Neuroprotective Effect of Subdural Infusion of Serp-1 in Spinal Cord Trauma
Abstract
1. Introduction
2. Experimental Section
2.1. Ethical Considerations
2.2. Serp-1 Protein Expression and Purification
2.3. Balloon Crush SCI in the Rat
2.4. Post-Operative Care and Clinical Testing
2.5. Pathology
Macrophage Counts in the Cavity of Injury
2.6. Statistical Analysis
3. Results
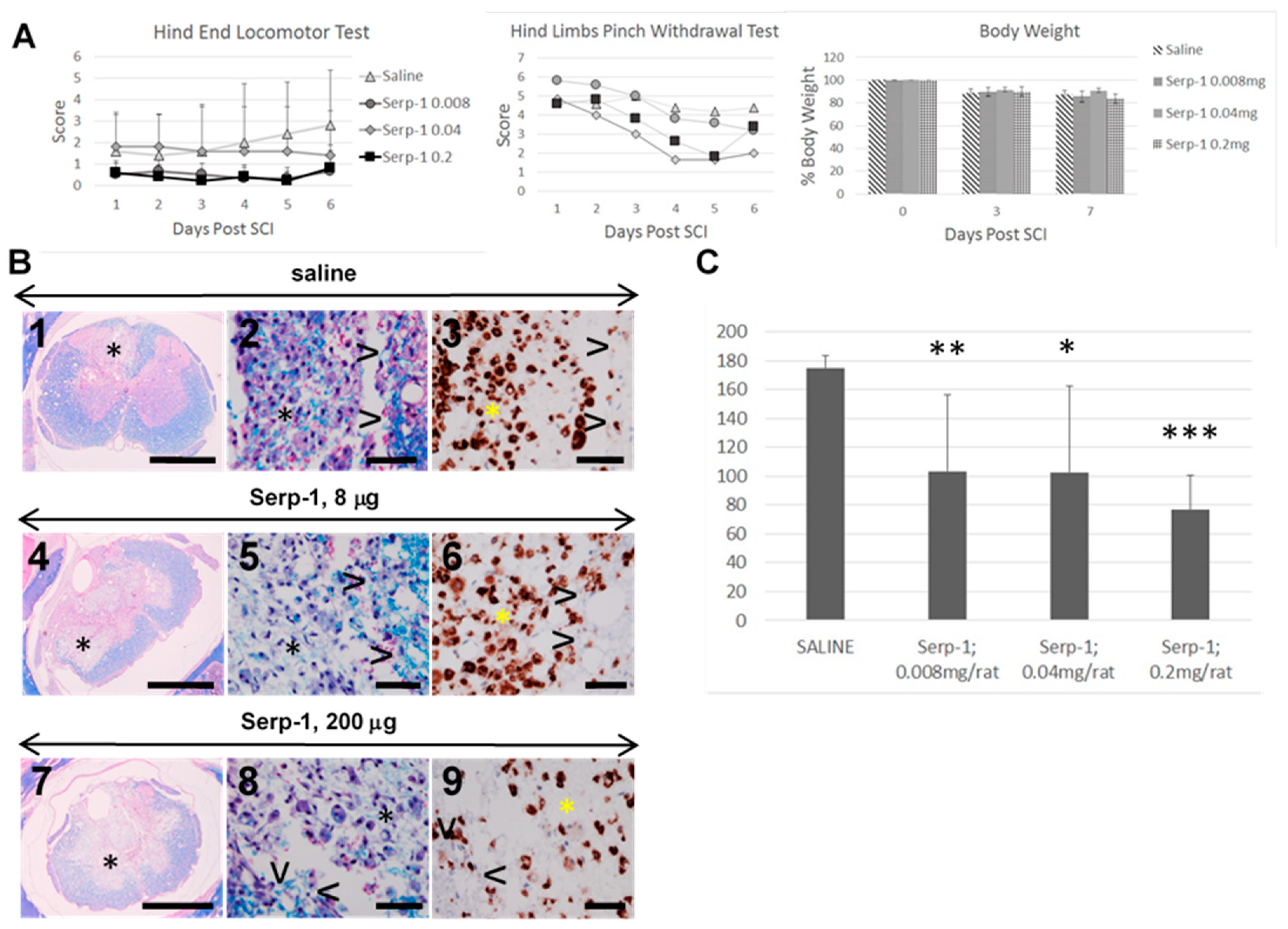
3.1. Clinical Testing, 7 Days Study
3.2. Histologic Analysis, 7 Days Study
3.3. Macrophage Counts, 7 Days Study
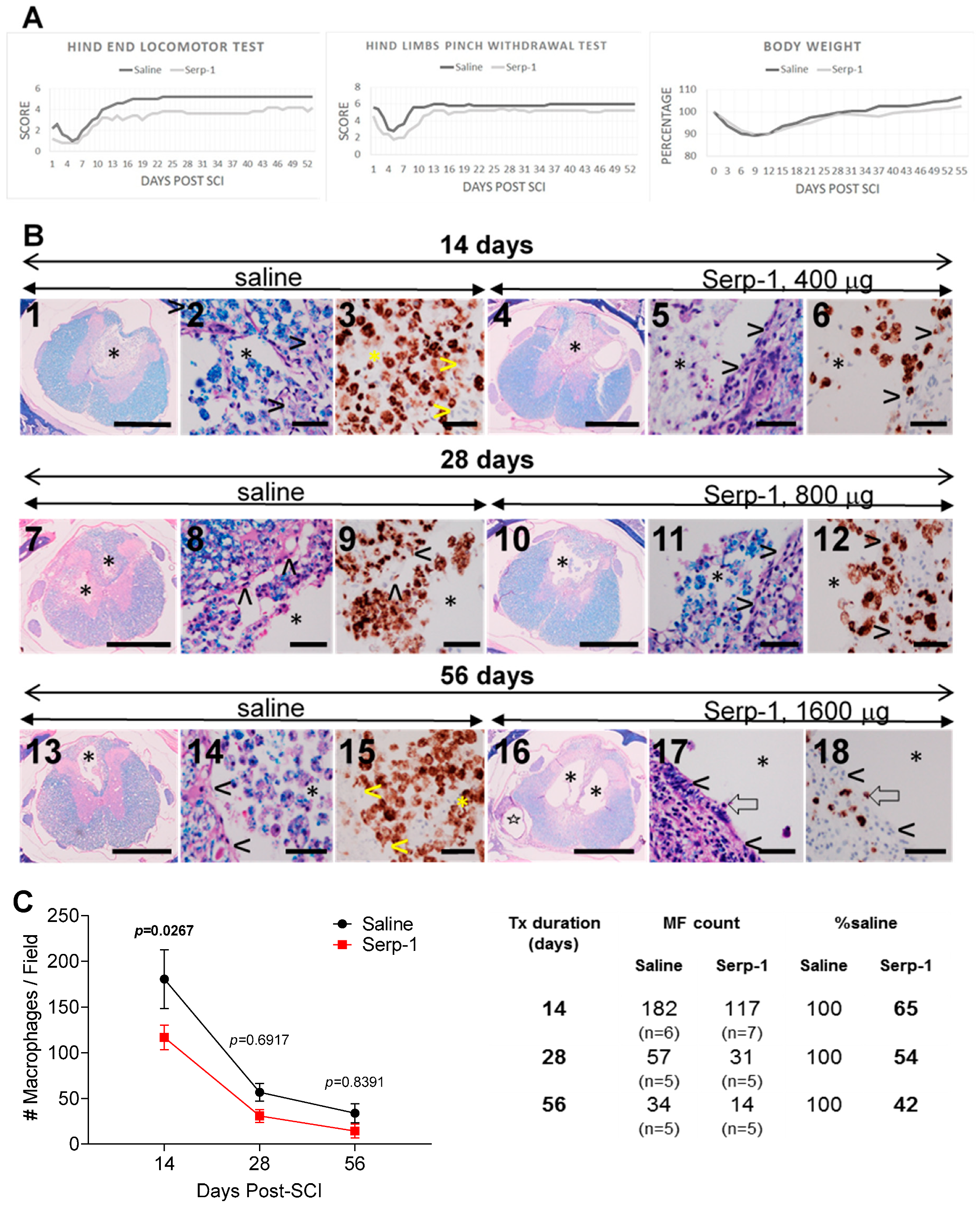
3.4. Clinical Testing, 56 Days Study
3.5. Histologic Analysis, 56 Days Study
3.6. Macrophage Counts, 56 Days Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kwiecien, J.M.; Jarosz, B.; Urdzikova, L.M.; Rola, R.; Dabrowski, W. Subdural infusion of dexamethasone inhibits leukomyelitis after acute spinal cord injury in a rat model. Folia Neuropathol. 2015, 1, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M.; Jarosz, B.; Oakden, W.; Klapec, M.; Stanisz, G.J.; Delaney, K.H.; Kotlinska-Hasiec, E.; Janik, R.; Rola, R.; Dabrowski, W. An in vivo model of anti-inflammatory activity of subdural dexamethasone following the spinal cord injury. Neurol. Neurochir. Pol. 2016, 50, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M.; Dabrowski, W.; Marzec-Kotarska, B.; Kwiecien-Delaney, C.J.; Yaron, J.R.; Zhang, L.; Schutz, L.; Lucas, A.R. Myxoma virus derived immune modulating proteins, M-T7 and Serp-1, reduce early inflammation after spinal cord injury in the rat model. Folia Neuropathol. 2019, 57, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M.; Dabrowski, W.; Dąbrowska-Bouta, B.; Sulkowski, G.; Oakden, W.; Kwiecien-Delaney, C.J.; Yaron, J.R.; Zhang, L.; Marzec-Kotarska, B.; Stanisz, G.J.; et al. Protracted inflammation extends damage after spinal cord injury. PLoS ONE 2020, 15, e0226584. [Google Scholar] [CrossRef]
- Barkho, B.Z.; Song, H.; Almone, J.B.; Smart, R.D.; Kuwabera, T.; Nakashima, K.; Gage, F.H.; Zhao, X. Identification of astrocyte-expressed factors that modulate neural stem/progenitor cell differentiation. Stem Cell Dev. 2006, 15, 407–421. [Google Scholar] [CrossRef]
- Wang, X.J.; Kong, K.M.; Qi, W.L.; Ye, W.L.; Song, P.S. Interleukin-1 beta induction of neuron apoptosis depends on p38 mitogen-activated protein kinase activity after spinal cord injury. Acta Pharmacol. Sin. 2005, 26, 934–942. [Google Scholar] [CrossRef]
- Kwiecien, J.M.; Yaron, J.R.; Zhang, L.; Delaney, K.H.; Lucas, A.R. Methods for Deriving Therapeutics from Viruses and Viral Components; Lucas, A.R., Ed.; Humana Press: New York, NY, USA, 2020; in press. [Google Scholar]
- Rahman, M.M.; Lucas, A.R.; McFadden, G. Viral TNF inhibitors as potential therapeutics. Adv. Exp. Med. Biol. 2009, 666, 64–77. [Google Scholar]
- Kwiecien, J.M.; Zhang, L.; Yaron, J.R.; Schutz, L.N.; Kwiecien-Delaney, C.J.; Enkidia, A.; Awo, E.A.; Burgin, M.; Dabrowski, W.; Lucas, A.R. Local chitosan-serpin injection after spinal cord injury reduces inflammatory damage and improves neurologic function. J. Clin. Med. 2020, 9, 1221. [Google Scholar] [CrossRef]
- Yaron, J.R.; Zhang, L.; Guo, Q.; Burgin, M.; Schutz, L.; Awo, E.; Wise, L.; Krause, K.; Ildefonso, C.; Kwiecien, J.M.; et al. Deriving immune modulating drugs from viruses-a new class of biologics. J. Clin. Med. 2020, 9, 972. [Google Scholar] [CrossRef]
- Viswanathan, K.; Bot, I.; Liu, L.; Dai, E.; Turner, P.C.; Togonu-Bickersteth, B.; Richardson, J.; Davids, J.A.; Williams, J.M.; Bartee, M.Y.; et al. Viral cross-class serpin inhibits vascular inflammation and T lymphocyte fratricide; a study in rodent models in vivo and human cell lines in vitro. PLoS ONE 2012, 7, e44694. [Google Scholar] [CrossRef]
- Tardif, J.-C.; L’Allier, P.L.; Grégoire, J.; Ibrahim, R.; McFadden, G.; Kostuk, W.; Knudtson, M.; Labinaz, M.; Waksman, R.; Pepine, C.J.; et al. A randomized controlled, phase 2 trial of the viral serpin Serp-1 in patients with acute coronary syndromes undergoing percutaneous coronary intervention. Circ. Cardiovasc. Interv. 2010, 3, 543–548. [Google Scholar] [CrossRef] [PubMed]
- The Food and Drug Administration, USA. Available online: https://www.fda.gov/drugs/development-approval-process-drugs (accessed on 25 April 2020).
- Kwiecien, J.M. Cellular mechanisms of white matter regeneration in adult dysmyelinated rat model. Folia Neuropathol. 2013, 51, 189–202. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M. Methods in Molecular Biology; Lucas, A.R., Ed.; Humana Press: New York, NY, USA, 2018; pp. 223–235. [Google Scholar]
- Barros, M.H.M.; Hauck, F.; Dreyer, J.H.; Kempkes, B.; Niedobitek, G. Macrophage polarisation: An immunohistochemical approach for identifying M1 and M2 macrophages. PLoS ONE 2013, 8, e80908. [Google Scholar] [CrossRef] [PubMed]
- Kigerl, K.A.; Gensel, J.C.; Ankeny, D.P.; Alexander, J.K.; Donnelly, D.J.; Popovich, P.G. Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J. Neurosci. 2009, 29, 13435–13444. [Google Scholar] [CrossRef] [PubMed]
- Bowes, A.L.; Yip, P.K. Modulating inflammatory cell responses to spinal cord injury: All in good time. J. Neurotrauma 2014, 31, 1753–1766. [Google Scholar] [CrossRef] [PubMed]
- Bracken, M.B.; Shepard, M.J.; Collins, W.F.; Holford, T.R.; Young, W.; Baskin, D.S.; Eisenberg, H.M.; Flamm, E.; Leo-Summers, L.; Maroon, J. A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. N. Engl. J. Med. 1990, 322, 1405–1411. [Google Scholar] [CrossRef]
- Schoenfeld, A.J.; Laughlin, M.D.; McCriskin, B.J.; Bader, J.O.; Waterman, B.R.; Belmont, P.J. Spinal Injuries in United States Military Personnel Deployed to Iraq and Afghanistan. Spine (Phila. Pa. 1976) 2013, 38, 1770–1778. [Google Scholar] [CrossRef]
- Shank, C.D.; Walters, B.C.; Hadley, M.N. Current Topics in the Management of Acute Traumatic Spinal Cord Injury. Neurocrit. Care 2019, 30, 261–271. [Google Scholar] [CrossRef]
- Ahuja, C.S.; Martin, A.R.; Fehlings, M. Recent advances in managing a spinal cord injury secondary to trauma. F1000Research 2016, 5, 1017. [Google Scholar] [CrossRef]
- Markandaya, M.; Stein, D.M.; Menaker, J. Acute Treatment Options for Spinal Cord Injury. Curr. Treat. Options Neurol. 2012, 14, 175–187. [Google Scholar] [CrossRef]
- Pettiford, J.N.; Bikhchandani, J.; Ostlie, D.J.; St. Peter, S.D.; Sharp, R.J.; Juang, D.; St Peter, S.D.; Sharp, R.J.; Juang, D. A review: The role of high dose methylprednisolone in spinal cord trauma in children. Pediatr. Surg. Int. 2012, 28, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Hurlbert, R.J.; Hadley, M.N.; Walters, B.C.; Aarabi, B.; Dhall, S.S.; Gelb, D.E.; Rozzelle, C.J.; Ryken, T.C.; Theodore, N. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013, 72, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Ulndreaj, A.; Badner, A.; Fehlings, M.G. Promising neuroprotective strategies for traumatic spinal cord injury with a focus on the differential effects among anatomical levels of injury. F1000Research 2017, 6, 1907. [Google Scholar] [CrossRef]
- Cordaro, M.; Casili, G.; Paterniti, I.; Cuzzocrea, S.; Esposito, E. Fumaric acid esters attenuate secondary degeneration after spinal cord injury. J. Neurotrauma 2017, 43, 3027–3040. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.; Varma, A.; Barry, J.; Vertegel, A.; Banik, N. Nanoparticle estrogen in rat spinal cord injury elicits rapid anti-inflammatory effects in plasma, cerebrospinal fluid, and tissue. J. Neurotrauma 2015, 32, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Ohta, S.; Iwashita, Y.; Kakinoki, R.; Noguchi, T.; Nakamura, T. Effects of continuous intravenous infusion of MCI-186 on functional recovery after spinal cord injury in rats. J. Neurotrauma 2011, 28, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Choi, H.Y.; Park, C.S.; Ju, B.G.; Yune, T.Y. Mithramycin A improves functional recovery by inhibiting BSCB disruption and hemorrhage after spinal cord injury. J. Neurotrauma 2018, 35, 508–520. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Cordaro, M.; Bruschetta, G.; Siracusa, R.; Crupi, R.; Esposito, E.; Cuzzocrea, S. N-palmitoylethanolamine-oxazoline as a new therapeutic strategy to control neuroinflammation: Neuroprotective effects in experimental models of spinal cord and brain injury. J. Neurotrauma 2017, 34, 2609–2623. [Google Scholar] [CrossRef]
- Hirst, J.A.; Howick, J.; Aronson, J.K.; Roberts, N.; Perera, R.; Koshiaris, C.; Henegan, C. The need for randomization in animal trials: An overview of systematic reviews. PLoS ONE 2014, 9, e98856. [Google Scholar] [CrossRef]
| Treatment | Duration Days | # Rats | Osmotic Pump | Total Serp-1 (mg) |
|---|---|---|---|---|
| Saline | 7 | 5 | 2ML1 | 0 |
| Serp-1, 0.008 mg | 7 | 6 | 2ML1 | 0.008 |
| Serp-1, 0.04 mg | 7 | 5 | 2ML1 | 0.04 |
| Serp-1, 0.2 mg | 7 | 6 | 2ML1 | 0.2 |
| Saline | 14 | 6 | 2ML4 | 0 |
| Serp-1, 0.2 mg/week | 14 | 7 | 2ML4 | 0.4 |
| Saline | 28 | 7 | 2ML4 | 0 |
| Serp-1, 0.2 mg/week | 28 | 6 | 2ML4 | 0.8 |
| Saline | 56 | 5 | 2ML4 × 2 | 0 |
| Serp-1, 0.2 mg/week | 56 | 5 | 2ML4 × 2 | 1.6 |
| Score | Description |
|---|---|
| 0 | Both hind legs have no motion, extended backwards. |
| 1 | One hind leg has flexing motion caudal to the level of the hip joint, with the plantar surface of the foot up, no weight support. |
| 2 | Both legs have flexing motion caudal to the hip, with the plantar surface of the foot up, no weight support or one leg has flexing motion beyond the hip, nobody support, the other leg no motion. |
| 3 | One leg has flexing motion beyond the hip, with the dorsal surface of the foot up, no weight support, the other leg has flexing motion caudal to the hip, with the plantar surface of the foot up; or one leg has flexing motion beyond the hip, with dorsal surface of the foot up, with body weight support but the other leg has no motion. |
| 4 | Both legs have flexing motion beyond the hip, with the dorsal surface of the foot up, but no body weight support; or one leg with the flexing motion beyond the hip with body support and the other leg with flexing motion caudal to the hip, with the plantar surface of the foot up, but no body weight support. |
| 5 | One leg has flexing motion beyond the hip with body weight support, the other leg flexing motion beyond the hip, with the dorsal surface of the foot up, but no body support. |
| 6 | Normal gait, no apparent weakness or proprioceptive deficits. |
| Score | Description |
|---|---|
| 0 | No toe retraction. |
| 1 | Weak retraction, no jerking. |
| 2 | Weak retraction with jerking. |
| 3 | Strong/normal retraction with jerking. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwiecien, J.M.; Dabrowski, W.; Kwiecien-Delaney, B.J.; Kwiecien-Delaney, C.J.; Siwicka-Gieroba, D.; Yaron, J.R.; Zhang, L.; Delaney, K.H.; Lucas, A.R. Neuroprotective Effect of Subdural Infusion of Serp-1 in Spinal Cord Trauma. Biomedicines 2020, 8, 372. https://doi.org/10.3390/biomedicines8100372
Kwiecien JM, Dabrowski W, Kwiecien-Delaney BJ, Kwiecien-Delaney CJ, Siwicka-Gieroba D, Yaron JR, Zhang L, Delaney KH, Lucas AR. Neuroprotective Effect of Subdural Infusion of Serp-1 in Spinal Cord Trauma. Biomedicines. 2020; 8(10):372. https://doi.org/10.3390/biomedicines8100372
Chicago/Turabian StyleKwiecien, Jacek M., Wojciech Dabrowski, Bryce J. Kwiecien-Delaney, Christian J. Kwiecien-Delaney, Dorota Siwicka-Gieroba, Jordan R. Yaron, Liqiang Zhang, Kathleen H. Delaney, and Alexandra R. Lucas. 2020. "Neuroprotective Effect of Subdural Infusion of Serp-1 in Spinal Cord Trauma" Biomedicines 8, no. 10: 372. https://doi.org/10.3390/biomedicines8100372
APA StyleKwiecien, J. M., Dabrowski, W., Kwiecien-Delaney, B. J., Kwiecien-Delaney, C. J., Siwicka-Gieroba, D., Yaron, J. R., Zhang, L., Delaney, K. H., & Lucas, A. R. (2020). Neuroprotective Effect of Subdural Infusion of Serp-1 in Spinal Cord Trauma. Biomedicines, 8(10), 372. https://doi.org/10.3390/biomedicines8100372








