Modulation of NMDA Receptor Activity in Fibromyalgia
Abstract
:1. Introduction
2. Methods
3. Results
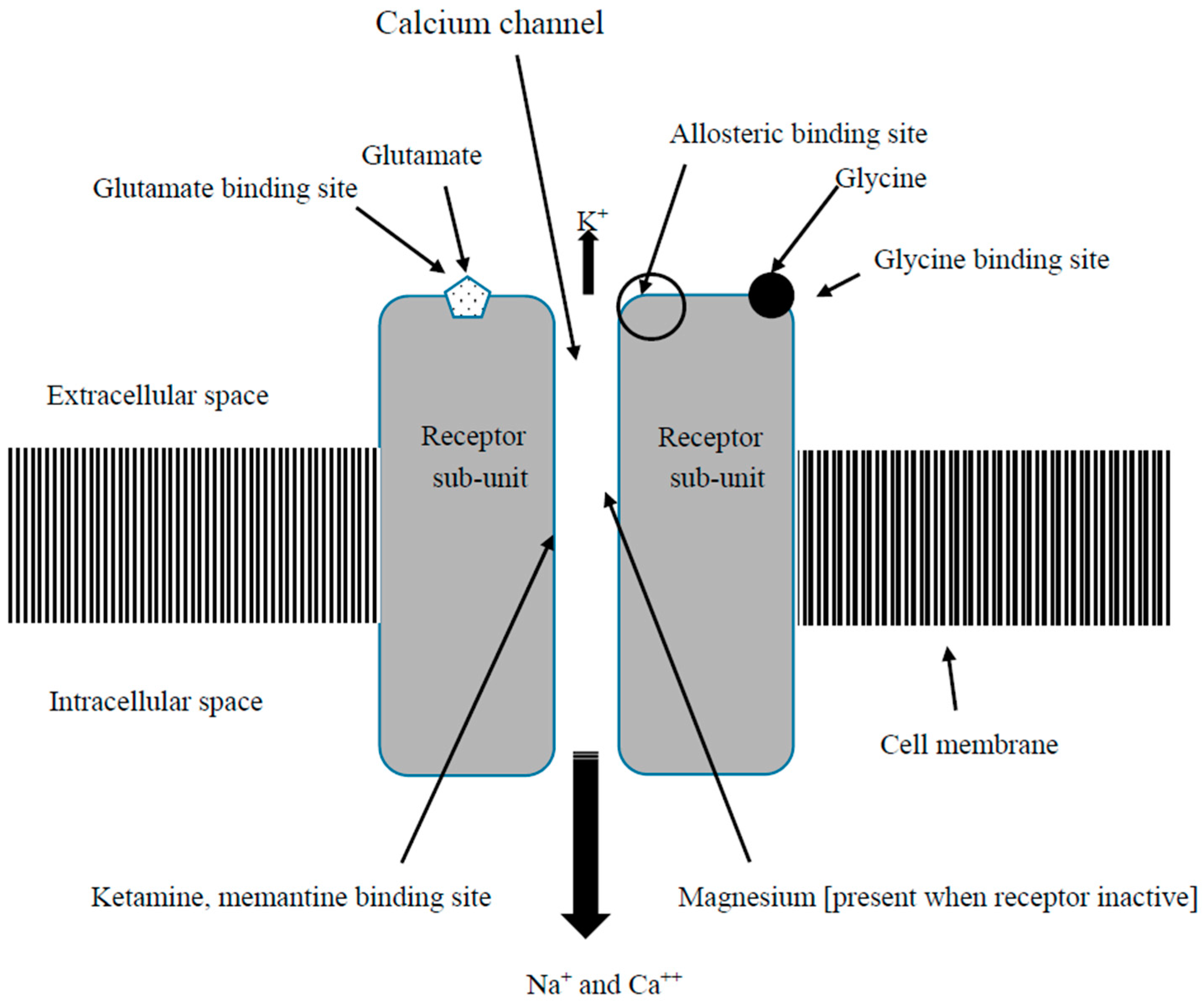
3.1. NMDAR Function in Fibromyalgia
3.2. NMDAR Inhibitors
3.2.1. Ketamine
3.2.2. Dextromethorphan
3.2.3. Memantine
3.2.4. Amantadine
3.2.5. Methadone
3.2.6. Guaifesin
3.3. Drugs with Indirect Effect on NMDAR Function
3.4. Non-Pharmacological Approaches
3.5. NMDA Antagonists in Fibromyalgia Management Guidelines
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lawson, K. Potential drug therapies for the treatment of fibromyalgia. Expert Opin. Investig. Drugs 2016, 25, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef] [PubMed]
- Yunus, M.B. Editorial review: An update on central sensitivity syndromes and the issues of nosology and psychobiology. Curr. Rheumatol. Rev. 2015, 11, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Kuner, R.; Flor, H. Structural plasticity and reorganisation in chronic pain. Nat. Rev. Neurosci. 2016, 18, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Dickenson, A.H.; Sullivan, A.F. NMDA receptors and central hyperalgesic states. Pain 1991, 46, 344–346. [Google Scholar] [CrossRef]
- Staud, R.; Vierck, C.J.; Robinson, M.E.; Price, D.D. Effects of the N-methyl-d-aspartate receptor antagonist dextromethorphan on temporal summation of pain are similar in fibromyalgia patients and normal control subjects. J. Pain 2005, 6, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.E. Elevated excitatory neurotransmitter levels in the fibromyalgia brain. Arthritis Res. Ther. 2010, 12, 141. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.E.; Sundgren, P.C.; Craig, A.D.; Kirshenbaum, E.; Sen, A.; Napadow, V.; Clauw, D.J. Elevated insular glutamate in fibromyalgia is associated with experimental pain. Arthritis Rheum. 2009, 60, 3146–3152. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.E.; Napadow, V.; Huggins, J.P.; Pauer, L.; Kim, J.; Hampson, J.; Sundgren, P.C.; Foerster, B.; Petrou, M.; Schmidt-Wilcke, T.; et al. Pregabalin rectifies aberrant brain chemistry, connectivity, and functional response in chronic pain patients. Anesthesiology 2013, 119, 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Fayed, N.; Garcia-Campayo, J.; Magallon, R.; Andres-Bergareche, H.; Luciano, J.V.; Andres, E.; Beltran, J. Localized 1h-NMR spectroscopy in patients with fibromyalgia: A controlled study of changes in cerebral glutamate/glutamine, inositol, choline, and n-acetylaspartate. Arthritis Res. Ther. 2010, 12, R134. [Google Scholar] [CrossRef] [PubMed]
- Pyke, T.; Osmotherly, P.G.; Baines, S. Measuring glutamate levels in the brains of fibromyalgia patients and a potential role for glutamate in the pathophysiology of fibromyalgia symptoms: A systematic review. Clin. J. Pain 2016. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.A.; Giovengo, S.L.; Russell, I.J.; Michalek, J.E. Changes in the concentrations of amino acids in the cerebrospinal fluid that correlate with pain in patients with fibromyalgia: Implications for nitric oxide pathways. Pain 2000, 87, 201–211. [Google Scholar] [CrossRef]
- Vyklicky, V.; Korinek, M.; Smejkalova, T.; Balik, A.; Krausova, B.; Kaniakova, M.; Lichnerova, K.; Cerny, J.; Krusek, J.; Dittert, I.; et al. Structure, function, and pharmacology of nmda receptor channels. Physiol. Res. 2014, 63, S191–S203. [Google Scholar] [PubMed]
- Iacobucci, G.J.; Popescu, G.K. NMDA receptors: Linking physiologic output to biophysical operation. Nat. Rev. Neurosci. 2017, 18, 236–249. [Google Scholar] [CrossRef] [PubMed]
- Hocking, G.; Visser, E.J.; Schug, S.A.; Cousins, M.J. Ketamine: Does life begin at 40? Pain Clin. Updates 2007, 15, 6. [Google Scholar]
- Staud, R. The neurobiology of chronic musculoskeletal pain (including chronic regional pain). In Fibromyalgia and Other Central Pain Syndromes; Wallace, D.J., Clauw, D., Eds.; Lippincott Williams &Wilkins: Philadelphia, PA, USA, 2005; Volume 1, pp. 45–62. [Google Scholar]
- McBain, C.J.; Mayer, M.L. N-methyl-d-aspartic acid receptor structure and function. Physiol. Rev. 1994, 74, 723–760. [Google Scholar] [PubMed]
- Kim, A.H.; Kerchner, G.A.; Choi, D.W. Blocking excitotoxicity. In CNS Neuroprotection; Marcoux, F.W., Choi, D.W., Eds.; Springer: New York, NY, USA, 2002; pp. 3–36. [Google Scholar]
- Mion, G.; Villevieille, T. Ketamine pharmacology: An update (pharmacodynamics and molecular aspects, recent findings). CNS Neurosci. Ther. 2013, 19, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Seeman, P.; Ko, F.; Tallerico, T. Dopamine receptor contribution to the action of pcp, lsd and ketamine psychotomimetics. Mol. Psychiatry 2005, 10, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Wood, P.B. A reconsideration of the relevance of systemic low-dose ketamine to the pathophysiology of fibromyalgia. J. Pain 2006, 7, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Niesters, M.; Martini, C.; Dahan, A. Ketamine for chronic pain: Risks and benefits. Br. J. Clin. Pharmacol. 2014, 77, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Persson, J. Ketamine in pain management. CNS Neurosci. Ther. 2013, 19, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Serafini, G.; Howland, R.H.; Rovedi, F.; Girardi, P.; Amore, M. The role of ketamine in treatment-resistant depression: A systematic review. Curr. Neuropharmacol. 2014, 12, 444–461. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.; Bengtsson, A.; Backman, E.; Henriksson, K.G.; Bengtsson, M. Pain analysis in patients with fibromyalgia. Effects of intravenous morphine, lidocaine, and ketamine. Scand. J. Rheumatol. 1995, 24, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.; Bengtsson, A.; Ahlner, J.; Henriksson, K.G.; Ekselius, L.; Bengtsson, M. Fibromyalgia—Are there different mechanisms in the processing of pain? A double blind crossover comparison of analgesic drugs. J. Rheumatol. 1997, 24, 1615–1621. [Google Scholar] [PubMed]
- Graven-Nielsen, T.; Aspegren Kendall, S.; Henriksson, K.G.; Bengtsson, M.; Sorensen, J.; Johnson, A.; Gerdle, B.; Arendt-Nielsen, L. Ketamine reduces muscle pain, temporal summation, and referred pain in fibromyalgia patients. Pain 2000, 85, 483–491. [Google Scholar] [CrossRef]
- Henriksson, K.G. Fibromyalgia—From syndrome to disease. Overview of pathogenetic mechanisms. J. Rehabil. Med. 2003, 35, 89–94. [Google Scholar] [CrossRef]
- Noppers, I.; Niesters, M.; Swartjes, M.; Bauer, M.; Aarts, L.; Geleijnse, N.; Mooren, R.; Dahan, A.; Sarton, E. Absence of long-term analgesic effect from a short-term S-ketamine infusion on fibromyalgia pain: A randomized, prospective, double blind, active placebo-controlled trial. Eur. J. Pain 2011, 15, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Littlejohn, G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat. Rev. Rheumatol. 2015, 11, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Sigtermans, M.J.; van Hilten, J.J.; Bauer, M.C.; Arbous, M.S.; Marinus, J.; Sarton, E.Y.; Dahan, A. Ketamine produces effective and long-term pain relief in patients with complex regional pain syndrome type 1. Pain 2009, 145, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Hanna, A.F.; Smith, A.G. Intravenous ketamine produces long-term pain relief in a patient with fibromyalgia. Fibrom. Open Access 2016, 1, 1–3. [Google Scholar]
- Marchetti, F.; Coutaux, A.; Bellanger, A.; Magneux, C.; Bourgeois, P.; Mion, G. Efficacy and safety of oral ketamine for the relief of intractable chronic pain: A retrospective 5-year study of 51 patients. Eur. J. Pain 2015, 19, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Cherry, D.A.; Plummer, J.L.; Gourlay, G.K.; Coates, K.R.; Odgers, C.L. Ketamine as an adjunct to morphine in the treatment of pain. Pain 1995, 62, 119–121. [Google Scholar] [CrossRef]
- Blonk, M.I.; Koder, B.G.; van den Bemt, P.M.; Huygen, F.J. Use of oral ketamine in chronic pain management: A review. Eur. J. Pain 2010, 14, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Li, J.H.; Vicknasingam, B.; Cheung, Y.W.; Zhou, W.; Nurhidayat, A.W.; Jarlais, D.C.; Schottenfeld, R. To use or not to use: An update on licit and illicit ketamine use. Subst. Abuse Rehabil. 2011, 2, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Siu, A.; Drachtman, R. Dextromethorphan: A review of N-methyl-d-aspartate receptor antagonist in the management of pain. CNS Drug Rev. 2007, 13, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.P.; Verdolin, M.H.; Chang, A.S.; Kurihara, C.; Morlando, B.J.; Mao, J. The intravenous ketamine test predicts subsequent response to an oral dextromethorphan treatment regimen in fibromyalgia patients. J. Pain 2006, 7, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.W.; Kotermanski, S.E. Mechanism of action of memantine. Curr. Opin. Pharmacol. 2006, 6, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Olivan-Blazquez, B.; Herrera-Mercadal, P.; Puebla-Guedea, M.; Perez-Yus, M.C.; Andres, E.; Fayed, N.; Lopez-Del-Hoyo, Y.; Magallon, R.; Roca, M.; Garcia-Campayo, J. Efficacy of memantine in the treatment of fibromyalgia: A double-blind, randomised, controlled trial with 6-month follow-up. Pain 2014, 155, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Sinis, N.; Birbaumer, N.; Gustin, S.; Schwarz, A.; Bredanger, S.; Becker, S.T.; Unertl, K.; Schaller, H.E.; Haerle, M. Memantine treatment of complex regional pain syndrome: A preliminary report of six cases. Clin. J. Pain 2007, 23, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Fayed, N.; Olivan-Blazquez, B.; Herrera-Mercadal, P.; Puebla-Guedea, M.; Perez-Yus, M.C.; Andres, E.; Lopez del Hoyo, Y.; Magallon, R.; Viguera, L.; Garcia-Campayo, J. Changes in metabolites after treatment with memantine in fibromyalgia. A double-blind randomized controlled trial with magnetic resonance spectroscopy with a 6-month follow-up. CNS Neurosci. Ther. 2014, 20, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Blanpied, T.A.; Clarke, R.J.; Johnson, J.W. Amantadine inhibits nmda receptors by accelerating channel closure during channel block. J. Neurosci. 2005, 25, 3312–3322. [Google Scholar] [CrossRef] [PubMed]
- Pud, D.; Eisenberg, E.; Spitzer, A.; Adler, R.; Fried, G.; Yarnitsky, D. The nmda receptor antagonist amantadine reduces surgical neuropathic pain in cancer patients: A double blind, randomized, placebo controlled trial. Pain 1998, 75, 349–354. [Google Scholar] [CrossRef]
- Fredheim, O.M.; Moksnes, K.; Borchgrevink, P.C.; Kaasa, S.; Dale, O. Clinical pharmacology of methadone for pain. Acta Anaesthesiol. Scand. 2008, 52, 879–889. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, B.; Almahrezi, A.; Schreier, G. Methadone in the treatment of neuropathic pain. Pain Res. Manag. 2003, 8, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Littlejohn, G.O.; Guymer, E.K.; Ngian, G.S. Is there a role for opioids in the treatment of fibromyalgia? Pain Manag. 2016, 6, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, M.; Showraki, A.; Emamghoreishi, M. Anticonvulsant effect of guaifenesin against pentylenetetrazol-induced seizure in mice. Iran. J. Med. Sci. 2013, 38, 116–121. [Google Scholar] [PubMed]
- Bennett, R.M.; De Campo, P.; Clark, S.R. A randomized, prospective, 12 month study to compare the efficiacy of guaifenesin versus placebo in the management of fibromyalgia. Arthritis Rheum. 1996, 39, S212. [Google Scholar]
- Golembiowska, K.; Dziubina, A. Involvement of adenosine in the effect of antidepressants on glutamate and aspartate release in the rat prefrontal cortex. Naunyn-Schmiedeberg's Arch. Pharmacol. 2001, 363, 663–670. [Google Scholar] [CrossRef]
- Nekovarova, T.; Yamamotova, A.; Vales, K.; Stuchlik, A.; Fricova, J.; Rokyta, R. Common mechanisms of pain and depression: Are antidepressants also analgesics? Front. Behav. Neurosci. 2014, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Puiu, T.; Kairys, A.E.; Pauer, L.; Schmidt-Wilcke, T.; Ichesco, E.; Hampson, J.P.; Napadow, V.; Clauw, D.J.; Harris, R.E. Association of alterations in gray matter volume with reduced evoked-pain connectivity following short-term administration of pregabalin in patients with fibromyalgia. Arthritis Rheumatol. 2016, 68, 1511–1521. [Google Scholar] [CrossRef] [PubMed]
- Calandre, E.P.; Rico-Villademoros, F.; Slim, M. α2Δ ligands, gabapentin, pregabalin and mirogabalin: A review of their clinical pharmacology and therapeutic use. Expert Rev. Neurother. 2016, 16, 1263–1277. [Google Scholar] [CrossRef] [PubMed]
- Holton, K.F.; Taren, D.L.; Thomson, C.A.; Bennett, R.M.; Jones, K.D. The effect of dietary glutamate on fibromyalgia and irritable bowel symptoms. Clin. Exp. Rheumatol. 2012, 30, 10–17. [Google Scholar] [PubMed]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Hauser, W.; Fluss, E.; Choy, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Clauw, D.J. Fibromyalgia: A clinical review. JAMA 2014, 311, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Ablin, J.; Fitzcharles, M.A.; Buskila, D.; Shir, Y.; Sommer, C.; Hauser, W. Treatment of fibromyalgia syndrome: Recommendations of recent evidence-based interdisciplinary guidelines with special emphasis on complementary and alternative therapies. Evid. Based Complement. Altern. Med. eCAM 2013, 2013, 485272. [Google Scholar] [CrossRef] [PubMed]
- Fitzcharles, M.A.; Ste-Marie, P.A.; Goldenberg, D.L.; Pereira, J.X.; Abbey, S.; Choiniere, M.; Ko, G.; Moulin, D.E.; Panopalis, P.; Proulx, J.; et al. Canadian pain society and canadian rheumatology association recommendations for rational care of persons with fibromyalgia: A summary report. J. Rheumatol. 2013, 40, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
| Drug | Analgesic Dose | Side Effects | Comment |
|---|---|---|---|
| Ketamine | Oral: 2 mg/kg IV: 0.2–0.75 mg/kg Continuous infusion: 2–7 mcg/kg/min | Psychomimetic—hallucinations, confusion, sedation, irrational behaviour | No studies of higher dose, longer duration regimens limit use. |
| Dextromethorphan | Oral: 45–400 mg/day | Drowsy, dizzy, anxiety, confusion | Few clinically useful studies, anecdotal use suggests limited effect. |
| Memantine | Oral: 10–30 mg/day | Hypertension, dizzy, drowsy, nausea, anxiety, hallucinations | Further studies may show this drug to be clinically useful. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Littlejohn, G.; Guymer, E. Modulation of NMDA Receptor Activity in Fibromyalgia. Biomedicines 2017, 5, 15. https://doi.org/10.3390/biomedicines5020015
Littlejohn G, Guymer E. Modulation of NMDA Receptor Activity in Fibromyalgia. Biomedicines. 2017; 5(2):15. https://doi.org/10.3390/biomedicines5020015
Chicago/Turabian StyleLittlejohn, Geoffrey, and Emma Guymer. 2017. "Modulation of NMDA Receptor Activity in Fibromyalgia" Biomedicines 5, no. 2: 15. https://doi.org/10.3390/biomedicines5020015
APA StyleLittlejohn, G., & Guymer, E. (2017). Modulation of NMDA Receptor Activity in Fibromyalgia. Biomedicines, 5(2), 15. https://doi.org/10.3390/biomedicines5020015





