Critical Success Factors and Traceability Technologies for Establishing a Safe Pharmaceutical Supply Chain
Abstract
1. Introduction
1.1. The State of the Literature
1.2. Review Objectives
- (i)
- Review the application of traceability technologies in various PSC phases to detect and solve counterfeit issues;
- (ii)
- Analyze the various barriers affecting the establishment of a safe PSC and the critical success factors for overcoming these barriers; and
- (iii)
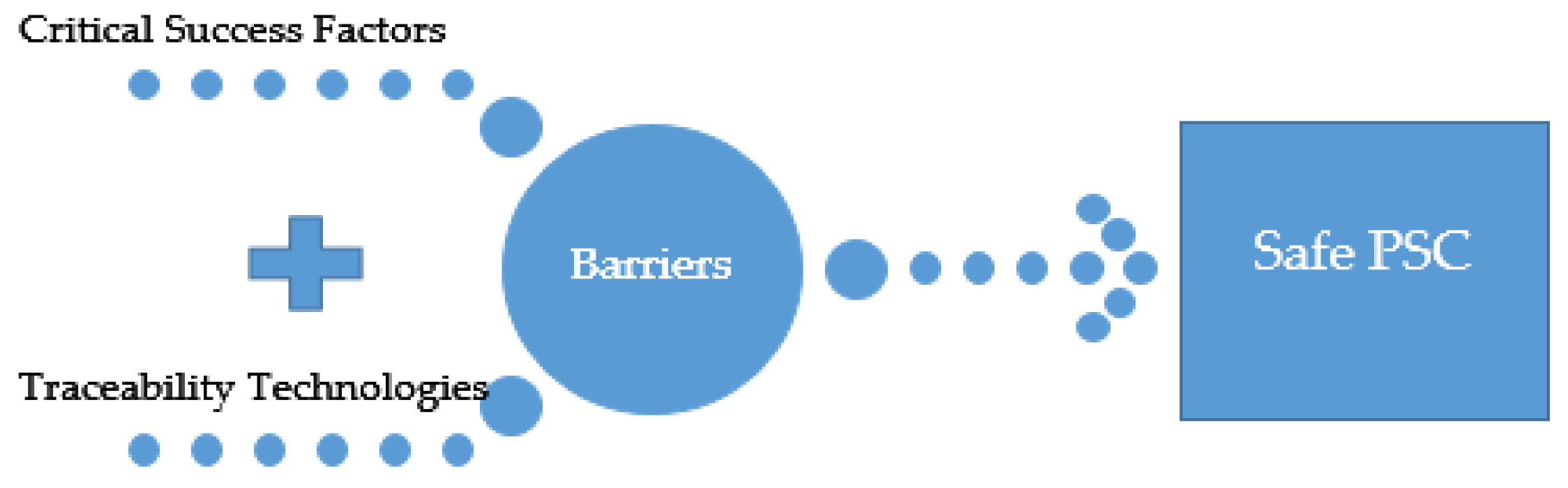
- Develop a conceptual framework and guidelines to demonstrate the influence of traceability technologies and success factors on overcoming the various barriers in different phases of the PSC.
2. Materials and Framework
2.1. Review Methodology
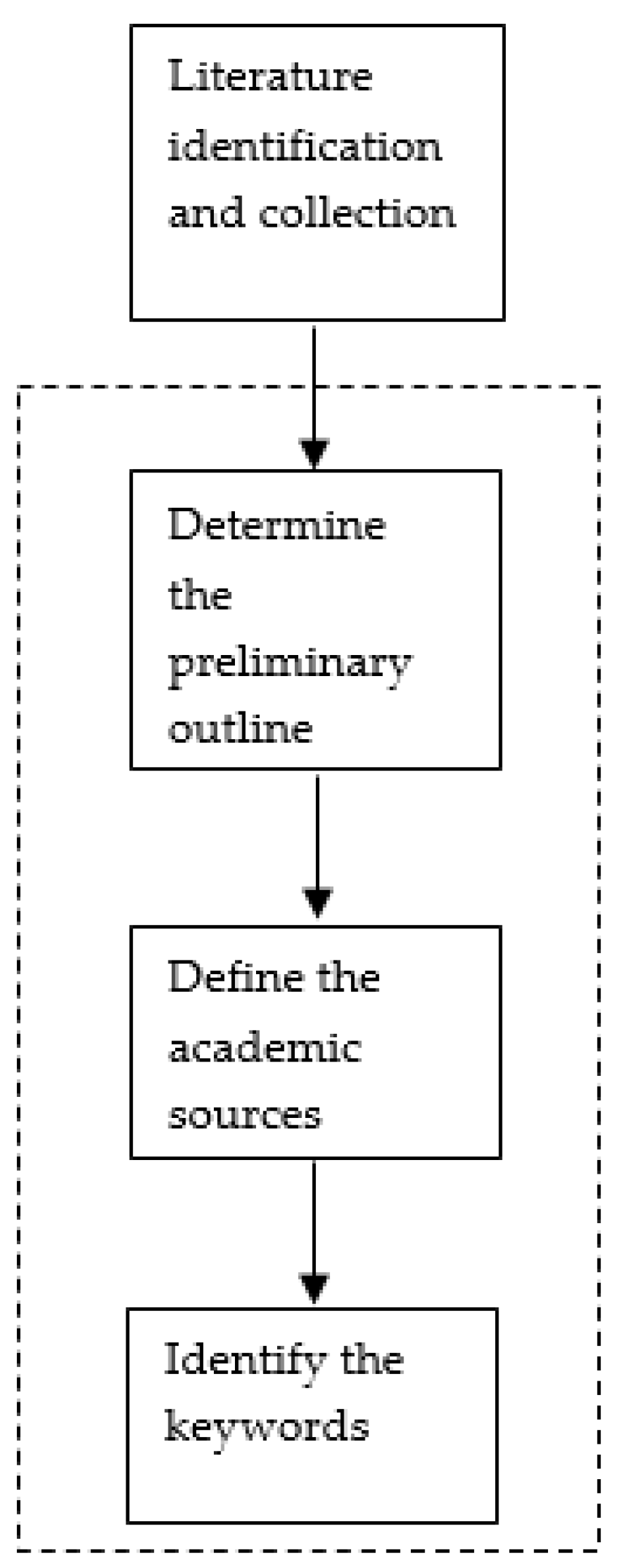
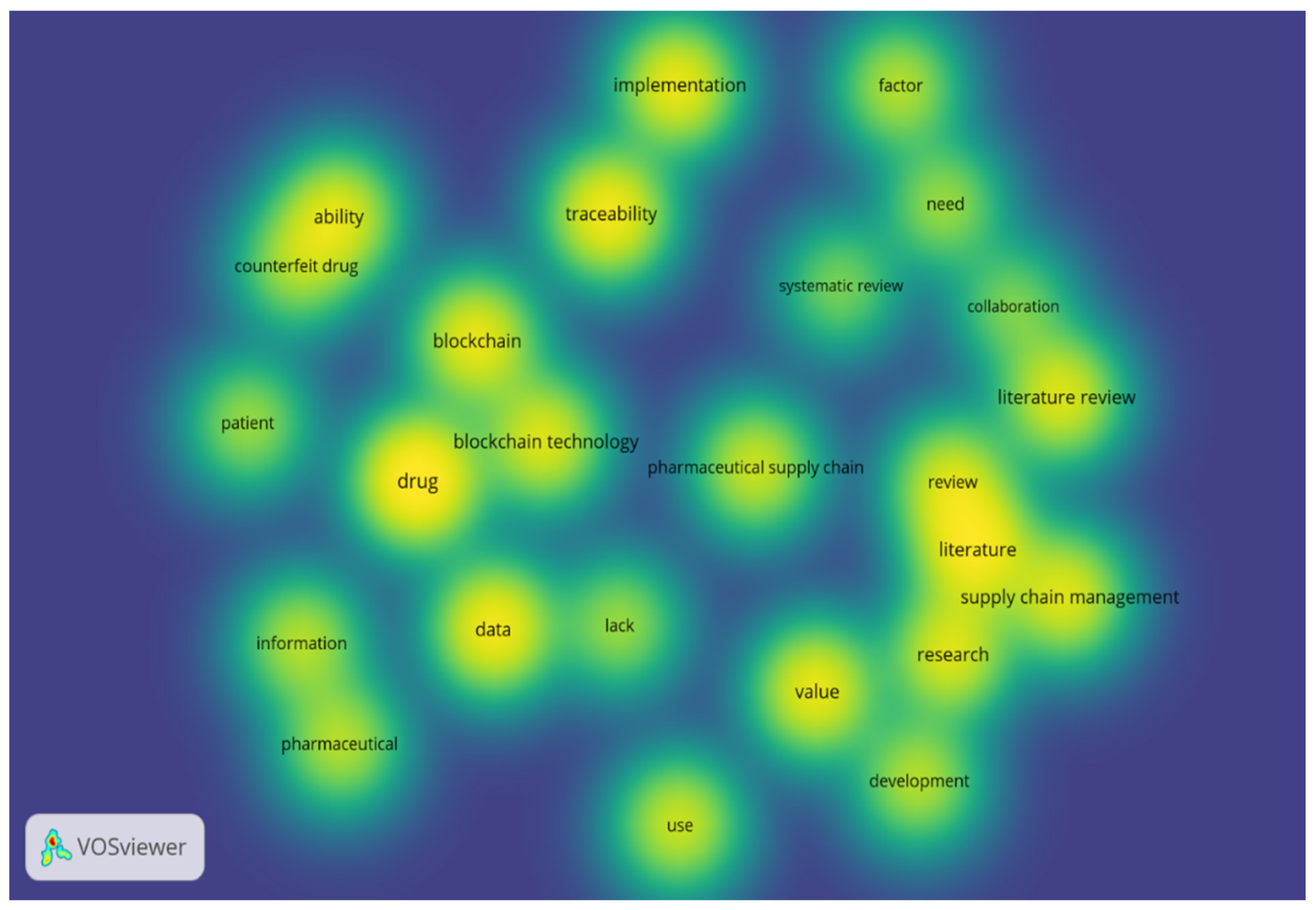
2.1.1. Literature Identification and Collection
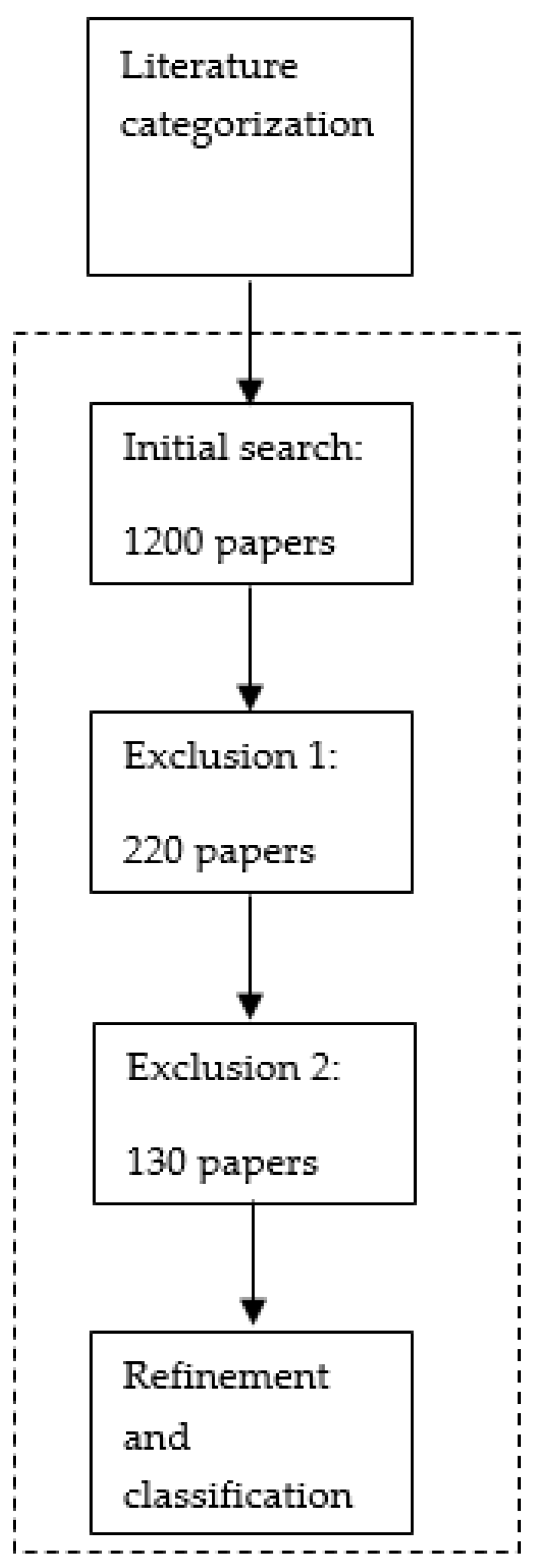
2.1.2. Literature Categorization
2.1.3. Literature Analysis, Evaluation, and Implications
3. Pharmaceutical Supply Chains
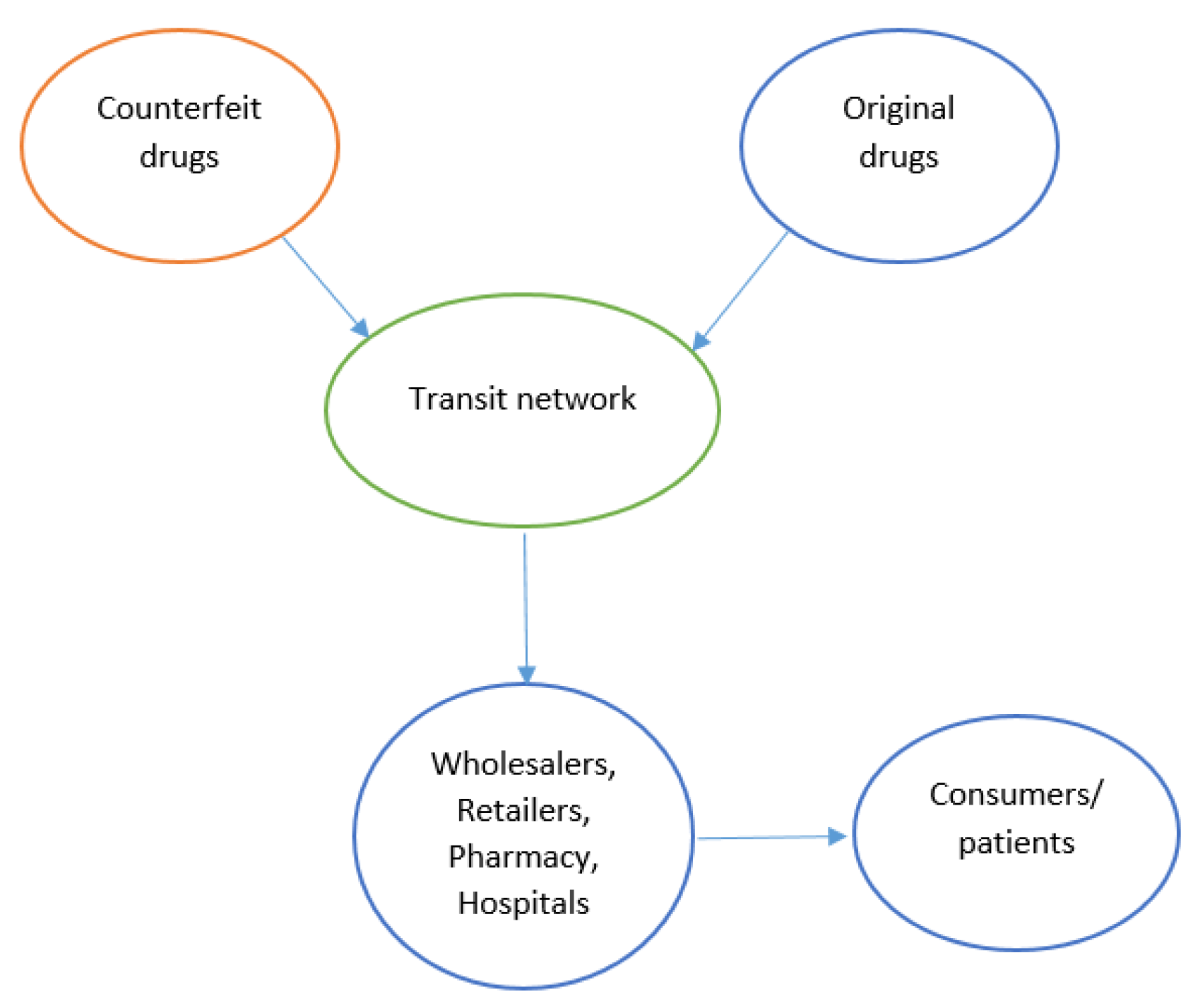
3.1. Issues of Counterfeit Medicines in the Legitimate PSC
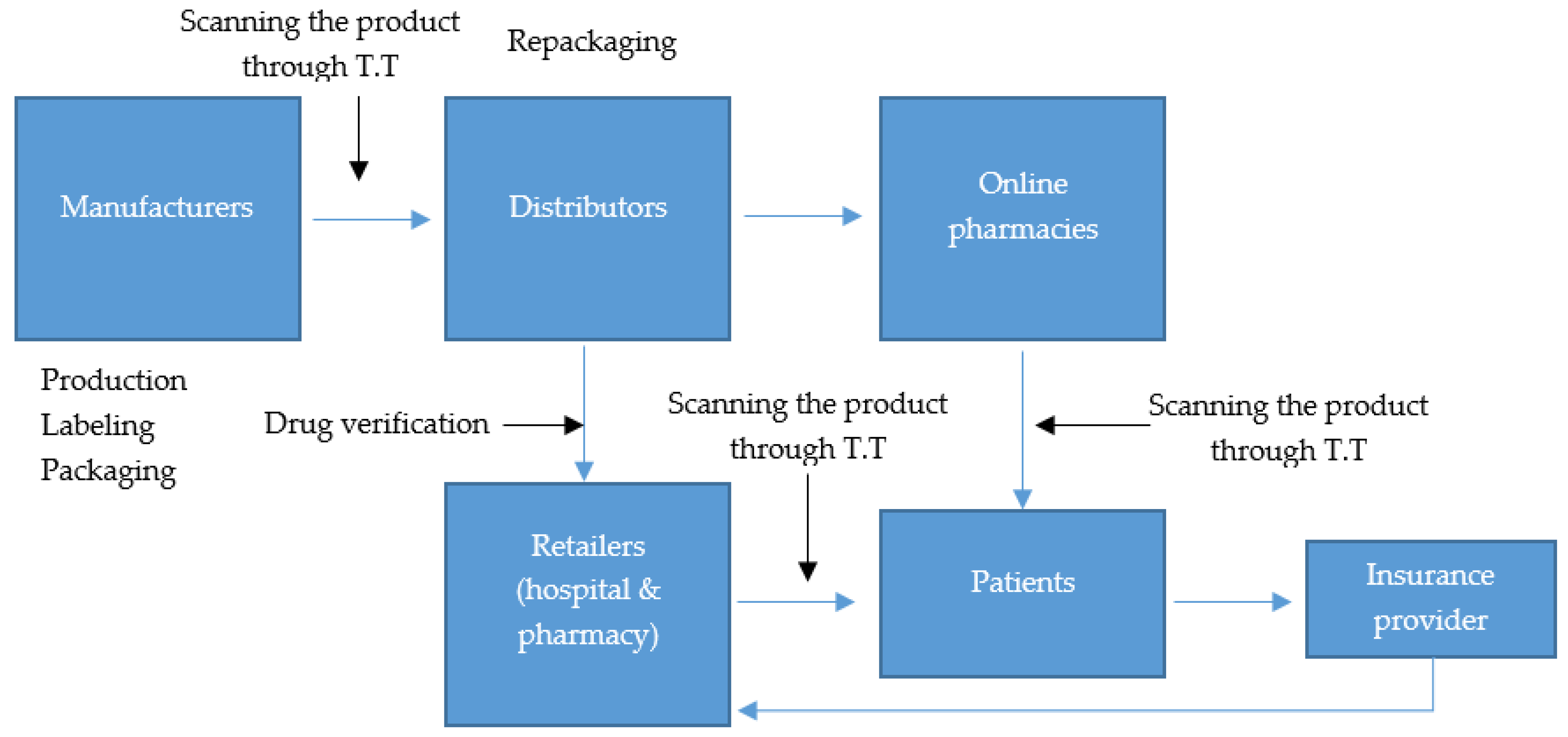
3.2. Traceability Technologies in Various Phases of the PSC
3.2.1. Definition of Traceability
3.2.2. Phases and Technologies
3.3. Barriers to Establishing a Safe Pharmaceutical Supply Chain
3.4. Critical Success Factors for Achieving a Safe Pharmaceutical Supply Chain
- a.
- Culture
- b.
- Trust
- c.
- Sharing information
- d.
- Cooperation
- e.
- Coordonation
4. Discussion
4.1. Conceptual Framework and Guidelines for a Safe PSC
- A PSC should be legitimate, regulated, and licensed by the health ministry or health body to supply authentic drugs to patients.
- In case of any suspicious activity concerning the packaging, pill size, color, texture, or taste compared to the previous prescription, the patient should report the matter or contact the manufacturers or the physician because these might be counterfeit medications.
- Healthcare and pharmacists can also assist patients in preventing counterfeit drugs from entering the PSC by purchasing from reliable and known sources. They can also warn patients against buying their medication online by telling them the dangers that they may encounter.
- Being cautious when shopping for medications online. Always shop in approved online pharmacies that display a Verified Internet Pharmacy Practice Sites (VIPPS) seal. Sites without this seal often sell counterfeit drugs.
- Implementing an effective PSC security system with several protective measures.
- Recommending ways in which health authorities can detect and deal with counterfeit drugs in the industry.
- Reaching a global common understanding to create awareness of what counterfeit drugs are.
- Developing a national action plan that creates prevention and detection measures to respond to counterfeit drugs.
- Connecting and collaborating with the patients and the supply chain partners in order to provide end-to-end visibility and different business interactions;
- Closely monitoring the manufacturing process to ensure product consistency, making sure that drugs are not contaminated, because an extra substance in the manufacturing process may be life threatening to the end beneficiaries;
- Having end-to-end traceability;
- Having a quick response to a change in demand. Responding to demand changes quickly and coming up with the best solutions is important in the pharmaceutical industry; and
- Having complete inventory visibility. Drug manufacturers should ensure complete downstream inventory visibility with good shipping practices. This eventually ensures that the products are reaching their intended destination.
4.2. Overall Findings
4.3. Managerial Implications
4.4. Theoretical Implications
4.5. Limitations and Scope of Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ghadge, A.; Duck, A.; Er, M.; Caldwell, N. Deceptive counterfeit risk in global supply chains. Supply Chain Forum Int. J. 2021, 22, 87–99. [Google Scholar] [CrossRef]
- Venhuis, B.J.; Oostlander, A.E.; Di Giorgio, D.; Mosimann, R.; du Plessis, I. Oncology drugs in the crosshairs of pharmaceutical crime. Lancet Oncol. 2018, 19, e209–e217. [Google Scholar] [CrossRef]
- Singh, R.; Dwivedi, A.D.; Srivastava, G. Internet of Things Based Blockchain for Temperature Monitoring and Counterfeit Pharmaceutical Prevention. Sensors 2020, 20, 3951. [Google Scholar] [CrossRef]
- Bansal, D.; Malla, S.; Gudala, K.; Tiwari, P. Anti-Counterfeit Technologies: A Pharmaceutical Industry Perspective. Sci. Pharm. 2013, 81, 1–13. [Google Scholar] [CrossRef]
- Zhang, H.; Hua, D.; Huang, C.; Samal, S.K.; Xiong, R.; Sauvage, F.; Braeckmans, K.; Remaut, K.; De Smedt, S. Materials and Technologies to Combat Counterfeiting of Pharmaceuticals: Current and Future Problem Tackling. Adv. Mater. 2020, 32, e1905486. [Google Scholar] [CrossRef]
- Yu, X.; Li, C.; Shi, Y.; Yu, M. Pharmaceutical supply chain in China: Current issues and implications for health system reform. Health Policy 2010, 97, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Coustasse, A.; Tomblin, S.; Slack, C. Impact of radio-frequency identification (RFID) technologies on the hospital supply chain: A literature review. Perspect. Health Inf. Manag. 2013, 10, 1–17. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3797551/ (accessed on 10 August 2021).
- Jaberidoost, M.; Nikfar, S.; Abdollahiasl, A.; Dinarvand, R. Pharmaceutical supply chain risks: A systematic review. DARU J. Pharm. Sci. 2013, 21, 69. [Google Scholar] [CrossRef]
- Kovacs, S.; Hawes, S.E.; Maley, S.N.; Mosites, E.; Wong, L.; Stergachis, A. Technologies for Detecting Falsified and Substandard Drugs in Low and Middle-Income Countries. PLoS ONE 2014, 9, e90601. [Google Scholar] [CrossRef]
- Ab Talib, M.S.; Hamid, A.B.A.; Thoo, A.C. Critical success factors of supply chain management: A literature survey and Pareto analysis. EuroMed J. Bus. 2015, 10, 234–263. [Google Scholar] [CrossRef]
- Hamilton, W.L.; Doyle, C.; Halliwell-Ewen, M.; Lambert, G. Public health interventions to protect against falsified medicines: A systematic review of international, national and local policies. Health Policy Plan. 2016, 31, 1448–1466. [Google Scholar] [CrossRef]
- Fadlallah, R.; El-Jardali, F.; Annan, F.; Azzam, H.; Akl, E.A. Strategies and Systems-Level Interventions to Combat or Prevent Drug Counterfeiting: A Systematic Review of Evidence Beyond Effectiveness. Pharm. Med. 2016, 30, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Sheikhi, M.; Goordarzi, M.; Shabani, M. Key Success Factors of drug Supply Chain Performance. In Proceedings of the International Conference on Industrial Engineering and Operations Management, Rabat, Morocco, 11–13 April 2017; Available online: http://ieomsociety.org/ieom2017/papers/371.pdf (accessed on 10 August 2021).
- Mackey, T.K.; Nayyar, G. A review of existing and emerging digital technologies to combat the global trade in fake medicines. Expert Opin. Drug Saf. 2017, 16, 587–602. [Google Scholar] [CrossRef]
- Ding, B. Pharma Industry 4.0: Literature review and research opportunities in sustainable pharmaceutical supply chains. Process. Saf. Environ. Prot. 2018, 119, 115–130. [Google Scholar] [CrossRef]
- De Lima, F.R.P.; Da Silva, A.L.; Filho, M.G.; Dias, E.M. Systematic review: Resilience enablers to combat counterfeit medicines. Supply Chain Manag. Int. J. 2018, 12, 117–135. [Google Scholar] [CrossRef]
- Da Silva, R.B.; De Mattos, C.A. Critical Success Factors of a Drug Traceability System for Creating Value in a Pharmaceutical Supply Chain (PSC). Int. J. Environ. Res. Public Health 2019, 16, 1972. [Google Scholar] [CrossRef] [PubMed]
- Bottoni, P.; Caroli, S. Fake pharmaceuticals: A review of current analytical approaches. Microchem. J. 2019, 149, 104053. [Google Scholar] [CrossRef]
- Bolla, A.S.; Patel, A.R.; Priefer, R. The silent development of counterfeit medications in developing countries—A systematic review of detection technologies. Int. J. Pharm. 2020, 587, 119702. [Google Scholar] [CrossRef]
- Yao, W.; Chu, C.H.; Li, Z. The use of RFID in healthcare: Benefits and barriers. In Proceedings of the 2010 IEEE International Conference on RFID-Technology and Applications, Guangzhou, China, 17–19 June 2010; pp. 128–134. [Google Scholar] [CrossRef]
- Tseng, J.-H.; Liao, Y.-C.; Chong, B.; Liao, S.-W. Governance on the Drug Supply Chain via Gcoin Blockchain. Int. J. Environ. Res. Public Health 2018, 15, 1055. [Google Scholar] [CrossRef]
- World Health Organization. Counterfeit Drugs: Guidelines for the Development of Measures to Combat Counterfeit Drugs (No. WHO/EDM/QSM/99.1); World Health Organization: Geneva, Switzerland, 1999; Available online: https://apps.who.int/iris/bitstream/handle/10665/65892/WHO_EDM_QSM_99.1.pdf (accessed on 10 August 2021).
- Modgil, S.; Singh, R.K.; Hannibal, C. Artificial intelligence for supply chain resilience: Learning from COVID-19. Int. J. Logist. Manag. 2021. [Google Scholar] [CrossRef]
- Burns, W. WHO launches taskforce to fight counterfeit drugs. Bull. World Health Organ. 2006, 84, 689–690. Available online: https://www.scielosp.org/pdf/bwho/2006.v84n9/689-690/en (accessed on 10 August 2021).
- Bryatov, S.R.; Borodinov, A.A. Blockchain technology in the pharmaceutical supply chain: Researching a business model based on Hyperledger Fabric. In Proceedings of the International Conference on Information Technology and Nanotechnology (ITNT), Samara, Russia, 21–24 May 2019; Available online: http://ceur-ws.org/Vol-2416/paper18.pdf (accessed on 10 August 2021).
- Abbas, K.; Afaq, M.; Khan, T.A.; Song, W.-C. A Blockchain and Machine Learning-Based Drug Supply Chain Management and Recommendation System for Smart Pharmaceutical Industry. Electronics 2020, 9, 852. [Google Scholar] [CrossRef]
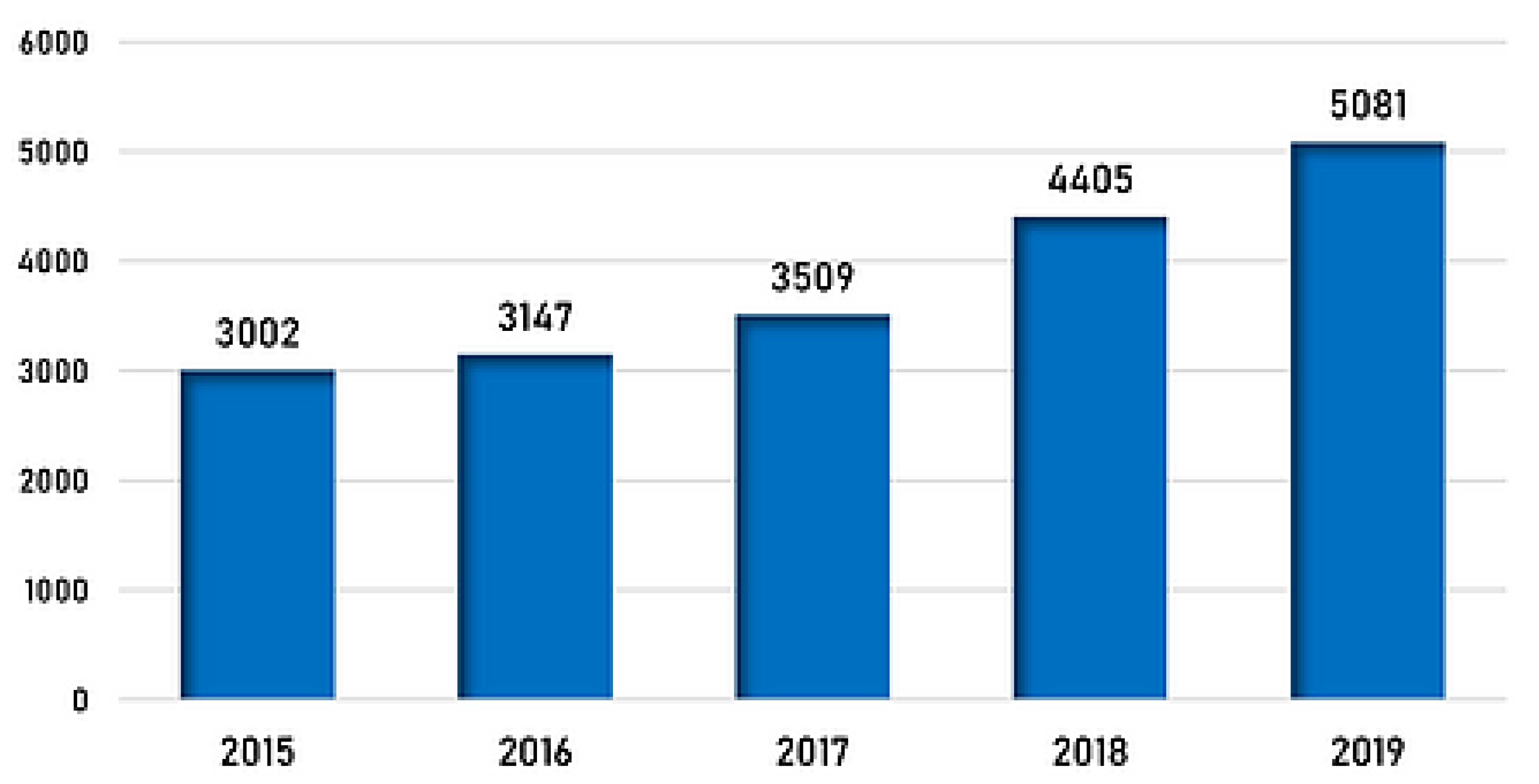
- PSI. Number of Incidents CY 2015–CY 2019 [Figure 8]. 2019. Available online: https://www.psi-inc.org/pharma-crime/incident-trends (accessed on 10 December 2020).
- Khan, S.; Haleem, A.; Khan, M.I.; Abidi, M.H.; Al-Ahmari, A. Implementing Traceability Systems in Specific Supply Chain Management (SCM) through Critical Success Factors (CSFs). Sustainability 2018, 10, 204. [Google Scholar] [CrossRef]
- King, B.; Zhang, X. Securing the pharmaceutical supply chain using RFID. In Proceedings of the 2007 International Conference on Multimedia and Ubiquitous Engineering (MUE’07), Seoul, Korea, 26–28 April 2007; pp. 23–28. [Google Scholar] [CrossRef]
- Theyel, G. Biomedical value chain traceability for innovation. In Proceedings of the 2017 IEEE Technology & Engineering Management Conference (TEMSCON), Santa Clara, CA, USA, 8–10 June 2017; pp. 295–300. [Google Scholar] [CrossRef]
- Cohen, D.F.; Kirshner, J. 6. The Cult of Energy Insecurity and Great Power Rivalry across the Pacific. In The Nexus of Economics, Security, and International Relations in East Asia; Stanford University Press: Palo Alto, CA, USA, 2020; pp. 144–176. [Google Scholar] [CrossRef]
- Barchetti, U.; Bucciero, A.; De Blasi, M.; Mainetti, L.; Patrono, L. RFID, EPC and B2B convergence towards an item-level traceability in the pharmaceutical supply chain. In Proceedings of the 2010 IEEE International Conference on RFID-Technology and Applications, Guangzhou, China, 17–19 June 2010; pp. 194–199. [Google Scholar] [CrossRef]
- Salim, M.; Widodo, R.; Noordin, M. Proof-of-Concept of Detection of Counterfeit Medicine through Polymeric Materials Analysis of Plastics Packaging. Polymers 2021, 13, 2185. [Google Scholar] [CrossRef] [PubMed]
- Modgil, S.; Sonwaney, V. Planning the application of blockchain technology in identification of counterfeit products: Sectorial prioritization. IFAC-PapersOnLine 2019, 52, 1–5. [Google Scholar] [CrossRef]
- Haji, M.; Kerbache, L.; Muhammad, M.; Al-Ansari, T. Roles of Technology in Improving Perishable Food Supply Chains. Logistics 2020, 4, 33. [Google Scholar] [CrossRef]
- Jamil, F.; Ahmad, S.; Iqbal, N.; Kim, D.-H. Towards a Remote Monitoring of Patient Vital Signs Based on IoT-Based Blockchain Integrity Management Platforms in Smart Hospitals. Sensors 2020, 20, 2195. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Jamil, F.; Ahmad, S.; Kim, D. A Novel Blockchain-Based Integrity and Reliable Veterinary Clinic Information Management System Using Predictive Analytics for Provisioning of Quality Health Services. IEEE Access 2021, 9, 8069–8098. [Google Scholar] [CrossRef]
- Shahbazi, Z.; Byun, Y.-C. A Procedure for Tracing Supply Chains for Perishable Food Based on Blockchain, Machine Learning and Fuzzy Logic. Electronics 2020, 10, 41. [Google Scholar] [CrossRef]
- Glover, D.G.; Hermans, J. Improving the traceability of the clinical trial supply chain. Appl. Clin. Trials 2017, 26, 36–38. [Google Scholar]
- Deisingh, A.K. Pharmaceutical counterfeiting. Analyst 2005, 130, 271–279. [Google Scholar] [CrossRef]
- Woods, J.; Iyengar-Emens, R. Blockchain to secure a more personalized pharma: Digital ledgers can detect fake drugs, enable virtual trials, and advance personalized medicine. Genet. Eng. Biotechnol. News 2019, 39, 27–29. [Google Scholar] [CrossRef]
- Schöner, M.M.; Kourouklis, D.; Sandner, P.; Gonzalez, E.; Förster, J. Blockchain Technology in the Pharmaceutical Industry; Frankfurt School Blockchain Center: Frankfurt, Germany, 2017; Available online: https://pdfs.semanticscholar.org/e091/f69e35d947d841c78c069e6c0e521df6180d.pdf (accessed on 10 August 2021).
- Huang, Y.; Wu, J.; Long, C. Drugledger: A practical blockchain system for drug Traceability and regulation. In Proceedings of the 2018 IEEE International Conference on Internet of Things (iThings) and IEEE Green Computing and Communications (GreenCom) and IEEE Cyber, Physical and Social Computing (CPSCom) and IEEE Smart Data (SmartData), Halifax, NS, Canada, 30 July–3 August 2018; pp. 1137–1144. [Google Scholar] [CrossRef]
- Kumar, A.; Choudhary, D.; Raju, M.S.; Chaudhary, D.K.; Sagar, R.K. Combating counterfeit drugs: A quantitative analysis on cracking down the fake drug industry by using blockchain technology. In Proceedings of the 2019 9th International Conference on Cloud Computing, Data Science & Engineering (Confluence), Noida, India, 10–11 January 2019; pp. 174–178. [Google Scholar] [CrossRef]
- Kumar, R.; Tripathi, R. Traceability of counterfeit medicine supply chain through Blockchain. In Proceedings of the 2019 11th International Conference on Communication Systems & Networks (COMSNETS), Bengaluru, India, 7–11 January 2019; pp. 568–570. [Google Scholar] [CrossRef]
- Shahbazi, Z.; Byun, Y.-C. Integration of Blockchain, IoT and Machine Learning for Multistage Quality Control and Enhancing Security in Smart Manufacturing. Sensors 2021, 21, 1467. [Google Scholar] [CrossRef]
- Gautam, C.S.; Utreja, A.; Singal, G.L. Spurious and counterfeit drugs: A growing industry in the developing world. Postgrad. Med. J. 2009, 85, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Klein, K.; Stolk, P. Challenges and Opportunities for the Traceability of (Biological) Medicinal Products. Drug Saf. 2018, 41, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Sharifian, R.; Ebrahimi, S.; Bastani, P. How radio frequency identification improves pharmaceutical industry: A comprehensive review literature. J. Pharm. Care 2015, 3, 26–33. Available online: http://jpc.tums.ac.ir/index.php/jpc/article/view/92 (accessed on 10 August 2021).
- Nilsson, E.; Nilsson, B.; Järpe, E. A pharmaceutical anti-counterfeiting method using time controlled numeric tokens. In Proceedings of the 2011 IEEE International Conference on RFID-Technologies and Applications, Sitges, Spain, 15–16 September 2011; pp. 343–347. [Google Scholar] [CrossRef]
- Ng, E.H.; Nepal, B.; Schott, E.; Keathley, H. Internet of things: Opportunities and applications in pharmaceutical manufacturing and logistics. In Proceedings of the American Society for Engineering Management 2018, Coeur d’Alene, ID, USA, 17–20 October 2018; Available online: https://www.researchgate.net/profile/Aniket-Marathe-4/publication/328269133_INTERNET_OF_THINGS_OPPORTUNITIES_AND_APPLICATIONS_IN_PHARMACEUTICAL_MANUFACTURING_AND_LOGISTICS/links/5c1fb8b1a6fdccfc7064a93e/INTERNET-OF-THINGS-OPPORTUNITIES-AND-APPLICATIONS-IN-PHARMACEUTICAL-MANUFACTURING-AND-LOGISTICS.pdf (accessed on 10 August 2021).
- Pérez, M.M.; González, G.V.; Dafonte, C. Safety and Traceability in Patient Healthcare through the Integration of RFID Technology for Intravenous Mixtures in the Prescription-Validation-Elaboration-Dispensation-Administration Circuit to Day Hospital Patients. Sensors 2016, 16, 1188. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen León-Araujo, M.; Gómez-Inhiesto, E.; Acaiturri-Ayesta, M.T. Implementation and evaluation of a RFID smart cabinet to improve traceability and the efficient consumption of high cost medical supplies in a large hospital. J. Med. Syst. 2019, 43, 178. Available online: https://link.springer.com/article/10.1007/s10916-019-1269-6 (accessed on 10 August 2021).
- Thatcher, C.; Acharya, S. Pharmaceutical uses of blockchain technology. In Proceedings of the 2018 IEEE International Conference on Advanced Networks and Telecommunications Systems (ANTS), Indore, India, 16–19 December 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Jamil, F.; Hang, L.; Kim, K.; Kim, D. A Novel Medical Blockchain Model for Drug Supply Chain Integrity Management in a Smart Hospital. Electronics 2019, 8, 505. [Google Scholar] [CrossRef]
- Imran; Iqbal, N.; Ahmad, S.; Kim, D. Health Monitoring System for Elderly Patients Using Intelligent Task Mapping Mechanism in Closed Loop Healthcare Environment. Symmetry 2021, 13, 357. [Google Scholar] [CrossRef]
- Iqbal, N.; Imran; Ahmad, S.; Ahmad, R.; Kim, D.-H. A Scheduling Mechanism Based on Optimization Using IoT-Tasks Orchestration for Efficient Patient Health Monitoring. Sensors 2021, 21, 5430. [Google Scholar] [CrossRef]
- Breen, L. A Preliminary Examination of Risk in the Pharmaceutical Supply Chain (PSC) in the National Health Service (NHS). J. Serv. Sci. Manag. 2008, 1, 193–199. [Google Scholar] [CrossRef]
- Tremblay, M. Medicines counterfeiting is a complex problem: A review of key challenges across the supply chain. Curr. Drug Saf. 2013, 8, 43–55. [Google Scholar] [CrossRef]
- Punniyamoorthy, M.; Thamaraiselvan, N.; Manikandan, L. Assessment of supply chain risk: Scale development and validation. Benchmarking Int. J. 2013, 20, 79–105. [Google Scholar] [CrossRef]
- Narayana, S.A.; Pati, R.K.; Vrat, P. Research on management issues in the pharmaceutical industry: A literature review. Int. J. Pharm. Health Mark. 2012, 6, 351–375. [Google Scholar] [CrossRef]
- Lee, K.; Kassab, Y.; Taha, N.; Zainal, Z. Factors Impacting Pharmaceutical Prices and Affordability: Narrative Review. Pharmacy 2020, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Du, Y.; Wang, Q.; Sun, C.; Ling, X.; Yu, B.; Tu, J.; Xiong, Y. Risk assessment of supply chain for pharmaceutical excipients with AHP-fuzzy comprehensive evaluation. Drug Dev. Ind. Pharm. 2016, 42, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Soon, J.M.; Manning, L. Developing anti-counterfeiting measures: The role of smart packaging. Food Res. Int. 2019, 123, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Kumar, R.; Kumar, P. Strategic issues in pharmaceutical supply chains: A review. Int. J. Pharm. Health Mark. 2016, 10, 234–257. [Google Scholar] [CrossRef]
- Mäkinen, M.M.; Rautava, P.T.; Forsström, J.J. Do online pharmacies fit European internal markets? Health Policy 2005, 72, 245–252. [Google Scholar] [CrossRef]
- Gray, N. The evolution of online pharmacies. Self Care J. 2011, 2, 76–86. Available online: https://selfcarejournal.com/wp-content/uploads/2015/09/Gray-2.376-86.pdf (accessed on 10 August 2021).
- Leontiadis, N.; Moore, T.; Christin, N. Pick your poison: Pricing and inventories at unlicensed online pharmacies. In Proceedings of the Fourteenth ACM Conference on Electronic Commerce, Philadelphia, PA, USA, 16–20 June 2013; pp. 621–638. [Google Scholar] [CrossRef]
- Shaikh, Z.A.; Buzdar, M.H.; Pahore, M.R.; Mirani, I.H.; Noor, M. The Emergent Business of Internet Pharmacies: Convenience, Risks, Regulatory Policies and Future. J. Pharm. Res. Int. 2019, 31, 1–6. [Google Scholar] [CrossRef]
- Stank, T.P.; Keller, S.B.; Daugherty, P.J. Supply chain collaboration and logistical service performance. J. Bus. Logist. 2001, 22, 29–48. [Google Scholar] [CrossRef]
- Barratt, M. Understanding the meaning of collaboration in the supply chain. Supply Chain Manag. Int. J. 2004, 9, 30–42. [Google Scholar] [CrossRef]
- Min, S.; Roath, A.S.; Daugherty, P.J.; Genchev, S.E.; Chen, H.; Arndt, A.D.; Richey, R.G. Supply chain collaboration: What’s happening? Int. J. Logist. Manag. 2005, 16, 237–256. [Google Scholar] [CrossRef]
- Matopoulos, A.; Vlachopoulou, M.; Manthou, V.; Manos, B. A conceptual framework for supply chain collaboration: Empirical evidence from the agri-food industry. Supply Chain Manag. Int. J. 2007, 12, 177–186. [Google Scholar] [CrossRef]
- Fawcett, S.E.; Magnan, G.M.; McCarter, M.W. Benefits, barriers, and bridges to effective supply chain management. Supply Chain Manag. Int. J. 2008, 13, 35–48. [Google Scholar] [CrossRef]
- De Vries, J.; Huijsman, R. Supply chain management in health services: An overview. Supply Chain Manag. Int. J. 2011, 16, 159–165. [Google Scholar] [CrossRef]
- Karlsen, K.; Sørensen, C.; Forås, F.; Olsen, P. Critical criteria when implementing electronic chain traceability in a fish supply chain. Food Control 2011, 22, 1339–1347. [Google Scholar] [CrossRef]
- Papert, M.; Rimpler, P.; Pflaum, A. Enhancing supply chain visibility in a pharmaceutical supply chain. Int. J. Phys. Distrib. Logist. Manag. 2016, 46, 859–884. [Google Scholar] [CrossRef]
- Tian, F. A supply chain traceability system for food safety based on HACCP, blockchain & Internet of Things. In Proceedings of the 2017 International Conference on Service Systems and Service Management, Honolulu, HI, USA, 25–30 June 2017; pp. 1–6. [Google Scholar] [CrossRef]
- Fernando, E. Success factor of implementation blockchain technology in pharmaceutical industry: A literature review. In Proceedings of the 2019 6th International Conference on Information Technology, Computer and Electrical Engineering (ICITACEE), Semarang, Indonesia, 26–27 September 2019; pp. 1–5. [Google Scholar] [CrossRef]
- Sodhi, M.S.; Tang, C.S. Research Opportunities in Supply Chain Transparency. Prod. Oper. Manag. 2019, 28, 2946–2959. [Google Scholar] [CrossRef]
- Hastig, G.M.; Sodhi, M.S. Blockchain for Supply Chain Traceability: Business Requirements and Critical Success Factors. Prod. Oper. Manag. 2019, 29, 935–954. [Google Scholar] [CrossRef]
- Rogerson, M.; Parry, G.C. Blockchain: Case studies in food supply chain visibility. Supply Chain Manag. Int. J. 2020, 25, 601–614. [Google Scholar] [CrossRef]
- Subramanian, L. Enabling health supply chains for improved well-being. Supply Chain Forum: Int. J. 2020, 21, 229–236. [Google Scholar] [CrossRef]
- Khanuja, A.; Jain, R.K. Supply chain integration: A review of enablers, dimensions and performance. Benchmarking Int. J. 2019, 27, 264–301. [Google Scholar] [CrossRef]
- Cagliano, A.C.; Mangano, G.; Rafele, C. Determinants of digital technology adoption in supply chain. An exploratory analysis. Supply Chain Forum: Int. J. 2021, 22, 100–114. [Google Scholar] [CrossRef]
- Huang, Y.; Han, W.; Macbeth, D.K. The complexity of collaboration in supply chain networks. Supply Chain Manag. Int. J. 2020, 25, 393–410. [Google Scholar] [CrossRef]
- Zekhnini, K.; Cherrafi, A.; Bouhaddou, I.; Benghabrit, Y.; Garza-Reyes, J.A. Supply chain management 4.0: A literature review and research framework. Benchmarking Int. J. 2020, 28, 465–501. [Google Scholar] [CrossRef]
- Razak, G.M.; Hendry, L.C.; Stevenson, M. Supply chain traceability: A review of the benefits and its relationship with supply chain resilience. Prod. Plan. Control 2021, 33, 1–21. [Google Scholar] [CrossRef]
- Sunny, J.; Undralla, N.; Pillai, V.M. Supply chain transparency through blockchain-based traceability: An overview with demonstration. Comput. Ind. Eng. 2020, 150, 106895. [Google Scholar] [CrossRef]
- Akhtar, M.M.; Rizvi, D.R. Traceability and detection of counterfeit medicines in pharmaceutical supply chain using blockchain-based architectures. In Sustainable and Energy Efficient Computing Paradigms for Society; Springer: Cham, Switzerland, 2021; pp. 1–31. [Google Scholar] [CrossRef]
- Soundarya, K.; Pandey, P.; Dhanalakshmi, R. A Counterfeit Solution for Pharma Supply Chain. EAI Endorsed Trans. Cloud Syst. 2018, 3, 11. [Google Scholar] [CrossRef][Green Version]


| Keywords | Search Strings | Databases |
|---|---|---|
| 1. Traceability technologies to detect counterfeit drugs in the pharmaceutical supply chain | Traceability technologies AND Drug counterfeiting detection AND Pharmaceutical supply chain | Scopus EBSCOhost Science direct Emerald insight Taylor & Francis Online Springer search IEEE |
| 2. Barriers to detecting counterfeit drugs in the pharmaceutical supply chain | Barriers (OR risks) AND Drug counterfeiting detection AND Pharmaceutical supply chain | |
| 3. Critical success factors for detecting counterfeit drugs in the pharmaceutical supply chain | Critical success factors (OR success factors) AND Drug counterfeiting detection AND Pharmaceutical supply chain |
| Authors | Suppliers of Raw Materials | Manufacturers | Packagers | Distributors/Wholesalers | Hospitals/Pharmacies | Patients | |
|---|---|---|---|---|---|---|---|
| Phases | |||||||
| Deisingh [40] | RFID coupled with EPC Pedigrees and mass serialization | ||||||
| King and Zhang [29] | RFID | ||||||
| Gautam et al. [47] | Simple barcode | ||||||
| Nilsson et al. [50] | RFID | ||||||
| Coustasse et al. [7] | RFID | ||||||
| Sharifian et al. [49] | RFID | ||||||
| Martínez et al. [52] | RFID | ||||||
| Glover and Hermans [39] | Blockchain | ||||||
| Schöner et al. [42] | Blockchain | ||||||
| Huang et al. [43] | Blockchain | Smartphones | |||||
| Klein and Stolk [48] | 2D data matrix | ||||||
| Ng et al. [51] | IoT sensors coupled with RFID | ||||||
| Thatcher and Acharya [54] | Blockchain | ||||||
| Tseng et al. [21] | Blockchain | ||||||
| Venhuis et al. [2] | Unique 2D barcodes | ||||||
| Bryatov and Borodinov [25] | Blockchain | ||||||
| Del Carmen León-Araujo et al. [53] | RFID | ||||||
| Jamil et al. [55] | Blockchain | ||||||
| Kumar et al. [44] | Blockchain | ||||||
| Kumar and Tripathi [45] | Blockchain with QR | RFID | |||||
| Woods and Iyengar-Emens [41] | Blockchain | ||||||
| Abbas et al. [26] | Blockchain | ||||||
| Singh et al. [3] | IoT sensors based Blockchain | ||||||
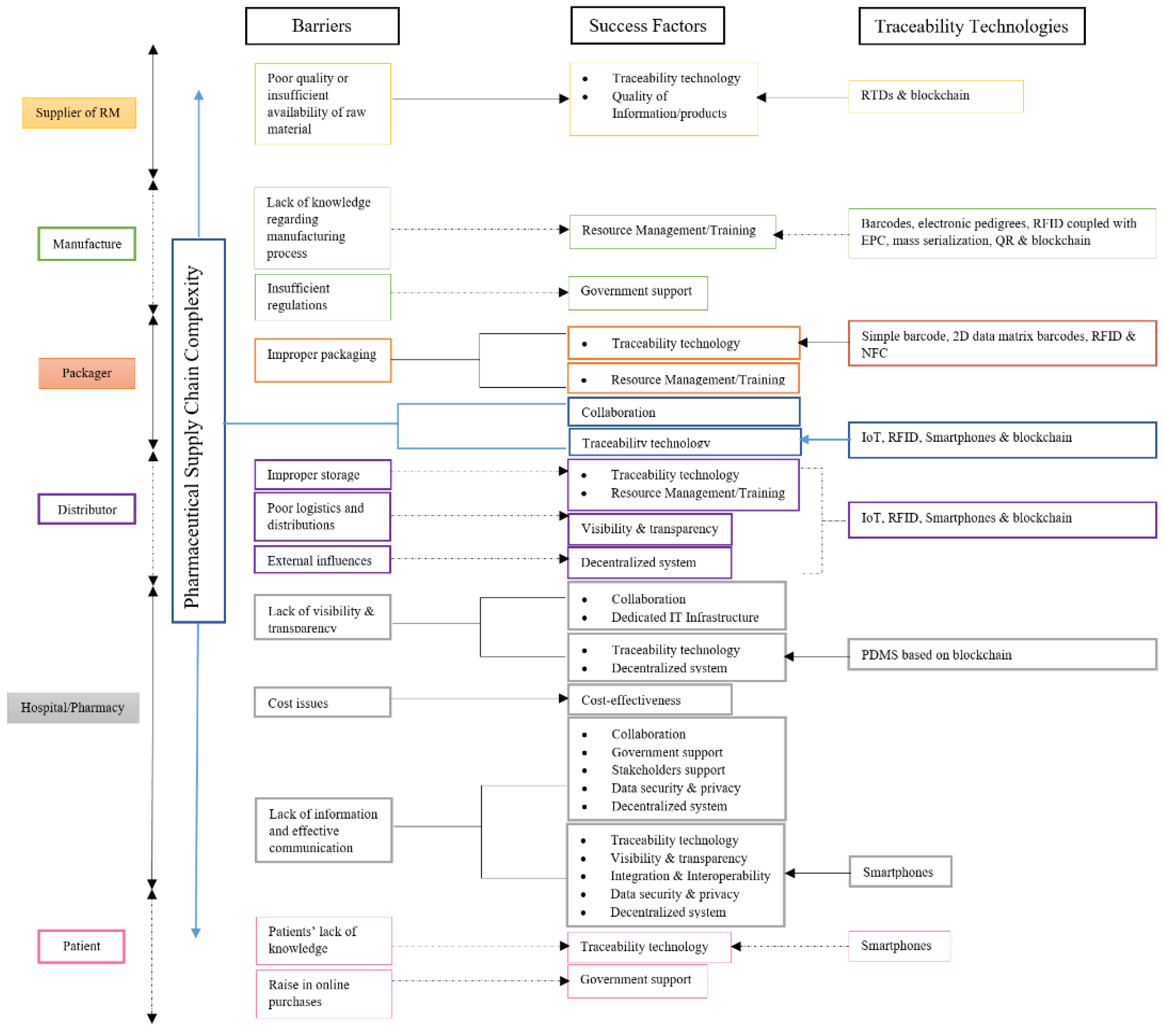
| Barriers | References |
| PSC complexity | Breen [58]; Jaberidoost et al. [8]; Tremblay [59]; Singh et al. [3] |
| Insufficient regulation | Breen [58]; Yu et al. [6]; Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Tremblay [59]; Venhuis et al. [2]; Kumar et al. [44] |
| Lack of visibility and transparency | Breen [58]; Yu et al. [6]; Jaberidoost et al. [8]; Tremblay [59]; Kumar et al. [44] |
| Cost issues | Breen [58]; Yu et al. [6]; Narayana et al. [61]; Bansal et al. [4]; Coustasse et al. [7]; Jaberidoost et al. [8]; Kumar et al. [44]; Lee et al. [62] |
| Lack of information and effective communication | Breen [58]; Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Tremblay [59]; Kumar et al. [44] |
| Poor quality or insufficient availability of raw material | Breen [58]; Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Li et al. [63]; Kumar et al. [44] |
| Lack of knowledge regarding the manufacturing process | Breen [58]; Narayana et al. [61]; Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Kumar et al. [44] |
| Improper packaging | Breen [58]; Tremblay [59]; Klein and Stolk [48]; Venhuis et al. [2]; Soon and Manning [64]; Salim et al. [33] |
| Improper storage | Breen [58]; Punniyamoorthy et al. [60]; Venhuis et al. [2]; Kumar et al. [44] |
| Poor logistics and distributions | Breen [58]; Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Singh et al. [65] |
| Patients’ lack of knowledge | Tremblay [59] |
| Rise in online purchases | Mäkinen et al. [66]; Gray [67]; Leontiadis [68]; Tremblay [59]; Shaikh et al. [69] |
| External influences | Jaberidoost et al. [8]; Punniyamoorthy et al. [60]; Kumar et al. [44] |
| Success Factors for Ideal Traceability | Stank et al. [70] | Barratt [71] | Min et al. [72] | Doukidis et al. [73] | Fawcett et al. [74] | De Vries and Huijsman [75] | Karlsen et al. [76] | Fadlallah et al. [12] | Papert et al. [77] | Tian [78] | Khan et al. [28] | De Lima et al. [16] | Silva and Mattos [17] | Fernando [79] | Sodhi and Tang [80] | Hastig and Sodhi [81] | Rogerson and Parry [82] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Collaboration and its drivers
| √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | ||||
| Visibility and transparency | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | |||||||
| Government support | √ | √ | √ | √ | √ | √ | |||||||||||
| Quality of information/ products | √ | √ | √ | √ | √ | ||||||||||||
| Integration and interoperability | √ | √ | √ | √ | √ | ||||||||||||
| Stakeholder support | √ | √ | √ | √ | √ | ||||||||||||
| Cost effectiveness | √ | √ | √ | √ | |||||||||||||
| Resource management/ training | √ | √ | √ | √ | |||||||||||||
| Data security and privacy | √ | √ | √ | √ | |||||||||||||
| Dedicated IT infrastructure | √ | √ | |||||||||||||||
| Decentralized system | √ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haji, M.; Kerbache, L.; Sheriff, K.M.M.; Al-Ansari, T. Critical Success Factors and Traceability Technologies for Establishing a Safe Pharmaceutical Supply Chain. Methods Protoc. 2021, 4, 85. https://doi.org/10.3390/mps4040085
Haji M, Kerbache L, Sheriff KMM, Al-Ansari T. Critical Success Factors and Traceability Technologies for Establishing a Safe Pharmaceutical Supply Chain. Methods and Protocols. 2021; 4(4):85. https://doi.org/10.3390/mps4040085
Chicago/Turabian StyleHaji, Mona, Laoucine Kerbache, K.M. Mahaboob Sheriff, and Tareq Al-Ansari. 2021. "Critical Success Factors and Traceability Technologies for Establishing a Safe Pharmaceutical Supply Chain" Methods and Protocols 4, no. 4: 85. https://doi.org/10.3390/mps4040085
APA StyleHaji, M., Kerbache, L., Sheriff, K. M. M., & Al-Ansari, T. (2021). Critical Success Factors and Traceability Technologies for Establishing a Safe Pharmaceutical Supply Chain. Methods and Protocols, 4(4), 85. https://doi.org/10.3390/mps4040085






