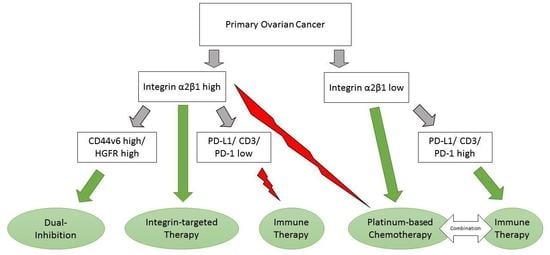
Integrin α2β1 Represents a Prognostic and Predictive Biomarker in Primary Ovarian Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Immunohistochemistry
2.3. Evaluation of Biomarker Expression
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristic
3.2. Prognostic and Predictive Impact of Integrin α2β1
3.3. Correlation of Integrin α2β1 with Other Biomarkers
3.4. Prognostic and Predictive Impact of Integrin α2β1 Combined with Other Biomarkers
3.5. Correlation of Integrin α2β1 and Immune Infiltrate
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- du Bois, A.; Reuss, A.; Pujade-Lauraine, E.; Harter, P.; Ray-Coquard, I.; Pfisterer, J. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: A combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: By the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d’Investigateurs Nationaux Pour les Etudes des Cancers de l’Ovaire (GINECO). Cancer 2009, 115, 1234–1244. [Google Scholar] [CrossRef]
- Waldron, L.; Haibe-Kains, B.; Culhane, A.C.; Riester, M.; Ding, J.; Wang, X.V.; Ahmadifar, M.; Tyekucheva, S.; Bernau, C.; Risch, T.; et al. Comparative meta-analysis of prognostic gene signatures for late-stage ovarian cancer. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Chen, R.; Bai, Y.; Lu, X. The prognostic value of tumor-infiltrating T lymphocytes in ovarian cancer. Oncotarget 2017, 8, 15621–15631. [Google Scholar] [CrossRef] [PubMed]
- Plotti, F.; Terranova, C.; Guzzo, F.; De Cicco Nardone, C.; Luvero, D.; Bartolone, M.; Dionisi, C.; Benvenuto, D.; Fabris, S.; Ciccozzi, M.; et al. Role of BRCA Mutation and HE4 in Predicting Chemotherapy Response in Ovarian Cancer: A Retrospective Pilot Study. Biomedicines 2021, 9, 55. [Google Scholar] [CrossRef]
- Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, D.K., AWMF). S3-Leitlinie Diagnostik, Therapie und Nachsorge Maligner Ovarialtumoren, Langversion 4.0. Available online: https://www.leitlinienprogramm-onkologie.de/leitlinien/ovarialkarzinom/ (accessed on 24 January 2021).
- National Institute for Health and Care Excellence. Olaparib for Maintenance Treatment of BRCA Mutation-Positive Advanced Ovarian, Fallopian Tube or Peritoneal Cancer after Response to First-Line Platinum-Based Chemotherapy. Available online: https://www.nice.org.uk/guidance/ta598 (accessed on 24 January 2021).
- Armstrong, D.K.; Alvarez, R.D.; Bakkum-Gamez, J.N.; Barroilhet, L.; Behbakht, K.; Berchuck, A.; Berek, J.S.; Chen, L.M.; Cristea, M.; DeRosa, M.; et al. NCCN Guidelines Insights: Ovarian Cancer, Version 1.2019. J. Natl. Compr. Cancer Netw. 2019, 17, 896–909. [Google Scholar] [CrossRef] [PubMed]
- Colombo, N.; Sessa, C.; du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann. Oncol. 2019, 30, 672–705. [Google Scholar] [CrossRef]
- Pignata, S.; Cecere, S.C.; Du Bois, A.; Harter, P.; Heitz, F. Treatment of recurrent ovarian cancer. Ann. Oncol. 2017, 28, viii51–viii56. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Bianconi, D.; Unseld, M.; Prager, G.W. Integrins in the Spotlight of Cancer. Int. J. Mol. Sci. 2016, 17, 2037. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, K.K.; Pal, S.; Moulik, S.; Chatterjee, A. Integrins and metastasis. Cell Adh. Migr. 2013, 7, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Alday-Parejo, B.; Stupp, R.; Rüegg, C. Are Integrins Still Practicable Targets for Anti-Cancer Therapy? Cancers 2019, 11, 978. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef]
- Aziz, M.; Agarwal, K.; Dasari, S.; Mitra, A.A.K. Productive Cross-Talk with the Microenvironment: A Critical Step in Ovarian Cancer Metastasis. Cancers 2019, 11, 1608. [Google Scholar] [CrossRef]
- Ghoneum, A.; Afify, H.; Salih, Z.; Kelly, M.; Said, N. Role of tumor microenvironment in ovarian cancer pathobiology. Oncotarget 2018, 9, 22832–22849. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Sawada, K.; Kimura, T. Potential of Integrin Inhibitors for Treating Ovarian Cancer: A Literature Review. Cancers 2017, 9, 83. [Google Scholar] [CrossRef]
- Heino, J. The collagen receptor integrins have distinct ligand recognition and signaling functions. Matrix Biol. 2000, 19, 319–323. [Google Scholar] [CrossRef]
- Humphries, J.D.; Byron, A.; Humphries, M.J. Integrin ligands at a glance. J. Cell Sci. 2006, 119, 3901–3903. [Google Scholar] [CrossRef] [PubMed]
- Van de Walle, G.R.; Vanhoorelbeke, K.; Majer, Z.; Illyés, E.; Baert, J.; Pareyn, I.; Deckmyn, H. Two functional active conformations of the integrin {alpha}2{beta}1, depending on activation condition and cell type. J. Biol. Chem. 2005, 280, 36873–36882. [Google Scholar] [CrossRef]
- Zutter, M.M.; Santoro, S.A. Widespread histologic distribution of the alpha 2 beta 1 integrin cell-surface collagen receptor. Am. J. Pathol. 1990, 137, 113–120. [Google Scholar]
- Naci, D.; Vuori, K.; Aoudjit, F. Alpha2beta1 integrin in cancer development and chemoresistance. Semin. Cancer Biol. 2015, 35, 145–153. [Google Scholar] [CrossRef]
- Zheng, W.; Ge, D.; Meng, G. Reversing microtubule-directed chemotherapeutic drug resistance by co-delivering alpha2beta1 inhibitor and paclitaxel with nanoparticles in ovarian cancer. Cell Biol. Int. 2020, 44, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Dotzer, K.; Schluter, F.; Schoenberg, M.B.; Bazhin, A.V.; von Koch, F.E.; Schnelzer, A.; Anthuber, S.; Grab, D.; Czogalla, B.; Burges, A.; et al. Immune Heterogeneity Between Primary Tumors and Corresponding Metastatic Lesions and Response to Platinum Therapy in Primary Ovarian Cancer. Cancers 2019, 11, 1250. [Google Scholar] [CrossRef] [PubMed]
- Sieh, W.; Kobel, M.; Longacre, T.A.; Bowtell, D.D.; deFazio, A.; Goodman, M.T.; Hogdall, E.; Deen, S.; Wentzensen, N.; Moysich, K.B.; et al. Hormone-receptor expression and ovarian cancer survival: An Ovarian Tumor Tissue Analysis consortium study. Lancet Oncol. 2013, 14, 853–862. [Google Scholar] [CrossRef]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O.; Fitzgibbons, P.; Hanna, W.; et al. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J. Clin. Oncol. 2018, 36, 2105–2122. [Google Scholar] [CrossRef] [PubMed]
- Ruschoff, J.; Dietel, M.; Baretton, G.; Arbogast, S.; Walch, A.; Monges, G.; Chenard, M.P.; Penault-Llorca, F.; Nagelmeier, I.; Schlake, W.; et al. HER2 diagnostics in gastric cancer-guideline validation and development of standardized immunohistochemical testing. Virchows Arch. 2010, 457, 299–307. [Google Scholar] [CrossRef]
- Yao, E.S.; Zhang, H.; Chen, Y.Y.; Lee, B.; Chew, K.; Moore, D.; Park, C. Increased beta1 integrin is associated with decreased survival in invasive breast cancer. Cancer Res. 2007, 67, 659–664. [Google Scholar] [CrossRef]
- Liu, Q.Z.; Gao, X.H.; Chang, W.J.; Gong, H.F.; Fu, C.G.; Zhang, W.; Cao, G.W. Expression of ITGB1 predicts prognosis in colorectal cancer: A large prospective study based on tissue microarray. Int. J. Clin. Exp. Pathol. 2015, 8, 12802–12810. [Google Scholar]
- Sun, Q.; Zhou, C.; Ma, R.; Guo, Q.; Huang, H.; Hao, J.; Liu, H.; Shi, R.; Liu, B. Prognostic value of increased integrin-beta 1 expression in solid cancers: A meta-analysis. OncoTargets Ther. 2018, 11, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Sawada, K.; Mitra, A.K.; Radjabi, A.R.; Bhaskar, V.; Kistner, E.O.; Tretiakova, M.; Jagadeeswaran, S.; Montag, A.; Becker, A.; Kenny, H.A.; et al. Loss of E-cadherin promotes ovarian cancer metastasis via alpha 5-integrin, which is a therapeutic target. Cancer Res. 2008, 68, 2329–2339. [Google Scholar] [CrossRef]
- Adachi, M.; Taki, T.; Higashiyama, M.; Kohno, N.; Inufusa, H.; Miyake, M. Significance of integrin alpha5 gene expression as a prognostic factor in node-negative non-small cell lung cancer. Clin. Cancer Res. 2000, 6, 96–101. [Google Scholar]
- Ren, J.; Xu, S.; Guo, D.; Zhang, J.; Liu, S. Increased expression of α5β1-integrin is a prognostic marker for patients with gastric cancer. Clin. Transl. Oncol. 2014, 16, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Chen, Z.; Wang, Z.H.; Wang, H.; Huang, L.M. Prognostic significance of α5β1-integrin expression in cervical cancer. Asian Pac. J. Cancer Prev. 2013, 14, 3891–3895. [Google Scholar] [CrossRef]
- Shield, K.; Riley, C.; Quinn, M.A.; Rice, G.E.; Ackland, M.L.; Ahmed, N. Alpha2beta1 integrin affects metastatic potential of ovarian carcinoma spheroids by supporting disaggregation and proteolysis. J. Carcinog. 2007, 6, 11. [Google Scholar] [CrossRef]
- Burnier, J.V.; Wang, N.; Michel, R.P.; Hassanain, M.; Li, S.; Lu, Y.; Metrakos, P.; Antecka, E.; Burnier, M.N.; Ponton, A.; et al. Type IV collagen-initiated signals provide survival and growth cues required for liver metastasis. Oncogene 2011, 30, 3766–3783. [Google Scholar] [CrossRef]
- Bartolomé, R.A.; Barderas, R.; Torres, S.; Fernandez-Aceñero, M.J.; Mendes, M.; García-Foncillas, J.; Lopez-Lucendo, M.; Casal, J.I. Cadherin-17 interacts with α2β1 integrin to regulate cell proliferation and adhesion in colorectal cancer cells causing liver metastasis. Oncogene 2014, 33, 1658–1669. [Google Scholar] [CrossRef] [PubMed]
- Eliceiri, B.P. Integrin and growth factor receptor crosstalk. Circ. Res. 2001, 89, 1104–1110. [Google Scholar] [CrossRef]
- Hamidi, H.; Ivaska, J. Every step of the way: Integrins in cancer progression and metastasis. Nat. Rev. Cancer 2018, 18, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Aoudjit, F.; Vuori, K. Integrin signaling inhibits paclitaxel-induced apoptosis in breast cancer cells. Oncogene 2001, 20, 4995–5004. [Google Scholar] [CrossRef] [PubMed]
- Duxbury, M.S.; Ito, H.; Benoit, E.; Waseem, T.; Ashley, S.W.; Whang, E.E. RNA interference demonstrates a novel role for integrin-linked kinase as a determinant of pancreatic adenocarcinoma cell gemcitabine chemoresistance. Clin. Cancer Res. 2005, 11, 3433–3438. [Google Scholar] [CrossRef] [PubMed]
- Sethi, T.; Rintoul, R.C.; Moore, S.M.; MacKinnon, A.C.; Salter, D.; Choo, C.; Chilvers, E.R.; Dransfield, I.; Donnelly, S.C.; Strieter, R.; et al. Extracellular matrix proteins protect small cell lung cancer cells against apoptosis: A mechanism for small cell lung cancer growth and drug resistance in vivo. Nat. Med. 1999, 5, 662–668. [Google Scholar] [CrossRef]
- Wilson, M.K.; Pujade-Lauraine, E.; Aoki, D.; Mirza, M.R.; Lorusso, D.; Oza, A.M.; du Bois, A.; Vergote, I.; Reuss, A.; Bacon, M.; et al. Fifth Ovarian Cancer Consensus Conference of the Gynecologic Cancer InterGroup: Recurrent disease. Ann. Oncol. 2017, 28, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Scalici, J.M.; Harrer, C.; Allen, A.; Jazaeri, A.; Atkins, K.A.; McLachlan, K.R.; Slack-Davis, J.K. Inhibition of alpha4beta1 integrin increases ovarian cancer response to carboplatin. Gynecol. Oncol. 2014, 132, 455–461. [Google Scholar] [CrossRef]
- Wantoch von Rekowski, K.; König, P.; Henze, S.; Schlesinger, M.; Zawierucha, P.; Januchowski, R.; Bendas, G. The Impact of Integrin-Mediated Matrix Adhesion on Cisplatin Resistance of W1 Ovarian Cancer Cells. Biomolecules 2019, 9, 788. [Google Scholar] [CrossRef] [PubMed]
- Baltes, F.; Pfeifer, V.; Silbermann, K.; Caspers, J.; Wantoch von Rekowski, K.; Schlesinger, M.; Bendas, G. β(1)-Integrin binding to collagen type 1 transmits breast cancer cells into chemoresistance by activating ABC efflux transporters. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118663. [Google Scholar] [CrossRef] [PubMed]
- Gibney, G.T.; Weiner, L.M.; Atkins, M.B. Predictive biomarkers for checkpoint inhibitor-based immunotherapy. Lancet Oncol. 2016, 17, e542–e551. [Google Scholar] [CrossRef]
- Schmid, M.C.; Avraamides, C.J.; Dippold, H.C.; Franco, I.; Foubert, P.; Ellies, L.G.; Acevedo, L.M.; Manglicmot, J.R.; Song, X.; Wrasidlo, W.; et al. Receptor tyrosine kinases and TLR/IL1Rs unexpectedly activate myeloid cell PI3kγ, a single convergent point promoting tumor inflammation and progression. Cancer Cell 2011, 19, 715–727. [Google Scholar] [CrossRef]
- Wu, A.; Zhang, S.; Liu, J.; Huang, Y.; Deng, W.; Shu, G.; Yin, G. Integrated Analysis of Prognostic and Immune Associated Integrin Family in Ovarian Cancer. Front. Genet. 2020, 11, 705. [Google Scholar] [CrossRef]
- Brown, N.F.; Marshall, J.F. Integrin-Mediated TGFbeta Activation Modulates the Tumour Microenvironment. Cancers 2019, 11, 1221. [Google Scholar] [CrossRef]
- Kandalaft, L.E.; Odunsi, K.; Coukos, G. Immunotherapy in Ovarian Cancer: Are We There Yet? J. Clin. Oncol. 2019, 37, 2460–2471. [Google Scholar] [CrossRef]
- Wang, W.; Liu, J.R.; Zou, W. Immunotherapy in Ovarian Cancer. Surg. Oncol. Clin. N. Am. 2019, 28, 447–464. [Google Scholar] [CrossRef]
- Attinger, A.; Hou, S.; Pesenti, E.A.; Mancini, L.; Mottl, H. Abstract LB-294: GBR 500, a monoclonal VLA-2 antibody inhibits tumor and metastasis growth but not extravasation in a prostate cancer animal model. In Proceedings of the AACR 102nd Annual Meeting 2011, Orlando, FL, USA, 2–6 April 2011. [Google Scholar]
- Milojkovic Kerklaan, B.; Slater, S.; Flynn, M.; Greystoke, A.; Witteveen, P.O.; Megui-Roelvink, M.; de Vos, F.; Dean, E.; Reyderman, L.; Ottesen, L.; et al. A phase I, dose escalation, pharmacodynamic, pharmacokinetic, and food-effect study of alpha2 integrin inhibitor E7820 in patients with advanced solid tumors. Investig. New Drugs 2016, 34, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Mita, M.; Kelly, K.R.; Mita, A.; Ricart, A.D.; Romero, O.; Tolcher, A.; Hook, L.; Okereke, C.; Krivelevich, I.; Rossignol, D.P.; et al. Phase I study of E7820, an oral inhibitor of integrin alpha-2 expression with antiangiogenic properties, in patients with advanced malignancies. Clin. Cancer Res. 2011, 17, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.Y.; Cho, W.D.; Hong, K.P.; Choi da, B.; Hong, J.W.; Kim, S.; Moon, Y.R.; Son, S.M.; Lee, O.J.; Lee, H.C.; et al. Novel monoclonal antibody against beta 1 integrin enhances cisplatin efficacy in human lung adenocarcinoma cells. J. Biomed. Res. 2016, 30, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Raab-Westphal, S.; Marshall, J.F.; Goodman, S.L. Integrins as Therapeutic Targets: Successes and Cancers. Cancers 2017, 9, 110. [Google Scholar] [CrossRef]
- Millard, M.; Odde, S.; Neamati, N. Integrin targeted therapeutics. Theranostics 2011, 1, 154–188. [Google Scholar] [CrossRef]
- Taylan, E.; Zayou, F.; Murali, R.; Karlan, B.Y.; Pandol, S.J.; Edderkaoui, M.; Orsulic, S. Dual targeting of GSK3B and HDACs reduces tumor growth and improves survival in an ovarian cancer mouse model. Gynecol. Oncol. 2020, 159, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Camblin, A.J.; Tan, G.; Curley, M.D.; Yannatos, I.; Iadevaia, S.; Rimkunas, V.; Mino-Kenudson, M.; Bloom, T.; Schoeberl, B.; Drummond, D.C.; et al. Dual targeting of IGF-1R and ErbB3 as a potential therapeutic regimen for ovarian cancer. Sci. Rep. 2019, 9, 16832. [Google Scholar] [CrossRef]
| Antigen | Clone | Species | Fixation | Use of Kit | wc (μg/mL) | Supplier | Cut-Off for Positivity |
|---|---|---|---|---|---|---|---|
| Primary antibodies | |||||||
| Integrin α2β1 | BHA2.1 | m | Acetone | - | 2.50 | Millipore, Burlington, MA, USA | ≥20% |
| ERα | 1D5 | m | Formalin | + | 2.50 | Dako, Santa Clara, CA, USA | ≥1% |
| PR | PgR 636 | m | Formalin | + | 2.50 | Dako, Santa Clara, CA, USA | ≥1% |
| HER-2/neu | 4B5 | r | Acetone | - | 1.50 | Ventana, Roche, Basel, CH | ≥10% (Intensity 2+/3+) |
| EGFR | H11 | m | Acetone | - | 2.94 | Dako, Santa Clara, CA, USA | ≥50% |
| HGFR | SP44 | r | Acetone | - | 2.12 | Spring Bioscience, Pleasanton, CA, USA | ≥50% |
| IGF1R | 23-41 | m | Acetone | + | 4.00 | invitrogen, Carlsbad, CA, USA | ≥80% |
| MUC-1 | Ma552 | m | Acetone | - | 0.50 | Monosan, Uden, NL | ≥70% |
| CD44v6 | VFF-18 | m | Acetone | - | 1.00 | affymetrix eBioscience, Santa Clara, CA, USA | ≥10% |
| Integrin αVβ3 | LM609 | m | Acetone | - | 5.00 | Millipore, Burlington, MA, USA | ≥20% |
| CD3 | UCHT1 | m | Acetone | - | 1.25 | BD Biosciences, Franklin Lakes, NJ, USA | |
| CD8 | C8/144B | m | Acetone | + | 3.00 | Dako, Santa Clara, CA, USA | |
| PD-1 | MIH4 | m | Acetone | + | 10.00 | affymetrix eBioscience, Santa Clara, CA, USA | |
| PD-L1 | MIH1 | m | Acetone | + | 10.00 | affymetrix eBioscience, Santa Clara, CA, USA | ≥1% |
| Positive controls | |||||||
| Epithelial Antigen | Ber-EP4 | m | Acetone | - | 2.50 | Dako, Santa Clara, CA, USA | |
| CD45 | 2B11 + PD7/26 | m | Acetone | - | 4.50 | Dako, Santa Clara, CA, USA | |
| Isotype controls | |||||||
| MOPC 21 | MOPC 21 | m | - | 5.00 | Sigma-Aldrich, St. Louis, MO, USA | ||
| MOPC 21 | m | + | 4.00 | Sigma-Aldrich, St. Louis, MO, USA | |||
| MOPC 21 | m | + | 10.00 | Sigma-Aldrich, St. Louis, MO, USA | |||
| DA1E | DA1E | r | - | 2.12 | Cell Signaling, Danvers, MA, USA | ||
| Biotin conjugated secondary antibodies | |||||||
| 111-065-114 | g anti r | 7.00 | Jackson Immunoresearch, West Grove, PA, USA | ||||
| 315-065-048 | r anti m | 0.75 | Jackson Immunoresearch, West Grove, PA, USA | ||||
| n or Value | % | ||
|---|---|---|---|
| Age | mean/median | 62/66 years | |
| range | 24–83 years | ||
| FIGO Stage | I or II | 0 | 0.0% |
| III | 34 | 70.8% | |
| IV | 14 | 29.2% | |
| pT | pT2 | 5 | 10.4% |
| pT3 | 43 | 89.6% | |
| pN | pN0 | 6 | 12.5% |
| pN1 | 31 | 64.6% | |
| Nx | 11 | 22.9% | |
| cM | cM0 | 34 | 70.8% |
| cM1 | 14 | 29.2% | |
| Primary Tumor Site | Ovarian | 39 | 81.3% |
| Fallopian Tube | 6 | 12.5% | |
| Peritoneal | 3 | 6.3% | |
| Histological Subtype | Serous | 44 | 91.7% |
| Other | 4 | 8.4% | |
| Grading | G1/G2 | 2 | 4.2% |
| G3 | 46 | 95.8% | |
| Ascites | yes | 40 | 83.3% |
| no | 8 | 16.7% | |
| Macroscopic Residual Tumor after Surgery | None | 35 | 72.9% |
| <1 cm | 6 | 12.5% | |
| >1 cm | 7 | 14.6% | |
| First-Line-Treatment | C | 4 | 8.3% |
| C + P | 15 | 31.3% | |
| C + P + B | 25 | 52.1% | |
| None | 4 | 8.3% | |
| Relapse after Chemotherapy | <6 months | 2 | 4.2% |
| 6–12 months | 12 | 25.0% | |
| >12 months | 28 | 58.3% | |
| none or non-sufficient chemotherapy | 6 | 12.5% |
| PFS | PFI | OS | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Log-Rank | MV Cox Regression | Log-Rank | MV Cox Regression | Log-Rank | ||||||
| MS | p | HR (CI 95%) | p | MS | p | HR (CI 95%) | p | MS | p | ||
| Age ≤ 62 years | 19 | 22 | 0.965 | 17 | 0.970 | nr | 0.193 | ||||
| Age > 62 years | 23 | 22 | 17 | 42 | |||||||
| <pT3c | 7 | 27 | 0.665 | 22 | 0.679 | 45 | 0.928 | ||||
| pT3c | 35 | 22 | 17 | 42 | |||||||
| pN0 | 5 | 29 | 0.163 | 17 | 0.145 | 45 | 0.929 | ||||
| pN1 | 28 | 22 | 22 | 42 | |||||||
| cM0 | 29 | 27 | 22 | nr | |||||||
| cM1 | 13 | 16 | 0.081 | 2.06 (0.92–4.62) | 0.081 | 11 | 0.068 | 2.10 (0.94–4.69) | 0.072 | 30 | 0.015 |
| G1/G2 | 2 | 14 | 0.579 | 8 | 0.610 | 30 | 0.843 | ||||
| G3 | 40 | 22 | 17 | 42 | |||||||
| Ascites absent | 6 | 35 | 0.147 | 30 | 0.139 | 42 | 0.408 | ||||
| Ascites present | 36 | 19 | 15 | 38 | |||||||
| MR Tumor absent | 30 | 27 | 22 | 45 | |||||||
| MR Tumor present | 12 | 13 | 0.008 | 2.19 (1.03–4.68) | 0.043 | 9 | 0.010 | 2.10 (0.99-4.51) | 0.057 | 26 | 0.041 |
| Integrin α2β1 low | 27 | 29 | 25 | 45 | |||||||
| Integrin α2β1 high | 15 | 16 | 0.035 | 2.46 (1.14–5.29) | 0.021 | 11 | 0.034 | 2.45 (1.14-5.26) | 0.022 | 30 | 0.155 |
| Integrin α2β1 | ||||||
|---|---|---|---|---|---|---|
| n | <20% | ≥20% | p# | |||
| Growth Factor-Receptor | ERα | 48 | 0.035 | |||
| <1% | 10 | 1 | ||||
| ≥1% | 20 | 17 | ||||
| PR | 48 | 0.127 | ||||
| <1% | 22 | 9 | ||||
| ≥1% | 8 | 9 | ||||
| Her-2/neu | 48 | 1 | ||||
| negative | 22 | 13 | ||||
| positive | 8 | 5 | ||||
| EGFR | 48 | 0.027 | ||||
| <50% | 27 | 11 | ||||
| ≥50% | 3 | 7 | ||||
| HGFR | 48 | 0.133 | ||||
| <50% | 16 | 5 | ||||
| ≥50% | 14 | 13 | ||||
| IGF1R | 48 | 0.451 | ||||
| <80% | 4 | 4 | ||||
| ≥80% | 26 | 14 | ||||
| Cell-Adhesion-Molecule | MUC-1 | 48 | 0.765 | |||
| <70% | 14 | 7 | ||||
| ≥70% | 16 | 11 | ||||
| CD44v6 | 48 | 0.103 | ||||
| <10% | 24 | 10 | ||||
| ≥10% | 6 | 8 | ||||
| Integrin αvβ3 | 48 | 0.19 | ||||
| <20% | 24 | 11 | ||||
| ≥20% | 6 | 7 | ||||
| PFS | PFI | OS | |||||
|---|---|---|---|---|---|---|---|
| n | MS | p * | MS | p * | MS | p * | |
| Integrin α2β1 high | 15 | 16 | 0.035 | 11 | 0.034 | 30 | 0.155 |
| Integrin α2β1 low | 27 | 29 | 25 | 45 | |||
| Integrin α2β1 high/ERα high | 14 | 16 | 0.078 | 11 | 0.073 | 30 | 0.287 |
| Remaining combinations # | 28 | 27 | 22 | 42 | |||
| Integrin α2β1 high/PR high | 8 | 16 | 0.574 | 1119 | 0.578 | 27 | 0.526 |
| Remaining combinations # | 34 | 24 | 19 | 42 | |||
| Integrin α2β1 high/Her-2/neu + | 5 | 21 | 0.043 | 15 | 0.037 | 36 | 0.698 |
| Remaining combinations # | 37 | 27 | 22 | 42 | |||
| Integrin α2β1 high/EGFR high | 6 | 14 | 0.289 | 8 | 0.290 | 30 | 0.482 |
| Remaining combinations # | 36 | 22 | 17 | 42 | |||
| Integrin α2β1 high/HGFR high | 11 | 15 | 0.004 | 10 | 0.004 | 27 | 0.054 |
| Remaining combinations # | 31 | 29 | 25 | 45 | |||
| Integrin α2β1 high/IGFR high | 11 | 16 | 0.045 | 11 | 0.043 | 36 | 0.381 |
| Remaining combinations # | 31 | 27 | 22 | 42 | |||
| Integrin α2β1 high/MUC-1 high | 9 | 14 | 0.063 | 9 | 0.055 | 27 | 0.257 |
| Remaining combinations # | 33 | 27 | 22 | 42 | |||
| Integrin α2β1 high/CD44v6 high | 6 | 13 | 0.000 | 9 | 0.001 | 19 | 0.025 |
| Remaining combinations # | 36 | 27 | 22 | 42 | |||
| Integrin α2β1 high/Integrin αvβ3 high | 5 | 35 | 0.322 | 30 | 0.320 | nr | 0.162 |
| Remaining combinations # | 37 | 22 | 17 | 42 | |||
| Immune Infiltrate | Integrin α2β1 | ||||
|---|---|---|---|---|---|
| n | <20% | ≥20% | p# | ||
| CD3 stromal | 48 | 0.034 | |||
| Low | 13 | 14 | |||
| High | 17 | 4 | |||
| CD3 intratumoral | 48 | 0.017 | |||
| Low | 12 | 14 | |||
| High | 18 | 4 | |||
| CD8 stromal | 48 | 0.133 | |||
| Low | 14 | 13 | |||
| High | 16 | 5 | |||
| CD8 intratumoral | 48 | 0.363 | |||
| Low | 17 | 13 | |||
| High | 13 | 5 | |||
| PD-1 stromal | 48 | 0.049 | |||
| Low | 18 | 16 | |||
| High | 12 | 2 | |||
| PD-1 intratumoral | 48 | 0.002 | |||
| Low | 15 | 17 | |||
| High | 15 | 1 | |||
| PD-L1 positivity | 48 | 0.005 | |||
| No | 7 | 12 | |||
| Yes | 23 | 6 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dötzer, K.; Schlüter, F.; Koch, F.E.v.; Brambs, C.E.; Anthuber, S.; Frangini, S.; Czogalla, B.; Burges, A.; Werner, J.; Mahner, S.; et al. Integrin α2β1 Represents a Prognostic and Predictive Biomarker in Primary Ovarian Cancer. Biomedicines 2021, 9, 289. https://doi.org/10.3390/biomedicines9030289
Dötzer K, Schlüter F, Koch FEv, Brambs CE, Anthuber S, Frangini S, Czogalla B, Burges A, Werner J, Mahner S, et al. Integrin α2β1 Represents a Prognostic and Predictive Biomarker in Primary Ovarian Cancer. Biomedicines. 2021; 9(3):289. https://doi.org/10.3390/biomedicines9030289
Chicago/Turabian StyleDötzer, Katharina, Friederike Schlüter, Franz Edler von Koch, Christine E. Brambs, Sabine Anthuber, Sergio Frangini, Bastian Czogalla, Alexander Burges, Jens Werner, Sven Mahner, and et al. 2021. "Integrin α2β1 Represents a Prognostic and Predictive Biomarker in Primary Ovarian Cancer" Biomedicines 9, no. 3: 289. https://doi.org/10.3390/biomedicines9030289
APA StyleDötzer, K., Schlüter, F., Koch, F. E. v., Brambs, C. E., Anthuber, S., Frangini, S., Czogalla, B., Burges, A., Werner, J., Mahner, S., & Mayer, B. (2021). Integrin α2β1 Represents a Prognostic and Predictive Biomarker in Primary Ovarian Cancer. Biomedicines, 9(3), 289. https://doi.org/10.3390/biomedicines9030289