Evaluation of Ethnic Variations in Visceral, Subcutaneous, Intra-Pancreatic, and Intra-Hepatic Fat Depositions by Magnetic Resonance Imaging among New Zealanders
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Anthropometric Measurements
2.3. MRI Acquisition
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Differences between the Ethnic Groups
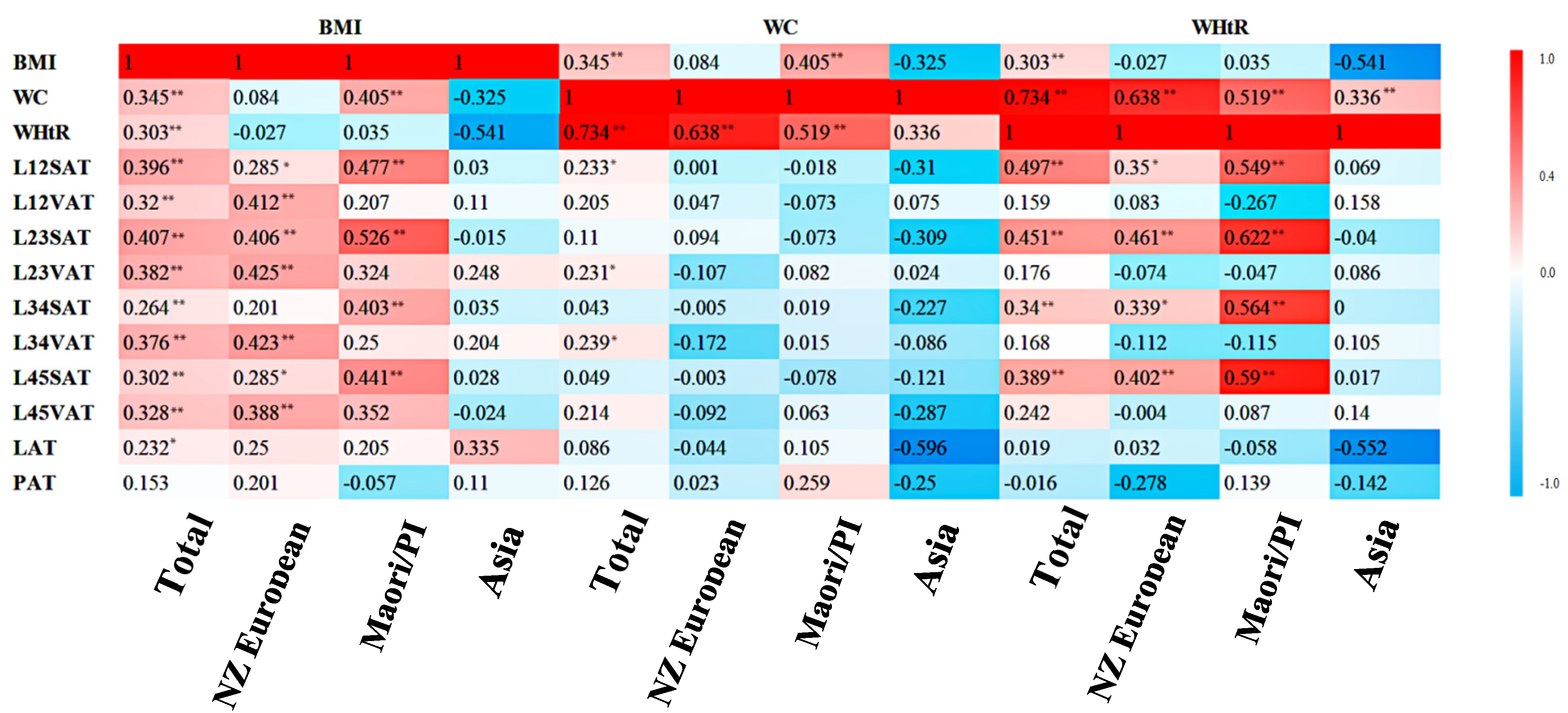
3.3. Correlations between Anthropometric Indices and MRI-Derived Fat Depositions across the Ethnicities
3.4. Usefulness of Measuring Fat Depositions at Different Levels
3.4.1. Body Mass Index
3.4.2. Waist Circumference
3.4.3. Waist to Height Ratio
3.5. Ethnicity-Specific Predictive Equations
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Singh, R.G.; Cervantes, A.; Kim, J.U.; Nguyen, N.N.; DeSouza, S.V.; Dokpuang, D.; Lu, J.; Petrov, M.S. Intrapancreatic fat deposition and visceral fat volume are associated with the presence of diabetes after acute pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G806–G815. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M. Subcutaneous and visceral adipose tissue: Structural and functional differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J. Am. Coll. Cardiol. 2014, 63, 2985–3023. [Google Scholar] [CrossRef] [PubMed]
- Gillies, N.; Pendharkar, S.A.; Asrani, V.M.; Mathew, J.; Windsor, J.A.; Petrov, M.S. Interleukin-6 is associated with chronic hyperglycemia and insulin resistance in patients after acute pancreatitis. Pancreatology 2016, 16, 748–755. [Google Scholar] [CrossRef] [PubMed]
- García Torres, D.; Castellanos González, M.F.; Cedeño Morales, R.; Benet Rodríguez, M.; Ramírez Arteaga, I. Adipose tissue as an endocrine gland. pathophysiological implications. Revista de Enfermedades no Transmisibles Finlay 2017, 7, 131–151. [Google Scholar]
- Singh, R.G.; Pendharkar, S.A.; Gillies, N.A.; Miranda-Soberanis, V.; Plank, L.D.; Petrov, M.S. Associations between circulating levels of adipocytokines and abdominal adiposity in patients after acute pancreatitis. Clin. Exp. Med. 2017, 17, 477–487. [Google Scholar] [CrossRef]
- Després, J.-P.; Lemieux, I. Abdominal obesity and metabolic syndrome. Nature 2006, 444, 881–887. [Google Scholar] [CrossRef]
- Ashwell, M.; Gunn, P.; Gibson, S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: Systematic review and meta-analysis. Obes. Rev. 2012, 13, 275–286. [Google Scholar] [CrossRef]
- Cheng, S.; Massaro, J.M.; Fox, C.S.; Larson, M.G.; Keyes, M.J.; McCabe, E.L.; Robins, S.J.; O’Donnell, C.J.; Hoffmann, U.; Jacques, P.F. Adiposity, cardiometabolic risk, and vitamin D status: The Framingham Heart Study. Diabetes 2010, 59, 242–248. [Google Scholar] [CrossRef]
- Catanzaro, R.; Cuffari, B.; Italia, A.; Marotta, F. Exploring the metabolic syndrome: Nonalcoholic fatty pancreas disease. World J. Gastroenterol. 2016, 22, 7660. [Google Scholar] [CrossRef]
- Coe, P.O.; Williams, S.R.; Morris, D.M.; Parkin, E.; Harvie, M.; Renehan, A.G.; O’Reilly, D.A. Development of MR quantified pancreatic fat deposition as a cancer risk biomarker. Pancreatology 2018, 18, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Sreedhar, U.L.; DeSouza, S.V.; Park, B.; Petrov, M.S. A Systematic Review of Intra-pancreatic Fat Deposition and Pancreatic Carcinogenesis. J. Gastrointest. Surg. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Petrov, M.S. Harnessing Analytic Morphomics for Early Detection of Pancreatic Cancer. Pancreas 2018, 47, 1051–1054. [Google Scholar] [CrossRef] [PubMed]
- Abate, N.; Burns, D.; Peshock, R.M.; Garg, A.; Grundy, S.M. Estimation of adipose tissue mass by magnetic resonance imaging: Validation against dissection in human cadavers. J. Lipid Res. 1994, 35, 1490–1496. [Google Scholar]
- Shuman, W.P.; Morris, L.; Leonetti, D.L.; Wahl, P.W.; Moceri, V.M.; Moss, A.A.; Fujimoto, W.Y. Abnormal body fat distribution detected by computed tomography in diabetic men. Investig. Radiol. 1986, 21, 483–487. [Google Scholar] [CrossRef]
- Seidell, J.C.; Oosterlee, A.; Thijssen, M.; Burema, J.; Deurenberg, P.; Hautvast, J.; Ruijs, J. Assessment of intra-abdominal and subcutaneous abdominal fat: Relation between anthropometry and computed tomography. Am. J. Clin. Nutr. 1987, 45, 7–13. [Google Scholar] [CrossRef]
- O’Connor, M.; Ryan, J.; Foley, S. Best single-slice location to measure visceral adipose tissue on paediatric CT scans and the relationship between anthropometric measurements, gender and VAT volume in children. Br. J. Radiol. 2015, 88, 20140711. [Google Scholar] [CrossRef]
- Irlbeck, T.; Massaro, J.; Bamberg, F.; O’donnell, C.; Hoffmann, U.; Fox, C. Association between single-slice measurements of visceral and abdominal subcutaneous adipose tissue with volumetric measurements: The Framingham Heart Study. Int. J. Obes. 2010, 34, 781–787. [Google Scholar] [CrossRef]
- Shen, W.; Punyanitya, M.; Chen, J.; Gallagher, D.; Albu, J.; Pi-Sunyer, X.; Lewis, C.; Grunfeld, C.; Heymsfield, S.; Heshka, S. Visceral adipose tissue: Relationships between single slice areas at different locations and obesity-related health risks. Int. J. Obes. 2007, 31, 763–769. [Google Scholar] [CrossRef]
- Kuk, J.L.; Church, T.S.; Blair, S.N.; Ross, R. Does measurement site for visceral and abdominal subcutaneous adipose tissue alter associations with the metabolic syndrome? Diabetes Care 2006, 29, 679–684. [Google Scholar] [CrossRef]
- Lovejoy, J.C.; Jacques, A.; Klemperer, M.; Tulley, R. Abdominal fat distribution and metabolic risk factors: Effects of race. Metabolism 1996, 45, 1119–1124. [Google Scholar] [CrossRef]
- Misra, A.; Wasir, J.S.; Vikram, N.K. Waist circumference criteria for the diagnosis of abdominal obesity are not applicable uniformly to all populations and ethnic groups. Nutrition 2005, 21, 969–976. [Google Scholar] [CrossRef]
- Kadowaki, T.; Sekikawa, A.; Murata, K.; Maegawa, H.; Takamiya, T.; Okamura, T.; El-Saed, A.; Miyamatsu, N.; Edmundowicz, D.; Kita, Y. Japanese men have larger areas of visceral adipose tissue than Caucasian men in the same levels of waist circumference in a population-based study. Int. J. Obes. 2006, 30, 1163–1165. [Google Scholar] [CrossRef] [PubMed]
- Fedorov, A.; Beichel, R.; Kalpathy-Cramer, J.; Finet, J.; Fillion-Robin, J.-C.; Pujol, S.; Bauer, C.; Jennings, D.; Fennessy, F.; Sonka, M. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn. Reson. Imaging 2012, 30, 1323–1341. [Google Scholar] [CrossRef]
- Fain, J.N.; Madan, A.K.; Hiler, M.L.; Cheema, P.; Bahouth, S.W. Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 2004, 145, 2273–2282. [Google Scholar] [CrossRef] [PubMed]
- Freedland, E.S. Role of a critical visceral adipose tissue threshold (CVATT) in metabolic syndrome: Implications for controlling dietary carbohydrates: A review. Nutr. Metab. 2004, 1, 1–24. [Google Scholar] [CrossRef]
- Preis, S.R.; Massaro, J.M.; Robins, S.J.; Hoffmann, U.; Vasan, R.S.; Irlbeck, T.; Meigs, J.B.; Sutherland, P.; D’Agostino Sr, R.B.; O’donnell, C.J. Abdominal subcutaneous and visceral adipose tissue and insulin resistance in the Framingham heart study. Obesity 2010, 18, 2191–2198. [Google Scholar] [CrossRef]
- Heni, M.; Machann, J.; Staiger, H.; Schwenzer, N.F.; Peter, A.; Schick, F.; Claussen, C.D.; Stefan, N.; Häring, H.U.; Fritsche, A. Pancreatic fat is negatively associated with insulin secretion in individuals with impaired fasting glucose and/or impaired glucose tolerance: A nuclear magnetic resonance study. Diabetes/Metab. Res. Rev. 2010, 26, 200–205. [Google Scholar] [CrossRef]
- Ozturk, K.; Dogan, T.; Celikkanat, S.; Ozen, A.; Demirci, H.; Kurt, O.; Turker, T.; Yilmaz, Y.; Uygun, A. The association of fatty pancreas with subclinical atherosclerosis in nonalcoholic fatty liver disease. Eur. J. Gastroenterol. Hepatol. 2018, 30, 411–417. [Google Scholar] [CrossRef]
- Cho, J.-Y.; Ye, S.-Y.; Kim, D.-H. The study on risk factor of metabolic diseases in pancreatic steatosis. J. Radiol. Sci. Technol. 2016, 39, 81–88. [Google Scholar] [CrossRef]
- Heid, I.M.; Jackson, A.U.; Randall, J.C.; Winkler, T.W.; Qi, L.; Steinthorsdottir, V.; Thorleifsson, G.; Zillikens, M.C.; Speliotes, E.K.; Mägi, R. Meta-analysis identifies 13 new loci associated with waist-hip ratio and reveals sexual dimorphism in the genetic basis of fat distribution. Nat. Genet. 2010, 42, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Schweiger, M.; Lass, A.; Zimmermann, R.; Eichmann, T.O.; Zechner, R. Neutral lipid storage disease: Genetic disorders caused by mutations in adipose triglyceride lipase/PNPLA2 or CGI-58/ABHD5. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E289–E296. [Google Scholar] [CrossRef] [PubMed]
- Cardel, M.; Higgins, P.; Willig, A.; Keita, A.; Casazza, K.; Gower, B.; Fernandez, J. African genetic admixture is associated with body composition and fat distribution in a cross-sectional study of children. Int. J. Obes. 2011, 35, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, X.; Peng, D.; Zhang, R.; Sun, X.; Chen, M.; Yan, J.; Wang, S.; Yan, D.; He, Z. Effects of obesity related genetic variations on visceral and subcutaneous fat distribution in a Chinese population. Sci. Rep. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- Savva, S.; Tornaritis, M.; Savva, M.; Kourides, Y.; Panagi, A.; Silikiotou, N.; Georgiou, C.; Kafatos, A. Waist circumference and waist-to-height ratio are better predictors of cardiovascular disease risk factors in children than body mass index. Int. J. Obes. 2000, 24, 1453–1458. [Google Scholar] [CrossRef]
- Camhi, S.M.; Bray, G.A.; Bouchard, C.; Greenway, F.L.; Johnson, W.D.; Newton, R.L.; Ravussin, E.; Ryan, D.H.; Smith, S.R.; Katzmarzyk, P.T. The relationship of waist circumference and BMI to visceral, subcutaneous, and total body fat: Sex and race differences. Obesity 2011, 19, 402–408. [Google Scholar] [CrossRef]
- Araneta, M.R.G.; Barrett-Connor, E. Ethnic differences in visceral adipose tissue and type 2 diabetes: Filipino, African-American, and white women. Obes. Res. 2005, 13, 1458–1465. [Google Scholar] [CrossRef]
- van Geenen, E.-J.M.; Smits, M.M.; Schreuder, T.C.; van der Peet, D.L.; Bloemena, E.; Mulder, C.J. Nonalcoholic fatty liver disease is related to nonalcoholic fatty pancreas disease. Pancreas 2010, 39, 1185–1190. [Google Scholar] [CrossRef]
- Smits, M.M.; Van Geenen, E.J. The clinical significance of pancreatic steatosis. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 169. [Google Scholar] [CrossRef]
- Garcia, B.C.; Wu, Y.; Nordbeck, E.B.; Musovic, S.; Olofsson, C.S.; Rorsman, P.; Asterholm, I.W. Pancreatic Adipose Tissue in Diet-Induced Type 2 Diabetes. Diabetes 2018, 67 (Suppl. 1), 2431. [Google Scholar] [CrossRef]
- Meyer, K.A.; Neeley, C.K.; Baker, N.A.; Washabaugh, A.R.; Flesher, C.G.; Nelson, B.S.; Frankel, T.L.; Lumeng, C.N.; Lyssiotis, C.A.; Wynn, M.L. Adipocytes promote pancreatic cancer cell proliferation via glutamine transfer. Biochem. Biophys. Rep. 2016, 7, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Michael, K.Y.; Ma, J.; Fisher, J.; Kreisberg, J.F.; Raphael, B.J.; Ideker, T. Visible machine learning for biomedicine. Cell 2018, 173, 1562–1565. [Google Scholar]
- Mamoshina, P.; Vieira, A.; Putin, E.; Zhavoronkov, A. Applications of deep learning in biomedicine. Mol. Pharm. 2016, 13, 1445–1454. [Google Scholar] [CrossRef]
- Erickson, B.J.; Korfiatis, P.; Akkus, Z.; Kline, T.L. Machine learning for medical imaging. Radiographics 2017, 37, 505–515. [Google Scholar] [CrossRef]
- Merriman, T.R.; Wilcox, P.L. Cardio-metabolic disease genetic risk factors among Māori and Pacific Island people in Aotearoa New Zealand: Current state of knowledge and future directions. Ann. Hum. Biol. 2018, 45, 202–214. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Total | NZ European | Māori/Pl | Asian | p-Value 1 |
|---|---|---|---|---|---|
| No. of participants | 104 | 53 | 30 | 21 | |
| Age (years) | 53.0 ± 1.6 | 56 ± 2.4 | 57.7 ± 2.0 | 39.1 ± 2.6 | 0.0001 *** |
| Sex | 0.092 | ||||
| Male | 66 | 31 | 21 | 14 | |
| Female | 38 | 22 | 9 | 7 | |
| Height (cm) | 171.4 ± 1.1 | 172.4 ± 1.2 | 172.5 ± 2.2 | 167.2 ± 2.1 | 0.109 |
| Weight (kg) | 82.6 ± 1.9 | 80.7 ± 2.1 | 93.7 ± 3.6 | 68.4 ± 4.0 | 0.0002 *** |
| WC (cm) | 100.0 ± 0.7 | 96.6 ± 0.6 | 109.0 ± 1.2 | 95.7 ± 0.5 | 0.0002 *** |
| BMI (kg/m2) | 28.1 ± 0.6 | 27.1 ± 0.6 | 31.7 ± 1.3 | 25.4 ± 1.0 | 0.0001 *** |
| WHtR | 58.6 ± 0.6 | 56.2 ± 0.5 | 63.6 ± 1.2 | 57.4 ± 0.7 | 0.0001 *** |
| L12SAT | 22.2 ± 1.0 | 20.3 ± 1.3 | 26.3 ± 2.3 | 21.1 ± 1.8 | 0.035 ** |
| L12VAT | 31.7 ± 1.0 | 29.2 ± 1.2 | 35.4 ± 1.4 | 32.6 ± 1.8 | 0.019 *** |
| L23SAT | 26.7 ± 1.1 | 25.1 ± 1.5 | 29.3 ± 2.4 | 27.0 ± 2.2 | 0.280 |
| L23VAT | 32.4 ± 1.0 | 29.2 ± 1.5 | 36.8 ± 1.7 | 34.0 ± 1.9 | 0.004 *** |
| L34SAT | 32.7 ± 1.1 | 31.1 ± 1.5 | 34.5 ± 2.3 | 34.0 ± 2.5 | 0.360 |
| L34VAT | 34.0 ± 1.1 | 31.8 ± 1.6 | 39.3 ± 2.4 | 39.0 ± 2.4 | 0.014 *** |
| L45SAT | 37.9 ± 1.2 | 36.6 ± 1.6 | 39.3 ± 2.4 | 39.1 ± 2.4 | 0.549 |
| L45VAT | 32.4 ± 1.0 | 30.4 ± 1.4 | 36.0 ± 1.6 | 32.6 ± 2.2 | 0.039 ** |
| SATA | 30.0 ± 1.1 | 28.5 ± 1.4 | 32.4 ± 2.2 | 30.3 ± 2.1 | 0.291 |
| VATA | 32.6 ± 0.9 | 30.2 ± 1.4 | 36.8 ± 1.4 | 32.9 ± 1.9 | 0.008 *** |
| LAT | 9.9 ± 1.0 | 8.2 ± 1.0 | 11.0 ± 1.9 | 13.5 ± 3.5 | 0.125 |
| PAT | 8.2 ± 0.4 | 7.5 ± 0.5 | 9.4 ± 0.6 | 8.0 ± 0.9 | 0.062 |
| Anthropometric Indices and Fat Deposition | NZ European vs. Māori/Pl | NZ European vs. Asian | Māori/Pl vs. Asian |
|---|---|---|---|
| BMI | 3.086 *** | 1.471 | 3.0478 *** |
| WC | 10.639 *** | 2.208 ** | 12.539 *** |
| WHtR | 6.796 *** | 0.785 | 5.096 *** |
| L12SAT | 2.362 *** | 0.353 | 1.757 |
| L12VAT | 2.874 *** | 1.287 | 1.101 |
| L23SAT | 1.684 | 0.651 | 0.864 |
| L23VAT | 3.07 *** | 1.767 | 1.028 |
| L34SAT | 0.199 | 1.043 | 0.278 |
| L34VAT | 1.478 *** | 0.224 | 2.227 ** |
| L45SAT | 1.184 | 0.818 | 0.256 |
| L45VAT | 2.431 *** | 0.860 | 1.148 |
| LAT | 1.565 | 1.454 * | 0.541 |
| PAT | 2.256 ** | 0.546 | 1.243 |
| Ethnicity Group | L12SAT | L23SAT | L34SAT | L45SAT | L12VAT | L23VAT | L34VAT | L45VAT |
|---|---|---|---|---|---|---|---|---|
| Unadjusted | ||||||||
| Total | 0.225 *** | 0.209 *** | 0.137 *** | 0.147 *** | 0.196 *** | 0.218 *** | 0.202 *** | 0.200 *** |
| (0.052) | (0.047) | (0.050) | (0.047) | (0.057) | (0.052) | (0.049) | (0.057) | |
| NZ European | 0.135 *** | 0.164 *** | 0.085 | 0.109 *** | 0.176 *** | 0.177 *** | 0.163 *** | 0.178 *** |
| (0.064) | (0.052) | (0.058) | (0.051) | (0.055) | (0.053) | (0.049) | (0.059) | |
| Māori/PI | 0.283 *** | 0.297 *** | 0.240 *** | 0.247 *** | 0.185 | 0.215 * | 0.203 | 0.282 * |
| (0.098) | (0.090) | (0.102) | (0.095) | (0.174) | (0.144) | (0.153) | (0.153) | |
| Asian | 0.018 | −0.007 | 0.015 | 0.012 | 0.062 | 0.135 | 0.091 | −0.011 |
| (0.135) | (0.110) | (0.096) | (0.097) | (0.127) | (0.121) | (0.101) | (0.108) | |
| Adjusted for Age and Sex | ||||||||
| Total | 0.310 *** | 0.203 *** | 0.337 *** | 0.229 *** | 0.207 *** | 0.237 *** | 0.201 *** | 0.237 *** |
| (0.187) | (0.239) | (0.085) | (0.124) | (0.089) | (0.135) | (0.085) | (0.139) | |
| NZ European | 0.221 *** | 0.296 *** | 0.165 ** | 0.226 *** | 0.190 *** | 0.204 *** | 0.204 *** | 0.223 *** |
| (0.108) | (0.300) | (0.056) | (0.165) | (0.126) | (0.146) | (0.115) | (0.169) | |
| Māori/PI | 0.209 | 0.318 | 0.077 | 0.131 | 0.315* | 0.271 ** | 0.275 * | 0.301 ** |
| (0.149) | (0.196) | (0.118) | (0.137) | (0.228) | (0.238) | (0.223) | (0.243) | |
| Asian | 0.073 | 0.038 | 0.062 | 0.063 | 0.064 | 0.174 | 0.105 | 0.004 |
| (−0.111) | (−0.119) | (−0.111) | (−0.112) | (−0.110) | (−0.031) | (−0.066) | (−0.123) | |
| Ethnicity Group | L12SAT | L23SAT | L34SAT | L45SAT | L12VAT | L23VAT | L34VAT | L45VAT |
|---|---|---|---|---|---|---|---|---|
| Unadjusted | ||||||||
| Total | 0.133 *** | 0.040 | 0.033 | 0.029 | 0.120 * | 0.111 * | 0.149 * | 0.0100 |
| (0.065) | (0.060) | (0.061) | (0.057) | (0.070) | (0.065) | (0.061) | (0.070) | |
| NZ European | 0.047 | 0.012 | 0.017 | 0.043 | 0.008 | 0.002 | 0.012 | −0.065 |
| (0.062) | (0.053) | (0.055) | (0.049) | (0.056) | (0.054) | (0.050) | (0.059) | |
| Māori/PI | −0.050 | −0.088 | −0.059 | −0.077 | −0.052 | −0.076 | 0.015 | 0.025 |
| (0.079) | (0.074) | (0.079) | (0.074) | (0.126) | (0.107) | (0.112) | (0.115) | |
| Asian | 0.008 | −0.004 | 0.012 | 0.024 | −0.012 | −0.028 | −0.005 | 0.016 |
| (0.056) | (0.046) | (0.040) | (0.040) | (0.053) | (0.052) | (0.043) | (0.045) | |
| Adjusted for Age and Sex | ||||||||
| Total | 0.196 *** | 0.153 *** | 0.115 ** | 0.140 *** | 0.083 | 0.073 | 0.096 * | 0.082 |
| (0.230) | (0.193) | (0.164) | (0.188) | (0.147) | (0.135) | (0.158) | (0.146) | |
| NZ European | 0.073 | 0.055 | 0.026 | 0.077 | 0.046 | 0.029 | 0.059 | −0.025 |
| (0.231) | (0.225) | (0.213) | (0.240) | (0.223) | (0.215) | (0.228) | (0.213) | |
| Māori/PI | 0.117 | 0.198 | 0.120 | 0.149 | −0.118 | −0.093 | −0.061 | 0.029 |
| (0.244) | (0.288) | (0.255) | (0.290) | (0.253) | (0.250) | (0.230) | (0.221) | |
| Asian | 0.034 | −0.06 | 0.016 | 0.020 | 0.066 | 0.017 | 0.041 | 0.068 |
| (0.300) | (0.297) | (−0.111) | (0.296) | (0.319) | (0.297) | (0.310) | (0.332) | |
| Ethnicity Group | L12SAT | L23SAT | L34SAT | L45SAT | L12VAT | L23VAT | L34VAT | L45VAT |
|---|---|---|---|---|---|---|---|---|
| Unadjusted | ||||||||
| Total | 0.235 *** | 0.187 *** | 0.159 *** | 0.167 *** | 0.069 | 0.063 | 0.078 * | 0.098 * |
| (0.044) | (0.041) | (0.043) | (0.040) | (0.053) | (0.049) | (0.046) | (0.052) | |
| NZ European | 0.0170 *** | 0.148 *** | 0.134 *** | 0.158 *** | 0.028 | 0.001 | 0.007 | −0.015 |
| (0.055) | (0.047) | (0.050) | (0.043) | (0.054) | (0.052) | (0.048) | (0.058) | |
| Māori/PI | 0.200 *** | 0.205 *** | 0.187 *** | 0.194 *** | −0.172 | −0.092 | −0.059 | 0.056 |
| (0.066) | (0.061) | (0.068) | (0.063) | (0.117) | (0.102) | (0.108) | (00.110) | |
| Asian | 0.087 | 0.027 | 0.035 | 0.035 | 0.042 | 0.009 | 0.040 | 0.094 |
| (0.102) | (0.084) | (0.074) | (0.074) | (0.098) | (0.096) | (0.079) | (0.081) | |
| Adjusted for Age and Sex | ||||||||
| Total | 0.150 ** | 0.160 ** | 0.149 ** | 0.263 *** | 0.078 | 0.070 | 0.110 | 0.059 |
| (0.057) | (0.058) | (0.054) | (0.122) | (0.028) | (0.027) | (0.041) | (0.023) | |
| NZ European | 0.091 | 0.034 | 0.038 | 0.086 | 0.032 | 0.031 | 0.059 | −0.054 |
| (−0.017) | (−0.041) | (−0.040) | (−0.011) | (−0.041) | (−0.041) | (−0.229) | (−0.033) | |
| Māori/PI | 0.120 | −0.016 | 0.073 | −0.001 | −0.109 | −0.091 | −0.029 | 0.003 |
| (0.007) | (−0.018) | (−0.006) | (−0.018) | (0.010) | (0.010) | (−0.016) | (−0.018) | |
| Asian | −0.024 | −0.052 | −0.009 | 0.017 | −0.002 | −0.023 | 0 | 0.017 |
| (−0.108) | (−0.076) | (−0.114) | (−0.111) | (−0.115) | (−0.106) | (−0.115) | (−0.108) | |
| Correlation Factors | VIF | Equation |
|---|---|---|
| Age, years | 1.061 * | L12VAT = 0.405 (age) − 0.565 (sex) + 0.347 (BMI) L23VAT = 0.460 (age) − 0.603 (sex) + 0.373 (BMI) L34VAT = 0.571 (age) − 0.612 (sex) + 0.348 (BMI) L45VAT = 0.464 (age) − 0.558 (sex) + 0.346 (BMI) |
| Sex | 2.987 * | |
| BMI, kg/m2 | 1.679 * | |
| WC, cm | 1.953 | |
| WHtR | 3.306 |
| Correlation Factors | VIF | Equation |
|---|---|---|
| Age, years | 1.057 | L23VAT = 0.615 (BMI) − 0.34 (sex) L34VAT = 0.518 (BMI) − 0.27 (sex) L45VAT = 0.642 (BMI) − 0.31 (sex) |
| Sex | 2.987 * | |
| BMI, kg/m2 | 1.679 * | |
| WC, cm | 1.953 | |
| WHtR | 3.306 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.Z.; Dokpuang, D.; Nemati, R.; He, K.H.; Zheng, A.B.; Petrov, M.S.; Lu, J. Evaluation of Ethnic Variations in Visceral, Subcutaneous, Intra-Pancreatic, and Intra-Hepatic Fat Depositions by Magnetic Resonance Imaging among New Zealanders. Biomedicines 2020, 8, 174. https://doi.org/10.3390/biomedicines8060174
Yang JZ, Dokpuang D, Nemati R, He KH, Zheng AB, Petrov MS, Lu J. Evaluation of Ethnic Variations in Visceral, Subcutaneous, Intra-Pancreatic, and Intra-Hepatic Fat Depositions by Magnetic Resonance Imaging among New Zealanders. Biomedicines. 2020; 8(6):174. https://doi.org/10.3390/biomedicines8060174
Chicago/Turabian StyleYang, John Zhiyong, Dech Dokpuang, Reza Nemati, Kevin Haokun He, Andy Baige Zheng, Maxim S. Petrov, and Jun Lu. 2020. "Evaluation of Ethnic Variations in Visceral, Subcutaneous, Intra-Pancreatic, and Intra-Hepatic Fat Depositions by Magnetic Resonance Imaging among New Zealanders" Biomedicines 8, no. 6: 174. https://doi.org/10.3390/biomedicines8060174
APA StyleYang, J. Z., Dokpuang, D., Nemati, R., He, K. H., Zheng, A. B., Petrov, M. S., & Lu, J. (2020). Evaluation of Ethnic Variations in Visceral, Subcutaneous, Intra-Pancreatic, and Intra-Hepatic Fat Depositions by Magnetic Resonance Imaging among New Zealanders. Biomedicines, 8(6), 174. https://doi.org/10.3390/biomedicines8060174







