The ATM Gene in Breast Cancer: Its Relevance in Clinical Practice
Abstract
:1. Introduction
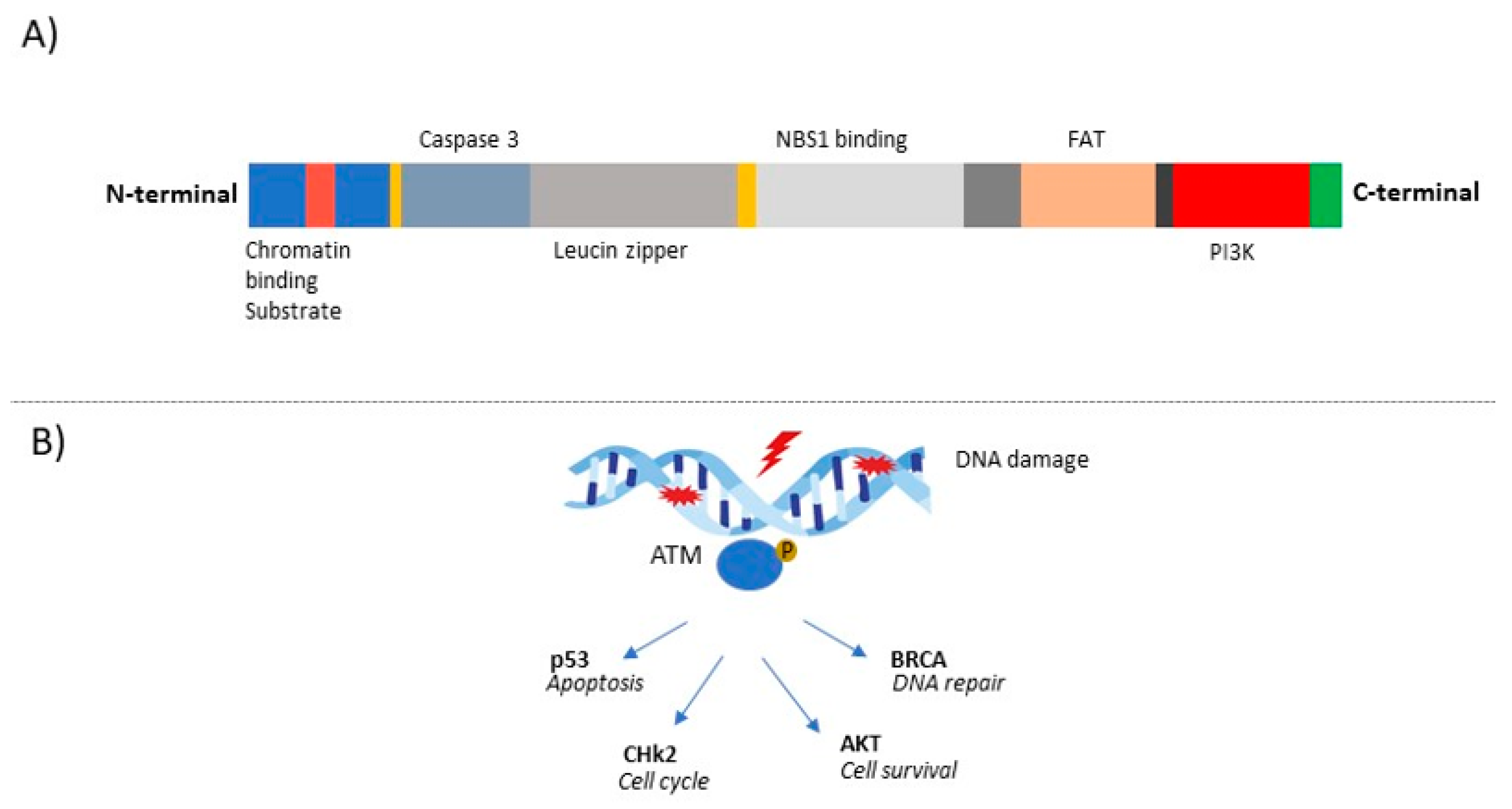
2. The ATM Gene and Its Role in Cancer
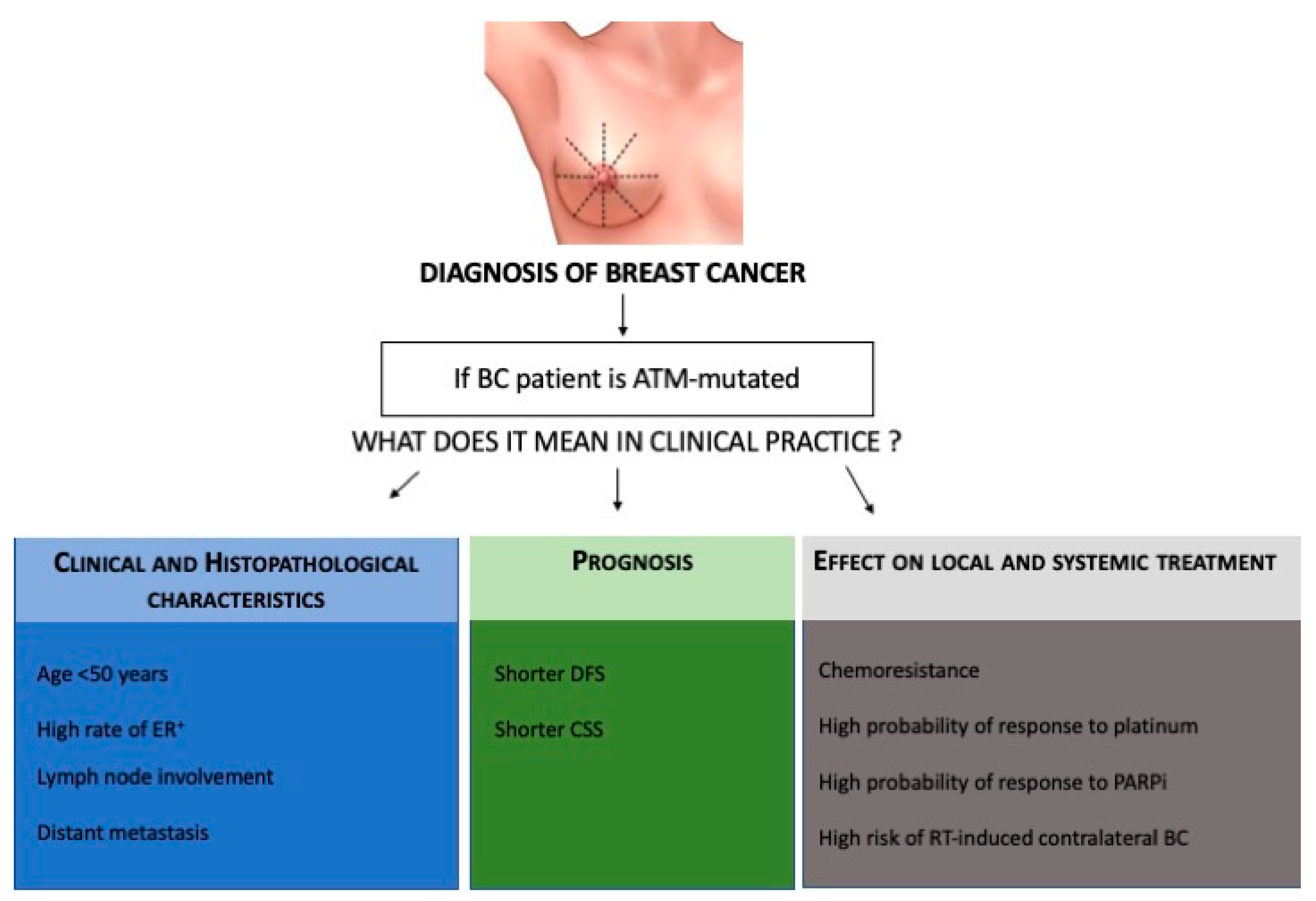
3. Role of ATM Gene Mutations in BC Susceptibility and Prognosis
4. Therapeutic Implications of ATM Gene Mutations in BC
5. Discussion: How Could the ATM Gene Mutation Influence BC Management?
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moslemi, M.; Moradi, Y.; Dehghanbanadaki, H.; Afkhami, H.; Khaledi, M.; Sedighimehr, N.; Fathi, J.; Sohrabi, E. The association between ATM variants and risk of breast cancer: A systematic review and meta-analysis. BMC Cancer 2021, 21, 27. [Google Scholar] [CrossRef]
- Dörk, T.; Bendix, R.; Bremer, M.; Rades, D.; Klöpper, K.; Nicke, M.; Skawran, B.; Hector, A.; Yamini, P.; Steinmann, D.; et al. Spectrum of ATM Gene Mutations in a Hospital-Based Series of Unselected Breast Cancer Patients. Cancer Res. 2001, 61, 7608–7615. [Google Scholar]
- Bernstein, J.L.; Teraoka, S.; Southey, M.C.; Jenkins, M.A.; Andrulis, I.L.; Knight, J.A.; John, E.M.; Lapinski, R.; Wolitzer, A.L.; Whittemore, A.S. Population-Based Estimates of Breast Cancer Risks Associated with ATM Gene Variants c. 7271T> G and c. 1066–6T> G (IVS10–6T> G) from the Breast Cancer Family Registry. Hum. Mutat. 2006, 27, 1122–1128. [Google Scholar] [CrossRef]
- Shiloh, Y. ATM and Related Protein Kinases: Safeguarding Genome Integrity. Nat. Rev. Cancer 2003, 3, 155–168. [Google Scholar] [CrossRef]
- Savitsky, K.; Bar-Shira, A.; Gilad, S.; Rotman, G.; Ziv, Y.; Vanagaite, L.; Tagle, D.A.; Smith, S.; Uziel, T.; Sfez, S.; et al. A Single Ataxia Elangiectasia Gene with a Product Similar to PI-3 Kinase. Science 1995, 268, 1749–1753. [Google Scholar] [CrossRef]
- Lee, J.H.; Paull, T.T. ATM Activation by DNA Double-Strand Breaks through the Mre11-Rad50-Nbs1 Complex. Science 2005, 308, 551–554. [Google Scholar] [CrossRef]
- Rotman, G.; Shiloh, Y. ATM: From Gene to Function. Hum. Mol. Genet. 1998, 7, 1555–1563. [Google Scholar] [CrossRef] [Green Version]
- Matsuoka, S.; Ballif, B.A.; Smogorzewska, A.; McDonald, E.R., 3rd; Hurov, K.E.; Luo, J.; Bakalarski, C.E.; Zhao, Z.; Solimini, N.; Lerenthal, Y.; et al. ATM and ATR Substrate Analysis Reveals Extensive Protein Networks Responsive to DNA Damage. Science 2007, 316, 1160–1166. [Google Scholar] [CrossRef] [Green Version]
- Thompson, D.; Duedal, S.; Kirner, J.; McGuffog, L.; Last, J.; Reiman, A.; Byrd, P.; Taylor, M.; Easton, D.F. Cancer Risks and Mortality in Heterozygous ATM Mutation Carriers. J. Natl. Cancer Inst. 2005, 97, 813–822. [Google Scholar] [CrossRef]
- Goldgar, D.E.; Healey, S.; Dowty, J.G.; Da Silva, L.; Chen, X.; Spurdle, A.B.; Terry, M.B.; Daly, M.J.; Buys, S.M.; Southey, M.C.; et al. Rare Variants in the ATM Gene and Risk of Breast Cancer. Breast Cancer Res. 2011, 13, R73. [Google Scholar] [CrossRef] [Green Version]
- Angele, S.; Hall, J. The ATM Gene and Breast Cancer: Is It Really a Risk Factor? Mutat. Res. 2000, 462, 167–178. [Google Scholar] [CrossRef]
- Broeks, A.; Urbanus, J.H.; Floore, A.N.; Dahler, E.C.; Klijn, J.G.; Rutgers, E.J.T.; Devilee, P.; Russell, N.S.; van Leeuwen, F.E.; Veer, L.J.V. ATM-Heterozygous Germline Mutations Contribute to Breast Cancer-Susceptibility. Am. J. Hum. Genet. 2000, 66, 494–500. [Google Scholar] [CrossRef] [Green Version]
- Fletcher, O.; Johnson, N.; Silva, I.D.S.; Orr, N.; Ashworth, A.; Nevanlinna, H.; Heikkinen, T.; Aittomäki, K.; Blomqvist, C.; Burwinkel, B.; et al. Missense Variants in ATM in 26,101 Breast Cancer Cases and 29,842 Controls. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2143–2151. [Google Scholar] [CrossRef] [Green Version]
- Thorstenson, Y.R.; Roxas, A.; Kroiss, R.; Jenkins, M.A.; Yu, K.M.; Bachrich, T.; Muhr, D.; Wayne, T.L.; Chu, G.; Davis, R.W.; et al. Contributions of ATM Mutations to Familial Breast and Ovarian Cancer. Cancer Res. 2003, 63, 3325–3333. [Google Scholar]
- Cremona, C.A.; Behrens, A. ATM Signalling and Cancer. Oncogene 2014, 33, 3351–3360. [Google Scholar] [CrossRef] [Green Version]
- Negrini, M.; Rasio, D.; Hampton, G.M.; Sabbioni, S.; Rattan, S.; Carter, S.L.; Rosenberg, A.L.; Schwartz, G.F.; Shiloh, Y.; Cavenee, W.K. Definition and Refinement of Chromosome 11 Regions of Loss of Heterozygosity in Breast Cancer: Identification of a New Region at 11q23.3. Cancer Res. 1995, 55, 3003–3007. [Google Scholar]
- Laake, K.; Launonen, V.; Niederacher, D.; Gudlaugsdottir, S.; Seitz, S.; Rio, P.; Champème, M.H.; Bièche, I.; Birnbaum, D.; White, G.; et al. Loss of Heterozygosity at 11q23.1 and Survival in Breast Cancer: Results of a Large European Study. Breast Cancer Somatic Genet. Consort. Genes Chromosomes Cancer 1999, 25, 212–221. [Google Scholar] [CrossRef]
- Bueno, R.C.; Canevari, R.A.; Villacis, R.A.R.; Domingues, M.A.C.; Caldeira, J.R.F.; Rocha, R.M.; Drigo, S.A.; Rogatto, S.R. ATM Down-Regulation Is Associated with Poor Prognosis in Sporadic Breast Carcinomas. Ann. Oncol. 2014, 25, 69–75. [Google Scholar] [CrossRef]
- Berstein, J.L.; Haile, R.W.; Stovall, M.; Boice, J.D., Jr.; Shore, R.E.; Langholz, B.; Thomas, D.C.; Bernstein, L.; Charles, F.L.; Olsen, J.H.; et al. Radiation Exposure, the ATM Gene, and Contralateral Breast Cancer in the Women’s Environmental Cancer and Radiation Epidemiology Study. J. Natl. Cancer Inst. 2010, 102, 475–483. [Google Scholar] [CrossRef]
- Abraham, R.T. PI 3-Kinase Related Kinases: ‘Big’ Players in Stress-Induced Signaling Pathways. DNA Repair 2004, 3, 883–887. [Google Scholar] [CrossRef]
- Hall, M.J.; Bernhisel, R.; Hughes, E.; Larson, K.; Rosenthal, E.T.; Singh, N.A.; Lancaster, J.M.; Kurian, A.W. Germline Pathogenic Variants in the Ataxia telangiectasia Mutated (ATM) Gene Are Associated with High and Moderate Risks for Multiple Cancers. Cancer Prev. Res. 2021, 14, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Ando, K.; Kernan, J.L.; Liu, P.H.; Sanda, T.; Logette, E.; Tschopp, J.; Look, A.T.; Wang, J.; Bouchier-Hayes, L.; Sidi, S. PIDD Death-Domain Phosphorylation by ATM Controls Prodeath versus Prosurvival PIDDosome Signaling. Mol. Cell 2012, 47, 681–693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, L.; Lin, C.; Wu, Z.; Gong, H.; Zeng, Y.; Wu, J.; Li, M.; Li, J. miR-18a Impairs DNA Damage Response Through Downregulation of Ataxia telangiectasia Mutated (ATM) Kinase. PLoS ONE 2011, 6, e25454. [Google Scholar] [CrossRef]
- Le Guezennec, X.; Bulavin, D.V. WIP1 Phosphatase at the Crossroads of Cancer and Aging. Trends Biochem. Sci. 2010, 35, 109–114. [Google Scholar] [CrossRef]
- Wang, L.; Mosel, A.J.; Oakley, G.G.; Peng, A. Deficient DNA Damage Signaling Leads to Chemoresistance to Cisplatin in Oral Cancer. Mol. Cancer Ther. 2012, 11, 2401–2409. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Xia, F.; Hermance, N.; Mabb, A.; Simonson, S.; Morrissey, S.; Gandhi, P.; Munson, M.; Miyamoto, S.; Kelliher, M.A. A Cytosolic ATM/NEMO/RIP1 Complex Recruits TAK1 to Mediate the NF-kappaB and p38 Mitogen-Activated Protein Kinase (MAPK)/MAPK-Activated Protein 2 Responses to DNA Damage. Mol. Cell. Biol. 2011, 31, 2774–2786. [Google Scholar] [CrossRef] [Green Version]
- Yin, H.; Glass, J. The Phenotypic Radiation Resistance of CD44 þ/CD24(-or Low) Breast Cancer Cells Is Mediated Through the Enhanced Activation of ATM Signaling. PLoS ONE 2011, 6, e24080. [Google Scholar] [CrossRef]
- Pazolli, E.; Alspach, E.; Milczarek, A.; Prior, J.; Piwnica-Worms, D.; Stewart, S.A. Chromatin Remodeling Underlies the Senescence-Associated Secretory Phenotype of Tumor Stromal Fibroblasts That Supports Cancer Progression. Cancer Res. 2012, 72, 2251–2261. [Google Scholar] [CrossRef] [Green Version]
- Stucci, L.S.; Tucci, M.; Passarelli, A.; Silvestris, F. Avβ3 Integrin: Pathogenetic Role in Osteotropic Tumors. Crit. Rev. Oncol. Hematol. 2015, 96, 183–193. [Google Scholar] [CrossRef]
- Tucci, M.; Passarelli, A.; Mannavola, F.; Felici, C.; Stucci, L.S.; Cives, M.; Silvestris, F. Immune System Evasion as Hallmark of Melanoma Progression: The Role of Dendritic Cells. Front. Oncol. 2019, 9, 1148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keimling, M.; Volcic, M.; Csernok, A.; Wieland, B.; Dörk, T.; Wiesmüller, L. Functional Characterization Connects Individual Patient Mutations in Ataxia telangiectasia Mutated (ATM) with Dysfunction of Specific DNA Double-Strand Break-Repair Signaling Pathways. FASEB J. 2011, 25, 3849–3860. [Google Scholar] [CrossRef] [PubMed]
- Hollestelle, A.; Wasielewski, M.; Martens, J.W.; Schutte, M. Discovering Moderate-Risk Breast Cancer Susceptibility Genes. Curr. Opin. Genet. Dev. 2010, 20, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Greenman, C.; Stephens, P.; Smith, R.; Dalgliesh, G.L.; Hunter, C.; Bignell, G.; Davies, H.; Teague, J.; Butler, A.; Stevens, C.; et al. Patterns of Somatic Mutation in Human Cancer Genomes. Nature 2007, 446, 153–158. [Google Scholar] [CrossRef] [Green Version]
- Roberts, N.J.; Jiao, Y.; Yu, J.; Kopelovich, L.; Petersen, G.M.; Bondy, M.L.; Gallinger, S.; Schwartz, A.G.; Syngal, S.; Cote, M.L.; et al. ATM Mutations in Patients with Hereditary Pancreatic Cancer. Cancer Discov. 2011, 2, 41–46. [Google Scholar] [CrossRef] [Green Version]
- Ding, L.; Getz, G.; Wheeler, D.A.; Mardis, E.R.; McLellan, M.D.; Cibulskis, K.; Sougnez, C.; Greulich, H.; Muzny, D.M.; Morgan, M.B.; et al. Somatic Mutations Affect Key Pathways in Lung Adenocarcinoma. Nature 2008, 455, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Sriramulu, S.; Ramachandran, M.; Subramanian, S.; Kannan, R.; Gopinath, M.; Sollano, J.; Bissi, L.; Banerjee, S.; Marotta, F.; Pathak, S. A Review on Role of ATM Gene in Hereditary Transfer of Colorectal Cancer. Acta Biomed. 2018, 89, 463–469. [Google Scholar]
- Squatrito, M.; Brennan, C.W.; Helmy, K.; Huse, J.T.; Petrini, J.H.; Holland, E.C. Loss of ATM/Chk2/p53 Pathway Components Accelerates Tumor Development and Contributes to Radiation Resistance in Gliomas. Cancer Cell 2010, 18, 619–629. [Google Scholar] [CrossRef] [Green Version]
- Swift, M.; Reitnauer, P.J.; Morrell, D.; Chase, C.L. Breast and Other Cancers in Families with Ataxia-telangiectasia. N. Engl. J. Med. 1987, 316, 1289–1294. [Google Scholar] [CrossRef]
- Chen, J.; Birkholtz, G.G.; Lindblom, P.; Rubio, C.; Lindblom, A. The Role of Ataxia-telangiectasia Heterozygotes in Familial Breast Cancer. Cancer Res. 1998, 58, 1376–1379. [Google Scholar]
- Milne, R.L. Variants in the ATM Gene and Breast Cancer Susceptibility. Genome Med. 2009, 1, 12. [Google Scholar] [CrossRef] [Green Version]
- Athma, P.; Rappaport, R.; Swift, M. Molecular Genotyping Shows That Ataxia-telangiectasia Heterozygotes Are Predisposed to Breast Cancer. Cancer Genet. Cytogenet. 1996, 92, 130–134. [Google Scholar] [CrossRef]
- Inskip, H.M.; Kinlen, L.J.; Taylor, A.M.R.; Woods, C.G.; Arlett, C.F. Risk of Breast Cancer and Other Cancers in Heterozygotes for Ataxia-Telangiectasia. Br. J. Cancer 1999, 79, 1304–1307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Easton, D.F.; Pharoah, P.D.; Antoniou, A.C.; Tischkowitz, M.; Tavtigian, S.V.; Nathanson, K.L.; Devilee, P.; Meindl, A.; Couch, F.J.; Southey, M.; et al. Gene-Panel Sequencing and the Prediction of Breast-Cancer Risk. N. Engl. J. Med. 2015, 372, 2243–2257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Os, N.J.; Roeleveld, N.; Weemaes, C.M.; Jongmans, M.C.; Janssens, G.O.; Taylor, A.M.; Hoogerbrugge, N.; Willemsen, M.A. Health Risks for Ataxia-telangiectasia Mutated Heterozygotes: A Systematic Review, Meta-Analysis and Evidence-Based Guideline. Clin. Genet. 2016, 90, 105–117. [Google Scholar] [CrossRef]
- Southey, M.C.; Goldgar, D.E.; Winqvist, R.; Pylkäs, K.; Couch, F.; Tischkowitz, M.; Foulkes, W.D.; Dennis, J.; Michailidou, K.; van Rensburg, E.J.; et al. PALB2, CHEK2 and ATM Rare Variants and Cancer Risk: Data from COGS. J. Med. Genet. 2016, 53, 800–811. [Google Scholar] [CrossRef] [Green Version]
- Marabelli, M.; Cheng, S.C.; Parmigiani, G. Penetrance of ATM Gene Mutations in Breast Cancer: A Meta-Analysis of Different Measures of Risk. Genet. Epidemiol. 2016, 40, 425–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitui, M.; Nahas, S.; Du, L.; Yang, Z.; Lai, C.; Nakamura, K.; Arroyo, S.; Scott, S.; Purayidom, A.; Concannon, P.; et al. Functional and Computational Assessment of Missense Variants in the Ataxia-telangiectasia Mutated (ATM) Gene: Mutations with Increased Cancer Risk. Hum. Mutat. 2009, 30, 12–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, L.-B.; Pan, X.-M.; Sun, H.; Wang, X.; Rao, L.; Li, L.-J.; Liang, W.-B.; Lv, M.-L.; Yang, W.-Z.; Zhang, L. The Association between ATM D1853N Polymorphism and Breast Cancer Susceptibility: A Meta-Analysis. J. Exp. Clin. Cancer Res. 2010, 29, 117. [Google Scholar] [CrossRef] [Green Version]
- Breast Cancer Association Consortium; Dorling, L.; Carvalho, S.; Allen, J.; González-Neira, A.; Luccarini, C.; Wahlström, C.; Pooley, K.A.; Parsons, M.T.; Fortuno, C.; et al. Easton DF. Breast Cancer Risk Genes-Association Analysis in More than 113,000 Women. N. Engl. J. Med. 2021, 384, 428–439. [Google Scholar]
- Meng, Z.H.; Ben, Y.; Li, Z.; Chew, K.; Ljung, B.-M.; Lagios, M.D.; Dairkee, S.H. Aberrations of Breast Cancer Susceptibility Genes Occur Early in Sporadic Breast Tumors and in Acquisition of Breast Epithelial Immortalization. Genes Chromosomes Cancer 2004, 41, 214–222. [Google Scholar] [CrossRef]
- Vo, Q.N.; Kim, W.J.; Cvitanovic, L.; Boudreau, D.A.; Ginzinger, D.G.; Brown, K.D. The ATM Gene Is a Target for Epigenetic Silencing in Locally Advanced Breast Cancer. Oncogene 2004, 23, 9432–9437. [Google Scholar] [CrossRef] [Green Version]
- Ng, W.L.; Yan, D.; Zhang, X.; Mo, Y.-Y.; Wang, Y. Over-Expression of miR-100 Is Responsible for the Low-Expression of ATM in the Human Glioma Cell Line: M059J. DNA Repair 2010, 9, 1170–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mannavola, F.; D’Oronzo, S.; Cives, M.; Stucci, L.S.; Ranieri, G.; Silvestris, F.; Tucci, M. Extracellular Vesicles and Epigenetic Modifications Are Hallmarks of Melanoma Progression. Int. J. Mol. Sci. 2019, 21, 52. [Google Scholar] [CrossRef] [Green Version]
- Pellerino, A.; Bruno, F.; Internò, V.; Rudà, R.; Soffietti, R. Current Clinical Management of Elderly Patients with Glioma. Expert Rev. Anticancer Ther. 2020, 20, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas, N. Comprehensive Molecular Portraits of Human Breast Tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef] [Green Version]
- Ye, C.; Cai, Q.; Dai, Q.; Shu, X.-O.; Shin, A.; Gao, Y.-T.; Zheng, W. Expression Patterns of the ATM Gene in Mammary Tissues and Their Associations with Breast Cancer Survival. Cancer 2007, 109, 1729–1735. [Google Scholar] [CrossRef] [PubMed]
- Tommiska, J.; Bartkova, J.; Heinonen, M.; Hautala, L.; Kilpivaara, O.; Eerola, H.; Aittomäki, K.; Hofstetter, B.; Lukas, J.; von Smitten, K.; et al. The DNA Damage Signalling Kinase ATM Is Aberrantly Reduced or Lost in BRCA1/BRCA2-Deficient and ER/PR/ERBB2-Triple-Negative Breast Cancer. Oncogene 2008, 27, 2501–2506. [Google Scholar] [CrossRef] [Green Version]
- Ding, S.L.; Sheu, L.F.; Yu, J.C.; Yang, T.L.; Chen, B.F.; Leu, F.J.; Shen, C.Y. Abnormality of the DNA Double-Strand-Break Checkpoint/Repair Genes, ATM, BRCA1 and TP53, in Breast Cancer Is Related to Tumour Grade. Br. J. Cancer 2004, 90, 1995–2001. [Google Scholar] [CrossRef] [Green Version]
- Jiang, H.; Reinhardt, H.C.; Bartkova, J.; Tommiska, J.; Blomqvist, C.; Nevanlinna, H.; Bartek, J.; Yaffe, M.B.; Hemann, M.T. The Combined Status of ATM and p53 Link Tumor Development with Therapeutic Response. Genes Dev. 2009, 23, 1895–1909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knappskog, S.; Chrisanthar, R.; Løkkevik, E.; Anker, G.; Østenstad, B.; Lundgren, S.; Risberg, T.; Mjaaland, I.; Leirvaag, B.; Miletic, H.; et al. Low Expression Levels of ATM May Substitute for CHEK2/TP53 Mutations Predicting Resistance Towards Anthracycline and Mitomycin Chemotherapy in Breast Cancer. Breast Cancer Res. 2012, 14, R47. [Google Scholar] [CrossRef] [Green Version]
- Konde, A.S.; Ivan, K.; Klavanian, J.; Rangarajan, T.; Jaiyesimi, I.A.; Zakalik, D. Heterozygous Germline ATM Mutations in Breast Cancer: A Single Academic Center Experience. J. Clin. Oncol. 2020, 38, 1537. [Google Scholar] [CrossRef]
- Stagni, V.; Manni, I.; Oropallo, V.; Mottolese, M.; Di Benedetto, A.; Piaggio, G.; Falcioni, R.; Giaccari, D.; Di Carlo, S.; Sperati, F.; et al. ATM Kinase Sustains HER2 Tumorigenicity in Breast Cancer. Nat. Commun. 2015, 6, 6886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, M.; Kipps, T.; Kurzrock, R. ATM Mutations in Cancer: Therapeutic Implications. Mol. Cancer Ther. 2016, 15, 1781–1791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, H.H.; Sun, X.; Nahas, S.A.; Teraoka, S.; Lai, C.-H.; Concannon, P.; Gatti, R.A. Improved Diagnostic Testing for Ataxia–telangiectasia by Immunoblotting of Nuclear Lysates for ATM Protein Expression. Mol. Genet. Metab. 2003, 80, 437–443. [Google Scholar] [CrossRef]
- Kabacik, S.; Ortega-Molina, A.; Efeyan, A.; Finnon, P.; Bouffler, S.; Serrano, M.; Badie, C. A Minimally Invasive Assay for Individual Assessment of the ATM/CHEK2/p53 Pathway Activity. Cell Cycle 2011, 10, 1152–1161. [Google Scholar] [CrossRef] [Green Version]
- Rigakos, G.; Razis, E. BRCAness: Finding the Achilles Heel in Ovarian Cancer. Oncologist 2012, 17, 956–962. [Google Scholar] [CrossRef] [Green Version]
- Pennington, K.P.; Walsh, T.; Harrell, M.I.; Lee, M.K.; Pennil, C.C.; Rendi, M.H.; Thorton, A.; Norquist, B.; Casadei, S.; Nord, A.S.; et al. Germline and Somatic Mutations in Homologous Recombination Genes Predict Platinum Response and Survival in Ovarian, Fallopian Tube, and Peritoneal Carcinomas. Clin. Cancer Res. 2014, 20, 764–775. [Google Scholar] [CrossRef] [Green Version]
- Williamson, C.T.; Kubota, E.; Hamill, J.D.; Klimowicz, A.; Ye, R.; Muzik, H.; Dean, M.; Tu, L.; Gilley, D.; Magliocco, A.M.; et al. Enhanced Cytotoxicity of PARP Inhibition in Mantle Cell Lymphoma Harbouring Mutations in Both ATM and p53. EMBO Mol. Med. 2012, 4, 515–527. [Google Scholar] [CrossRef]
- Kubota, E.; Williamson, C.T.; Ye, R.; Elegbede, A.; Peterson, L.; Lees-Miller, S.P.; Bebb, D.G. Low ATM Protein Expression and Depletion of p53 Correlates with Olaparib Sensitivity in Gastric Cancer Cell Lines. Cell Cycle 2014, 13, 2129–2137. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Zou, W.; Zhang, J.; Zhang, Y.; Xu, Q.; Li, S.; Chen, C. Mechanisms of CDK4/6 Inhibitor Resistance in Luminal Breast Cancer. Front. Pharmacol. 2020, 11, 580251. [Google Scholar] [CrossRef]
- Anurag, M.; Punturi, N.; Hoog, J.; Bainbridge, M.N.; Ellis, M.J.; Haricharan, S. Comprehensive Profiling of DNA Repair Defects in Breast Cancer Identifies a Novel Class of Endocrine Therapy Resistance Drivers. Clin. Cancer Res. 2018, 24, 4887–4899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haricharan, S.; Punturi, N.; Singh, P.; Holloway, K.R.; Anurag, M.; Schmelz, J.; Schmidt, C.; Lei, J.T.; Suman, V.; Hunt, K.; et al. Loss of MutL Disrupts CHK2-Dependent Cell-Cycle Control Through CDK4/6 to Promote Intrinsic Endocrine Therapy Resistance in Primary Breast Cancer. Cancer Discov. 2017, 7, 1168–1183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef] [Green Version]
- Jones, C.D.; Blades, K.; Foote, K.M.; Guichard, S.M.; Jewsbury, P.J.; McGuire, T. Discovery of AZD6738, a Potent and Selective Inhibitor with the Potential to Test the Clinical Efficacy of ATR Kinase Inhibition in Cancer Patients. Cancer Res. 2013, 73, 2348. [Google Scholar]
- Vendetti, F.P.; Lau, A.; Schamus, S.; Conrads, T.P.; O’Connor, M.J.; Bakkenist, C.J. The Orally Active and Bioavailable ATR Kinase Inhibitor AZD6738 Potentiates the Anti-Tumor Effects of Cisplatin to Resolve ATM-Deficient Non-Small Cell Lung Cancer In Vivo. Oncotarget 2015, 6, 44289–44305. [Google Scholar] [CrossRef] [Green Version]
- Choi, M.; Fecteau, J.-F.; Brown, J.; Lau, A.; Kipps, T.J. Induction of Proliferation Sensitizes Chronic Lymphocytic Leukemic Cells to Apoptosis Mediated by the ATR Inhibitor AZD6738. Cancer Res. 2014, 74, 5485. [Google Scholar]
- Kwok, M.; Davies, N.; Agathanggelou, A.; Smith, E.; Oldreive, C.; Petermann, E.; Stewart, G.; Brown, J.; Lau, A.; Pratt, G.; et al. ATR Inhibition Induces Synthetic Lethality and Overcomes Chemore- Sistance in TP53- or ATM-Defective Chronic Lymphocytic Leukemia Cells. Blood 2016, 127, 582–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, Y.; Palii, S.S.; Innes, C.L.; Paules, R.S. Depletion of ATR Selectively Sensitizes ATM-Deficient Human Mammary Epithelial Cells to Ionizing Radiation and DNA-Damaging Agents. Cell Cycle 2014, 13, 3541–3550. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stucci, L.S.; Internò, V.; Tucci, M.; Perrone, M.; Mannavola, F.; Palmirotta, R.; Porta, C. The ATM Gene in Breast Cancer: Its Relevance in Clinical Practice. Genes 2021, 12, 727. https://doi.org/10.3390/genes12050727
Stucci LS, Internò V, Tucci M, Perrone M, Mannavola F, Palmirotta R, Porta C. The ATM Gene in Breast Cancer: Its Relevance in Clinical Practice. Genes. 2021; 12(5):727. https://doi.org/10.3390/genes12050727
Chicago/Turabian StyleStucci, Luigia Stefania, Valeria Internò, Marco Tucci, Martina Perrone, Francesco Mannavola, Raffaele Palmirotta, and Camillo Porta. 2021. "The ATM Gene in Breast Cancer: Its Relevance in Clinical Practice" Genes 12, no. 5: 727. https://doi.org/10.3390/genes12050727