Crystal Structure of 9-Dibenzylsulfide-7,8-dicarba-nido-undecaborane 9-Bn2S-7,8-C2B9H11
Abstract
1. Introduction
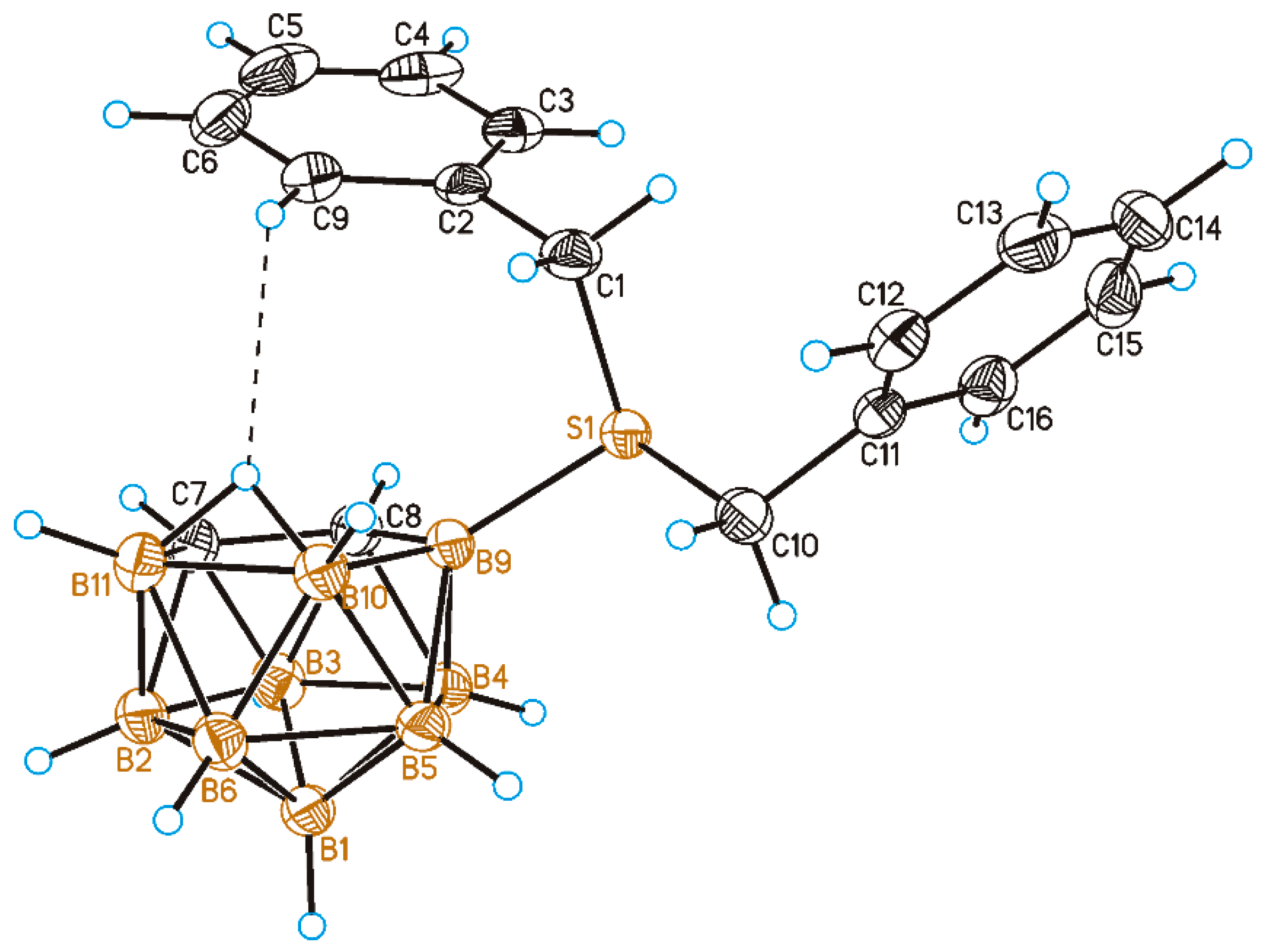
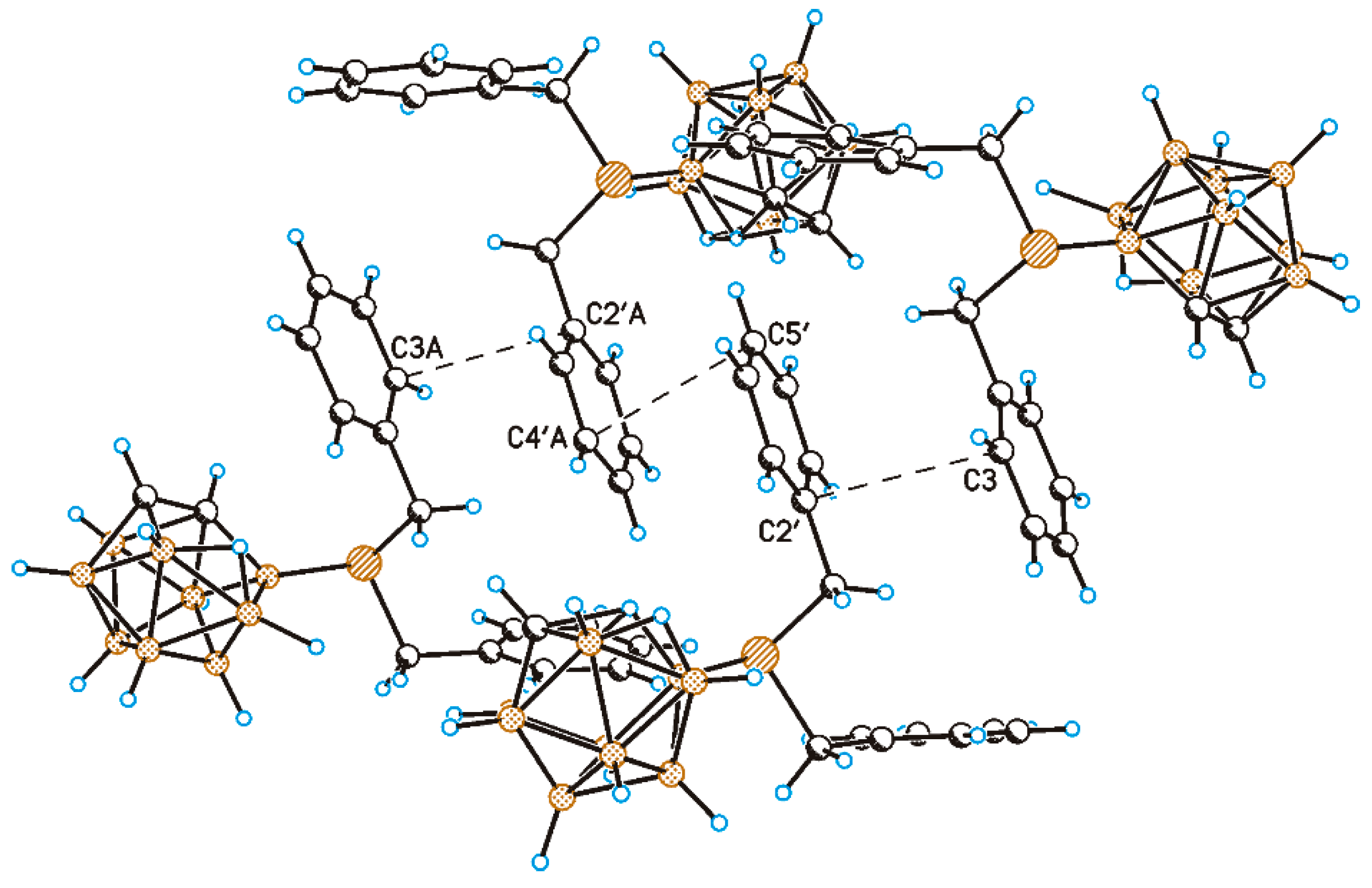
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Timofeev, S.V.; Sivaev, I.B.; Prikaznova, E.A.; Bregadze, V.I. Transition metal complexes with charge-compensated dicarbol-lide ligands. J. Organomet. Chem. 2014, 751, 221–250. [Google Scholar] [CrossRef]
- Vinogradov, M.M.; Zakharova, M.V.; Timofeev, S.V.; Loginov, D.A.; Sivaev, I.B.; Nelyubina, Y.V.; Starikova, Z.A.; Bregadze, V.I.; Kudinov, A.R. The C-substituted charge-compensated dicarbollide [7-SMe2-7,8-C2B9H10]−: Synthesis and room-temperature rearrangement of the iridium complex. Inorg. Chem. Commun. 2015, 51, 80–82. [Google Scholar] [CrossRef]
- Vinogradov, M.M.; Nelyubina, Y.V.; Pavlov, A.A.; Novikov, V.V.; Shvydkiy, N.V.; Kudinov, A.R. Polyhedral rearrange-ments in the complexes of rhodium and iridium with isomeric carborane anions [7,8-Me2-X-SMe2-7,8-nido-C2B9H8]− (X = 9 and 10). Organometalics 2017, 36, 791–800. [Google Scholar] [CrossRef]
- Timofeev, S.V.; Zhidkova, O.B.; Sivaev, I.B.; Starikova, Z.A.; Suponitsky, K.Y.; Yan, H.; Bregadze, V.I. Synthesis of rhodacarboranes containing σ- and π-carboranyl ligands in one molecule. J. Organomet. Chem. 2018, 867, 342–346. [Google Scholar] [CrossRef]
- Vinogradov, M.M.; Loginov, D.A. Rhoda- and iridacarborane halide complexes: Synthesis, structure and application in homogeneous catalysis. J. Organomet. Chem. 2020, 910, 121135. [Google Scholar] [CrossRef]
- Vinogradov, M.M.; Nesterov, I.D.; Nelyubina, Y.V.; Pavlov, A.A. Pathway bifurcations in the cage rearrangement of metallacarboranes: Experimental and computational evidence. Dalton Trans. 2021, 50, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Timofeev, S.V.; Zhidkova, O.B.; Prikaznova, E.A.; Sivaev, I.B.; Semioshkin, A.; Godovikov, I.A.; Starikova, Z.A.; Bregadze, V.I. Direct synthesis of nido-carborane derivatives with pendant functional groups by copper-promoted reactions with di-methylalkylamines. J. Organomet. Chem. 2014, 757, 21–27. [Google Scholar] [CrossRef]
- Zakharova, M.V.; Sivaev, I.B.; Anufriev, S.A.; Timofeev, S.V.; Suponitsky, K.Y.; Godovikov, I.A.; Bregadze, V.I. A new approach to the synthesis of functional derivatives of nido-carborane: Alkylation of [9-MeS-nido-7,8-C2B9H11]−. Dalton Trans. 2014, 43, 5044–5053. [Google Scholar] [CrossRef]
- Anufriev, S.A.; Sivaev, I.B.; Suponitsky, K.Y.; Bregadze, V.I. Practical synthesis of 9-methylthio-7,8-nido-carborane [9-MeS-7,8-C2B9H11]−. Some evidences of BH···X hydride-halogen bonds in 9-XCH2(Me)S-7,8-C2B9H11 (X = Cl, Br, I). J. Organomet. Chem. 2017, 849–850, 315–323. [Google Scholar] [CrossRef]
- Anufriev, S.A.; Zakharova, M.V.; Sivaev, I.B.; Bregadze, V.I. New carborane-containing acids and amines. Russ. Chem. Bull. 2017, 66, 1643–1649. [Google Scholar] [CrossRef]
- Anufriev, S.A.; Sivaev, I.B.; Suponitsky, K.Y.; Godovikov, I.A.; Bregadze, V.I. Synthesis of 10-methylsulfide and 10-alkylmethylsulfonium nido-carborane derivatives: B–H···π Interactions between the B–H–B hydrogen atom and alkyne group in 10-RC≡CCH2S(Me)-7,8-C2B9H11. Eur. J. Inorg. Chem. 2017, 4436–4443. [Google Scholar] [CrossRef]
- Erokhina, S.A.; Stogniy, M.Y.; Suponitsky, K.Y.; Kosenko, I.D.; Sivaev, I.B.; Bregadze, V.I. Synthesis of new nido-carborane based carboxylic acids and amines. Polyhedron 2018, 153, 145–151. [Google Scholar] [CrossRef]
- Druzina, A.A.; Zhidkova, O.B.; Dudarova, N.V.; Kosenko, I.D.; Ananyev, I.V.; Timofeev, S.V.; Bregadze, V.I. Synthesis and structure of nido-carboranyl azide and its “click” reactions. Molecules 2021, 26, 530. [Google Scholar] [CrossRef]
- Cowie, J.; Hamilton, E.J.M.; Laurie, J.C.V.; Welch, A.J. Structure of 10,11-μ-hydro-9-dimethylsulfido-7,8-dicarba-nido-undecaborane(11). Acta Cryst. C 1988, 44, 1648–1650. [Google Scholar] [CrossRef]
- Rosair, G.M.; Welch, A.J.; Weller, A.S.; Zahn, S.K. Sterically encumbered charge-compensated carbaboranes: Synthesis and reactivity molecular structures of 7-Ph-11-SMe2-7,8-nido-C2B9H10 and 1-Ph-3,3-(CO)2-7-SMe2-3,1,2-closo-RhC2B9H8. J. Organomet. Chem. 1997, 536–537, 299–308. [Google Scholar] [CrossRef]
- Ellis, D.; Rosair, G.M.; Robertson, S.; Welch, A.J. 7,8-Diphenyl-9-dimethylsulfido-10,11-μ-hydro-7,8-dicarba-nido-undecaborane(9). Acta Cryst. C 2000, 56, 1399–1400. [Google Scholar] [CrossRef]
- Chen, M.; Zhao, D.; Xu, J.; Li, C.; Lu, C.; Yan, H. Electrooxidative B-H functionalization of nido-carboranes. Angew. Chem. Int. Ed. 2021, 60, 7838–7844. [Google Scholar] [CrossRef] [PubMed]
- Timofeev, S.V.; Rudakov, D.A.; Rakova, E.A.; Glukhov, I.V.; Starikova, Z.A.; Bragin, V.I.; Godovikov, I.A.; Shirokii, V.L.; Potkin, V.I.; Maier, N.A.; et al. Synthesis and structure of halogen derivatives of 9-dimethylsulfonium-7,8-dicarba-nido-undecaborane [9-Me2S-7,8-C2B9H11]. J. Organomet. Chem. 2007, 692, 5133–5140. [Google Scholar] [CrossRef]
- Grüner, B.; Holub, J.; Plešek, J.; Štíbr, B.; Thornton-Pett, M.; Kennedy, J.D. Dimethylsulfide-dicarbaborane chemistry. Isolation and characterisation of isomers [9-(SMe2)-nido-7,8-C2B9H10-X-Me] (where X = 1, 2, 3 and 4) and some related compounds. An unusual skeletal rearrangement. Dalton Trans. 2007, 4859–4865. [Google Scholar] [CrossRef]
- Kazheva, O.N.; Rudakov, D.A.; Shilov, G.V.; D’yachenko, O.A.; Potkin, V.I. Structure of 6,11-dichloro-9-dimethylthio-7,8-dicarba-nido-undecaborane [6,11-Cl2-9-SMe2-7,8-C2B9H9]. J. Struct. Chem. 2013, 54, 349–354. [Google Scholar] [CrossRef]
- Anufriev, S.A.; Erokhina, S.A.; Sivaev, I.B.; Bregadze, V.I. On the reaction of nido-carborane with thiourea. Russ. Chem. Bull. 2016, 65, 2704–2707. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A.; Kudin, K.N., Jr.; Burant, J.C.; Millam, J.M.; et al. Gaussian 03; Revision E.01; Gaussian, Inc.: Wallingford, UK, 2004. [Google Scholar]
- Anufriev, S.A.; Erokhina, S.A.; Suponitsky, K.Y.; Godovikov, I.A.; Filippov, O.A.; Fabrizi de Biani, F.; Corsini, M.; Chizhov, A.O.; Sivaev, I.B. Methylsulfanyl-stabilized rotamers of cobalt bis(dicarbollide). Eur. J. Inorg. Chem. 2017, 4444–4451. [Google Scholar] [CrossRef]
- Suponitsky, K.Y.; Anisimov, A.A.; Anufriev, S.A.; Sivaev, I.B.; Bregadze, V.I. 1,12-Diiodo-ortho-carborane: A classic textbook example of the dihalogen bond. Crystals 2021, 11, 396. [Google Scholar] [CrossRef]
- Suponitsky, K.Y.; Burakov, N.I.; Kanibolotsky, A.L.; Mikhailov, V.A. Multiple noncovalent bonding in halogen complexes with oxygen organics. I. Tertiary amides. J. Phys. Chem. A 2016, 120, 4179–4190. [Google Scholar] [CrossRef] [PubMed]
- Suponitsky, K.Y.; Masunov, A.E.; Antipin, M.Y. Computational search for nonlinear optical materials: Are polarization functions important in the hyperpolarizability predictions of molecules and aggregates? Mendeleev Commun. 2009, 19, 311–313. [Google Scholar] [CrossRef]
- Bader, R.F.W. Atoms in Molecules. In A Quantum Theory; Clarendon Press: Oxford, UK, 1990. [Google Scholar]
- Keith, T.A. AIMAll (Version 15.05.18); TK Gristmill Software: Overland Park, KS, USA, 2015. [Google Scholar]
- Espinosa, E.; Molins, E.; Lecomte, C. Hydrogen bond strengths revealed by topological analyses of experimentally observed electron densities. Chem. Phys. Lett. 1998, 285, 170–173. [Google Scholar] [CrossRef]
- Espinosa, E.; Alkorta, I.; Rozas, I.; Elguero, J.; Molins, E. About the evaluation of the local kinetic, potential and total energy densities in closed-shell interactions. Chem. Phys. Lett. 2001, 336, 457–461. [Google Scholar] [CrossRef]
- Lyssenko, K.A. Analysis of supramolecular architectures: Beyond molecular packing diagrams. Mendeleev. Commun. 2012, 22, 1–7. [Google Scholar] [CrossRef]
- Palysaeva, N.V.; Gladyshkin, A.G.; Vatsadze, I.A.; Suponitsky, K.Y.; Dmitriev, D.E.; Sheremetev, A.B. N-(2-Fluoro-2,2-dinitroethyl)azoles: Novel assembly of diverse explosophoric building blocks for energetic compounds design. Org. Chem. Front. 2019, 6, 249–255. [Google Scholar] [CrossRef]
- Dalinger, I.L.; Suponitsky, K.Y.; Shkineva, T.K.; Lempert, D.B.; Sheremetev, A.B. Bipyrazole bearing ten nitro groups—Novel highly dense oxidizer for forward-looking rocket propulsions. J. Mater. Chem. A 2018, 6, 14780–14786. [Google Scholar] [CrossRef]
- APEX2 and SAINT; Bruker AXS Inc.: Madison, WI, USA, 2014.
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
| Torsion Angle | Molecule A | Molecule A′ | Calculation |
|---|---|---|---|
| C(8)-B(9)-S(1)-C(1) | 91.2(2) | 113.4(2) | 112.9 |
| C(8)-B(9)-S(1)-C(10) | −163.5(2) | −140.7(2) | −141.0 |
| B(9)-S(1)-C(1)-C(2) | −65.8(2) | −84.2(2) | −77.8 |
| B(9)-S(1)-C(10)-C(11) | −165.0(2) | 179.7(2) | −177.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anufriev, S.A.; Anisimov, A.A.; Sivaev, I.B.; Bregadze, V.I. Crystal Structure of 9-Dibenzylsulfide-7,8-dicarba-nido-undecaborane 9-Bn2S-7,8-C2B9H11. Molbank 2021, 2021, M1230. https://doi.org/10.3390/M1230
Anufriev SA, Anisimov AA, Sivaev IB, Bregadze VI. Crystal Structure of 9-Dibenzylsulfide-7,8-dicarba-nido-undecaborane 9-Bn2S-7,8-C2B9H11. Molbank. 2021; 2021(2):M1230. https://doi.org/10.3390/M1230
Chicago/Turabian StyleAnufriev, Sergey A., Alexei A. Anisimov, Igor B. Sivaev, and Vladimir I. Bregadze. 2021. "Crystal Structure of 9-Dibenzylsulfide-7,8-dicarba-nido-undecaborane 9-Bn2S-7,8-C2B9H11" Molbank 2021, no. 2: M1230. https://doi.org/10.3390/M1230
APA StyleAnufriev, S. A., Anisimov, A. A., Sivaev, I. B., & Bregadze, V. I. (2021). Crystal Structure of 9-Dibenzylsulfide-7,8-dicarba-nido-undecaborane 9-Bn2S-7,8-C2B9H11. Molbank, 2021(2), M1230. https://doi.org/10.3390/M1230








