5,9,11-Trihydroxy-2,2-dimethyl-3-(2-methylbut-3-en-2-yl)pyrano[2,3-a]xanthen-12(2H)-one from the Stem Bark of Calophyllum tetrapterum Miq.
Abstract
:1. Introduction
2. Result and Discussion
3. Experiment Section
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Anti-HIV Reverse Transcriptase Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ito, C.; Itoigawa, M.; Mishina, Y.; Filho, V.C.; Mukainaka, T.; Tokuda, H.; Nishino, H.; Furukawa, H. Chemical constituents of Calophyllum brasiliensis: Structure elucidation of seven xanthones and their cancer chemopreventive activity. J. Nat. Prod. 2002, 65, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.J.; Mei, W.L.; Zhong, H.M.; Zeng, Y.B.; Wu, X.D.; Dai, H.F. A new prenylated xanthone from the branches of Calophyllum inophyllum. J. Asian Nat. Prod. Res. 2011, 13, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Tanjung, M.; Saputri, R.D.; Tjahjandarie, T.S. 5,9,11-Trihydroxy-2,2-dimethyl-10-(3′-methyl-2′-butenyl)-3-(2″-methyl-3″-butenyl)pyrano[2,3-a]xanthen-12(2H)-one from the stem bark of Calophyllum pseudomole. Molbank 2016, 2016, M906. [Google Scholar] [CrossRef]
- Mah, S.H.; Ee, G.C.L.; Teh, S.S.; Sukari, M.A. Calophyllum inophyllum and Calophyllum soulattri source of anti-proliferative xanthones and their structure-activity relationships. Nat. Prod. Res. 2013, 27, 98–101. [Google Scholar]
- Joshi, S.P.; Kulkarni, S.R.; Phalgune, U.D.; Puranik, V.G. New dipyranocoumarin from the leaves of Calophyllum apetalum Willd. Nat. Prod. Res. 2013, 27, 1896–1901. [Google Scholar] [CrossRef] [PubMed]
- Daud, S.B.; Ee, G.C.L.; Malek, E.A.; Teh, S.S.; See, I. A new coumarin from Calophyllum hosei. Nat. Prod. Res. 2014, 28, 1534–1538. [Google Scholar] [CrossRef] [PubMed]
- Guilet, D.G.; Helesbeux, J.J.; Seraphin, D.; Sevenet, T.; Richomme, P.; Bruneton, J. Novel cytotoxic 4-phenilcoumarins from Calophyllum dispar. J. Nat. Prod. 2001, 64, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Cottiglia, F.; Dhanapal, B.; Sticher, O.; Heilmann, J. New chromanone acids with antibacterial activity from the bark of Calophyllum brasiliense. J. Nat. Prod. 2004, 67, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Reyes, M.H.; Basualdo, M.C.; Abe, F.; Estrada, M.J.; Soler, C.; Chilpa, R.R. HIV-1 inhibitory compounds from Calophyllum brasiliense leaves. Biol. Pharm. Bull. 2004, 27, 1471–1475. [Google Scholar] [CrossRef]
- Ha, L.D.; Hansen, P.E.; Duus, F.; Pham, H.D.; Nguyen, L.D. A new chromanone acid from the bark of Calophyllum dryobalanoides. Phytochem. Lett. 2012, 5, 287–291. [Google Scholar] [CrossRef]
- Lim, C.K.; Subramaniam, H.; Say, Y.H.; Jong, V.Y.M.; Khaledi, H.; Chee, C.F. A new chromanone acid from the stem bark of Calophyllum teysmannii. Nat. Prod. Res. 2015, 29, 1970–1977. [Google Scholar] [CrossRef] [PubMed]
- Ferchichi, L.; Derbre, S.; Mahmood, K.; Toure, K.; Guilet, D.; Litaudon, M.; Awang, K.; Hadi, A.H.A.; Ray, A.M.L.; Richomme, P. Bioguided fractionation and isolation of natural inhibitors of advanced glycation end-products (AGEs) from Calophyllum flavoramulum. Phytochemistry 2012, 78, 98–106. [Google Scholar] [CrossRef] [PubMed]
- McKee, T.C.; Covington, C.D.; Fuller, R.W.; Bokesch, H.R.; Young, S.; Cardellina, J.H.; Kadushin, M.R.; Soejarto, D.D.; Stevens, P.F.; Cragg, G.M.; et al. Pyranocoumarins from tropical species of the genus Calophyllum: Chemotaxonomic study of extracts in the national cancer institute collection. J. Nat. Prod. 1998, 61, 1252–1256. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Zeng, Y-B.; Mei, W.L.; Zhao, Y-X.; Deng, Y-Y.; Dai, H-F. Cytotoxic prenylated xanthones from Calophyllum inophyllum. J. Asian Nat. Prod. Res. 2008, 10, 993–997. [Google Scholar] [CrossRef] [PubMed]
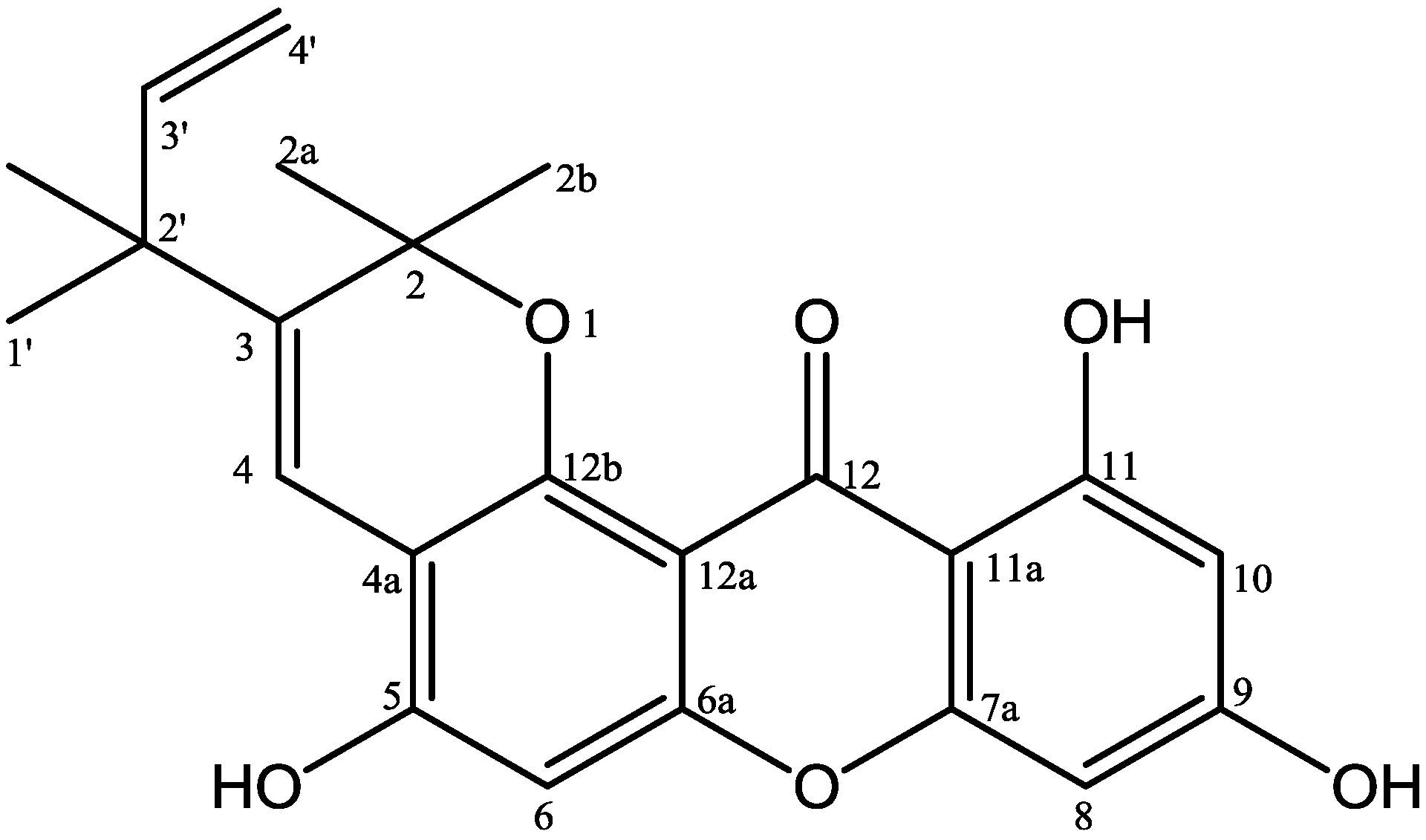
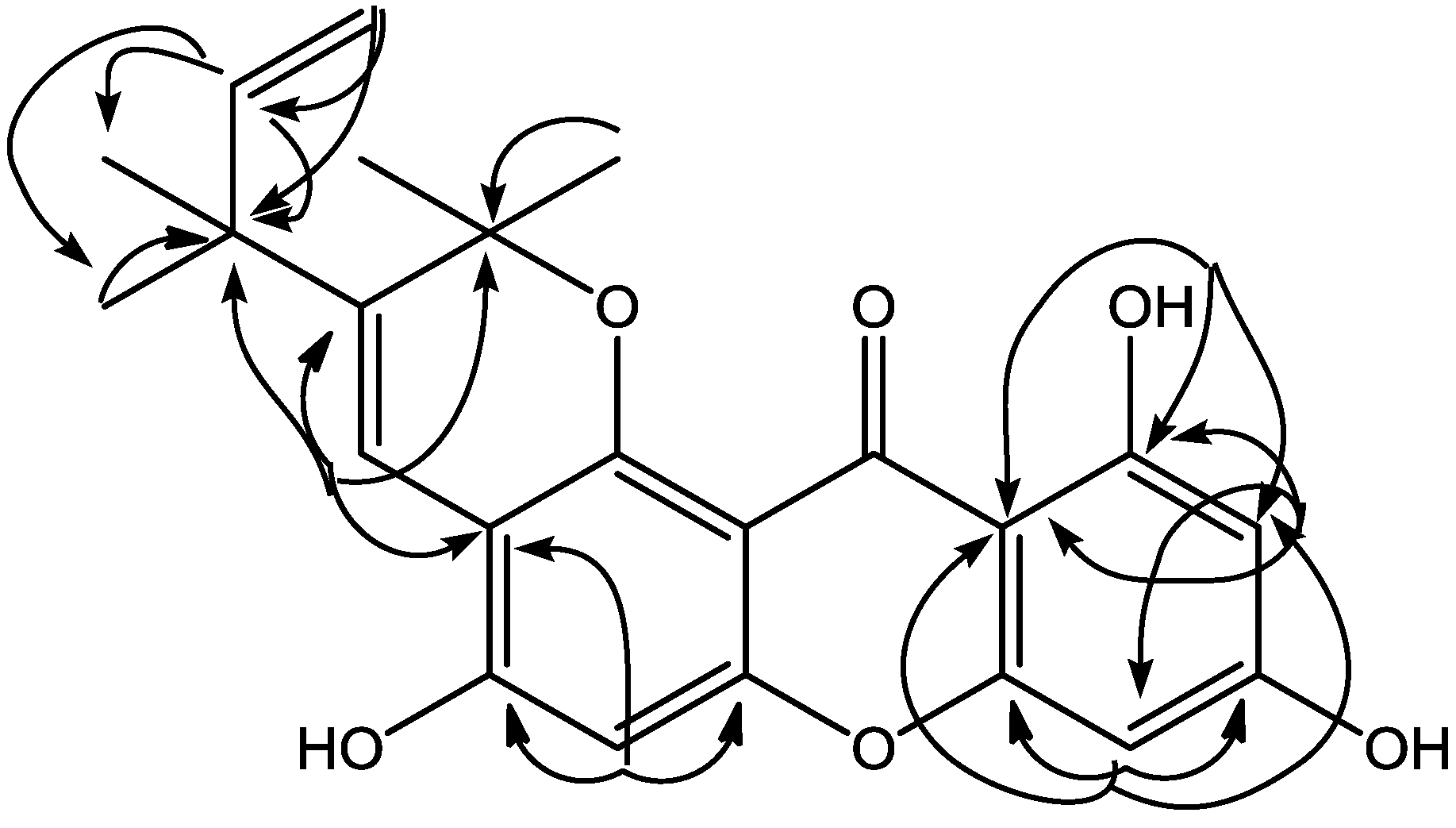
| No. C | δH (Mult, J Hz) | δC | HMBC |
|---|---|---|---|
| 2 | - | 80.4 | - |
| 2a | 1.50 (s, 3H) | 27.3 | C-2; C-2b |
| 2b | 1.50 (s, 3H) | 27.3 | C-2; C-2a |
| 3 | - | 137.8 | - |
| 4 | 8.17 (s, 1H) | 118.7 | C-2; C-3; C-4a; C-2′ |
| 4a | - | 108.3 | - |
| 5 | - | 155.6 | - |
| 6 | 6.80 (s, 1H) | 103.0 | C-4a; C-5; C-6a |
| 6a | - | 153.6 | - |
| 7a | - | 158.1 | - |
| 8 | 6.33 (d, 2.2, 1H) | 93.9 | C-7a; C-9; C-10; C-11a |
| 9 | - | 165.4 | - |
| 10 | 6.20 (d, 2.2, 1H) | 98.7 | C-8, C-11, C-11a |
| 11 | - | 164.7 | - |
| 11a | - | 103.9 | - |
| 12 | - | 183.1 | - |
| 12a | - | 122.8 | - |
| 12b | - | 149.9 | - |
| 1′ | 1.41 (s) | 28.6 | C-2′; C-3′, 2′-CH3 |
| 2′ | - | 42.7 | - |
| 2′-CH3 | 1.41 (s) | 28.6 | C-1′; C-2′, C-3′ |
| 3′ | 6.02 (dd, 10.6; 17.6, 1H) | 147.9 | C-1′; C-2′, 2′-CH3 |
| 4′ | 5.16 (dd, 1.1; 17.5, 1H) 5.08 (dd, 1.1; 10.6, 1H) | 112.3 | C-2′, C-3′ |
| 11-OH | 13.48 (s, 1H) | - | C-10; C-11; C-11a |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tjahjandarie, T.S.; Saputri, R.D.; Tanjung, M. 5,9,11-Trihydroxy-2,2-dimethyl-3-(2-methylbut-3-en-2-yl)pyrano[2,3-a]xanthen-12(2H)-one from the Stem Bark of Calophyllum tetrapterum Miq. Molbank 2017, 2017, M936. https://doi.org/10.3390/M936
Tjahjandarie TS, Saputri RD, Tanjung M. 5,9,11-Trihydroxy-2,2-dimethyl-3-(2-methylbut-3-en-2-yl)pyrano[2,3-a]xanthen-12(2H)-one from the Stem Bark of Calophyllum tetrapterum Miq. Molbank. 2017; 2017(1):M936. https://doi.org/10.3390/M936
Chicago/Turabian StyleTjahjandarie, Tjitjik Srie, Ratih Dewi Saputri, and Mulyadi Tanjung. 2017. "5,9,11-Trihydroxy-2,2-dimethyl-3-(2-methylbut-3-en-2-yl)pyrano[2,3-a]xanthen-12(2H)-one from the Stem Bark of Calophyllum tetrapterum Miq." Molbank 2017, no. 1: M936. https://doi.org/10.3390/M936
APA StyleTjahjandarie, T. S., Saputri, R. D., & Tanjung, M. (2017). 5,9,11-Trihydroxy-2,2-dimethyl-3-(2-methylbut-3-en-2-yl)pyrano[2,3-a]xanthen-12(2H)-one from the Stem Bark of Calophyllum tetrapterum Miq. Molbank, 2017(1), M936. https://doi.org/10.3390/M936






