Antimalarial Activity of Axidjiferosides, New β-Galactosylceramides from the African Sponge Axinyssa djiferi
Abstract
:Abbreviations
| COSY | homonuclear correlation spectroscopy |
| FAME | fatty acid methyl esters |
| LCB | long chain base |
| GC-MS | gas chromatography-mass spectrometry |
| GL | glycolipid(s) |
| GSL | glycosphingolipid(s) |
| amu | atomic mass unit |
| HMBC | heteronuclear multiple bond coherence |
| HSQC | heteronuclear single quantum coherence |
| IC50 | 50% inhibitory concentration |
| ESI-MS | electrospray ionization mass spectrometry |
| TLC | thin layer chromatography |
| dw | dry weight |
| MEC | minimum effective concentration |
1. Introduction
2. Results and Discussion
2.1. Glycolipid Isolation and Structure Determination
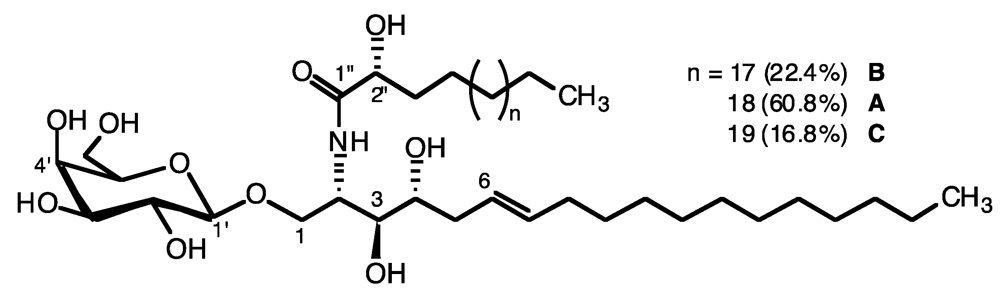
| Position | δH ppm, mult., J in Hz | δC ppm |
|---|---|---|
| 1a | 3.88 (dd, J = 10.6/3.1) | 66.19 |
| 1b | 3.70 (dd, J = 10.6/2.7) | - |
| 2 | 4.33 (m) | 48.09 |
| 2-NH | 6.81 (d, J = 9.0) | - |
| 3 | 5.12 (m) | 71.95 |
| 4 | 4.95 (m) | 72.64 |
| 5 | 2.40 (m) | 20.75 |
| 6 | 5.50 (dt, J = 6.8/15.0) | 124.03 |
| 7 | 5.28 (dt, J = 7.0/15.0) | 134.58 |
| 8 | 1.85 (m) | 24.86 |
| terminal methyl | 0.87 (t, J = 6.8) | 19.23 |
| acetates | 2.05/2.25/2.17/2.06/2.11/1.99/2.07 (7 s) | 169.44/169.86/170.0/170.06/170.18/170.36/170.74 |
| CH2 (C9–C17) | 1.27 (m) | 29.21–30.06 |
| CH2 (C4″–C20″) | 1.32 (m) | 29.21–30.06 |
| 1′ | 4.47 (d, J = 7.8) | 100.66 |
| 2′ | 5.17 (m) | 73.88 |
| 3′ | 5.02 (dd, J = 10.4/3.3) | 70.79 |
| 4′ | 5.38 (d, J = 3.3) | 66.96 |
| 5′ | 3.95 (t, J = 6.7) | 70.79 |
| 6′ | 4.15 (d, J = 6.7) | 61.06 |
| 1″ | - | 171.08 |
| 2″ | 5.14 (t, J = 3.6) | 68.6 |
| 3″ | 1.31 (m) | 34.41 |
2.2. Antimalarial, Antileishmanial, Antitrypanosomal and Antiproliferative Activity
| Plasmodium falciparum FCB1 | 0.53 ± 0.2 |
|---|---|
| Leishmania donovani | IC50 > 100 |
| NSCLC-N6 | IC50 > 35 |
| A 549 | IC50 > 35 |
| KMS-11 | IC50 > 60 |
| GBM | IC50 > 60 |
| HCT-116 | IC50 > 60 |
| Trypanosoma brucei | MEC > 100 |
3. Experimental Section
3.1. General Procedures
3.2. Animal Material

3.3. Lipid Extraction and Axidjiferoside Isolation
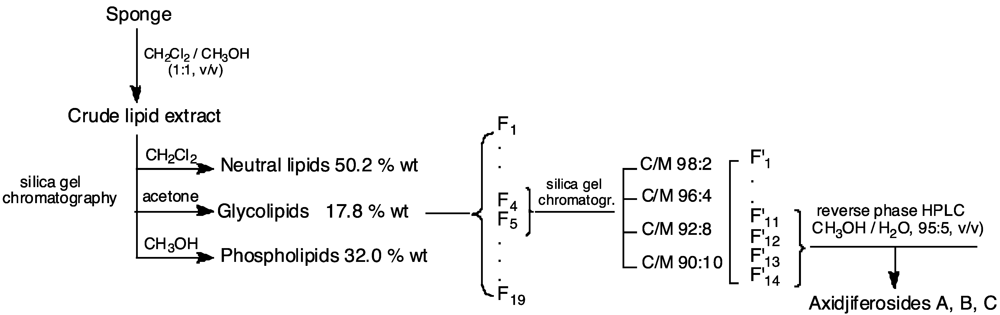
3.4. Acid Methanolysis of Axidjiferosides
3.5. Antiplasmodial Activity of Axidjiferosides
3.6. Antileishmanial Activity of Axidjiferosides
3.7. Antitrypanosomal Activity of Axidjiferosides
3.8. Antiproliferative Activity of Axidjiferosides
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Tuteja, R. Malaria—An overview. FEBS J. 2007, 274, 4670–4609. [Google Scholar]
- World Health Organization, World Malaria Report 2011; WHO Press: Geneva, Switzerland, 2011; pp. 1–248.
- Moorthy, V.; Smith, P.G.; Kieny, M.P. A vaccine against malaria: A substantial step forward. Lancet 2009, 373, 1411–1412. [Google Scholar] [CrossRef]
- Nayyar, G.M.L.; Breman, J.G.; Newton, P.N. Poor-quality antimalarial drugs in southeast Asia and Africa. Lancet 2012, 12, 488–496. [Google Scholar]
- Bell, D.R.; Jorgensen, P.; Christophel, E.M.; Palmer, K.L. Malaria risk: Estimation of the malaria burden. Nature 2005, 437, E3–E4. [Google Scholar]
- Petersen, I.; Eastman, R.; Lanzer, M. Drug-resistant malaria: Molecular mechanisms and implications for public health. FEBS Lett. 2011, 585, 1551–1562. [Google Scholar] [CrossRef]
- Fidock, D.A.; Rosenthal, P.J.; Croft, S.L.; Brun, R.; Nwaka, S. Antimalarial drug discovery: Efficacy models for compound screening. Nat. Rev. Drug Discov. 2004, 3, 509–520. [Google Scholar] [CrossRef]
- Grellier, P.; Deregnaucourt, C.; Florent, I. Advances in Antimalarial Drug Evaluation and New Targets for Antimalarials. In Malaria Parasites; Omolade, O., Ed.; InTech Europe: Rijeka, Croatia, 2012; pp. 321–350. [Google Scholar]
- Kaur, K.; Jain, M.; Kaur, T.; Jain, R. Antimalarials from nature. Bioorg. Med. Chem. 2009, 17, 3229–3256. [Google Scholar] [CrossRef]
- Gademann, K.; Kobylinska, J. Antimalarial natural products of marine and freshwater origin. Chem. Rec. 2009, 9, 187–198. [Google Scholar] [CrossRef]
- Fattorusso, E.; Taglialatela-Scafati, O. Marine antimalarials. Mar. Drugs 2009, 7, 130–152. [Google Scholar] [CrossRef]
- De Mesquita, M.L.; Grellier, P.; Blond, A.; Brouard, J.P.; de Paula, J.E.; Espinola, L.S.; Mambu, L. New ether diglycosides from Matayba guianensis with antiplasmodial activity. Bioorg. Med. Chem. 2005, 13, 4499–4503. [Google Scholar] [CrossRef]
- Gonzalez-Aseguinolaza, G.; de Oliveira, C.; Tomaska, M.; Hong, S.; Bruna Romero, O.; Nakayama, T.; Taniguchi, M.; Bendelac, A.; van Kaer, L.; Koezuka, Y.; et al. α-Galactosylceramide-activated Vα14 natural killer T cells mediate protection against murine malaria. Proc. Natl. Acad. Sci. USA 2000, 97, 8461–8466. [Google Scholar] [CrossRef]
- Laurent, D.; Pietra, F. Antiplasmodial marine natural products in the perspective of current chemotherapy and prevention of malaria—A review. Mar. Biotechnol. 2006, 8, 433–447. [Google Scholar] [CrossRef]
- Kornprobst, J.M. Encyclopedia of Marine Natural Products; Wiley-Blackwell: Weinheim, Germany, 2010; Volume 2, pp. 577–582. [Google Scholar]
- Fattorusso, E.; Mangoni, A. Marine glycolipids. Prog. Chem. Org. Nat. Prod. 1997, 72, 215–301. [Google Scholar]
- Jin, W.; Rinehart, K.L.; Jares-Erijman, E.A. Ophidiacerebrosides: Cytotoxic glycosphingolipids containing a novel sphingosine from a sea star. J. Org. Chem. 1994, 59, 144–147. [Google Scholar] [CrossRef]
- Costantino, V.; Fattorusso, E.; Mangoni, A.; di Rosa, M.; Ianaro, A. Glycolipids from sponges. 6. Plakoside A and B, two unique prenylated glycosphingolipids with immunosuppressive activity from the marine sponge Plakortis simplex. J. Am. Chem. Soc. 1997, 119, 12465–12470. [Google Scholar]
- Costantino, V.; Fattorusso, E.; Mangoni, A. Glycolipids from sponges. Part 9: Plakoside C and D, two further prenylated glycosphingolipids from the marine sponge Ectyoplasia ferox. Tetrahedron 2000, 56, 5953–5957. [Google Scholar]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A.; Freigang, S.; Teyton, L. Corrugoside: A new immunostimulatory α-galactoglycosphingolipid from the marine sponge Axinella corrugata. Bioorg. Med. Chem. 2008, 16, 2077–2085. [Google Scholar] [CrossRef]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A.; Teta, R. Amphiceramide A and B, novel glycosphingolipids from the marine sponge Amphimedon compressa. Eur. J. Org. Chem. 2009, 2009, 2112–2119. [Google Scholar] [CrossRef]
- Farokhi, F.; Wielgosz-Collin, G.; Robic, A.; Debitus, C.; Malleter, M.; Roussakis, C.; Kornprobst, J.M.; Barnathan, G. Antiproliferative activity against human non-small cell lung cancer of two O-alkyl-diglycosylglycerols from the marine sponges Myrmekioderma dendyi and Trikentrion laeve. Eur. J. Med. Chem. 2012, 49, 406–410. [Google Scholar] [CrossRef]
- Farokhi, F.; Wielgosz-Collin, G.; Clément, M.; Kornprobst, J.M.; Barnathan, G. Cytotoxicity on human cancer cells of Ophidiacerebrosides isolated from the African starfish Narcissia canariensis. Mar. Drugs 2010, 8, 2988–2998. [Google Scholar] [CrossRef]
- Hirsch, S.; Kashman, Y. New glycosphingolipids from marine organisms. Tetrahedron 1989, 45, 3897–3906. [Google Scholar]
- Boury-Esnault, N.; Marschal, C.; Kornprobst, J.M.; Barnathan, G. A new species of Axinyssa Lendenfeld, 1897 (Porifera, Demospongiae, Halichondrida) from the Senegalese coast. Zootaxa 2002, 117, 1–8. [Google Scholar]
- Genin, E.; Njinkoué, J.M.; Wielgosz-Collin, G.; Houssay, C.; Kornprobst, J.M.; Debitus, C.; Bonin, M.; Micouin, L.; Boury-Esnault, N.; Hooper, J.N.A.; et al. Glycolipids from marine sponges: Monoglycosylceramides and alkyldiglycosylglycerols: Isolation, characterization and biological activity. Boll. Musei Ist. Biol. Univ. Genova 2004, 68, 327–334. [Google Scholar]
- Kang, S.S.; Kim, J.S.; Son, K.H.; Kim, H.P.; Chang, H.W. Cyclooxygenase-2 inhibitory cerebrosides from Phytolaccae radix. Chem. Pharm. Bull. 2001, 49, 321–323. [Google Scholar] [CrossRef]
- Frappier, F.; Jossang, A.; Soudon, J.; Calvo, F.; Rasoanaivo, P.; Rastimamanga-Urveg, S.; Saez, J.; Schrevel, J.; Grellier, P. Bisbenzylisoquinolines as modulators of chloroquine resistance in Plasmodium falciparum and multidrug resistance in tumor cells. Antimicrob. Agents Chemother. 1996, 6, 1476–1481. [Google Scholar]
- Labaied, M.; Dagan, A.; Dellinger, M.; Geze, M.; Egee, S.; Thomas, S.L.; Wang, C.; Gatt, S.; Grellier, P. Anti-Plasmodium activity of ceramide analogs. Malaria J. 2004, 3, 49–59. [Google Scholar] [CrossRef] [Green Version]
- Roussakis, C.; Gratas, C.; Audouin, A.F.; le Boterff, J.; Dabouis, C.; André, M.J.; Moyon, E.; Vo, N.H.; Pradal, G.; Verbist, J.F. Study of in vitro drug sensitivity on a newly established cell line from a primary bronchial epidermoid carcinoma of human origin (NSCLCN6). Anticancer Res. 1991, 11, 2239–2244. [Google Scholar]
- Li, Y.; Matsunaga, S.; Fusetani, N. Halicylindrosides, antifungal and cytotoxic cerebrosides from the marine sponge Halichondria cylindrata. Tetrahedron 1995, 51, 2273–2280. [Google Scholar] [CrossRef]
- Giard, D.J.; Aaronson, S.A.; Todaro, G.J.; Arnstein, P.; Kersey, J.H.; Dosik, H.; Parks, W.P. In vitro cultivation of human tumors: Establishment of cell lines derived from a series of solid tumors. J. Natl. Cancer Inst. 1973, 51, 1417–1423. [Google Scholar]
- Cartron, J.P.; Juin, P.; Oliver, L.; Martin, S.; Meflah, K.; Vallette, F.M. Non-redundant role of Bax and Bak in Bid-mediated apoptosis. Mol. Cell. Biol. 2003, 23, 4701–4712. [Google Scholar] [CrossRef]
- Brattain, M.G.; Fine, W.D.; Khaled, F.M.; Thompson, J.; Brattain, D.E. Heterogeneity of malignant cells from a human colonic carcinoma. Cancer Res. 1981, 41, 1751–1756. [Google Scholar]
- Namba, M.; Ohtsuki, T.; Mori, M.; Togawa, A.; Wada, H.; Sugihara, T.; Yawata, Y.; Kimoto, T. Establishment of five human myeloma cell lines. In Vitro Cell. Dev. Biol. 1989, 25, 723–729. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Farokhi, F.; Grellier, P.; Clément, M.; Roussakis, C.; Loiseau, P.M.; Genin-Seward, E.; Kornprobst, J.-M.; Barnathan, G.; Wielgosz-Collin, G. Antimalarial Activity of Axidjiferosides, New β-Galactosylceramides from the African Sponge Axinyssa djiferi. Mar. Drugs 2013, 11, 1304-1315. https://doi.org/10.3390/md11041304
Farokhi F, Grellier P, Clément M, Roussakis C, Loiseau PM, Genin-Seward E, Kornprobst J-M, Barnathan G, Wielgosz-Collin G. Antimalarial Activity of Axidjiferosides, New β-Galactosylceramides from the African Sponge Axinyssa djiferi. Marine Drugs. 2013; 11(4):1304-1315. https://doi.org/10.3390/md11041304
Chicago/Turabian StyleFarokhi, Fereshteh, Philippe Grellier, Monique Clément, Christos Roussakis, Philippe M. Loiseau, Emilie Genin-Seward, Jean-Michel Kornprobst, Gilles Barnathan, and Gaëtane Wielgosz-Collin. 2013. "Antimalarial Activity of Axidjiferosides, New β-Galactosylceramides from the African Sponge Axinyssa djiferi" Marine Drugs 11, no. 4: 1304-1315. https://doi.org/10.3390/md11041304
APA StyleFarokhi, F., Grellier, P., Clément, M., Roussakis, C., Loiseau, P. M., Genin-Seward, E., Kornprobst, J.-M., Barnathan, G., & Wielgosz-Collin, G. (2013). Antimalarial Activity of Axidjiferosides, New β-Galactosylceramides from the African Sponge Axinyssa djiferi. Marine Drugs, 11(4), 1304-1315. https://doi.org/10.3390/md11041304






