Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies
Abstract
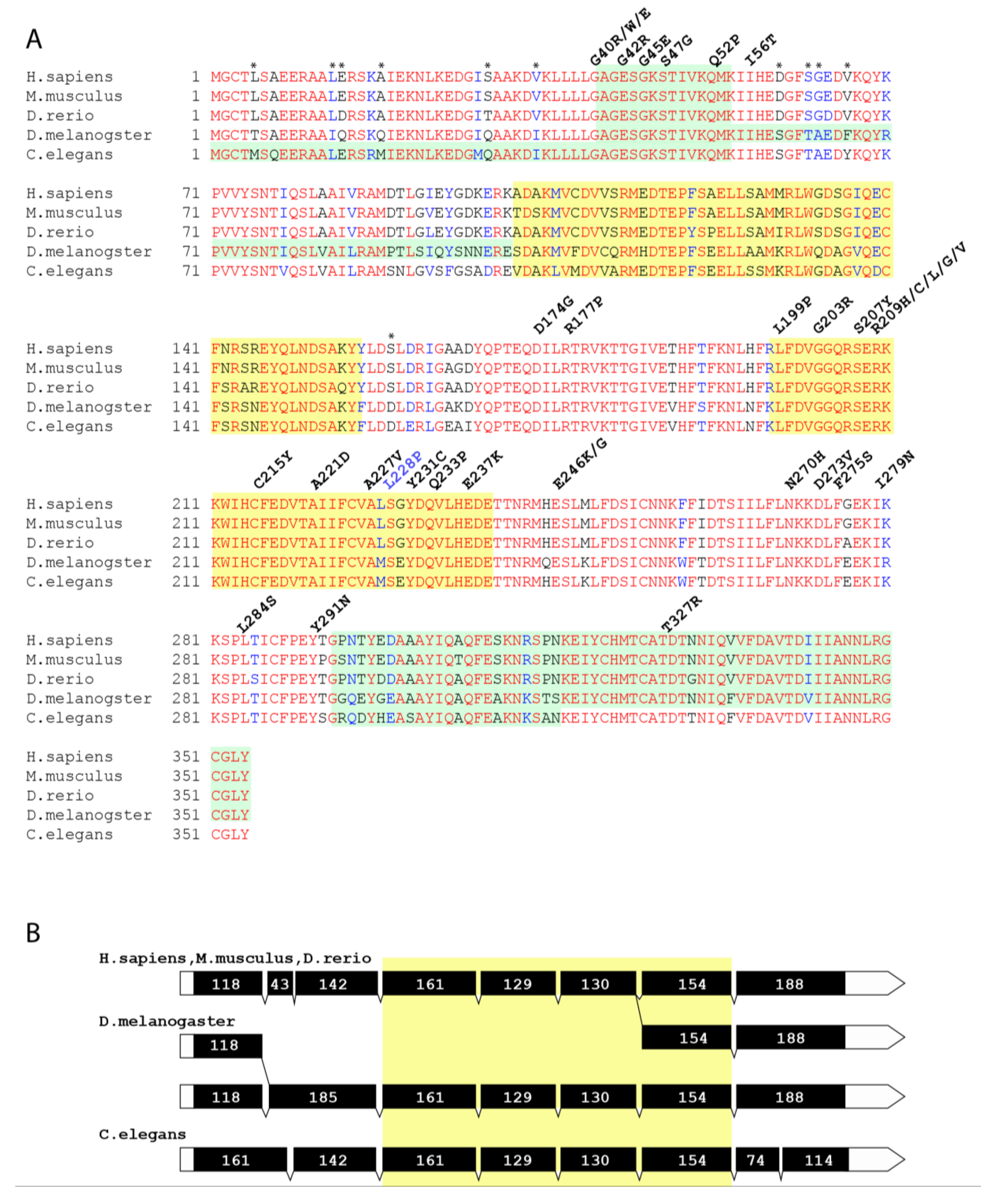
1. Introduction
2. Materials and Methods
2.1. Plasmids
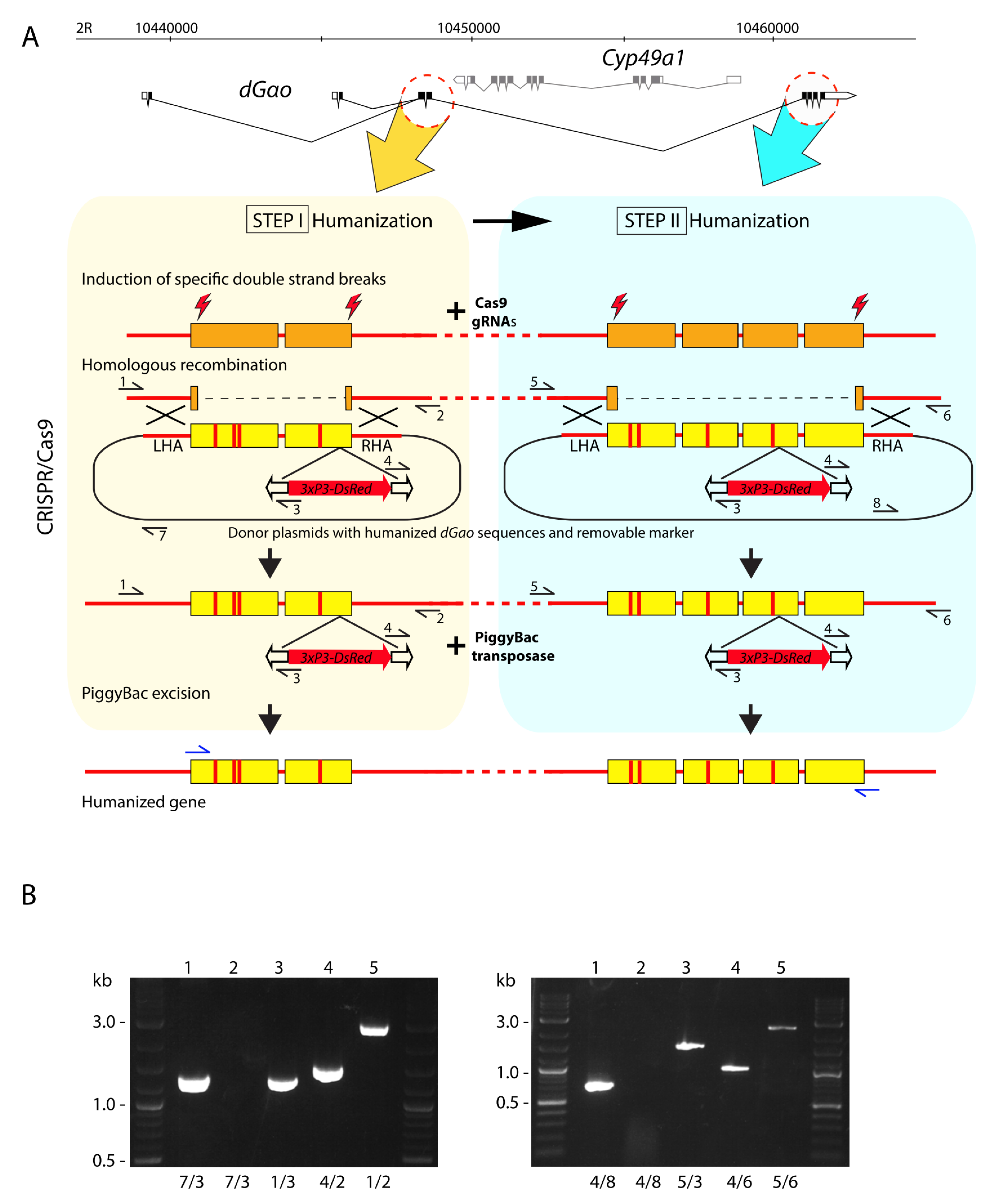
2.1.1. Donor Plasmid pLdhGao23R for the First Round of Humanization dGαo
2.1.2. Donor Plasmid for the Second Round of Humanization dGαo
2.1.3. Plasmids Providing Expression of gRNAs under the Control of the Drosophila U6:3 Promoter
2.2. Flies, Germline Transformation
2.3. Molecular Analysis
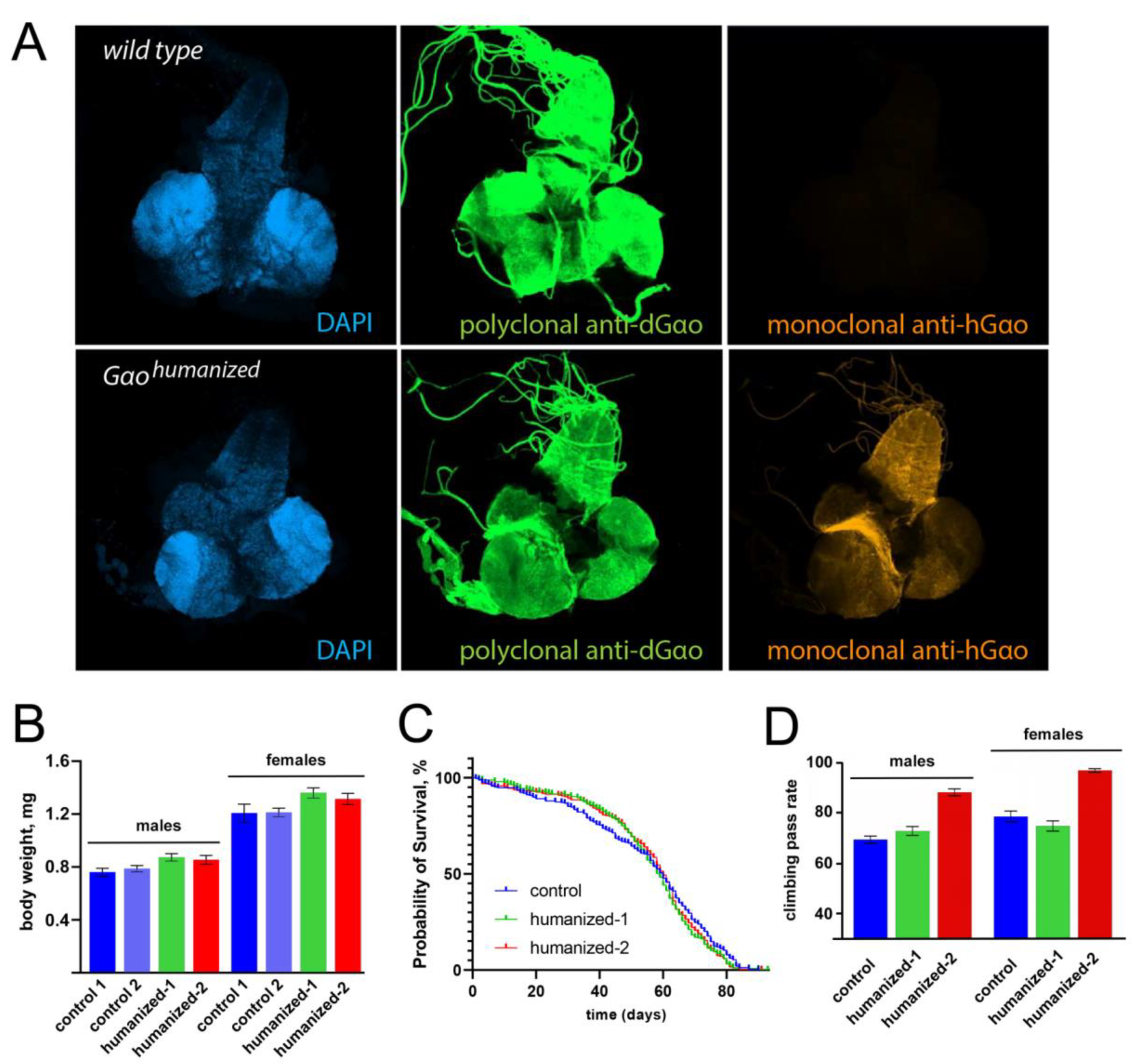
2.4. Immunochemistry and Microscopy
2.5. Behavioural Assays
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Staley, K. Molecular mechanisms of epilepsy. Nat. Neurosci. 2015, 18, 367–372. [Google Scholar] [CrossRef]
- Krook-Magnuson, E.; Soltesz, I. Beyond the hammer and the scalpel: Selective circuit control for the epilepsies. Nat. Neurosci. 2015, 18, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Ran, X.; Li, J.; Shao, Q.; Chen, H.; Lin, Z.; Sun, Z.S.; Wu, J. EpilepsyGene: A genetic resource for genes and mutations related to epilepsy. Nucleic Acids Res. 2015, 43, D893–D899. [Google Scholar] [CrossRef] [PubMed]
- Noebels, J. Pathway-driven discovery of epilepsy genes. Nat. Neurosci. 2015, 18, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Grone, B.P.; Baraban, S.C. Animal models in epilepsy research: Legacies and new directions. Nat. Neurosci. 2015, 18, 339–343. [Google Scholar] [CrossRef]
- McTague, A.; Howell, K.B.; Cross, J.H.; Kurian, M.A.; Scheffer, I.E. The genetic landscape of the epileptic encephalopathies of infancy and childhood. Lancet Neurol. 2016, 15, 304–316. [Google Scholar] [CrossRef]
- Nakamura, K.; Kodera, H.; Akita, T.; Shiina, M.; Kato, M.; Hoshino, H.; Terashima, H.; Osaka, H.; Nakamura, S.; Tohyama, J.; et al. De Novo mutations in GNAO1, encoding a Galphao subunit of heterotrimeric G proteins, cause epileptic encephalopathy. Am. J. Hum. Genet. 2013, 93, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Schirinzi, T.; Garone, G.; Travaglini, L.; Vasco, G.; Galosi, S.; Rios, L.; Castiglioni, C.; Barassi, C.; Battaglia, D.; Gambardella, M.L.; et al. Phenomenology and clinical course of movement disorder in GNAO1 variants: Results from an analytical review. Parkinsonism. Relat. Disord. 2019, 61, 19–25. [Google Scholar] [CrossRef]
- Menke, L.A.; Engelen, M.; Alders, M.; Odekerken, V.J.; Baas, F.; Cobben, J.M. Recurrent GNAO1 Mutations Associated With Developmental Delay and a Movement Disorder. J. Child. Neurol. 2016, 31, 1598–1601. [Google Scholar] [CrossRef]
- Schorling, D.C.; Dietel, T.; Evers, C.; Hinderhofer, K.; Korinthenberg, R.; Ezzo, D.; Bonnemann, C.G.; Kirschner, J. Expanding Phenotype of De Novo Mutations in GNAO1: Four New Cases and Review of Literature. Neuropediatrics 2017. [Google Scholar] [CrossRef]
- Talvik, I.; Moller, R.S.; Vaher, M.; Vaher, U.; Larsen, L.H.; Dahl, H.A.; Ilves, P.; Talvik, T. Clinical Phenotype of De Novo GNAO1 Mutation: Case Report and Review of Literature. Child. Neurol. Open. 2015, 2, 2329048X15583717. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, M.S.; Jackson, V.E.; Scerri, T.S.; Van Reyk, O.; Coleman, M.; Braden, R.O.; Turner, S.; Rigbye, K.A.; Boys, A.; Barton, S.; et al. Severe childhood speech disorder: Gene discovery highlights transcriptional dysregulation. Neurology 2020, 94, e2148–e2167. [Google Scholar] [CrossRef] [PubMed]
- Fredriksson, R.; Schioth, H.B. The repertoire of G-protein-coupled receptors in fully sequenced genomes. Mol. Pharmacol. 2005, 67, 1414–1425. [Google Scholar] [CrossRef] [PubMed]
- Milligan, G.; Kostenis, E. Heterotrimeric G-proteins: A short history. Br. J. Pharmacol. 2006, 147 (Suppl. 1), S46–S55. [Google Scholar] [CrossRef]
- Wettschureck, N.; Offermanns, S. Mammalian G proteins and their cell type specific functions. Physiol. Rev. 2005, 85, 1159–1204. [Google Scholar] [CrossRef]
- Sternweis, P.C.; Robishaw, J.D. Isolation of two proteins with high affinity for guanine nucleotides from membranes of bovine brain. J. Biol. Chem. 1984, 259, 13806–13813. [Google Scholar]
- Strathmann, M.; Simon, M.I. G protein diversity: A distinct class of alpha subunits is present in vertebrates and invertebrates. Proc. Natl. Acad. Sci. USA 1990, 87, 9113–9117. [Google Scholar] [CrossRef]
- Wolfgang, W.J.; Quan, F.; Goldsmith, P.; Unson, C.; Spiegel, A.; Forte, M. Immunolocalization of G protein alpha-subunits in the Drosophila CNS. J. Neurosci. 1990, 10, 1014–1024. [Google Scholar] [CrossRef]
- Greif, G.J.; Sodickson, D.L.; Bean, B.P.; Neer, E.J.; Mende, U. Altered regulation of potassium and calcium channels by GABA(B) and adenosine receptors in hippocampal neurons from mice lacking Galpha(o). J. Neurophysiol. 2000, 83, 1010–1018. [Google Scholar] [CrossRef][Green Version]
- Jiang, M.; Gold, M.S.; Boulay, G.; Spicher, K.; Peyton, M.; Brabet, P.; Srinivasan, Y.; Rudolph, U.; Ellison, G.; Birnbaumer, L. Multiple neurological abnormalities in mice deficient in the G protein Go. Proc. Natl. Acad. Sci. USA 1998, 95, 3269–3274. [Google Scholar] [CrossRef]
- Campbell, V.; Berrow, N.; Dolphin, A.C. GABAB receptor modulation of Ca2+ currents in rat sensory neurones by the G protein G(0): Antisense oligonucleotide studies. J. Physiol. 1993, 470, 1–11. [Google Scholar] [CrossRef]
- Strathmann, M.; Wilkie, T.M.; Simon, M.I. Alternative splicing produces transcripts encoding two forms of the alpha subunit of GTP-binding protein Go. Proc. Natl. Acad. Sci. USA 1990, 87, 6477–6481. [Google Scholar] [CrossRef]
- Fremion, F.; Astier, M.; Zaffran, S.; Guillen, A.; Homburger, V.; Semeriva, M. The heterotrimeric protein Go is required for the formation of heart epithelium in Drosophila. J. Cell Biol. 1999, 145, 1063–1076. [Google Scholar] [CrossRef]
- Valenzuela, D.; Han, X.; Mende, U.; Fankhauser, C.; Mashimo, H.; Huang, P.; Pfeffer, J.; Neer, E.J.; Fishman, M.C. G alpha(o) is necessary for muscarinic regulation of Ca2+ channels in mouse heart. Proc. Natl. Acad. Sci. USA 1997, 94, 1727–1732. [Google Scholar] [CrossRef]
- Egger-Adam, D.; Katanaev, V.L. Trimeric G protein-dependent signaling by Frizzled receptors in animal development. Front. Biosci. 2008, 13, 4740–4755. [Google Scholar] [CrossRef]
- Koval, A.; Purvanov, V.; Egger-Adam, D.; Katanaev, V.L. Yellow submarine of the Wnt/Frizzled signaling: Submerging from the G protein harbor to the targets. Biochem. Pharmacol. 2011, 82, 1311–1319. [Google Scholar] [CrossRef]
- Entschladen, F.; Zanker, K.S.; Powe, D.G. Heterotrimeric G protein signaling in cancer cells with regard to metastasis formation. Cell Cycle 2011, 10, 1086–1091. [Google Scholar] [CrossRef]
- Song, L.; Yu, B.; Yang, Y.; Liang, J.; Zhang, Y.; Ding, L.; Wang, T.; Wan, X.; Yang, X.; Tang, J.; et al. Identification of functional cooperative mutations of GNAO1 in human acute lymphoblastic leukemia. Blood 2020. [Google Scholar] [CrossRef]
- Milligan, G.; Streaty, R.A.; Gierschik, P.; Spiegel, A.M.; Klee, W.A. Development of opiate receptors and GTP-binding regulatory proteins in neonatal rat brain. J. Biol. Chem. 1987, 262, 8626–8630. [Google Scholar]
- Strittmatter, S.M.; Valenzuela, D.; Kennedy, T.E.; Neer, E.J.; Fishman, M.C. G0 is a major growth cone protein subject to regulation by GAP-43. Nature 1990, 344, 836–841. [Google Scholar] [CrossRef]
- Andreopoulos, S.; Li, P.P.; Warsh, J.J. Developmental expression of G alpha o and G alpha s isoforms in PC12 cells: Relationship to neurite outgrowth. Dev. Brain Res. 1995, 88, 30–36. [Google Scholar] [CrossRef]
- Asano, T.; Morishita, R.; Sano, M.; Kato, K. The GTP-binding proteins, Go and Gi2, of neural cloned cells and their changes during differentiation. J. Neurochem. 1989, 53, 1195–1198. [Google Scholar] [CrossRef]
- Zubiaur, M.; Neer, E.J. Nerve growth factor changes G protein levels and localization in PC12 cells. J. Neurosci. Res. 1993, 35, 207–217. [Google Scholar] [CrossRef]
- Strittmatter, S.M.; Fishman, M.C.; Zhu, X.P. Activated mutants of the alpha subunit of G(o) promote an increased number of neurites per cell. J. Neurosci. 1994, 14, 2327–2338. [Google Scholar] [CrossRef]
- Guillen, A.; Semeriva, M.; Bockaert, J.; Homburger, V. The transduction signalling protein Go during embryonic development of Drosophila melanogaster. Cell Signal. 1991, 3, 341–352. [Google Scholar] [CrossRef]
- Wolfgang, W.J.; Quan, F.; Thambi, N.; Forte, M. Restricted spatial and temporal expression of G-protein alpha subunits during Drosophila embryogenesis. Development 1991, 113, 527–538. [Google Scholar]
- Choi, J.M.; Kim, S.S.; Choi, C.I.; Cha, H.L.; Oh, H.H.; Ghil, S.; Lee, Y.D.; Birnbaumer, L.; Suh-Kim, H. Development of the main olfactory system and main olfactory epithelium-dependent male mating behavior are altered in Go-deficient mice. Proc. Natl. Acad. Sci. USA 2016. [Google Scholar] [CrossRef]
- Lee, M.S.; Jun, D.H.; Hwang, C.I.; Park, S.S.; Kang, J.J.; Park, H.S.; Kim, J.; Kim, J.H.; Seo, J.S.; Park, W.Y. Selection of neural differentiation-specific genes by comparing profiles of random differentiation. Stem Cells 2006, 24, 1946–1955. [Google Scholar] [CrossRef]
- Jiang, M.; Bajpayee, N.S. Molecular mechanisms of go signaling. Neurosignals 2009, 17, 23–41. [Google Scholar] [CrossRef]
- Luchtenborg, A.M.; Solis, G.P.; Egger-Adam, D.; Koval, A.; Lin, C.; Blanchard, M.G.; Kellenberger, S.; Katanaev, V.L. Heterotrimeric Go protein links Wnt-Frizzled signaling with ankyrins to regulate the neuronal microtubule cytoskeleton. Development 2014, 141, 3399–3409. [Google Scholar] [CrossRef]
- Lin, C.; Koval, A.; Tishchenko, S.; Gabdulkhakov, A.; Tin, U.; Solis, G.P.; Katanaev, V.L. Double suppression of the Galpha protein activity by RGS proteins. Mol. Cell 2014, 53, 663–671. [Google Scholar] [CrossRef]
- Lin, C.; Katanaev, V.L. Kermit interacts with galphao, vang, and motor proteins in Drosophila planar cell polarity. PLoS ONE 2013, 8, e76885. [Google Scholar] [CrossRef]
- Purvanov, V.; Koval, A.; Katanaev, V.L. A direct and functional interaction between Go and Rab5 during G protein-coupled receptor signaling. Sci. Signal. 2010, 3, ra65. [Google Scholar] [CrossRef]
- Egger-Adam, D.; Katanaev, V.L. The trimeric G protein Go inflicts a double impact on axin in the Wnt/frizzled signaling pathway. Dev. Dyn. 2010, 239, 168–183. [Google Scholar] [CrossRef]
- Kopein, D.; Katanaev, V.L. Drosophila GoLoco-protein pins is a target of Galpha(o)-mediated G protein-coupled receptor signaling. Mol. Biol. Cell 2009, 20, 3865–3877. [Google Scholar] [CrossRef]
- Solis, G.P.; Bilousov, O.; Koval, A.; Luchtenborg, A.M.; Lin, C.; Katanaev, V.L. Golgi-Resident Galphao Promotes Protrusive Membrane Dynamics. Cell 2017, 170, 939–955 e924. [Google Scholar] [CrossRef]
- Solis, G.P.; Valnohova, J.; Alvarez, C.; Katanaev, V.L. Local and substrate-specific S-palmitoylation determines subcellular localization of Gαo. bioRxiv 2020. [Google Scholar] [CrossRef]
- Feng, H.; Sjogren, B.; Karaj, B.; Shaw, V.; Gezer, A.; Neubig, R.R. Movement disorder in GNAO1 encephalopathy associated with gain-of-function mutations. Neurology 2017, 89, 762–770. [Google Scholar] [CrossRef]
- Bolus, H.; Crocker, K.; Boekhoff-Falk, G.; Chtarbanova, S. Modeling Neurodegenerative Disorders in Drosophila melanogaster. Int. J. Mol. Sci. 2020, 21, 3055. [Google Scholar] [CrossRef]
- Bellen, H.J.; Wangler, M.F.; Yamamoto, S. The fruit fly at the interface of diagnosis and pathogenic mechanisms of rare and common human diseases. Hum. Mol. Genet. 2019, 28, R207–R214. [Google Scholar] [CrossRef]
- Luchtenborg, A.M.; Katanaev, V.L. Lack of evidence of the interaction of the Abeta peptide with the Wnt signaling cascade in Drosophila models of Alzheimer’s disease. Mol. Brain 2014, 7, 81. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Katanaev, V.L.; Kryuchkov, M.V. The eye of Drosophila as a model system for studying intracellular signaling in ontogenesis and pathogenesis. Biochemistry (Mosc) 2011, 76, 1556–1581. [Google Scholar] [CrossRef] [PubMed]
- Katanaev, V.L.; Kryuchkov, M.; Averkov, V.; Nikolaeva, K.; Klimova, N.; Khaustov, S.; Solis, G. HumanaFly: High-throughput transgenesis and expression of breast cancer transcripts in Drosophila eye discovers the RPS12-Wingless signaling axis. Unpublished manuscript.
- Takai, A.; Yamaguchi, M.; Yoshida, H.; Chiyonobu, T. Investigating Developmental and Epileptic Encephalopathy Using Drosophila melanogaster. Int. J. Mol. Sci. 2020, 21, 6442. [Google Scholar] [CrossRef]
- Gratz, S.J.; Ukken, F.P.; Rubinstein, C.D.; Thiede, G.; Donohue, L.K.; Cummings, A.M.; O’Connor-Giles, K.M. Highly specific and efficient CRISPR/Cas9-catalyzed homology-directed repair in Drosophila. Genetics 2014, 196, 961–971. [Google Scholar] [CrossRef]
- Gloor, G.B.; Preston, C.R.; Johnson-Schlitz, D.M.; Nassif, N.A.; Phillis, R.W.; Benz, W.K.; Robertson, H.M.; Engels, W.R. Type I repressors of P element mobility. Genetics 1993, 135, 81–95. [Google Scholar]
- Ali, Y.O.; Escala, W.; Ruan, K.; Zhai, R.G. Assaying locomotor, learning, and memory deficits in Drosophila models of neurodegeneration. J. Vis. Exp. 2011. [Google Scholar] [CrossRef]
- Consortium, G.T. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef]
- Berghammer, A.J.; Klingler, M.; Wimmer, E.A. A universal marker for transgenic insects. Nature 1999, 402, 370–371. [Google Scholar] [CrossRef]
- Takano-Shimizu-Kouno, T.; Ohsako, T. Humanized Flies and Resources for Cross-Species Study. Adv. Exp. Med. Biol. 2018, 1076, 277–288. [Google Scholar] [CrossRef]
- Kachroo, A.H.; Laurent, J.M.; Yellman, C.M.; Meyer, A.G.; Wilke, C.O.; Marcotte, E.M. Evolution. Systematic humanization of yeast genes reveals conserved functions and genetic modularity. Science 2015, 348, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Wangler, M.F.; Yamamoto, S.; Chao, H.T.; Posey, J.E.; Westerfield, M.; Postlethwait, J.; Members of the Undiagnosed Diseases, N.; Hieter, P.; Boycott, K.M.; Campeau, P.M.; et al. Model Organisms Facilitate Rare Disease Diagnosis and Therapeutic Research. Genetics 2017, 207, 9–27. [Google Scholar] [CrossRef]
- Chang, J.C.; Morton, D.B. Drosophila lines with mutant and wild type human TDP-43 replacing the endogenous gene reveals phosphorylation and ubiquitination in mutant lines in the absence of viability or lifespan defects. PLoS ONE 2017, 12, e0180828. [Google Scholar] [CrossRef]
- Syrbe, S.; Hedrich, U.B.S.; Riesch, E.; Djemie, T.; Muller, S.; Moller, R.S.; Maher, B.; Hernandez-Hernandez, L.; Synofzik, M.; Caglayan, H.S.; et al. De novo loss- or gain-of-function mutations in KCNA2 cause epileptic encephalopathy. Nat. Genet. 2015, 47, 393–399. [Google Scholar] [CrossRef]
- Papazian, D.M.; Schwarz, T.L.; Tempel, B.L.; Jan, Y.N.; Jan, L.Y. Cloning of genomic and complementary DNA from Shaker, a putative potassium channel gene from Drosophila. Science 1987, 237, 749–753. [Google Scholar] [CrossRef]
- Ueda, A.; Wu, C.F. Distinct frequency-dependent regulation of nerve terminal excitability and synaptic transmission by IA and IK potassium channels revealed by Drosophila Shaker and Shab mutations. J. Neurosci. 2006, 26, 6238–6248. [Google Scholar] [CrossRef]
- Calhoun, J.D.; Vanoye, C.G.; Kok, F.; George, A.L., Jr.; Kearney, J.A. Characterization of a KCNB1 variant associated with autism, intellectual disability, and epilepsy. Neurol. Genet. 2017, 3, e198. [Google Scholar] [CrossRef]
- Suzuki, D.T.; Grigliatti, T.; Williamson, R. Temperature-sensitive mutations in Drosophila melanogaster. VII. A mutation (para-ts) causing reversible adult paralysis. Proc. Natl. Acad. Sci. USA 1971, 68, 890–893. [Google Scholar] [CrossRef]
- Schutte, S.S.; Schutte, R.J.; Barragan, E.V.; O’Dowd, D.K. Model systems for studying cellular mechanisms of SCN1A-related epilepsy. J. Neurophysiol. 2016, 115, 1755–1766. [Google Scholar] [CrossRef]
- van der Bliek, A.M.; Meyerowitz, E.M. Dynamin-like protein encoded by the Drosophila shibire gene associated with vesicular traffic. Nature 1991, 351, 411–414. [Google Scholar] [CrossRef]
- Nakashima, M.; Kouga, T.; Lourenco, C.M.; Shiina, M.; Goto, T.; Tsurusaki, Y.; Miyatake, S.; Miyake, N.; Saitsu, H.; Ogata, K.; et al. De novo DNM1 mutations in two cases of epileptic encephalopathy. Epilepsia 2016, 57, e18–e23. [Google Scholar] [CrossRef] [PubMed]
- Howlett, I.C.; Rusan, Z.M.; Parker, L.; Tanouye, M.A. Drosophila as a model for intractable epilepsy: Gilgamesh suppresses seizures in para(bss1) heterozygote flies. G3 (Bethesda) 2013, 3, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.; Howlett, I.C.; Rusan, Z.M.; Tanouye, M.A. Seizure and epilepsy: Studies of seizure disorders in Drosophila. Int. Rev. Neurobiol. 2011, 99, 1–21. [Google Scholar] [CrossRef]
- Yamaguchi, M. (Ed.) Drosophila Models for Human Diseases; Springer: Singapore, 2018; Advances in Experimental Medicine and Biology; Volume 1076, pp. 1–308. ISBN 978-981-13-0528-3. [Google Scholar] [CrossRef]
- Pandey, U.B.; Nichols, C.D. Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol. Rev. 2011, 63, 411–436. [Google Scholar] [CrossRef]
- Sonoshita, M.; Cagan, R.L. Modeling Human Cancers in Drosophila. Curr Top. Dev. Biol. 2017, 121, 287–309. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savitsky, M.; Solis, G.P.; Kryuchkov, M.; Katanaev, V.L. Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies. Biomedicines 2020, 8, 395. https://doi.org/10.3390/biomedicines8100395
Savitsky M, Solis GP, Kryuchkov M, Katanaev VL. Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies. Biomedicines. 2020; 8(10):395. https://doi.org/10.3390/biomedicines8100395
Chicago/Turabian StyleSavitsky, Mikhail, Gonzalo P. Solis, Mikhail Kryuchkov, and Vladimir L. Katanaev. 2020. "Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies" Biomedicines 8, no. 10: 395. https://doi.org/10.3390/biomedicines8100395
APA StyleSavitsky, M., Solis, G. P., Kryuchkov, M., & Katanaev, V. L. (2020). Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies. Biomedicines, 8(10), 395. https://doi.org/10.3390/biomedicines8100395






