Broiler White Striping: A Review of Its Etiology, Effects on Production, and Mitigation Efforts
Abstract
:1. Introduction
2. Recent History of Chicken Production
3. Muscle Growth Physiology
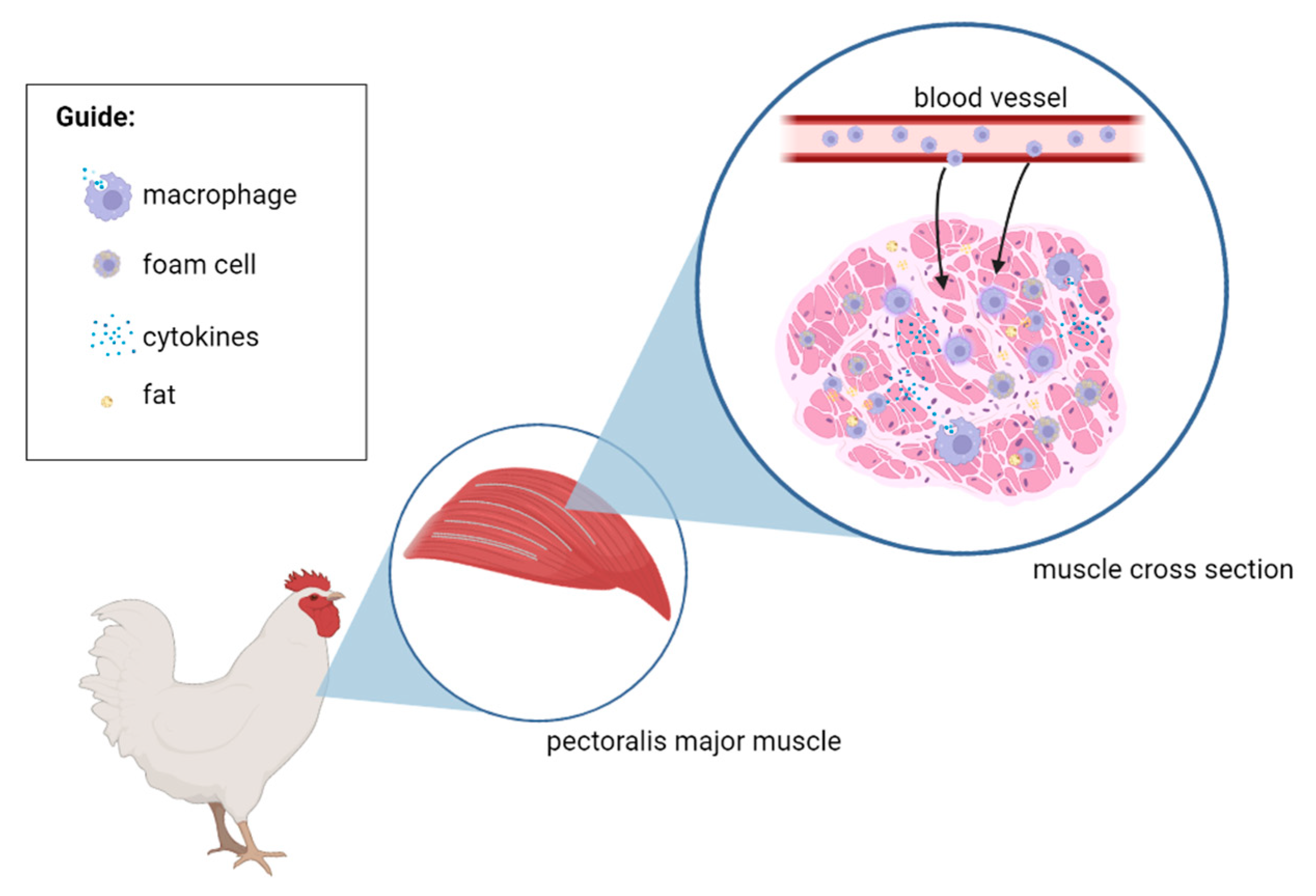
4. The Suspected Pathological Process of White Striping
5. The Causes of White Striping
6. Visualization, Detection, and Measurement of White Striping
7. The Effects of White Striping on Meat Quality and Production
8. Strategies Attempted to Reduce White Striping
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- USDA National Agricultural Statistics Service Charts and Maps: Broilers Pounds Produced United States, 1971–2021. Available online: https://www.nass.usda.gov/Charts_and_Maps/Poultry/brlprd.php (accessed on 30 March 2023).
- National Chicken Council Per Capita Consumption of Poultry and Livestock, 1965 to Forecast 2022. Available online: https://www.nationalchickencouncil.org/about-the-industry/statistics/per-capita-consumption-of-poultry-and-livestock-1965-to-estimated-2012-in-pounds/ (accessed on 30 March 2023).
- National Chicken Council, U.S. Broiler Performance. Available online: https://www.nationalchickencouncil.org/about-the-industry/statistics/u-s-broiler-performance/ (accessed on 30 March 2023).
- Kuttappan, V.A.; Owens, C.M.; Coon, C.; Hargis, B.M.; Vazquez-Anon, M. Incidence of broiler breast myopathies at 2 different ages and its impact on selected raw meat quality parameters. Poult. Sci. 2017, 96, 3005–3009. [Google Scholar] [CrossRef] [PubMed]
- Praud, C.; Jimenez, J.; Pampouille, E.; Courousse, N.; Godet, E.; Le Bihan-Duval, E.; Berri, C. Molecular Phenotyping of White Striping and Wooden Breast Myopathies in Chicken. Front. Physiol. 2020, 11, 633. [Google Scholar] [CrossRef]
- Hansen, P.L.; Mighell, R.L. Economic Choices in Broiler Production; USDA Technical Bulletin No. 1154; U.S. Government Printing Office: Washington, DC, USA, 1956. [Google Scholar]
- Smith, R.K.; Froehlich, P.; Battles, R.U.; Lingard, H.T.; Mace, A.T.; Mendum, S.W.; Porteus, H.S.; Schaben, L.J.; Shaw, E.E.; Sims, G.J.U.S. Department of Agriculture Agricultural Statistics, 1949; U.S. Government Printing Office: Washington, DC, USA, 1949. [Google Scholar]
- National Chicken Council, U.S. Chicken Industry History. Available online: https://www.nationalchickencouncil.org/about-the-industry/history/ (accessed on 30 March 2023).
- Chicken Feed. Available online: https://globalhealthnow.org/object/chicken-feed (accessed on 30 March 2023).
- Kains, M.G. Profitable Poultry Production; Orange Judd Company: New York, NY, USA, 1910. [Google Scholar]
- Babatunde, O.O.; Park, C.S.; Adeola, O. Nutritional Potentials of Atypical Feed Ingredients for Broiler Chickens and Pigs. Animals 2021, 11, 1196. [Google Scholar] [CrossRef]
- Siegel, P.B. Evolution of the Modern Broiler and Feed Efficiency. Annu. Rev. Anim. Biosci. 2014, 2, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Havenstein, G.B.; Ferket, P.R.; Qureshi, M.A. Carcass composition and yield of 1957 vs. 2001 broilers when fed representative 1957 and 2001 broiler diets. Poult. Sci. 2003, 10, 1509–1518. [Google Scholar] [CrossRef]
- Rauw, W.M.; Kanis, E.; Noordhuizen-Stassen, E.N.; Grommers, F.J. Undesirable side effects of selection for high production efficiency in farm animals: A review. Livest. Prod. Sci. 1998, 56, 15–33. [Google Scholar] [CrossRef]
- Kuttappan, V.A.; Brewer, V.B.; Apple, J.K.; Waldroup, P.W.; Owens, C.M. Influence of growth rate on the occurrence of white striping in broiler breast fillets. Poult. Sci. 2012, 91, 2677–2685. [Google Scholar] [CrossRef]
- Alnahhas, N.; Berri, C.; Chabault, M.; Chartrin, P.; Boulay, M.; Bourin, M.C.; Le Bihan-Duval, E. Genetic parameters of white striping in relation to body weight, carcass composition, and meat quality traits in two broiler lines divergently selected for the ultimate pH of the pectoralis major muscle. BMC Genet. 2016, 17, 61. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jin, P.; Duan, R.; Chen, E.H. Mechanisms of myoblast fusion during muscle development. Curr. Opin. Genet. Dev. 2015, 32, 162–170. [Google Scholar] [CrossRef]
- Pallafacchina, G.; Blaauw, B.; Schiaffino, S. Role of satellite cells in muscle growth and maintenance of muscle mass. Nutr. Metab. Cardiovas. 2013, 23, S12–S18. [Google Scholar] [CrossRef]
- Bentzinger, C.F.; Wang, Y.X.; Rudnicki, M.A. Building Muscle: Molecular Regulation of Myogenesis. CSH Perspect. Biol. 2012, 4, a008342. [Google Scholar] [CrossRef] [PubMed]
- Girardi, F.; Taleb, A.; Ebrahimi, M.; Datye, A.; Gamage, D.G.; Peccate, C.; Giordani, L.; Millay, D.P.; Gilbert, P.M.; Cadot, B.; et al. TGF beta signaling curbs cell fusion and muscle regeneration. Nat. Commun. 2021, 12, 750. [Google Scholar] [CrossRef] [PubMed]
- Hindi, S.M.; Tajrishi, M.M.; Kumar, A. Signaling Mechanisms in Mammalian Myoblast Fusion. Sci. Signal. 2013, 6, re2. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, S.F. Developmental Biology, 7th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2003. [Google Scholar]
- Alberts, B.J.A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Velleman, S.G. Muscle development in the embryo and hatchling. Poult. Sci. 2007, 86, 1050–1054. [Google Scholar] [CrossRef]
- Latroche, C.; Gitiaux, C.; Chretien, F.; Desguerre, I.; Mounier, R.; Chazaud, B. Skeletal Muscle Microvasculature: A Highly Dynamic Lifeline. Physiology 2015, 30, 417–427. [Google Scholar] [CrossRef]
- Mounier, R.; Chretien, F.; Chazaud, B. Blood Vessels and the Satellite Cell Niche. Curr. Top. Dev. Biol. 2011, 96, 121–138. [Google Scholar]
- Ehmsen, J.T.; Hoke, A. Cellular and molecular features of neurogenic skeletal muscle atrophy. Exp. Neurol. 2020, 331, 113379. [Google Scholar] [CrossRef]
- Carlson, B.M. The biology of long-term denervated skeletal muscle. Eur. J. Transl. Myol. 2014, 24, 5–11. [Google Scholar] [CrossRef]
- Vanhatalo, O.E.; Henderson, J.D.; De la Torre, U.; Garrity, C.R.; Pechanec, M.Y.; Mienaltowski, A.; King, A.J.; Mienaltowski, M.J. Research Note: Evaluation of the incidence of white striping and underlying myopathic abnormalities affected by fast weight gain in commercially fed broiler chickens. Poult. Sci. 2021, 100, 101020. [Google Scholar] [CrossRef]
- Smith, T.W.; Rasmusson, R.L.; Lobaugh, L.A.; Lieberman, M. Na+/K+ Pump Inhibition Induces Cell Shrinkage in Cultured Chick Cardiac Myocytes. Basic Res. Cardiol. 1993, 88, 411–420. [Google Scholar] [CrossRef]
- Sarhan, M.; Land, W.G.; Tonnus, W.; Hugo, C.P.; Linkermann, A. Origin and Consequences of Necroinflammation. Physiol. Rev. 2018, 98, 727–780. [Google Scholar] [CrossRef]
- Urciuolo, A.; Quarta, M.; Morbidoni, V.; Gattazzo, F.; Molon, S.; Grumati, P.; Montemurro, F.; Tedesco, F.S.; Blaauw, B.; Cossu, G.; et al. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 2013, 4, 1964. [Google Scholar] [CrossRef] [PubMed]
- Ayansola, H.; Liao, C.Y.; Dong, Y.Y.; Yu, X.X.; Zhang, B.K.; Wang, B. Prospect of early vascular tone and satellite cell modulations on white striping muscle myopathy. Poult. Sci. 2021, 100, 100945. [Google Scholar] [CrossRef] [PubMed]
- Alexakis, C.; Partridge, T.; Bou-Gharios, G. Implication of the satellite cell in dystrophic muscle fibrosis: A self-perpetuating mechanism of collagen overproduction. Am. J. Physiol. Cell Physiol. 2007, 293, C661–C669. [Google Scholar] [CrossRef]
- Wang, T.X.; Yao, W.L.; Li, J.; He, Q.Y.; Shao, Y.F.; Huang, F.R. Acetyl-CoA from inflammation-induced fatty acids oxidation promotes hepatic malate-aspartate shuttle activity and glycolysis. Am. J. Physiol. Endoc. Metab. 2018, 315, E496–E510. [Google Scholar] [CrossRef] [PubMed]
- Afshinnia, F.; Rajendiran, T.M.; Soni, T.; Byun, J.; Wernisch, S.; Sas, K.M.; Hawkins, J.; Bellovich, K.; Gipson, D.; Michailidis, G.; et al. Impaired β-Oxidation and Altered Complex Lipid Fatty Acid Partitioning with Advancing CKD. J. Am. Soc. Nephrol. 2018, 29, 295–306. [Google Scholar] [CrossRef]
- Ley, K.; Laudanna, C.; Cybulsky, M.I.; Nourshargh, S. Getting to the site of inflammation: The leukocyte adhesion cascade updated. Nat. Rev. Immunol. 2007, 7, 678–689. [Google Scholar] [CrossRef]
- Yoon, K.W. Dead cell phagocytosis and innate immune checkpoint. BMB Rep. 2017, 50, 496–503. [Google Scholar] [CrossRef]
- Guerrini, V.; Gennaro, M.L. Foam Cells: One Size Doesn’t Fit All. Trends Immunol. 2019, 40, 1163–1179. [Google Scholar] [CrossRef]
- Poznyak, A.V.; Nikiforov, N.G.; Starodubova, A.V.; Popkova, T.V.; Orekhov, A.N. Macrophages and Foam Cells: Brief Overview of Their Role, Linkage, and Targeting Potential in Atherosclerosis. Biomedicines 2021, 9, 1221. [Google Scholar] [CrossRef]
- Olkowski, A.A.; Duke, T.; Wojnarowicz, C. The aetiology of hypoxaemia in chickens selected for rapid growth. Comp. Biochem. Phys. A 2005, 141, 122–131. [Google Scholar] [CrossRef]
- Hudson, N.J.; Bottje, W.G.; Hawken, R.J.; Kong, B.; Okimoto, R.; Reverter, A. Mitochondrial metabolism: A driver of energy utilisation and product quality? Anim. Prod. Sci. 2017, 57, 2204–2215. [Google Scholar] [CrossRef]
- Nadella, V.; Mohanty, A.; Sharma, L.; Yellaboina, S.; Mollenkopf, H.J.; Mazumdar, V.B.; Palaparthi, R.; Mylavarapu, M.B.; Maurya, R.; Kurukuti, S.; et al. Inhibitors of Apoptosis Protein Antagonists (Smac Mimetic Compounds) Control Polarization of Macrophages during Microbial Challenge and Sterile Inflammatory Responses. Front. Immunol. 2018, 8, 1792. [Google Scholar] [CrossRef] [PubMed]
- Snyder, C.M.; Chandel, N.S. Mitochondrial Regulation of Cell Survival and Death During Low-Oxygen Conditions. Antioxid. Redox Signal. 2009, 11, 2673–2683. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Pachouri, U.C.; Khaidem, D.C.; Kundu, A.; Chopra, C.; Singh, P. Mitochondrial DNA Damage and Diseases. F1000Research 2015, 4, 176. [Google Scholar] [CrossRef]
- Marsch, E.; Sluimer, J.C.; Daemen, M.J.A.P. Hypoxia in atherosclerosis and inflammation. Curr. Opin. Lipidol. 2013, 24, 393–400. [Google Scholar] [CrossRef]
- Boerboom, G.; van Kempen, T.; Navarro-Villa, A.; Perez-Bonilla, A. Unraveling the cause of white striping in broilers using metabolomics. Poult. Sci. 2018, 97, 3977–3986. [Google Scholar] [CrossRef] [PubMed]
- Brookheart, R.T.; Michel, C.I.; Schaffer, J.E. As a Matter of Fat. Cell Metab. 2009, 10, 9–12. [Google Scholar] [CrossRef]
- Havenstein, G.B.; Ferket, P.R.; Qureshi, M.A. Growth, livability, and feed conversion of 1957 vs. 2001 broilers when fed representative 1957 and 2001 broiler diets. Poult. Sci. 2003, 10, 1500–1508. [Google Scholar] [CrossRef]
- Bailey, R.A.; Watson, K.A.; Bilgili, S.F.; Avendano, S. The genetic basis of pectoralis major myopathies in modern broiler chicken lines. Poult. Sci. 2015, 12, 2870–2879. [Google Scholar] [CrossRef]
- Panisson, J.C.; Bassi, L.S.; Barrilli, L.E.N.; Dias, R.C.; Majorka, A.; Krabbe, E.L.; Lopes, L.; Oliveira, S.G. Energy and nutrient intake on white striping, wooden breastand carcass composition from three genetic lineages at different ages. Animal Feed Sci. Technol. 2022, 285, 115225. [Google Scholar] [CrossRef]
- Pampouille, E.; Berri, C.; Boitard, S.; Hennequet-Antier, C.; Beauclerq, S.A.; Godet, E.; Praud, C.; Jego, Y.; Le Bihan-Duval, E. Mapping QTL for white striping in relation to breast muscle yield and and meat quality traits in broiler chickens. BMC Genom. 2018, 19, 202. [Google Scholar] [CrossRef] [PubMed]
- Lake, J.A.; Dekkers, J.C.M.; Abasht, B. Genetic basis and identification of candidate genes for wooden breast and white striping in commercial broiler chickens. Sci. Rep. 2021, 11, 6785. [Google Scholar] [CrossRef]
- USDA Directive: Ante-Mortem and Post-Mortem Poultry Inspection—Revision 1. Available online: https://www.fsis.usda.gov/policy/fsis-directives/6100.3 (accessed on 30 March 2023).
- Silva, S.R.; Pinheiro, V.M.; Guedes, C.M.; Mourao, J.L. Prediction of carcase and breast weights and yields in broiler chickens using breast volume determined in vivo by real-time ultrasonic measurement. Br. Poult. Sci. 2006, 47, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Mastelini, S.M.; Campos, G.F.C.; Barbon, A.P.A.D.; Prudencio, S.H.; Shimokomaki, M.; Soares, A.L.; Barbon, S. White striping degree assessment using computer vision system and consumer acceptance test. Asian Australas. J. Anim. Sci. 2019, 32, 1015–1026. [Google Scholar] [CrossRef]
- Traffano-Schiffo, M.V.; Castro-Giraldez, M.; Colom, R.J.; Fito, P.J. Development of a Spectrophotometric System to Detect White Striping Physiopathy in Whole Chicken Carcasses. Sensors 2017, 17, 1024. [Google Scholar] [CrossRef]
- Kuttappan, V.A.; Hargis, B.M.; Owens, C.M. White striping and woody breast myopathies in the modern poultry industry: A review. Poult. Sci. 2016, 95, 2724–2733. [Google Scholar] [CrossRef]
- Trocino, A.; Piccirillo, A.; Birolo, M.; Radaelli, G.; Bertotto, D.; Filiou, E.; Petracci, M.; Xiccato, G. Effect of genotype, gender and feed restriction on growth, meat quality and the occurrence of white striping and wooden breast in broiler chickens. Poult. Sci. 2015, 94, 2996–3004. [Google Scholar] [CrossRef]
- Russo, E.; Drigo, M.; Longoni, C.; Pezzotti, R.; Fasoli, P.; Recordati, C. Evaluation of White Striping prevalence and predisposing factors in broilers at slaughter. Poult. Sci. 2015, 94, 1843–1848. [Google Scholar] [CrossRef]
- Brouckaert, G.; Kalai, M.; Krysko, D.V.; Saelens, X.; Vercammen, D.; Ndlovu, M.; Haegeman, G.; D’Herde, K.; Vandenabeele, P. Phagocytosis of Necrotic Cells by Macrophages Is Phosphatidylserine Dependent and Does Not Induce Inflammatory Cytokine Production. Mol. Biol. Cell 2004, 15, 1089–1100. [Google Scholar] [CrossRef]
- Haschek, W.M.; Rousseaux, C.G.; Wallig, M.A.; Bolon, B.; Ochoa, R.; Mahler, B.W. Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Prisco, F.; De Biase, D.; Piegari, G.; D’Aquino, I.; Lama, A.; Comella, F.; Mercogliano, R.; Dipineto, L.; Papparella, S.; Paciello, O. Pathologic characterization of white striping myopathy in broiler chickens. Poult. Sci. 2021, 100, 101150. [Google Scholar] [CrossRef]
- Lake, J.A.; Abasht, B. Glucolipotoxicity: A Proposed Etiology for Wooden Breast and Related Myopathies in Commercial Broiler Chickens. Front Physiol. 2020, 11, 169. [Google Scholar] [CrossRef]
- Petracci, M.; Mudalal, S.; Babini, E.; Cavani, C. Effect of white striping on chemical composition and nutritional value of chicken breast meat. Ital. J. Anim. Sci. 2014, 13, 3138. [Google Scholar] [CrossRef]
- Marchesi, J.A.P.; Ibelli, A.M.G.; Peixoto, J.O.; Cantao, M.E.; Pandolfi, J.R.C.; Marciano, C.M.M.; Zanella, R.; Settles, M.L.; Coutinho, L.L.; Ledur, M.C. Whole transcriptome analysis of the pectoralis major muscle reveals molecular mechanisms involved with white striping in broiler chickens. Poult. Sci. 2019, 98, 590–601. [Google Scholar] [CrossRef]
- Mudalal, S.; Lorenzi, M.; Soglia, F.; Cavani, C.; Petracci, M. Implications of white striping and wooden breast abnormalities on quality traits of raw and marinated chicken meat. Animal 2015, 9, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Park, C.H.; Kong, C.; Kim, Y.S.; Choi, Y.M. Muscle fiber and fresh meat characteristics of white-striping chicken breasts, and its effects on palatability of sous-vide cooked meat. Poult. Sci. 2021, 100, 101177. [Google Scholar] [CrossRef] [PubMed]
- Brambila, G.S.; Bowker, B.C.; Zhuang, H. Comparison of sensory texture attributes of broiler breast fillets with different degrees of white striping. Poult. Sci. 2016, 95, 2472–2476. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Bai, L.; He, Z.; Sun, J.; Tan, X.; Zhao, D.; Feng, F.; Liu, D.; Zhao, G.; Wen, J.; et al. Intergrated metabolomics and lipidomics evaluate the alterations of flavor precursors in chicken breast muscle with white striping symptom. Front. Physiol. 2022, 13, 1079667. [Google Scholar] [CrossRef]
- Liu, T.; Xia, N.; Wang, Q.; Chen, D. Identification of the Non-Volatile Taste-Active Components in Crab Sauce. Foods 2019, 8, 324. [Google Scholar] [CrossRef]
- Abo Ghanima, M.M.; Abd El-Hack, M.E.; Abougabal, M.S.; Taha, A.E.; Tufarelli, V.; Laudadio, V.; Naiel, M.A.E. Growth, carcass traits, immunity, and oxidative status of broilers exposed to continuous or intermittent lighting programs. Anim. Biosci. 2021, 34, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.M. Slow and steady wins the race: The behaviour and welfare of commercial faster growing broiler breeds compared to a commercial slower growing breed. PLoS ONE 2020, 15, e0231006. [Google Scholar] [CrossRef]
- Bodle, B.C.; Alvarado, C.; Shirley, R.B.; Mercier, Y.; Lee, J.T. Evaluation of different dietary alterations in their ability to mitigate the incidence and severity of woody breast and white striping in commercial male broilers. Poult. Sci. 2018, 97, 3298–3310. [Google Scholar] [CrossRef] [PubMed]
- Bautista-Ortega, J.; Ruiz-Feria, C.A. L-Arginine and antioxidant vitamins E and C improve the cardiovascular performance of broiler chickens grown under chronic hypobaric hypoxia. Poult. Sci. 2010, 89, 2141–2146. [Google Scholar] [CrossRef]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Bio. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef]
- Jin, C.L.; Ye, J.L.; Yang, J.Z.; Gao, C.Q.; Yan, H.C.; Li, H.C.; Wang, X.Q. mTORC1 Mediates Lysine-Induced Satellite Cell Activation to Promote Skeletal Muscle Growth. Cells 2019, 8, 1549. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, U.; Ipek, E.; Ozdemir, O.S.; Aydin, A.K.; Raza, I.; Calik, A.; Kuter, E.; Ozsoy, S.Y.; Cengiz, O. Intermittent dilution of dietary digestible lysine lowers the incidence of white striping by suppressing the growth, lipid synthesis, and muscle damage in broiler chickens. J. Sci. Food Agr. 2023, 103, 283–297. [Google Scholar] [CrossRef]
- Meloche, K.J.; Fancher, B.I.; Emmerson, D.A.; Bilgili, S.F.; Dozier, W.A. Effects of reduced digestible lysine density on myopathies of the Pectoralis major muscles in broiler chickens at 48 and 62 days of age. Poult. Sci. 2018, 97, 3311–3324. [Google Scholar] [CrossRef]
- Sachs, N.J.; Hampton, A.R.; Foster, K.K.; Pechanec, M.Y.; Henderson, J.D.; King, A.J.; Mienaltowski, M.J. The effects of an alternative diet regimen with natural methionine ingredients on white striping breast myopathy in broiler chickens. Poult. Sci. 2019, 98, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Kuttappan, V.A.; Goodgame, S.D.; Bradley, C.D.; Mauromoustakos, A.; Hargis, B.M.; Waldroup, P.W.; Owens, C.M. Effect of different levels of dietary vitamin E (DL-alpha-tocopherol acetate) on the occurrence of various degrees of white striping on broiler breast fillets. Poult. Sci. 2012, 91, 3230–3235. [Google Scholar] [CrossRef]
- Meloche, K.J.; Fancher, B.I.; Emmerson, D.A.; Bilgili, S.F.; Dozier, W.A. Effects of quantitative nutrient allocation on myopathies of the Pectoralis major muscles in broiler chickens at 32, 43, and 50 days of age. Poult. Sci. 2018, 97, 1786–1793. [Google Scholar] [CrossRef]

| Effect of White Striping on Meat Quality and Production | Study |
|---|---|
| Decreased production | Marchesi et al., 2019 [66] |
| Increased fat content | Petracci et al., 2014 [65] |
| Decreased palatability | Kuttappan et al., 2012 [15] |
| Decreased protein content | Petracci et al., 2014 [65] |
| Increased calorie | Kuttappan et al., 2012 [15] |
| Increased pH level | Mudalal et al., 2015 [69] |
| Decreased drip loss | Mudalal et al., 2015 [69] |
| Indicator of hypoxia | Boerboom et al., 2018 [47] |
| Indicator of decreased β oxidation activity | Boerboom et al., 2018 [47] |
| Increased collagen content | Petracci et al., 2014 [65] |
| Decreased juiciness | Lee et al., 2021 [68] |
| Increased chewiness | Brambila et al., 2016 [69] |
| Strategy | Mechanism | Outcome | Study |
|---|---|---|---|
| Increase digestible arginine | Arginine aids vasodilation; improved muscle oxygenation | Effects not significant | Bodle et al., 2018 [74] |
| Vitamin C supplementation | Free radicals scavenging; cytoprotection | Effects not significant | |
| Increase vitamin premix supplementation | Antioxidants; cytoprotection | Effects not significant | |
| Reducing dietary amino acid density in the grower phase | Muscle growth decrease; improved hemodynamics/circulation | Effects not significant | |
| Decrease lysine supplementation | Lessens proteins synthesis; reduces growth, vasoconstriction, and hypoxia | Lower white striping incidence and less tissue damage | Ahsan et al., 2022 [78] Meloche et al., 2018 [79] |
| Alternative methionine source | Legume antioxidant properties | Lower white striping incidence | Sachs et al., 2019 [80] |
| Vitamin E supplementation | Antioxidants; cytoprotection | WS incidence decreased | Kuttapan et al., 2012 [81] |
| Decrease feed intake | Less energy for muscle growth | WS incidence decreased | Meloche et al., 2018 [79] |
| Feed restriction | Less energy for muscle growth | WS incidence increased | Trocino et al., 2015 [59] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Mienaltowski, M.J. Broiler White Striping: A Review of Its Etiology, Effects on Production, and Mitigation Efforts. Poultry 2023, 2, 292-304. https://doi.org/10.3390/poultry2020022
Lee J, Mienaltowski MJ. Broiler White Striping: A Review of Its Etiology, Effects on Production, and Mitigation Efforts. Poultry. 2023; 2(2):292-304. https://doi.org/10.3390/poultry2020022
Chicago/Turabian StyleLee, Jessie, and Michael J. Mienaltowski. 2023. "Broiler White Striping: A Review of Its Etiology, Effects on Production, and Mitigation Efforts" Poultry 2, no. 2: 292-304. https://doi.org/10.3390/poultry2020022





