1-(8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl)-2-(2-chlorophenyl)ethanone
Abstract
: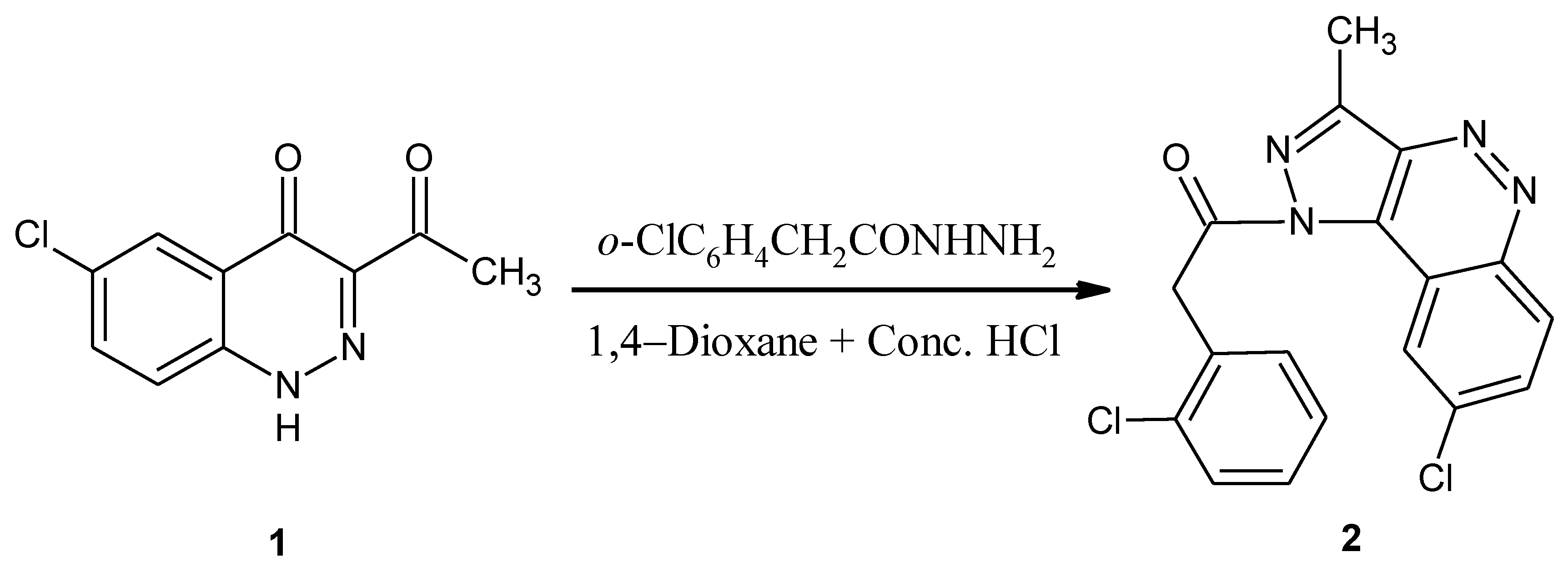
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgments
References
- Nargund, L.V.G.; Badiger, V.V.; Yarnal, S.M. Synthesis and antimicrobial and anti-inflammatory activities of substituted 2-mercapto-3-(n-aryl)pyrimido[5,4-c]cinnolin-4-(3H)-ones. J. Pharm. Sci. 1992, 81, 365–366. [Google Scholar] [CrossRef] [PubMed]
- Kametani, T.; Kigasawa, K.; Hiiragi, M.; Ishimaru, H.; Wagatsuma, N.; Kohagisawa, T.; Nakamura, T. Novel Type of Analgesic- Synthesis and Analgesic Activity. Heterocycles 1980, 14, 449–451. [Google Scholar] [CrossRef]
- Ruchelman, A.L.; Singh, S.K.; Ray, A.; Wu, X.; Yang, J.-M.; Zhou, N.; Liu, A.; Liu, L.F.; LaVoie, E.J. 11H-Isoquino[4,3-c]cinnolin-12-ones: Novel anticancer agents with potent topoisomerase I-targeting activity and cytotoxicity. Bioorg. Med. Chem. 2004, 12, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Pinna, G.A.; Salis, E.; Berta, D.; Gavini, E.; D’Amico, M. Synthesis and pharmacological evaluaion of 4a-methyl-4,4a,5,6-tetrahydrothieno[2,3-h]cinnolin-3(2H)-ones. Farmaco 1997, 52, 29–33. [Google Scholar] [PubMed]
- Stanczak, A.; Kwapiszewski, W.; Szadowska, A.; Pakulska, W. Synthesis and action on the central nervous system of some N2-substituted cinnoline derivatives. Pharmazie 1994, 49, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bawa, S.; Drabu, S. N-[(2-chloro-6-mthylquinolin-3-yl)methyl]aniline. Molbank 2009, 2009, M618. [Google Scholar] [CrossRef]
- Mishra, R.; Siddiqui, A.A.; Kumar, R.; Kumar, S. 6-Oxo-3-phenyl-5,6-dihydropyridazine-1(4H)-carbohydrazide. Molbank 2010, 2010, M652. [Google Scholar] [CrossRef]
- Bawa, S.; Kumar, R.; Chawla, G.; Kumar, S.; Mishra, R. (8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl) (pyridin-4-yl)methanone. Molbank 2010, 2010, M688. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bawa, S.; Tonk, R.K.; Chawla, G.; Kumar, S.; Afzal, O. 1-(8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl)-2-(2-chlorophenyl)ethanone. Molbank 2011, 2011, M744. https://doi.org/10.3390/M744
Bawa S, Tonk RK, Chawla G, Kumar S, Afzal O. 1-(8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl)-2-(2-chlorophenyl)ethanone. Molbank. 2011; 2011(4):M744. https://doi.org/10.3390/M744
Chicago/Turabian StyleBawa, Sandhya, Rajiv K. Tonk, Gita Chawla, Suresh Kumar, and Obaid Afzal. 2011. "1-(8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl)-2-(2-chlorophenyl)ethanone" Molbank 2011, no. 4: M744. https://doi.org/10.3390/M744
APA StyleBawa, S., Tonk, R. K., Chawla, G., Kumar, S., & Afzal, O. (2011). 1-(8-Chloro-3-methyl-1H-pyrazolo[4,3-c]cinnolin-1-yl)-2-(2-chlorophenyl)ethanone. Molbank, 2011(4), M744. https://doi.org/10.3390/M744




