Abstract
Aims: To date, precision medicine has played a pivotal role in the clinical administration of solid-tumor patients. In this scenario, a rapidly increasing number of predictive biomarkers have been approved in diagnostic practice or are currently being investigated in clinical trials. A pitfall in molecular testing is the diagnostic routine sample available to analyze predictive biomarkers; a scant tissue sample often represents the only diagnostical source of nucleic acids with which to conduct molecular analysis. At the sight of these critical issues, next-generation sequencing (NGS) platforms emerged as referral testing strategies for the molecular analysis of predictive biomarkers in routine practice, but the need for highly skilled personnel and extensive working time drastically impacts the widespread diffusion of this technology in diagnostic settings. Here, we technically validate a fully integrated NGS platform on diagnostic routine tissue samples previously tested with an NGS-based diagnostic workflow by a referral institution. Methods: A retrospective series of n = 64 samples (n = 32 DNA, n = 32 RNA samples), previously tested using a customized NGS assay (SiRe™ and SiRe fusion), was retrieved from the internal archive of the University of Naples Federico II. Each sample was tested by adopting an Oncomine Precision Assay (OPA), which is able to detect 2769 molecular actionable alterations [hotspot mutations, copy number variations (CNV) and gene fusions] on fully integrated NGS platforms (Genexus, Thermo Fisher Scientific (Waltham, MA, USA). The concordance rate between these technical approaches was determined. Results: The Genexus system successfully carried out molecular analysis in all instances. A concordance rate of 96.9% (31 out of 32) was observed between the OPA and SiRe™ panels both for DNA- and RNA-based analysis. A negative predictive value of 100% and a positive predictive value of 96.9% (62 out of 64) were assessed. Conclusions: A fully automatized Genexus system combined with OPA (Thermo Fisher Scientific) may be considered a technically valuable, time-saving sequencing platform to test predictive biomarkers in diagnostic routine practice.
1. Introduction
In recent decades, personalized medicine has laid the basis for a novel therapeutical option for solid-tumor patients [1,2]. Currently, target therapy is routinely available for the clinical administration of several solid-tumor patients, including metastatic colorectal cancer (mCRC), melanoma (MM), non-small cell lung cancer (NSCLC), gastrointestinal stromal tumor (GIST), and breast cancer (BC) patients [3,4,5,6,7,8,9]. In particular, an increasing number of predictive biomarkers are being approved in clinical practice to provide lung cancer patients diagnosed with the NSCLC type with the best therapeutical option [8,9]. In this evolving scenario, the minimal request in terms of predictive biomarkers to clinically administrate solid-tumor patients has been regulated by international societies [10,11,12,13,14]. The most common diagnostic sample available to approach diagnosis and molecular tests in the advanced tumor stage consists of a “scant sample” with a low abundance of neoplastic cells to successfully carry out mandatory gene testing [15,16,17]. In this scenario, cytological specimens and small biopsies represent the most common biological source to accurately perform molecular analysis. In addition, cell block (CB), a hybrid preparation where the aspirated material is processed following standardized formalin fixation and paraffin embedding (FFPE), represents an alternative source of neoplastic cells affected by the lowest quality and quantity of nucleic acids adopted in molecular tests [18,19]. Despite tissue specimens being considered the “gold standard” for molecular testing, a non-negligible percentage of patients do not have access to molecular tests due to insufficient diagnostic material [16,17]. In this scenario, liquid biopsy becomes an integrating biological source for successfully performing molecular analysis when tissue is not available. Moreover, circulating tumor DNA (ctDNA) isolated from peripheral blood is a reliable source for detecting target molecular alterations [20,21]. At the sight of these aspects, single plex technology results are inadequate to successfully analyze the minimum gene panel established for each solid tumor. In this heterogeneous landscape of biological sources, next-generation sequencing (NGS) platforms play a crucial role in the molecular analysis of predictive biomarkers [22,23,24]. This technology allows us to simultaneously analyze very low-frequency clinically relevant biomarkers using very low amounts of nucleic acids in a single run [22,23]. Remarkably, NGS systems are scalable, decreasing reaction costs in accordance with the number of samples processed in each run [24]. On the other hand, an adequate number of samples may be collected in more than 30 days for a non-negligible number of small–medium institutions involved in molecular tests, thereby saving on technical costs. This aspect drastically impacts turnaround time (TAT), resulting in a delay in the clinical administration of tumor patients [24,25]. In this scenario, the Ion Torrent™ Genexus™ Integrated Sequencer (Genexus; Thermo Fisher Scientific, Waltham, MA, USA) was designed to automatically carry out the entire NGS workflow (from tissue and liquid biopsy-derived nucleic acids extraction to data analysis) without other manual operations [26,27,28]. This technology allows us to successfully carry out the molecular analysis of a small batch of diagnostic specimens [1,2,3,4,5,6,7,8] without impacting the turnaround time (TAT) of the diagnostic workflow. We aimed to evaluate the concordance rate between the Genexus system and Ion Torrent S5™ Plus (Thermo Fisher Scientific, Waltham, MA, USA) on a retrospective series of extracted genomic DNA (gDNA) from solid-tumor patients previously tested in our diagnostic routine.
2. Study Design
A retrospective series of n = 64 previously extracted DNA and RNA specimens from solid-tumor patients (n = 16 CRC, n = 13 NSCLC, n = 2 BC and n = 1 MM and n = 32 NSCLC cases for DNA- and RNA-related molecular analysis, respectively) was retrieved from the internal archive of the predictive molecular pathology laboratory of the University of Naples Federico II. Clinical pathological data are listed in Table 1 and Table 2.

Table 1.
Clinical characteristics of archival cases and corresponding requests on DNA-based molecular alterations.

Table 2.
Clinical characteristics of archival cases and corresponding requests on RNA-based molecular alterations.
Each sample was previously tested by adopting a customized NGS assay (SiRe™ and SiRe fusion) that covers n = 568 clinically relevant alterations in BRAF, EGFR, KRAS, NRAS, PIK3CA, c-KIT, PDGFRA and ALK, ROS1, RET, and NTRK gene fusions, as well as MET exon 14 skipping alterations, which is routinely employed in the molecular testing of solid-tumor patients [29]. The Oncomine Precision Assay (OPA), able to detect 2769 molecular actionable alterations [hotspot mutations, copy number variations (CNV) and gene fusions], was combined with the Genexus (Thermo Fisher Scientific) platform to assess the molecular profile of selected samples [26,27]. The concordance rate of the OPA in the Genexus system with SiRe™ on the S5 Plus platform was investigated. All information regarding human material were managed using anonymous numerical codes, and all samples were handled in compliance with the Helsinki Declaration (http://www.wma.net/en/30publications/10policies/b3/, accessed on 1 September 2023).
3. Material and Methods
3.1. Routine Sample Processing Strategy
Nucleic acids were previously purified from n = 4 representative slides of neoplastic area (>10%). Specifically, a QIAamp DNA Mini Kit (Qiagen, Crawley, West Sussex, UK) was utilised following manufacturer instructions. DNA quantification was successfully carried out in all cases, adopting a Qubit fluorimeter (Thermo Fisher) or a TapeStation 4200 microfluidic platform (Agilent Technologies, Santa Clara, CA, USA) following manufacturer instructions. In the instance of an inadequate amount of nucleic acids, we maximized for volume input. Conversely, RNA volume was maximized for cDNA synthesis. Selected samples were routinely analyzed with SiRe™ and SiRe fusion panels using the Ion S5™ Plus software (Thermo Fisher Scientific) to assess mutational status in clinically relevant biomarkers for NSCLC patients [29,30]. Briefly, 15 μL of extracted DNA/cDNA was dispensed into the Ion Kit-Chef system (Thermo Fisher Scientific) for library preparation. A total of n = 8 samples was simultaneously processed following previously validated thermal conditions. After pooling, a templating procedure was carried out for n = 16 libraries by using the Ion 510™, Ion 520™ and Ion 530™ Kit-Chef (Thermo Fisher Scientific) according to manufacturer instructions on a 520 chip (Thermo Fisher Scientific). Data were inspected by adopting designed bed files on proprietary Torrent Suite software [v.5.0.2]. In detail, variant inspection was performed with a variant caller plug-in (v.5.0.2.1), which is able to filter variants with ≥5× allele coverage and a quality score ≥20, within an amplicon that covered at least 500× alleles.
3.2. Genexus Analysis
A series of n = 64 extracted gDNA and gRNA samples from solid-tumor patients was retrospectively tested in the Genexus (Thermo Fisher Scientific) system. The platform enables entire NGS workflows (from library preparation to data interpretation) within 24 h. The OPA assay includes the most clinically relevant actionable genes (EGFR, BRAF, KRAS, ALK, ROS1, NTRK, and RET) for NSCLC patients [27,28]. Briefly, samples were created on a dedicated server and assigned to a new run. The Genexus platform was loaded with OPA primers, strip solutions, strip reagents, and supplies according to manufacturer instructions. A total of 10 ng was required by the OPA assay on the Genexus platform. Accordingly, each sample was diluted and immediately dispensed on a 96-well plate, following manufacturer instructions. Finally, nucleic acids were sequenced on a GX5TM chip that allows for the simultaneous processing of n = 8 samples in a single line with an OPA assay. Data analysis was performed using proprietary Genexus software (1.0). Particularly, detected alterations were annotated by adopting Oncomine Knowledgebase Reporter Software (Oncomine Reporter 5.0). In addition, BAM files were also visually inspected with the Golden Helix Genome Browser v.2.0.7 (Bozeman, MT, USA) in hotspot regions in EGFR, KRAS, and BRAF lung cancer-addicted molecular alterations.
4. Results
4.1. Hotspot Mutations
Overall, the Genexus system successfully carried out molecular analysis in all DNA series. In detail, a median number of total reads, mapped reads, mean read length, percent reads on target, mean depth, uniformity of amplicon coverage of 1,134,878.2 (ranging from 424,900.0 to 1,791,041.0), 1,074,345.7 (ranging from 365,139.0 to 1,756,414.0), 90.9 bp (ranging from 71 to 103 bp), 88.3% (ranging from 77.7 to 93.7%), 3602.9 (ranging from 994.00 to 6097.0) and 98.2% (ranging from 96.7 to 99.4%) were detected, respectively (Table 3).

Table 3.
Technical parameters from DNA-based analysis by using S5 Plus (Ion Reporter 5.2.0.1) and Genexus systems.
Remarkably, n = 29 out of 32 (90.6%) patients [n = 16 CRC, n = 10 NSCLC, n = 2 BC and n = 1 MM] showed molecular alterations covered by OPA reference genes. Of note, 24 out of 29 (82.7%) cases highlighted clinically relevant molecular alterations referenced by the SiRe™ panel. In particular, n = 3 out 29 EGFR mutations [n = 1 exon 19 c.2300_2308dup p.A767_V769dup; n = 1 exon 21 c.2573T>G p.L858R and a concomitant EGFR exon 20 c.2369C>T p.T790M+ exon 21 c.2573T>G p.L858R]; n = 13 out of 29 KRAS molecular alterations [n = 3 exon 2 c.35G>A p.G12D; n = 2 exon 2 c.34G>T p.G12C; n = 2 exon 2 c.35G>A p.G12V; n = 1 exon 2 c.38G>A p.G13D; n = 1 exon 3 c.182A>T p.Q61L]; n = 1 exon 3 c.181C>A p.Q61K; n = 1 exon 4 c.436G>A p.A146T and n = 2 concomitant KRAS exon 2 c.35G>A p.G12D+ c.38G>A p.G13D; KRAS exon 2 c.38G>A p.G13D+ c.38_39delinsAA p.G13E]; n = 3 out of 29 BRAF mutations [n = 2 exon 15 c.1799T>A p.V600E and n = 1 exon 15 c.1801A>G p.K601E]; n = 4 out of 29 PIK3CA hotspot mutations [n = 2 exon 9 c.1633G>A p.E545K and n = 2 exon 20 c.3140A>G p.H1047R]; n = 3 out 29 NRAS mutations [n = 2 exon 3 c.181C>A p.Q61K and n = 1 exon 3 c.182A>G p.Q61R]; and n = 1 out of 29 c-KIT molecular alterations [exon 11 c.1727T>C p.L576P] were detected (Table 4).

Table 4.
Comparison of DNA-related molecular alterations between S5 Plus and Genexus platforms.
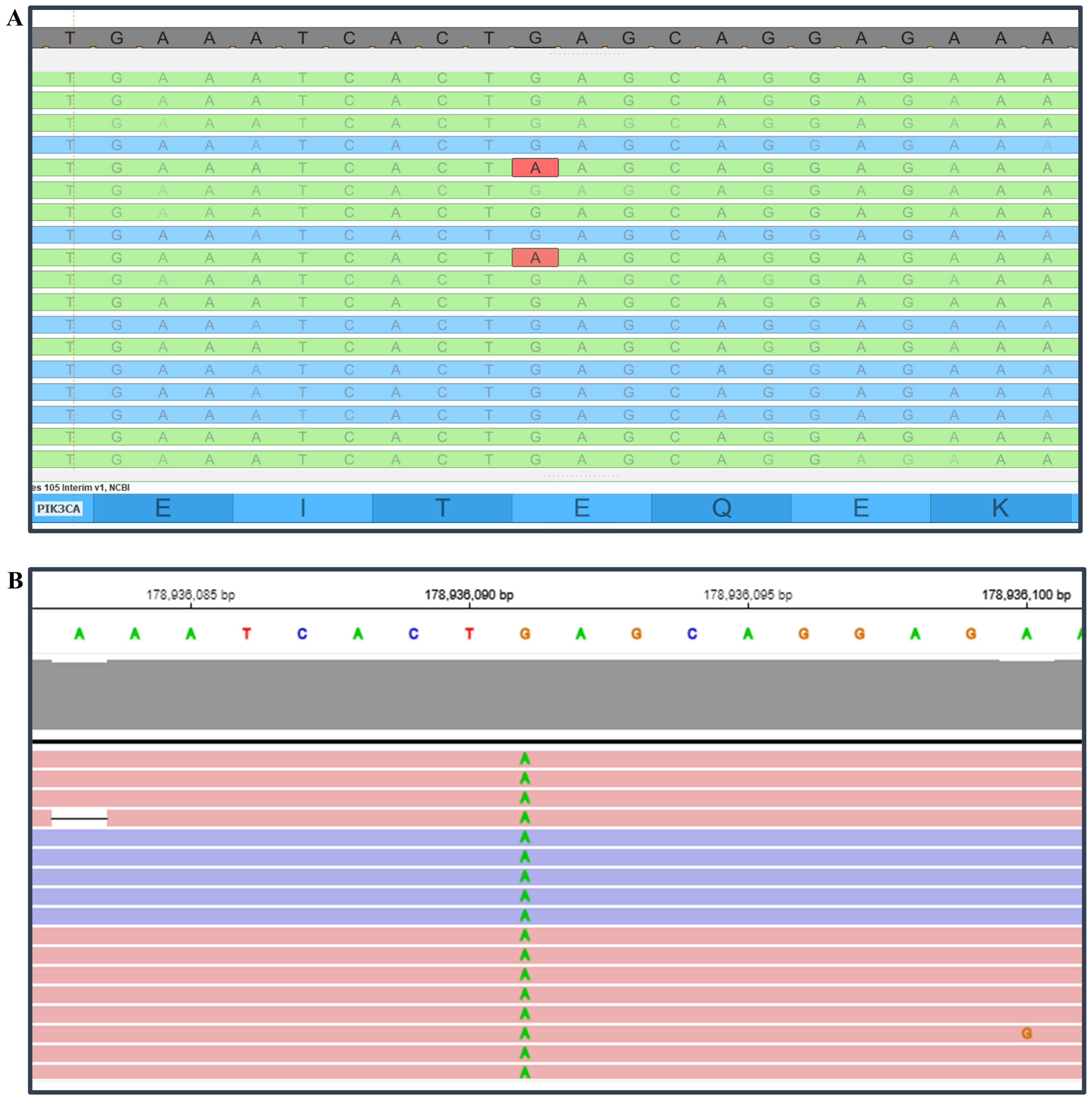
The molecular profile detected by OPA on the Genexus platform matched with the Sire panel on the S5 Plus system in 31 out of 32 patients (96.9%). Remarkably, positive results previously identified adopting the SiRe panel were confirmed in 23 out of 24 (95.8%) patients. Particularly, ID#19 showed an exon 9 PIK3CA p.E545K hotspot mutation not observed by using the S5 system with a standardized clinical cut-off (MAF = ≥5.0%) (Figure 1).
Figure 1.
PIK3CA p.E545K hotspot mutations manually inspected with Golden Helix Genome Browser v.2.0.7 (Bozeman, MT, USA) (A) and automatically annotated on proprietary Genexus software (B).
No significant variations in accordance with histological groups, mutation type and mutant allele fraction levels between Genexus and the previously tested samples on the S5 platform were identified. In addition, the OPA assay also identified n = 16 out of 32 (50.0%) DNA-based molecular alterations in other genes not covered by the SiRe panel. Moreover, 12 out of 16, 1 out of 16, and 1 out of 16 highlighted TP53, CTNNB1 and MTOR hotspot molecular alterations, respectively. Moreover, concomitant TP53 (exon 7 p.G279E plus exon 5 p.V197M) and TP53 (exon 4 p.R175H) in association with CTNNB1 (exon 3 p.S45F) hotspot mutations were identified in ID#2 and ID#16 cases (Table 5).

Table 5.
Expanded list of molecular alterations covered by OPA on the Genexus platform.
4.2. Fusions Rearrangements
Regarding RNA samples, the Genexus platform successfully analyzed all retrieved cases. Briefly, a median number of total reads, mapped reads and mean read length of 1,721,491.0 (ranging from 1,471,817.00 to 2,462,555.00), 158,230.4 (ranging from 37,387.0 to 1,029,745.00), 98.8 bp (ranging from 91 to 104 bp) were identified, respectively (Table 6).

Table 6.
Technical parameters from RNA-based analysis by using S5 Plus and Genexus systems.
Of note, 10 out of 32 (31.2%) patients highlighted aberrant transcripts by using the Genexus platform. Among them, 5 out of 10 and 2 out of 10 patients showed ALK and RET rearrangements, respectively. Moreover, three patients were positive for ROS1, NTRK aberrant transcripts and MET Δ 14 skipping mutations, respectively (Table 7). Interestingly, rearranged genes were identified by OPA on the Genexus platform in 9 out of 10 (90.0%) retrieved cases, showing a concordance rate of 96.9% (31 out of 32 cases) with the SiRe panel in the S5 system. Particularly, ID#1 was positive for a NTRK3–KANK1 fusion transcript not previously detected with the SiRe panel on the S5 platform. No significant variations were observed in accordance with histological groups, rearranged genes, fusion partners, and mapped read levels between Genexus and previously tested samples on the S5 platform.

Table 7.
Comparison of RNA-related molecular alterations between S5 Plus and Genexus platforms.
5. Discussion
In the era of personalized medicine, the rapidly increasing number of predictive biomarkers approved in clinical practice has revolutionized the treatment strategy for solid-tumor patients [1,2,9]. Although there is a widespread diffusion of single-gene testing platforms in the vast majority of laboratories involved in molecular tests, low multiplexing biomarker analysis discourages their implementation as pivotal diagnostic platforms in clinical practice [23,24]. As regards NGS techniques, they allow us to simultaneously cover clinically relevant molecular alterations from a plethora of diagnostic routine specimens, saving technical costs and maintaining adequate TAT [31]. Moreover, NGS platforms may also benefit from automatized technical procedures that allow for accurate and reproducible analysis, resulting in low bench-working time [31]. The Genexus system consists of a scalable, versatile, and fully automatized sequencer that is able to carry out each technical procedure without manual operations [32]. This system is built to integrate analytical procedures (nucleic acid extraction, library preparation, template generation, sequencing) with data analysis by adopting pre-customized pipeline analysis. Accordingly, automatized data analysis carried out by proprietary software supports healthcare professional figures involved in molecular testing. This approach allows us to save time by accurately interpreting molecular records, in comparison with semi-automatized procedures. As regards the NGS-based multiplexing strategy, it is considered a reliable technical approach that is able to decrease technical costs in molecular tests. Here, we have validated the Genexus system in our diagnostic routine by comparing its analytical performance in a retrospective series of clinical cases previously analyzed with a custom NGS panel in the S5 system. As expected, all diagnostic specimens (n = 64) were successfully analyzed by using this fully automatized system. Overall, a concordance rate of 96.9% (62 out of 64) was reached by adopting the Sire panel in the S5 system as the reference standard. Interestingly, molecular analysis was unmatched with previously archived data in only two cases (DNA-ID#19 and RNA-ID#1). Of note, sample DNA-ID#19 derived from a BC patient had a positive result for PIK3CA exon 9 p.E545K hotspot alteration in the Genexus system, with a mutant allele fraction (MAF) of 7.2%. Following the manufacturer’s clinical cut-off (MAF ≥ 5%), previous analysis did not show any clinically relevant molecular alteration. By conducting a visual inspection of raw data, the same alteration at 0.9% was detected. This event may occur in residual scant samples where mutated alleles may encounter decreasing VAF levels [33]. Similarly, RNA-ID#1 showed NTRK3 (ex14)—KANK1 (ex3), an aberrant transcript not previously detected with the standard reference approach. In this case, NTRK3 was not covered by reference range of the SiRe fusion panel.
In a non-negligible percentage of cases, synchronous lesions may be observed in CRC patients. In this scenario, NGS may be considered an affordable technical strategy to comprehensively conduct the molecular assessment of CRC patients where heterogeneous specimens are clinically available [28]. DNA-ID#11 and DNA-ID#2 represent synchronous lesions of a CRC elected to molecular testing. Interestingly, both S5 and Genexus systems revealed KRAS exon 2 p.G12C and PIK3CA exon 20 p.H1047R hotspot mutations, demonstrating a common origin of these lesions. Moreover, NGS systems overcome technical issues from the analysis of “complex” molecular alteration. Case DNA-ID#22 confirmed two concomitant KRAS exon 2 hotspot mutations (p.G13D+p.G13E) on the Genexus platform, previously detected by reference technology. Although this study provides encouraging results for the implementation of the Genexus system in the clinical routine setting of solid-tumor patients, some limitations may be identified. Firstly, this technical report aims to compare the analytical parameters of two NGS-based technologies using a series of diagnostic routine specimens without any clinical considerations. Secondly, this retrospective study is based on the analysis of a small group of cases retrieved from the internal archive of the University of Naples Federico II. All these crucial points warrant further analysis, but this preliminary data may suggest that a fully automatized Genexus system integrated with commercially available OPA (Thermo Fisher Scientific) represents a technically affordable, time-saving sequencing platform that enables us to analyze clinically relevant molecular alterations in diagnostic routine specimens.
Author Contributions
Conceptualization, C.D.L., F.P., G.T. and U.M.; methodology, C.D.L., F.P., G.R., M.N., P.P., M.R., F.C., L.P., C.S., D.C., G.G., A.I., G.T. and U.M.; software, C.D.L., F.P., G.T. and U.M.; validation, C.D.L., F.P., G.R., M.N., P.P., M.R., F.C., L.P., C.S., D.C., G.G., A.I., G.T. and U.M.; formal analysis, C.D.L., F.P., G.R., M.N., P.P., M.R., F.C., L.P., C.S., D.C., G.G., A.I., G.T. and U.M.; data curation, C.D.L., F.P., G.T. and U.M.; writing—original draft preparation, C.D.L. and F.P.; writing—review and editing, G.T. and U.M.; visualization, C.D.L., F.P., G.R., M.N., P.P., M.R., F.C., L.P., C.S., D.C., G.G., A.I., G.T. and U.M.; supervision, G.T. and U.M.; project administration, G.T. and U.M. All authors have read and agreed to the published version of the manuscript.
Funding
1. Monitoraggio ambientale, studio ed approfondimento della salute della popolazione residente in aree a rischio—In attuazione della D.G.R. Campanian.180/2019. 2. POR Campania FESR 2014–2020 Progetto “Sviluppo di Approcci Terapeutici Innovativi per patologie Neoplastiche resistenti ai trattamenti—SATIN”. 3. This work has been partly supported by a grant from the Italian Health Ministry’s research program (ID: NET-2016-02363853). National Center for Gene Therapy and Drugs based on RNA Technology MUR-CN3 CUP E63C22000940007 to DS.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Written informed consent has been obtained from the patient(s) to publish this paper.
Data Availability Statement
Not applicable.
Conflicts of Interest
Pasquale Pisapia has received personal fees as speaker bureau from Novartis for work performed outside of the curr ent study. Umberto Malapelle has received personal fees (as consultant and/or speaker bureau) from Boehringer Ingelheim, Roche, MSD, Amgen, Thermo Fisher Scientific, Eli Lilly, Diaceutics, GSK, Merck and AstraZeneca, Janssen, Diatech, Novartis and Hedera unrelated to the current work. Giancarlo Troncone reports personal fees (as speaker bureau or advisor) from Roche, MSD, Pfizer, Boehringer Ingelheim, Eli Lilly, BMS, GSK, Menarini, AstraZeneca, Amgen and Bayer, unrelated to the current work. The remaining authors declare no conflict of interest.
References
- Yates, L.R.; Seoane, J.; Le Tourneau, C.; Siu, L.L.; Marais, R.; Michiels, S.; Soria, J.C.; Campbell, P.; Normanno, N.; Scarpa, A.; et al. The European Society for Medical Oncology (ESMO) Precision Medicine Glossary. Ann. Oncol. 2018, 29, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Ciardiello, F.; Arnold, D.; Casali, P.G.; Cervantes, A.; Douillard, J.Y.; Eggermont, A.; Eniu, A.; McGregor, K.; Peters, S.; Piccart, M.; et al. Delivering precision medicine in oncology today and in future-the promise and challenges of personalised cancer medicine: A position paper by the European Society for Medical Oncology (ESMO). Ann. Oncol. 2014, 25, 1673–1678. [Google Scholar] [CrossRef]
- Di Nicolantonio, F.; Martini, M.; Molinari, F.; Sartore-Bianchi, A.; Arena, S.; Saletti, P.; De Dosso, S.; Mazzucchelli, L.; Frattini, M.; Siena, S.; et al. Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J. Clin. Oncol. 2008, 26, 5705–5712. [Google Scholar] [CrossRef] [PubMed]
- Lièvre, A.; Bachet, J.B.; Boige, V.; Cayre, A.; Le Corre, D.; Buc, E.; Ychou, M.; Bouché, O.; Landi, B.; Louvet, C.; et al. KRAS mutations as an independent prognostic factor in patients with advanced colorectal cancer treated with cetuximab. J. Clin. Oncol. 2008, 26, 374–379. [Google Scholar] [CrossRef]
- Chapman, P.B.; Hauschild, A.; Robert, C.; Haanen, J.B.; Ascierto, P.; Larkin, J.; Dummer, R.; Garbe, C.; Testori, A.; Maio, M.; et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 2011, 364, 2507–2516. [Google Scholar] [CrossRef] [PubMed]
- Antonescu, C.R. Targeted therapies in gastrointestinal stromal tumors. Semin. Diagn. Pathol. 2008, 25, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Fusco, N.; Malapelle, U.; Fassan, M.; Marchiò, C.; Buglioni, S.; Zupo, S.; Criscitiello, C.; Vigneri, P.; Dei Tos, A.P.; Maiorano, E.; et al. PIK3CA Mutations as a Molecular Target for Hormone Receptor-Positive, HER2-Negative Metastatic Breast Cancer. Front. Oncol. 2021, 11, 644737. [Google Scholar] [CrossRef]
- Planchard, D.; Popat, S.; Kerr, K.; Novello, S.; Smit, E.F.; Faivre-Finn, C.; Mok, T.S.; Reck, M.; Van Schil, P.E.; Hellmann, M.D.; et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018, 29 (Suppl. 4), iv192–iv237. [Google Scholar] [CrossRef]
- Yang, S.R.; Schultheis, A.M.; Yu, H.; Mandelker, D.; Ladanyi, M.; Büttner, R. Precision medicine in non-small cell lung cancer: Current applications and future directions. Semin. Cancer Biol. 2022, 84, 184–198. [Google Scholar] [CrossRef]
- Lindeman, N.I.; Cagle, P.T.; Aisner, D.L.; Arcila, M.E.; Beasley, M.B.; Bernicker, E.H.; Colasacco, C.; Dacic, S.; Hirsch, F.R.; Kerr, K.; et al. Updated Molecular Testing Guideline for the Selection of Lung Cancer Patients for Treatment with Targeted Tyrosine Kinase Inhibitors: Guideline from the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. Arch. Pathol. Lab. Med. 2018, 142, 321–346. [Google Scholar]
- Kalemkerian, G.P.; Narula, N.; Kennedy, E.B.; Biermann, W.A.; Donington, J.; Leighl, N.B.; Lew, M.; Pantelas, J.; Ramalingam, S.S.; Reck, M.; et al. Molecular Testing Guideline for the Selection of Patients With Lung Cancer for Treatment With Targeted Tyrosine Kinase Inhibitors: American Society of Clinical Oncology Endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 911–919. [Google Scholar] [PubMed]
- Jennings, L.J.; Arcila, M.E.; Corless, C.; Kamel-Reid, S.; Lubin, I.M.; Pfeifer, J.; Temple-Smolkin, R.L.; Voelkerding, K.V.; Nikiforova, M.N. Guidelines for Validation of Next-Generation Sequencing-Based Oncology Panels: A Joint Consensus Recommendation of the Association for Molecular Pathology and College of American Pathologists. J. Mol. Diagn. 2017, 19, 341–365. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, A.R.; Hamilton, S.R.; Allegra, C.J.; Grody, W.; Cushman-Vokoun, A.M.; Funkhouser, W.K.; Kopetz, S.E.; Lieu, C.; Lindor, N.M.; Minsky, B.D.; et al. Molecular Biomarkers for the Evaluation of Colorectal Cancer: Guideline from the American Society for Clinical Pathology, College of American Pathologists, Association for Molecular Pathology, and the American Society of Clinical Oncology. J. Clin. Oncol. 2017, 35, 1453–1486. [Google Scholar] [CrossRef] [PubMed]
- Mok, T.S.; Wu, Y.L.; Thongprasert, S.; Yang, C.H.; Chu, D.T.; Saijo, N.; Sunpaweravong, P.; Han, B.; Margono, B.; Ichinose, Y.; et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 2009, 361, 947–957. [Google Scholar] [CrossRef]
- Kris, M.G.; Johnson, B.E.; Berry, L.D.; Kwiatkowski, D.J.; Iafrate, A.J.; Wistuba, I.I.; Varella-Garcia, M.; Franklin, W.A.; Aronson, S.L.; Su, P.F.; et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA 2014, 311, 1998–2006. [Google Scholar] [CrossRef]
- Meric-Bernstam, F.; Brusco, L.; Shaw, K.; Horombe, C.; Kopetz, S.; Davies, M.A.; Routbort, M.; Piha-Paul, S.A.; Janku, F.; Ueno, N.; et al. Feasibility of Large-Scale Genomic Testing to Facilitate Enrollment onto Genomically Matched Clinical Trials. J. Clin. Oncol. 2015, 33, 2753–2762. [Google Scholar] [CrossRef]
- Sundaresan, T.K.; Sequist, L.V.; Heymach, J.V.; Riely, G.J.; Jänne, P.A.; Koch, W.H.; Sullivan, J.P.; Fox, D.B.; Maher, R.; Muzikansky, A.; et al. Detection of T790M, the Acquired Resistance EGFR Mutation, by Tumor Biopsy versus Noninvasive Blood-Based Analyses. Clin. Cancer Res. 2016, 22, 1103–1110. [Google Scholar] [CrossRef]
- Roy-Chowdhuri, S.; Chow, C.W.; Kane, M.K.; Yao, H.; Wistuba, I.I.; Krishnamurthy, S.; Stewart, J.; Staerkel, G. Optimizing the DNA yield for molecular analysis from cytologic preparations. Cancer Cytopathol. 2016, 124, 254–260. [Google Scholar] [CrossRef]
- Pepe, F.; De Luca, C.; Smeraglio, R.; Pisapia, P.; Sgariglia, R.; Nacchio, M.; Russo, M.; Serra, N.; Rocco, D.; Battiloro, C.; et al. Performance analysis of SiRe next-generation sequencing panel in diagnostic setting: Focus. on NSCLC routine samples. J. Clin. Pathol. 2019, 72, 38–45. [Google Scholar] [CrossRef]
- Velizheva, N.P.; Rechsteiner, M.P.; Wong, C.E.; Zhong, Q.; Rössle, M.; Bode, B.; Moch, H.; Soltermann, A.; Wild, P.J.; Tischler, V. Cytology smears as excellent starting material for next-generation sequencing-based molecular testing of patients with adenocarcinoma of the lung. Cancer Cytopathol. 2017, 125, 30–40. [Google Scholar] [CrossRef]
- Heitzer, E.; Haque, I.S.; Roberts, C.E.S.; Speicher, M.R. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat. Rev. Genet. 2019, 20, 71–88. [Google Scholar] [CrossRef] [PubMed]
- Pisapia, P.; Pepe, F.; Iaccarino, A.; Sgariglia, R.; Nacchio, M.; Conticelli, F.; Salatiello, M.; Tufano, R.; Russo, G.; Gragnano, G.; et al. Next Generation Sequencing in Cytopathology: Focus on Non-Small Cell Lung Cancer. Front. Med. 2021, 8, 633923. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, H.; Tanishima, S.; Fujii, K.; Mori, R.; Okada, C.; Yanagita, E.; Shibata, Y.; Matsuoka, R.; Amano, T.; Yamada, T.; et al. Clinical impact of a cancer genomic profiling test using an in-house comprehensive targeted sequencing system. Cancer Sci. 2020, 111, 3926–3937. [Google Scholar] [CrossRef] [PubMed]
- Kou, T.; Kanai, M.; Yamamoto, Y.; Kamada, M.; Nakatsui, M.; Sakuma, T.; Mochizuki, H.; Hiroshima, A.; Sugiyama, A.; Nakamura, E.; et al. Clinical sequencing using a next-generation sequencing-based multiplex gene assay in patients with advanced solid tumors. Cancer Sci. 2017, 108, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Pisapia, P.; Pepe, F.; Baggi, A.; Barberis, M.; Galvano, A.; Gristina, V.; Mastrilli, F.; Novello, S.; Pagni, F.; Pasini, S.; et al. Next generation diagnostic algorithm in non-small cell lung cancer predictive molecular pathology: The KWAY Italian multicenter cost evaluation study. Crit. Rev. Oncol. Hematol. 2022, 169, 103525. [Google Scholar] [CrossRef]
- Low, S.K.; Ariyasu, R.; Uchibori, K.; Hayashi, R.; Chan, H.T.; Chin, Y.M.; Akita, T.; Harutani, Y.; Kiritani, A.; Tsugitomi, R.; et al. Rapid genomic profiling of circulating tumor DNA in non-small cell lung cancer using Oncomine Precision Assay. with Genexus™ integrated sequencer. Transl. Lung Cancer Res. 2022, 11, 711–721. [Google Scholar] [CrossRef]
- Ilié, M.; Hofman, V.; Bontoux, C.; Heeke, S.; Lespinet-Fabre, V.; Bordone, O.; Lassalle, S.; Lalvée, S.; Tanga, V.; Allegra, M.; et al. Setting Up an Ultra-Fast Next-Generation Sequencing Approach as Reflex Testing at Diagnosis of Non-Squamous Non-Small Cell Lung Cancer; Experience of a Single Center (LPCE, Nice, France). Cancers 2022, 14, 2258. [Google Scholar] [CrossRef]
- Sheffield, B.S.; Beharry, A.; Diep, J.; Perdrizet, K.; Iafolla, M.A.J.; Raskin, W.; Dudani, S.; Brett, M.A.; Starova, B.; Olsen, B.; et al. Point of Care Molecular Testing: Community-Based Rapid Next-Generation Sequencing to Support Cancer Care. Curr. Oncol. 2022, 29, 1326–1334. [Google Scholar] [CrossRef]
- Malapelle, U.; Mayo de-Las-Casas, C.; Rocco, D.; Garzon, M.; Pisapia, P.; Jordana-Ariza, N.; Russo, M.; Sgariglia, R.; De Luca, C.; Pepe, F.; et al. Development of a gene panel for next-generation sequencing of clinically relevant mutations in cell-free DNA from cancer patients. Br. J. Cancer. 2017, 116, 802–810. [Google Scholar] [CrossRef]
- Malapelle, U.; Pepe, F.; Pisapia, P.; Sgariglia, R.; Nacchio, M.; De Luca, C.; Lacalamita, R.; Tommasi, S.; Pinto, R.; Palomba, G.; et al. Harmonization of Next-Generation Sequencing Procedure in Italian Laboratories: A Multi-Institutional Evaluation of the SiRe® Panel. Front. Oncol. 2020, 10, 236. [Google Scholar] [CrossRef]
- Mosele, F.; Remon, J.; Mateo, J.; Westphalen, C.B.; Barlesi, F.; Lolkema, M.P.; Normanno, N.; Scarpa, A.; Robson, M.; Meric-Bernstam, F.; et al. Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: A report from the ESMO Precision Medicine Working Group. Ann. Oncol. 2020, 31, 1491–1505. [Google Scholar] [CrossRef] [PubMed]
- Werner, R.; Connolly, A.; Bennett, M.; Hand, C.K.; Burke, L. Implementation of an ISO15189 accredited next-generation sequencing service with the fully automated Ion Torrent Genexus: The experience of a clinical diagnostic laboratory. J. Clin. Pathol. 2022, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rivera, D.; Paudice, M.; Gismondi, V.; Anselmi, G.; Vellone, V.G.; Varesco, L.; Ligurian BRCA Working Group. Implementing NGS-based BRCA tumour tissue testing in FFPE ovarian carcinoma specimens: Hints from a real-life experience within the framework of expert recommendations. J. Clin. Pathol. 2021, 74, 596–603. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).