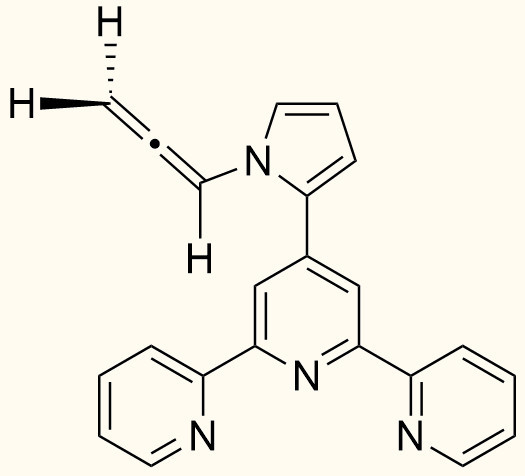
4′-(N-(Propan-1,2-dienyl)pyrrol-2-yl)-2,2′:6′,2″-terpyridine
Abstract
1. Introduction
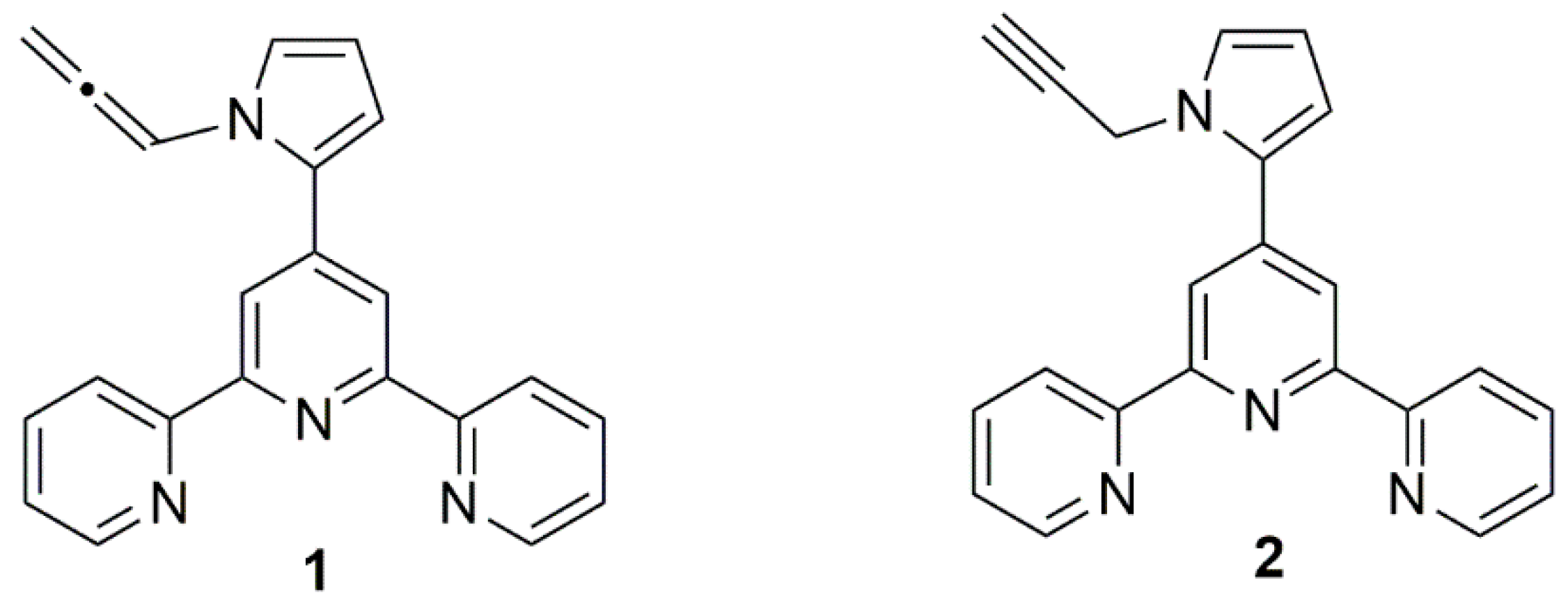
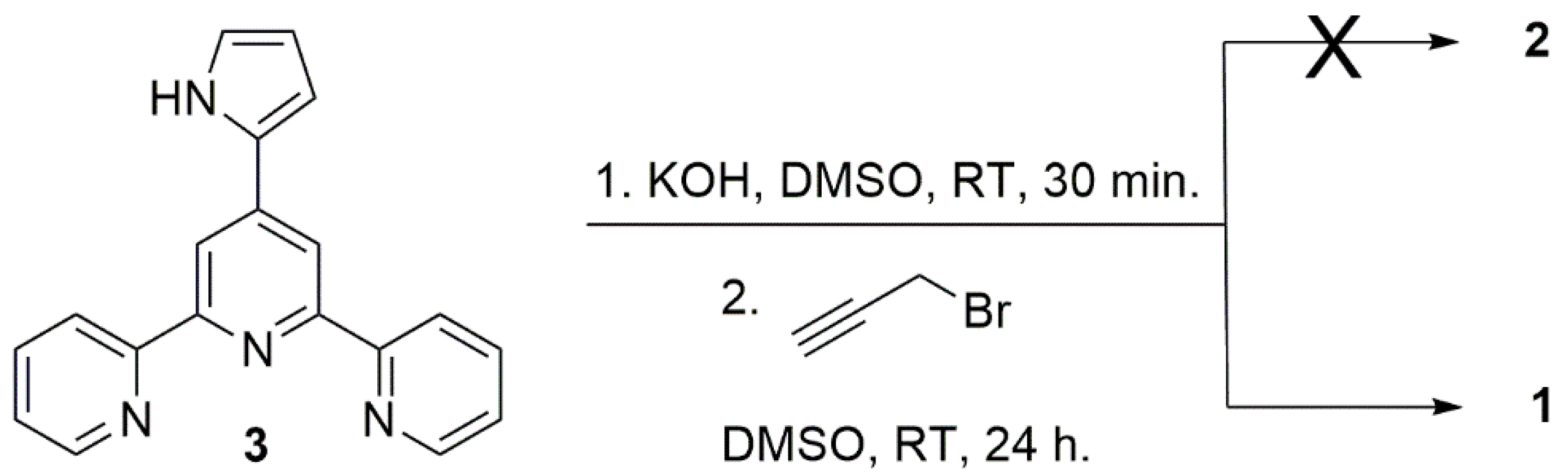
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Schubert, U.S.; Hofmeier, H.; Newkome, G.R. Modern Terpyridine Chemistry; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Husson, J.; Knorr, M. Syntheses and applications of furanyl-functionalised 2,2′:6′,2″-terpyridines. Beilstein J. Org. Chem. 2012, 8, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Husson, J.; Knorr, M. 2,2′:6′,2″-Terpyridines Functionalized with Thienyl Substituents: Synthesis and Applications. J. Heterocyclic Chem. 2012, 49, 453–478. [Google Scholar] [CrossRef]
- Dehaudt, J.; Husson, J.; Guyard, L. A more efficient synthesis of 4,4′,4″-tricarboxy-2,2′:6′,2″-terpyridine. Green Chem. 2011, 13, 3337–3340. [Google Scholar] [CrossRef]
- Momeni, B.Z.; Doustkhahvajari, F. Heteroleptic complexes of silver(I) featuring 4′-hydroxy and 4′-(2-furyl)-2,2′:6′,2″-terpyridine: An easy route for synthesis of silver nanoparticles. Inorg. Chim. Acta. 2019, 487, 145–152. [Google Scholar] [CrossRef]
- Shen, Y.; Shao, T.; Fang, B.; Du, W.; Zhang, M.; Liu, J.; Liu, T.; Tian, X.; Zhang, Q.; Wang, A.; et al. Visualization of mitochondrial DNA in living cells with super-resolution microscopy using thiophene-based terpyridine Zn(II) complexes. Chem. Commun. 2018, 54, 11288–11291. [Google Scholar] [CrossRef]
- Husson, J.; Guyard, L. 4′-(5-Methylfuran-2-yl)-2,2′:6′,2″-terpyridine: A New Ligand Obtained from a Biomass-Derived Aldehyde with Potential Application in Metal-Catalyzed Reactions. Molbank 2018, 2018, M1032. [Google Scholar] [CrossRef]
- Njogu, E.M.; Martincigh, B.S.; Omondi, B.; Nyamori, V.O. Synthesis, characterization, antimicrobial screening and DNA binding of novel silver(I)-thienylterpyridine and silver(I)-furylterpyridine. Appl. Organomet. Chem. 2018, 32, E4554. [Google Scholar] [CrossRef]
- Liang, Y.W.; Strohecker, D.; Lynch, V.; Holliday, B.J.; Jones, R.A. A Thiophene-Containing Conductive Metallopolymer Using an Fe(II) Bis(terpyridine) Core for Electrochromic Materials. ACS Appl. Mater. Interfaces 2016, 8, 34568–34580. [Google Scholar] [CrossRef]
- Fernandes, S.S.M.; Besley, M.; Ciarrocchi, C.; Licchelli, M.; Raposo, M.M.M. Terpyridine derivatives functionalized with (hetero)aromatic groups and the corresponding Ru complexes: Synthesis and characterization as SHG chromophores. Dyes Pigment. 2018, 150, 49–58. [Google Scholar] [CrossRef]
- Czerwinska, K.; Machura, B.; Kula, S.; Krompiec, S.; Erfurt, K.; Roma-Rodrigues, C.; Fernandes, A.R.; Shul’pina, L.; Ikonnikov, N.S.; Shul’pin, G.B. Copper(II) complexes of functionalized 2,2′:6′,2″-terpyridines and 2,6-di(thiazol-2-yl)pyridine: Structure, spectroscopy, cytotoxicity and catalytic activity. Dalton Trans. 2017, 46, 9591–9604. [Google Scholar] [CrossRef]
- Klemens, T.; Switlicka-Olszewska, A.; Machura, B.; Grucela, M.; Schab-Balcerzak, E.; Smolarek, K.; Mackowski, S.; Szlapa, A.; Kula, S.; Krompiec, S.; et al. Rhenium(I) terpyridine complexes—Synthesis, photophysical properties and application in organic light emitting devices. Dalton Trans. 2016, 45, 1746–1762. [Google Scholar] [CrossRef] [PubMed]
- Naidji, B.; Husson, J.; Et Taouil, A.; Brunol, E.; Sanchez, J.-B.; Berger, F.; Rauch, J.-Y.; Guyard, L. Terpyridine-based metallopolymer thin films as active layer in ammonia sensor device. Synth. Met. 2016, 221, 214–219. [Google Scholar] [CrossRef]
- Husson, J.; Guyard, L. A new and facile method for the functionalization of a Merrifield resin with terpyridines: Application as a heterogeneous catalyst for the synthesis of biaryls in environmentally friendly solvents. Green Process. Synth. 2016, 5, 331–336. [Google Scholar] [CrossRef]
- Busemann, A.; Araman, C.; Flaspohler, I.; Pratesi, A.; Zhou, X.-Q.; van Rixel, V.H.S.; Siegler, M.A.; Messori, L.; van Kasteren, S.I.; Bonnet, S. Alkyne Functionalization of a Photoactivated Ruthenium Polypyridyl Complex for Click-Enabled Serum Albumin Interaction Studies. Inorg. Chem. 2020, 59, 7710–7720. [Google Scholar] [CrossRef] [PubMed]
- Husson, J.; Guyard, L. Synthesis of new 4′-(N-alkylpyrrol-2-yl)-2,2′:6′,2″-terpyridines via N-alkylation of a pyrrole moiety. Heterocycl. Commun. 2015, 21, 199–202. [Google Scholar] [CrossRef]
- Tarasova, O.A.; Brandsma, L.; Trofimov, B.A. Facile One-Pot Syntheses of 1-Allenylpyrroles. Synthesis 1993, 571–572. [Google Scholar] [CrossRef]
- Tarasova, O.A.; Taherirastgar, F.; Verkruijsse, H.D.; Mal’kina, A.G.; Brandsma, L.; Trofimov, B.A. Metallation and functionalization of 1-allenylpyrrole. Recl. Trav. Chim. Pays Bas 1996, 115, 145–147. [Google Scholar] [CrossRef]
- Duan, G.-J.; Ling, J.-B.; Wang, W.-P.; Luo, Y.-C.; Xu, P.-F. Organocatalytic formal [2+2] cycloaddition initiated by vinylogous Friedel-Crafts alkylation: Enantioselective synthesis of substituted cyclobutene derivatives. Chem. Commun. 2013, 49, 4625–4627. [Google Scholar] [CrossRef]
- Zhao, Y.; Yuan, Y.; Xu, M.; Zheng, Z.; Zhang, R.; Li, Y. Selective synthesis of pyrrolo[1,2-a]azepines or 4,6-dicarbonyl indoles via tandem reactions of alkynone with pyrrole derivatives. Org. Biomol. Chem. 2017, 15, 6328–6332. [Google Scholar] [CrossRef]
- Turnu, F.; Luridiana, A.; Cocco, A.; Porcu, S.; Frongia, A.; Sarais, G.; Secci, F. Catalytic Tandem Friedel-Crafts Alkylation/C4-C3 Ring-Contraction Reaction: An Efficient Route for the Synthesis of Indolyl Cyclopropanecarbaldehydes and Ketones. Org. Lett. 2019, 21, 7329–7332. [Google Scholar] [CrossRef]
- Endo, T.; Tomita, I. Novel Polymerization Methods for Allene Derivatives. Prog. Polym. Sci. 1997, 22, 565–600. [Google Scholar] [CrossRef]
- Rivera-Fuentes, P.; Diederich, F. Allenes in Molecular Materials. Angew. Chem. Int. Ed. 2012, 51, 2818–2828. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Husson, J.; Guyard, L. 4′-(N-(Propan-1,2-dienyl)pyrrol-2-yl)-2,2′:6′,2″-terpyridine. Molbank 2020, 2020, M1142. https://doi.org/10.3390/M1142
Husson J, Guyard L. 4′-(N-(Propan-1,2-dienyl)pyrrol-2-yl)-2,2′:6′,2″-terpyridine. Molbank. 2020; 2020(2):M1142. https://doi.org/10.3390/M1142
Chicago/Turabian StyleHusson, Jérôme, and Laurent Guyard. 2020. "4′-(N-(Propan-1,2-dienyl)pyrrol-2-yl)-2,2′:6′,2″-terpyridine" Molbank 2020, no. 2: M1142. https://doi.org/10.3390/M1142
APA StyleHusson, J., & Guyard, L. (2020). 4′-(N-(Propan-1,2-dienyl)pyrrol-2-yl)-2,2′:6′,2″-terpyridine. Molbank, 2020(2), M1142. https://doi.org/10.3390/M1142