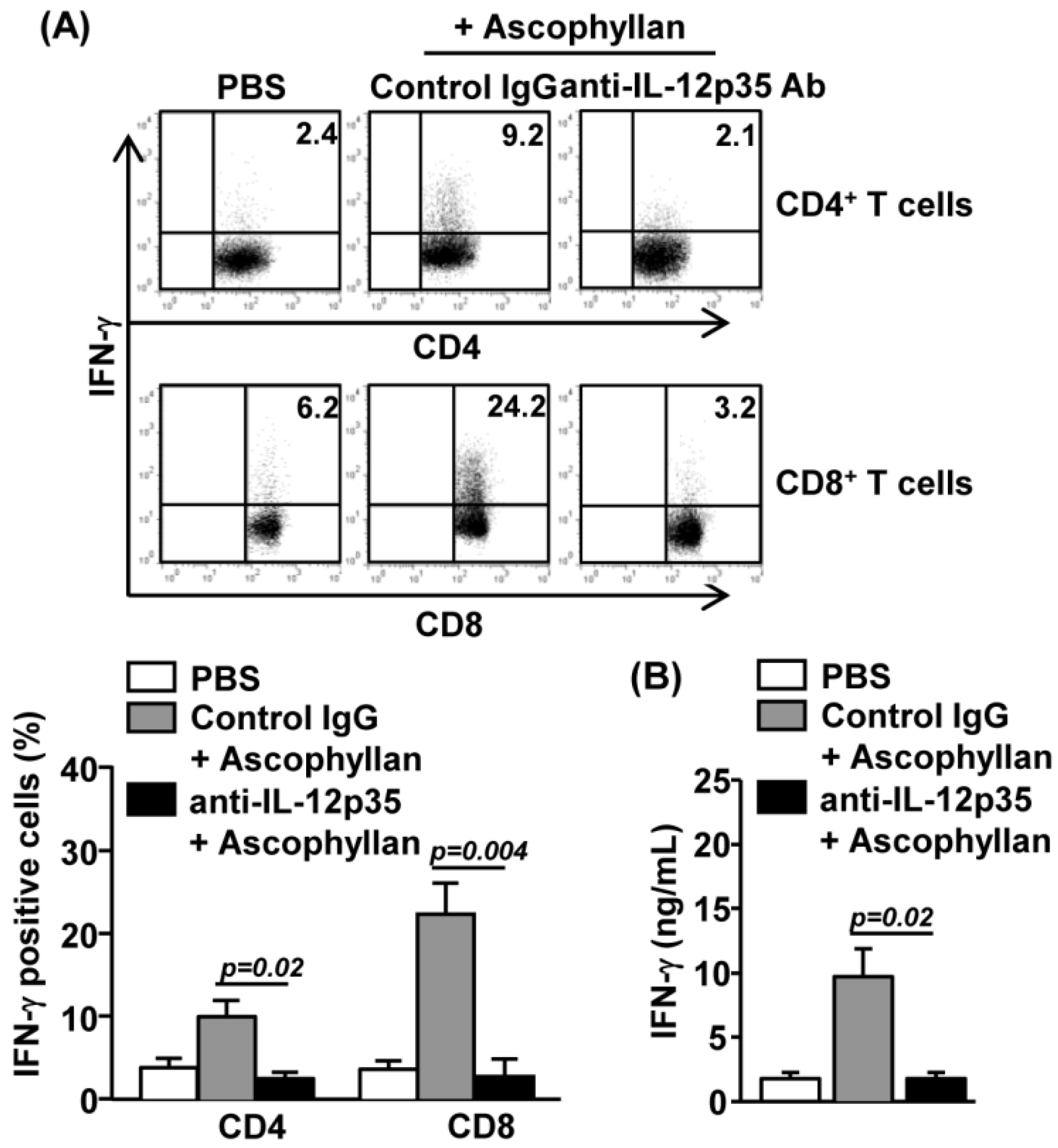
2.5. Ascophyllan-Induced Generation of Th1 and Tc1 is Dependent on IL-12
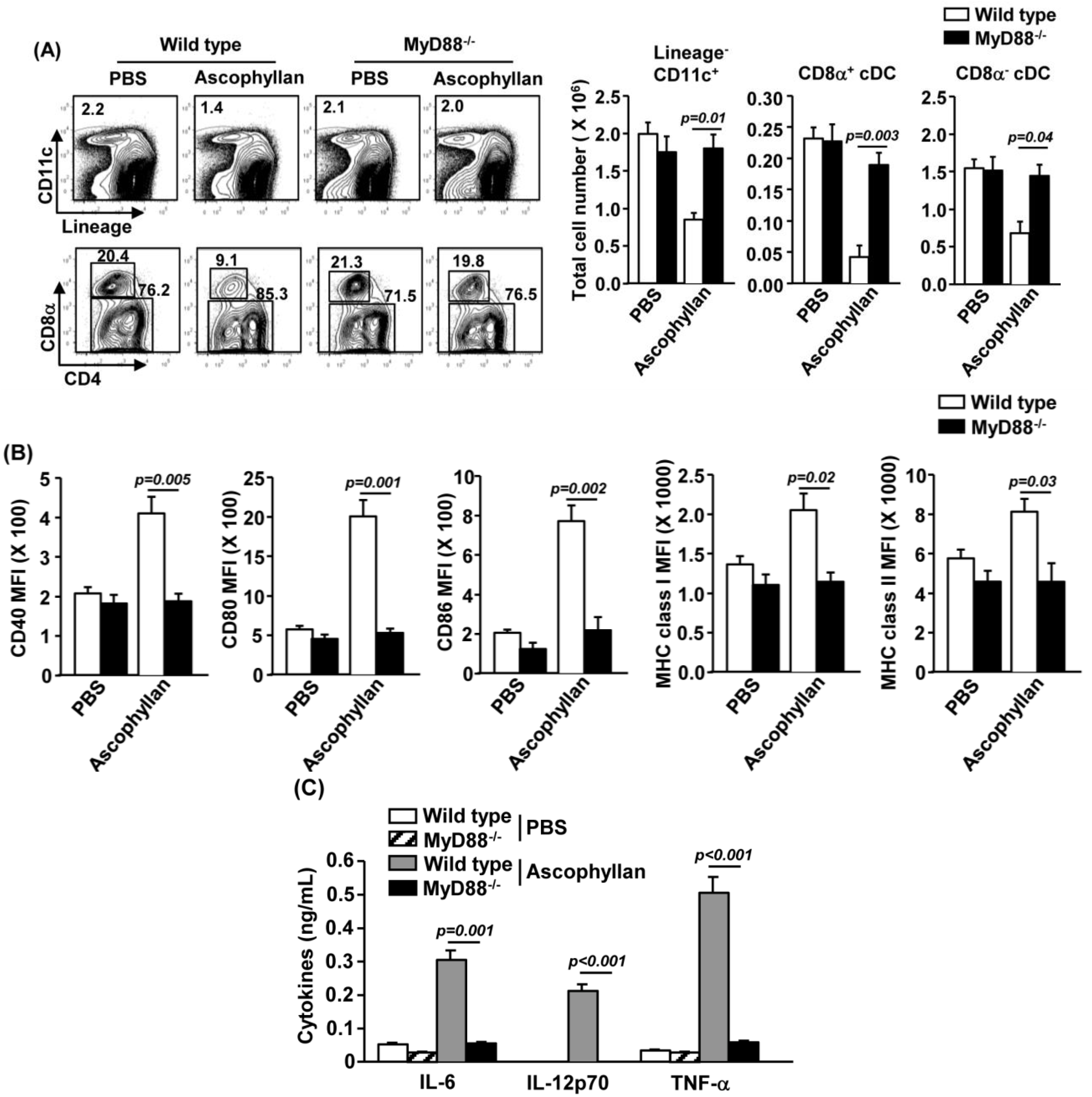
2.6. MyD88 Signaling Pathway is Essential for Ascophyllan-Induced DC Maturation
3. Experimental Section
3.1. Mice
3.2. Chemicals and Cytokines
3.3. Antibodies
3.4. Flow Cytometry Analysis
3.5. In Vitro BMDC Generation
3.6. Spleen cDC Analysis
3.7. Apoptosis Assay
3.8. Ex Vivo T Cell Stimulation and Intracellular Cytokine Staining
3.9. ELISA Assay
3.10. Real-Time qPCR
3.11. Histology and in Situ Apoptosis Detection
3.12. Statistical Analysis
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ramberg, J.E.; Nelson, E.D.; Sinnott, R.A. Immunomodulatory dietary polysaccharides: A systematic review of the literature. Nutr. J. 2010, 9. [Google Scholar] [CrossRef]
- Leung, M.Y.; Liu, C.; Koon, J.C.; Fung, K.P. Polysaccharide biological response modifiers. Immunol. Lett. 2006, 105, 101–114. [Google Scholar]
- Nakayasu, S.; Soegima, R.; Yamaguchi, K.; Oda, T. Biological activities of fucose-containing polysaccharide ascophyllan isolated from the brown alga Ascophyllum nodosum. Biosci. Biotechnol. Biochem. 2009, 73, 961–964. [Google Scholar] [CrossRef]
- Larsen, B.; Haug, A.; Painter, T. Sulphated polysaccharides in brown algae. 3. The native state of dfucoidan in Ascophyllum nodosum and Fucus vesiculosus. Acta Chem. Scand. 1970, 24, 3339–3352. [Google Scholar]
- Jiang, Z.; Okimura, T.; Yamaguchi, K.; Oda, T. The potent activity of sulfated polysaccharide, ascophyllan, isolated from Ascophyllum nodosum to induce nitric oxide and cytokine production from mouse macrophage RAW264.7 cells: Comparison between ascophyllan and fucoidan. Nitric Oxide 2011, 25, 407–415. [Google Scholar]
- Nakano, K.; Kim, D.; Jiang, Z.; Ueno, M.; Okimura, T.; Yamaguchi, K.; Oda, T. Immunostimulatory activities of the sulfated polysaccharide ascophyllan from Ascophyllum nodosum in in vivo and in vitro systems. Biosci. Biotechnol. Biochem. 2012, 76, 1573–1576. [Google Scholar]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar]
- Fujii, S.; Liu, K.; Smith, C.; Bonito, A.J.; Steinman, R.M. The linkage of innate to adaptive immunity via maturing dendritic cells in vivo requires CD40 ligation in addition to antigen presentation and CD80/86 costimulation. J. Exp. Med. 2004, 199, 1607–1618. [Google Scholar]
- Pooley, J.L.; Heath, W.R.; Shortman, K. Cutting edge: Intravenous soluble antigen is presented to CD4 T cells by CD8-dendritic cells, but cross-presented to CD8 T cells by CD8+ dendritic cells. J. Immunol. 2001, 166, 5327–5330. [Google Scholar] [CrossRef]
- Schnorrer, P.; Behrens, G.M.; Wilson, N.S.; Pooley, J.L.; Smith, C.M.; El-Sukkari, D.; Davey, G.; Kupresanin, F.; Li, M.; Maraskovsky, E.; et al. The dominant role of CD8+ dendritic cells in cross-presentation is not dictated by antigen capture. Proc. Natl. Acad. Sci. USA 2006, 103, 10729–10734. [Google Scholar] [CrossRef]
- Shortman, K.; Heath, W.R. The CD8+ dendritic cell subset. Immunol. Rev. 2010, 234, 18–31. [Google Scholar] [CrossRef]
- Villadangos, J.A.; Schnorrer, P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nat. Rev. Immunol. 2007, 7, 543–555. [Google Scholar] [CrossRef]
- Watts, C. The exogenous pathway for antigen presentation on major histocompatibility complex class II and CD1 molecules. Nat. Immunol. 2004, 5, 685–692. [Google Scholar]
- Moser, M.; Murphy, K.M. Dendritic cell regulation of TH1-TH2 development. Nat. Immunol. 2000, 1, 199–205. [Google Scholar]
- Kurts, C.; Kosaka, H.; Carbone, F.R.; Miller, J.F.; Heath, W.R. Class I-restricted cross-presentation of exogenous self-antigens leads to deletion of autoreactive CD8(+) T cells. J. Exp. Med. 1997, 186, 239–245. [Google Scholar] [CrossRef]
- Tan, J.K.; O’Neill, H.C. Maturation requirements for dendritic cells in T cell stimulation leading to tolerance versus immunity. J. Leukoc. Biol. 2005, 78, 319–324. [Google Scholar]
- Fitton, J.H. Therapies from fucoidan; multifunctional marine polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar]
- Kwak, J.Y. Fucoidan as a marine anticancer agent in preclinical development. Mar. Drugs 2014, 12, 851–870. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar]
- Kim, M.H.; Joo, H.G. Immunostimulatory effects of fucoidan on bone marrow-derived dendritic cells. Immunol. Lett. 2008, 115, 138–143. [Google Scholar]
- Yang, M.; Ma, C.; Sun, J.; Shao, Q.; Gao, W.; Zhang, Y.; Li, Z.; Xie, Q.; Dong, Z.; Qu, X. Fucoidan stimulation induces a functional maturation of human monocyte-derived dendritic cells. Int. Immunopharmacol. 2008, 8, 1754–1760. [Google Scholar] [CrossRef]
- Jin, J.O.; Park, H.Y.; Xu, Q.; Park, J.I.; Zvyagintseva, T.; Stonik, V.A.; Kwak, J.Y. Ligand of scavenger receptor class A indirectly induces maturation of human blood dendritic cells via production of tumor necrosis factor-alpha. Blood 2009, 113, 5839–5847. [Google Scholar] [CrossRef]
- Jiang, Z.; Okimura, T.; Yokose, T.; Yamasaki, Y.; Yamaguchi, K.; Oda, T. Effects of sulfated fucan, ascophyllan, from the brown Alga Ascophyllum nodosum on various cell lines: A comparative study on ascophyllan and fucoidan. J. Biosci. Bioeng. 2010, 110, 113–117. [Google Scholar] [CrossRef]
- Vremec, D.; Pooley, J.; Hochrein, H.; Wu, L.; Shortman, K. CD4 and CD8 expression by dendritic cell subtypes in mouse thymus and spleen. J. Immunol. 2000, 164, 2978–2986. [Google Scholar] [CrossRef]
- Takeda, K.; Akira, S. TLR signaling pathways. Semin. Immunol. 2004, 16, 3–9. [Google Scholar]
- Gordon, S. Pattern recognition receptors: Doubling up for the innate immune response. Cell 2002, 111, 927–930. [Google Scholar] [CrossRef]
- Calzas, C.; Goyette-Desjardins, G.; Lemire, P.; Gagnon, F.; Lachance, C.; van Calsteren, M.R.; Segura, M. Group B Streptococcus and Streptococcus suis capsular polysaccharides induce chemokine production by dendritic cells via Toll-like receptor 2- and MyD88-dependent and -independent pathways. Infect. Immun. 2013, 81, 3106–3118. [Google Scholar] [CrossRef]
- Lin, C.Y.; Lu, M.C.; Su, J.H.; Chu, C.L.; Shiuan, D.; Weng, C.F.; Sung, P.J.; Huang, K.J. Immunomodulatory effect of marine cembrane-type diterpenoids on dendritic cells. Mar. Drugs 2013, 11, 1336–1350. [Google Scholar]
- Wallner, S.; Lutz-Nicoladoni, C.; Tripp, C.H.; Gastl, G.; Baier, G.; Penninger, J.M.; Stoitzner, P.; Wolf, D. The role of the E3 ligase Cbl-B in murine dendritic cells. PLoS One 2013, 8, e65178. [Google Scholar]
- Ochi, A.; Nguyen, A.H.; Bedrosian, A.S.; Mushlin, H.M.; Zarbakhsh, S.; Barilla, R.; Zambirinis, C.P.; Fallon, N.C.; Rehman, A.; Pylayeva-Gupta, Y.; et al. MyD88 inhibition amplifies dendritic cell capacity to promote pancreatic carcinogenesis via Th2 cells. J. Exp. Med. 2012, 209, 1671–1687. [Google Scholar] [CrossRef]
- Macedo, G.C.; Magnani, D.M.; Carvalho, N.B.; Bruna-Romero, O.; Gazzinelli, R.T.; Oliveira, S.C. Central role of MyD88-dependent dendritic cell maturation and proinflammatory cytokine production to control Brucella abortus infection. J. Immunol. 2008, 180, 1080–1087. [Google Scholar] [CrossRef]
- Sathe, P.; Pooley, J.; Vremec, D.; Mintern, J.; Jin, J.O.; Wu, L.; Kwak, J.Y.; Villadangos, J.A.; Shortman, K. The acquisition of antigen cross-presentation function by newly formed dendritic cells. J. Immunol. 2011, 186, 5184–5192. [Google Scholar]
- Vremec, D.; O’Keeffe, M.; Wilson, A.; Ferrero, I.; Koch, U.; Radtke, F.; Scott, B.; Hertzog, P.; Villadangos, J.; Shortman, K. Factors determining the spontaneous activation of splenic dendritic cells in culture. Innate Immun. 2011, 17, 338–352. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, W.; Du, J.-Y.; Jiang, Z.; Okimura, T.; Oda, T.; Yu, Q.; Jin, J.-O. Ascophyllan Purified from Ascophyllum nodosum Induces Th1 and Tc1 Immune Responses by Promoting Dendritic Cell Maturation. Mar. Drugs 2014, 12, 4148-4164. https://doi.org/10.3390/md12074148
Zhang W, Du J-Y, Jiang Z, Okimura T, Oda T, Yu Q, Jin J-O. Ascophyllan Purified from Ascophyllum nodosum Induces Th1 and Tc1 Immune Responses by Promoting Dendritic Cell Maturation. Marine Drugs. 2014; 12(7):4148-4164. https://doi.org/10.3390/md12074148
Chicago/Turabian StyleZhang, Wei, Jiang-Yuan Du, Zedong Jiang, Takasi Okimura, Tatsuya Oda, Qing Yu, and Jun-O Jin. 2014. "Ascophyllan Purified from Ascophyllum nodosum Induces Th1 and Tc1 Immune Responses by Promoting Dendritic Cell Maturation" Marine Drugs 12, no. 7: 4148-4164. https://doi.org/10.3390/md12074148
APA StyleZhang, W., Du, J.-Y., Jiang, Z., Okimura, T., Oda, T., Yu, Q., & Jin, J.-O. (2014). Ascophyllan Purified from Ascophyllum nodosum Induces Th1 and Tc1 Immune Responses by Promoting Dendritic Cell Maturation. Marine Drugs, 12(7), 4148-4164. https://doi.org/10.3390/md12074148




