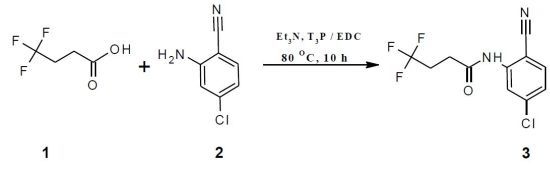
N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide
Abstract
:Experimental
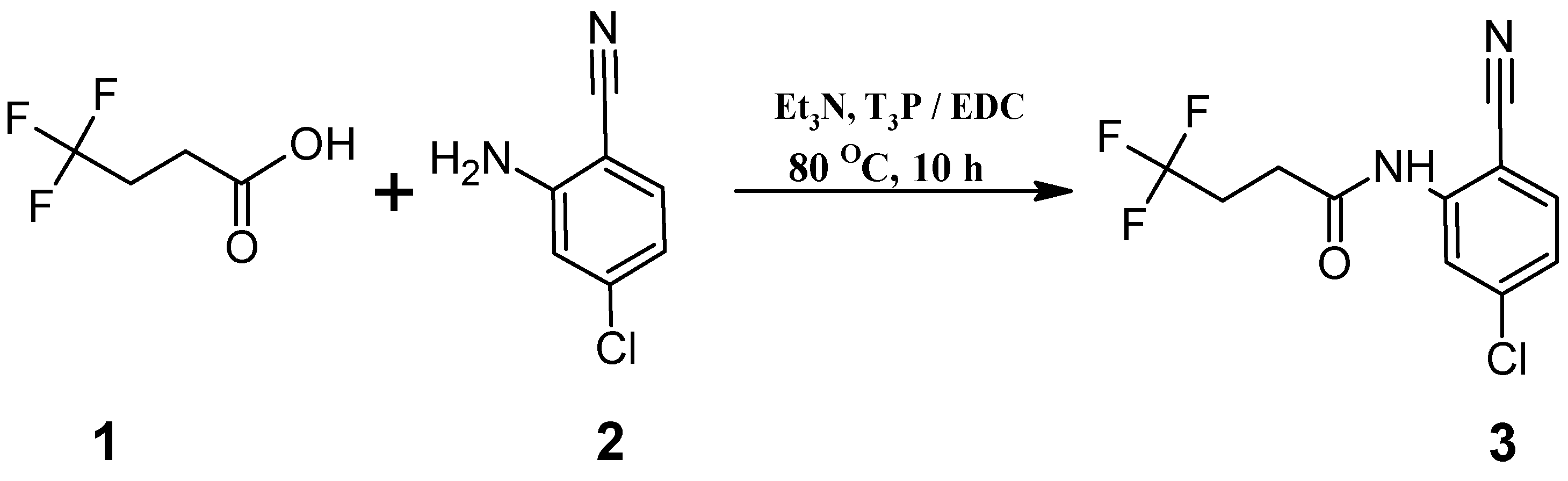
Synthesis of N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide (3)
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgments
References
- Abdou, I.M.; Saleh, A.M.; Hussein, H.F. Synthesis and antitumor activity of 5-Trifluoromethyl-2,4-dihydropyrazol-3-one nucleus. Molecules 2004, 9, 109–116. [Google Scholar]
- Bukhtiarova, T.A.; Trinus, F.P.; Danilenko, V.F.; Danilenko, G.I.; Ovrutskii, V.M.; Sharykina, N.I. Structure and anti-inflammatory activity of isonicotinic and nicotinic amides. Pharm. Chem. J. 1997, 31, 597–599. [Google Scholar] [CrossRef]
- Geronikaki, A.; Litina, D.H.; Chatziopoulos, C.; Soloupis, G. Synthesis and biological evaluation of new 4,5-disubstituted-thiazolyl amides, derivatives of 4-hydroxy-piperidine or of 4-N-methyl piperazine. Molecules 2003, 8, 472–479. [Google Scholar] [CrossRef]
- Milyutin, A.V.; Amirova, L.R.; Krylova, I.V.; Nazmetdinov, F.Y.; Novoselova, G.N.; Andreichikov, Y.S.; Kolla, V.E. Synthesis, properties, and biological activity of 3-Pyridylamides of 4-aryl-2-hydroxy-4-oxo-2-butenic (aroylpyruvic) acids. Pharm. Chem. J. 1997, 31, 30–33. [Google Scholar] [CrossRef]
- Igosheva, E.V.; Tolmacheva, I.A.; Vikharev, Yu.B.; Grishko, V.V.; Savinova, O.V.; Boreko, E.I.; Eremin, V.F. Synthesis and biological activity of 28-Methoxy-28-oxo-1-cyano-2,3-sec-LUP-20(29)-en-3-oic acid. Chem. Nat. Compd. 2012, 48, 426–430. [Google Scholar] [CrossRef]
- Galeeva, R.N.; Rudometova, E.V.; Gavrilov, M.Y.; Kolla, V.E.; Kon’shin, M.E.; Nazmetdinov, F.Y. Synthesis and pharmacological activity of 2-arylamino-6,7-dihidro-5H-pyridine-3-carboxylic acid amides. Pharm. Chem. J. 1997, 31, 89–90. [Google Scholar] [CrossRef]
- Kolotova, N.V.; Dolzhenko, A.V.; Koz’minykh, V.O.; Kotegov, V.P.; Godina, A.T. Substituted amides and hydrazides of 1,4-dicarboxylic acids. Part 8. Synthesis and hypoglycemic activity of substituted succinic acid amides and hydrazides. Pharm. Chem. J. 1999, 33, 635–637. [Google Scholar] [CrossRef]
- Kolotova, N.V.; Koz’minykh, E.N.; Kolla, V.E.; Syropyatov, B. Ya.; Voronina, E.V.; Koz’minykh, V.O. Substituted amides and hydrazides of 1,4-dicarboxylic acids. Part 7. Synthesis and pharmacological activity of some maleic, succinic, and phthalic acid acylhydrazides. Pharm. Chem. J. 1999, 33, 248–254. [Google Scholar] [CrossRef]
- Walter, H. New carboxylic acid amides of the pyrrole series: Synthetic routes and biological aspects. Z. Naturforsch. B Chem. Sci. 2008, 63b, 351–362. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Manojkumar, K.E.; Sreenivasa, S.; Shivaraja, G.; Rao, T.M.C. N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide. Molbank 2013, 2013, M803. https://doi.org/10.3390/M803
Manojkumar KE, Sreenivasa S, Shivaraja G, Rao TMC. N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide. Molbank. 2013; 2013(3):M803. https://doi.org/10.3390/M803
Chicago/Turabian StyleManojkumar, Karikere Ekanna, Swamy Sreenivasa, Govindaiah Shivaraja, and Tadimety Madhu Chakrapani Rao. 2013. "N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide" Molbank 2013, no. 3: M803. https://doi.org/10.3390/M803
APA StyleManojkumar, K. E., Sreenivasa, S., Shivaraja, G., & Rao, T. M. C. (2013). N-(2-Chloro-5-cyanophenyl)-4,4,4-trifluorobutanamide. Molbank, 2013(3), M803. https://doi.org/10.3390/M803