Properties of Nanohybrid Dental Composites—A Comparative In Vitro Study
Abstract
1. Introduction
2. Materials and Methods
- -
- Clearfil Majesty ES-2, Kuraray Noritake Dental Inc., Nagoya, Japan, (CM);
- -
- Harmonize Universal, Kerr Ltd., Uxbridge, UK, (HU);
- -
- PS2 experimental composite developed by Raluca Ripan Institute for Research in Chemistry, Babeș-Bolyai University, Cluj-Napoca, Romania, (PS2).
- Sample size calculation
- Statistical analysis
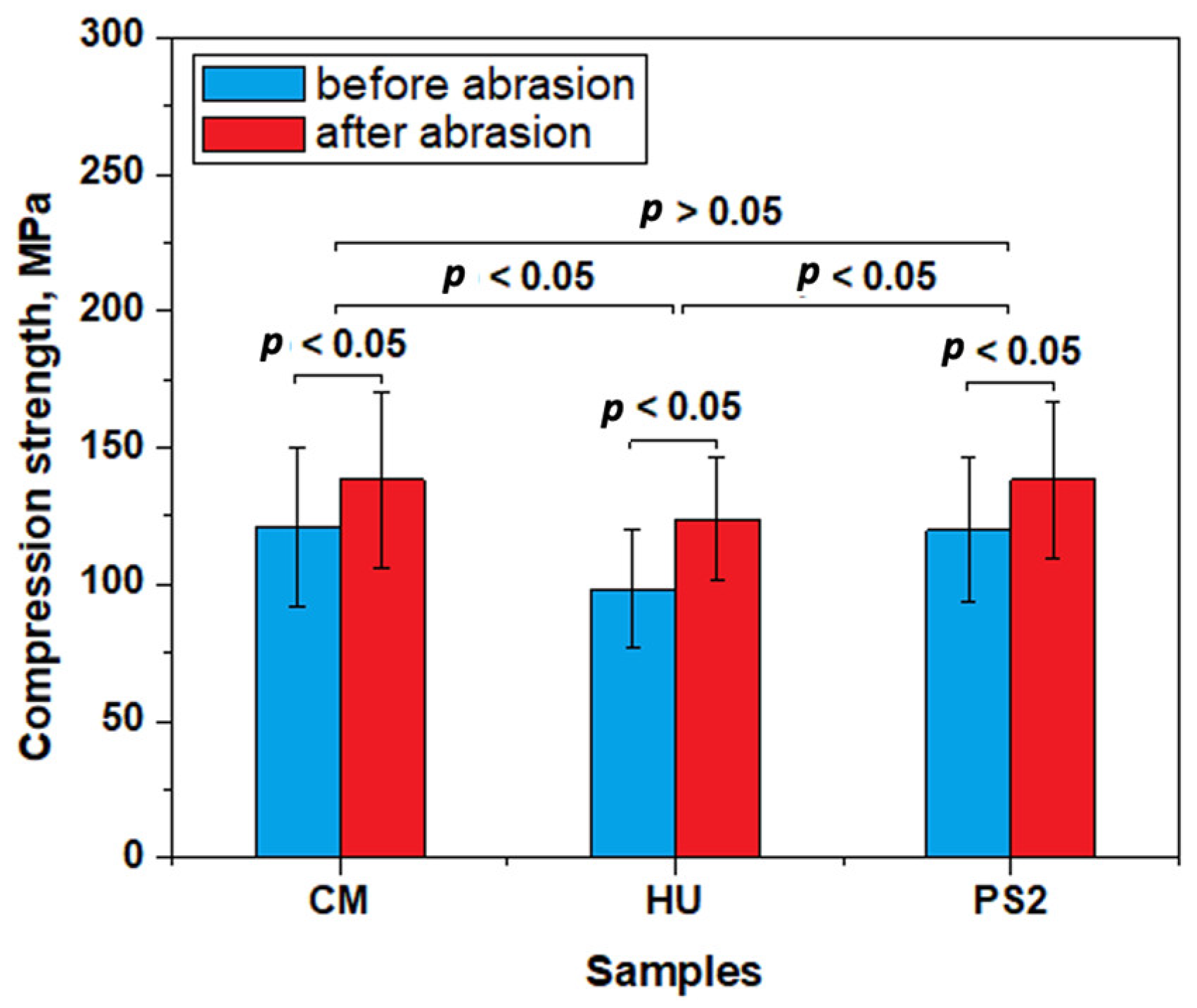
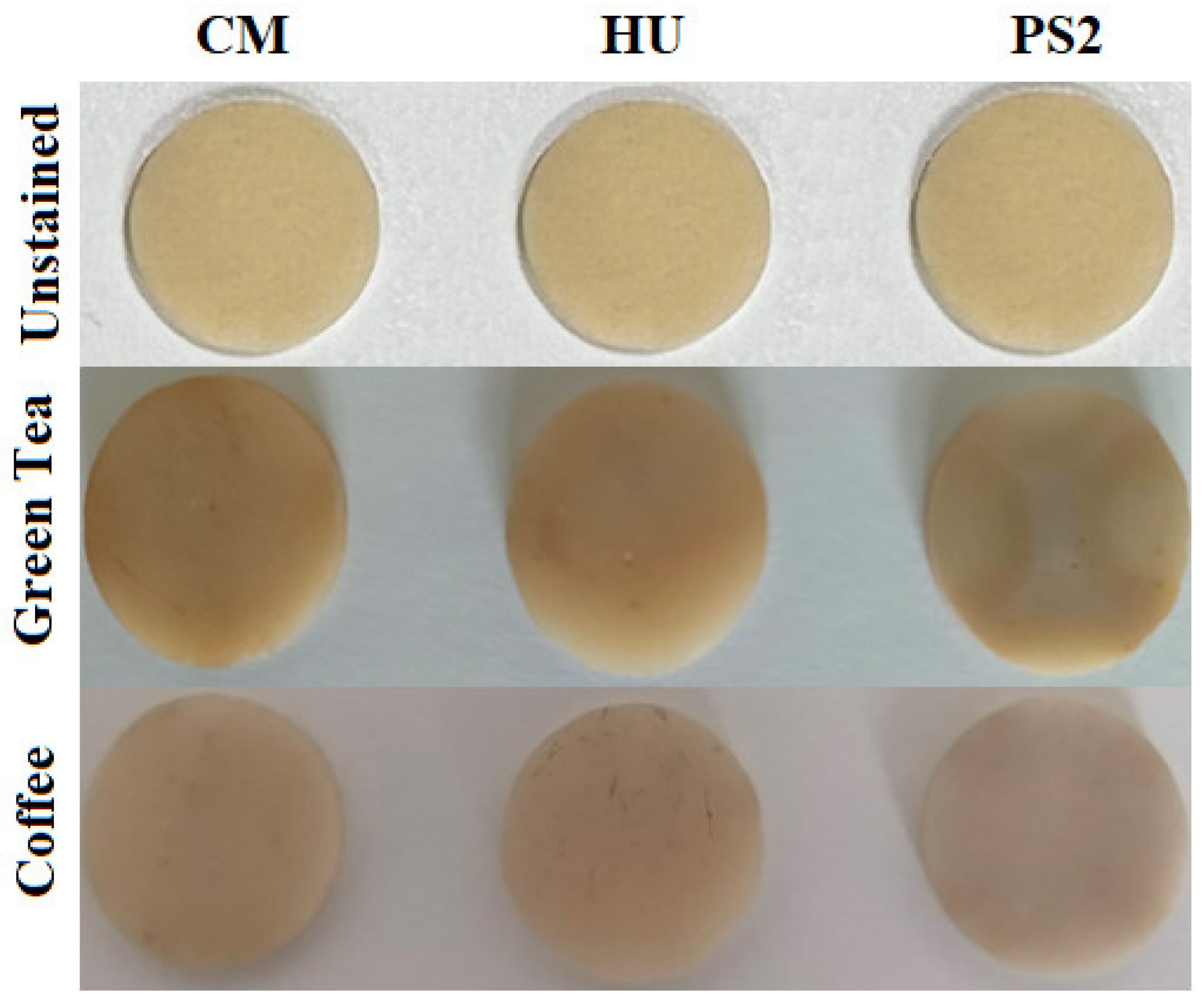
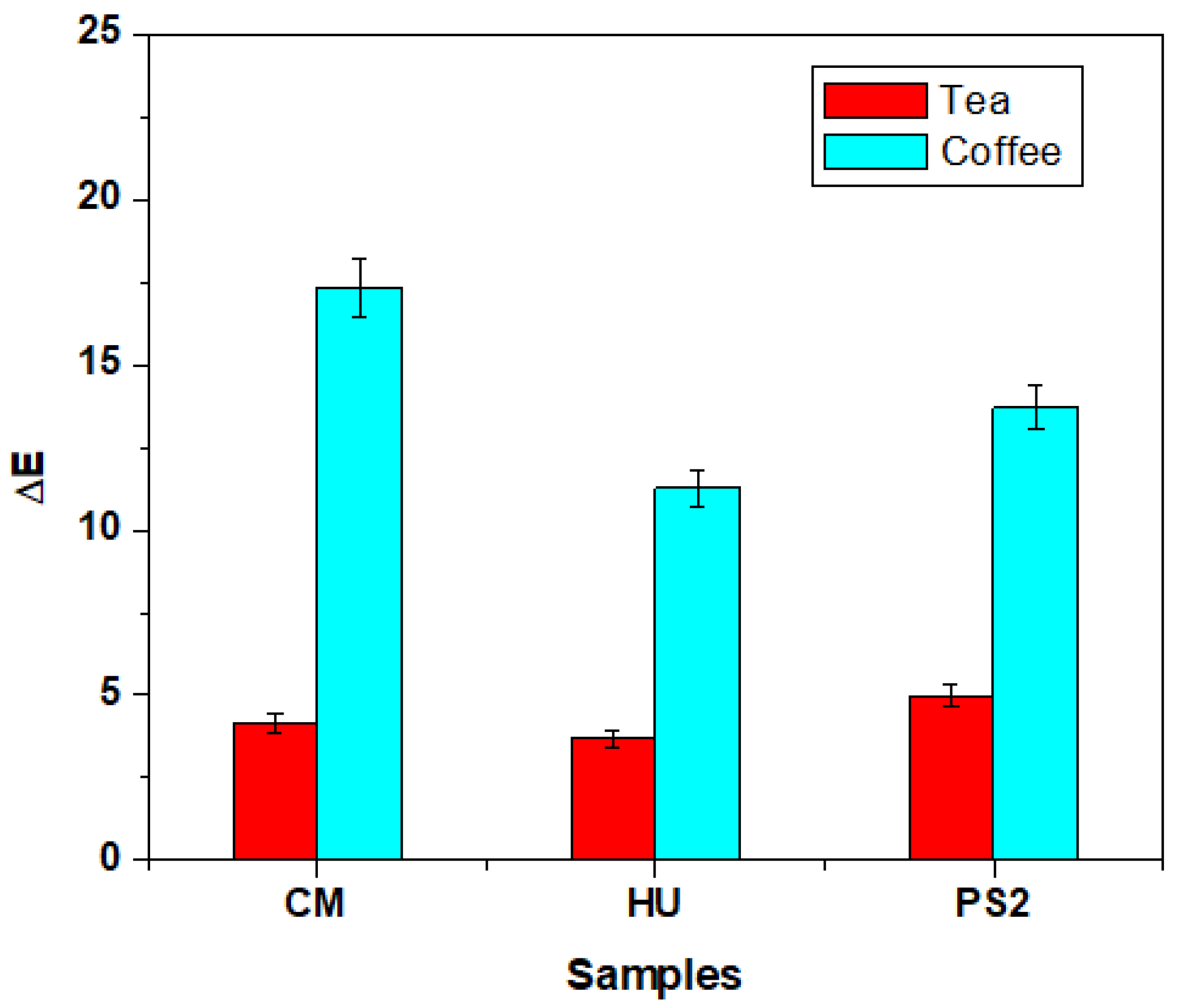
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Salernitano, E.; Migliaresi, C. Composite Materials for Biomedical Applications: A Review. J. Appl. Biomater. Biomech. 2003, 1, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Battistella, E.; Varoni, E.; Cochis, A.; Palazzo, B.; Rimondini, L. Degradable Polymers May Improve Dental Practice. J. Appl. Biomater. Biomech. 2011, 9, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Görkem, U.; Güzel, K.; Sönmez, I. Assessment of monomer release from 3 different fissure sealants. J. Appl. Biomater. Funct. Mater. 2018, 16, 90–96. [Google Scholar] [CrossRef]
- Yao, S.; Qin, L.; Ma, L.; Zhang, X.; Jiang, H.; Zhang, J.; Zhou, C.; Wu, J. Novel antimicrobial and self-healing dental resin to combat secondary caries and restoration fracture. Dent. Mater. 2023, 11, 1040–1050. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.; Meena, A.; Lee, H.-H.; Lee, S.-Y.; Park, S.-J. Tribological behavior of dental resin composites: A comprehensive review. Tribol. Intern. 2023, 190, 109017. [Google Scholar] [CrossRef]
- Korkut, E.; Torlak, E.; Altunsoy, M. Antimicrobial and Mechanical Properties of Dental Resin Composite Containing Bioactive Glass. J. Appl. Biomater. Funct. Mater. 2016, 14, e296–e301. [Google Scholar] [CrossRef]
- Tan, Q.; Li, B.; Wang, Z.; Chen, Q. Investigation of physio-mechanical properties of cross-linked Bis-GMA/TEGDMA dental resins: A molecular dynamics study. Mater. Today Commun. 2023, 35, 105926. [Google Scholar] [CrossRef]
- Monjarás-Ávila, A.J.; Sanchez-Olivares, G.; Calderas, F.; Moreno, L.; Zamarripa-Calderón, J.-E.; Cuevas-Suárez, C.E.; Rivera-Gonzaga, A. Sodium montmorillonite concentration effect on Bis-GMA/TEGDMA resin to prepare clay polymer nanocomposites for dental applications. Appl. Clay Sci. 2020, 196, 105755. [Google Scholar] [CrossRef]
- De Santis, R.; Gloria, A.; Sano, H. Effect of Light Curing and Dark Reaction Phases on the Thermomechanical Properties of a Bis-GMA Based Dental Restorative Material. J. Appl. Biomater. Biomech. 2009, 7, 132–140. [Google Scholar] [CrossRef]
- Barreto Girão, L.; Ohana de Lima Martins, J.; Lemos, J.V.M. Influence of the degree of conversion and Bis-GMA residues of bulk fill resins on tissue toxicity in an subcutaneous model in rats. J. Appl. Biomater. Funct. Mater. 2020, 18. [Google Scholar] [CrossRef]
- Majori, M.; Bedini, R.; Altamura, C.; Filippini, P.; Caiazza, S. Mechanical and Ultrastructural Evaluation of Quartz Post-Endodontic Reconstructions. J. Appl. Biomater. Biomech. 2004, 2, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, E.; Sáenz de Viteri, V.; Igartua, A.; Martinetti, R.; Dolcini, L.; Barandika, G. Structural Characterization and Mechanical Performance of Calcium Phosphate Scaffolds and Natural Bones: A Comparative Study. J. Appl. Biomater. Biomech. 2010, 8, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Lan, T.H.; Chen, Y.-F.; Wang, Y.-Y.; Chou, M.M.C. Evaluation of the Feasibility of NaCaPO4-Blended Zirconia as a New CAD/CAM Material for Dental Restoration. Materials 2021, 14, 3819. [Google Scholar] [CrossRef]
- Hsu, S.-M.; Alsafadi, M.; Vasconez, C.; Fares, C.; Craciun, V.; O’Neill, E.; Ren, F.; Clark, A.; Esquivel-Upshaw, J. Qualitative Analysis of Remineralization Capabilities of Bioactive Glass (NovaMin) and Fluoride on Hydroxyapatite (HA) Discs: An In Vitro Study. Materials 2021, 14, 3813. [Google Scholar] [CrossRef] [PubMed]
- Vallerini, B.F.; Silva, L.D.; Villas-Bôas, M.O.C.; Peitl, O.; Zanotto, E.D.; Pinelli, L.A.P. Microstructure and mechanical properties of an experimental lithium disilicate dental glass-ceramic. Ceram. Int. 2023, 50, 188–196. [Google Scholar] [CrossRef]
- Aminoroaya, A.; Bagheri, R.; Khorasani, S.N.; Talebi, Z.; Derakhshanfar, P.; Neisiany, R.E. Mesoporous silica aerogel reinforced dental composite: Effects of microstructure and surface modification. J. Mechan Behav. Biomed. Mater. 2022, 125, 104947. [Google Scholar] [CrossRef]
- Niu, H.; Yang, D.-L.; Fu, J.-W.; Gao, T.; Wang, J.-X. Mechanical behavior and reinforcement mechanism of nanoparticle cluster fillers in dental resin composites: Simulation and experimental study. Dental Mater. 2022, 38, 1801–1811. [Google Scholar] [CrossRef]
- Elfakhri, F.; Alkahtani, R.; Li, C.; Khaliq, J. Influence of filler characteristics on the performance of dental composites: A comprehensive review. Ceram. Int. 2022, 48, 27280–27294. [Google Scholar] [CrossRef]
- Soares, P.M.; da Rosa, L.S.; Pereira, G.K.R.; Valandro, L.F.; Rippe, M.P.; Piva, A.M.d.O.D.; Feilzer, A.J.; Kleverlaan, C.J.; Tribst, J.P.M. Mechanical Behavior of Repaired Monolithic Crowns: A 3D Finite Element Analysis. Dent. J. 2023, 11, 254. [Google Scholar] [CrossRef]
- Mohamad-Kharib, A.; Chamorro-Petronacci, C.; Pérez-Jardón, A.; Castelo-Baz, P.; Martin-Biedma, B.; Ginzo-Villamayor, M.J.; García-García, A. Staining Susceptibility of Microhybrid and Nanohybrid Composites on Exposure to Different Color Solutions. Appl. Sci. 2023, 13, 11211. [Google Scholar] [CrossRef]
- Mourouzis, P.; Koulaouzidou, E.A.; Palaghias, G.; Helvatjoglu-Antoniades, M. Color Match of Resin Composites to Intact Tooth Structure. J. Appl. Biomater. Funct. Mater. 2015, 13, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Sarosi, C.; Moldovan, M.; Soanca, A.; Roman, A.; Gherman, T.; Trifoi, A.; Chisnoiu, R.M. Effects of monomer composition of urethane methacrylate based resins on the C= C degree of conversion, residual monomer content and mechanical properties. Polymers 2021, 13, 4415. [Google Scholar] [CrossRef] [PubMed]
- Prodan, D.; Moldovan, M.; Chisnoiu, A.M.; Saroși, C.; Cuc, S.; Filip, M.; Cimpean, S.I. Development of New Experimental Dental Enamel Resin Infiltrants-Synthesis and Characterization. Materials 2023, 15, 803. [Google Scholar] [CrossRef]
- Paolone, G.; Mazzitelli, C.; Boggio, F.; Breschi, L.; Vichi, A.; Gherlone, E.; Cantatore, G. Effect of Different Artificial Staining Procedures on the Color Stability and Translucency of a Nano-Hybrid Resin-Based Composite. Materials 2023, 16, 2336. [Google Scholar] [CrossRef] [PubMed]
- Garoushi, S.; Lassila, L.V.J.; Vallittu, P.K. Influence of nanometer scale particulate fillers on some properties of microfilled composite resin. J. Mater. Sci. Mater. Med. 2011, 22, 1645–1651. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, R.; Burrow, M.F.; Tyas, M.J. Surface characteristics of aesthetic restorative materials—An SEM study. J. Oral Rehabil. 2007, 34, 68–76. [Google Scholar] [CrossRef]
- Singh, T.; Mahalakshmi, V.; Sahu, S.; Chatterjee, S.; Khan, A.M.; Haqh, M.F.; Singh, V., Sr. A Comparative Evaluation of the Effect of Different Beverages on Colour Stability and Surface Micromorphology of Nanocomposite Restorative Material. Cureus 2023, 15, e41905. [Google Scholar] [CrossRef]
- Alrefaie, T.; Abdou, A.; Almasabi, W.; Qi, F.; Nakamoto, A.; Nakajima, M.; Otsuki, M.; Shimada, Y. Effect of Water Storage and Bleaching on Light Transmission Properties and Translucency of Nanofilled Flowable Composite. Materials 2023, 16, 10. [Google Scholar] [CrossRef]
- Alagha, E.I. Effect of Different Bonding Techniques on Marginal Adaptation of Harmonize Composite Resin at Different Storage Times. J. Pharm. Bioallied Sci. 2021, 13 (Suppl. S1), S586–S589. [Google Scholar] [CrossRef]
- Murata, M.; Nakanishi, Y.; Kusano, K.; Hirose, Y.; Tazaki, J.; Akazawa, T.; Mizoguchi, I. Immediate Tooth Autotransplantation with Root Canal Filling and Partially Demineralized Dentin/Cementum Matrix into Congenital Missing Tooth Region. J. Funct. Biomater. 2022, 13, 82. [Google Scholar] [CrossRef]
- Păstrav, M.; Moldovan, M.; Chisnoiu, A.; Saroși, C.; Miuța, F.; Păstrav, O.; Chisnoiu, R. Influence of Filler, Monomer Matrix and Silane Coating On Composite Resin Adhesion. Stud. Univ. Babes-Bolyai Chem. 2021, 66, 225–233. [Google Scholar] [CrossRef]
- Ilie, N.; Stawarczyk, B. Efficiency of different repair kits on bonding to aged dental resin composite substrates. Int. J. Adhes. Adhes. 2015, 58, 7–12. [Google Scholar] [CrossRef]
- Alhotan, A.; Raszewski, Z.; Alamoush, R.A.; Chojnacka, K.; Mikulewicz, M.; Haider, J. Influence of Storing Composite Filling Materials in a Low-pH Artificial Saliva on Their Mechanical Properties—An In Vitro Study. J. Funct. Biomater. 2023, 14, 328. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.; Chung, S.H.; Um, S.-H.; Rhee, S.-H. Effect of grain size of dental zirconia on shear bond strength of composite resin cement. J. Appl. Biomater. Funct. Mater. 2020, 18. [Google Scholar] [CrossRef]
- Salari, S.; Dadkan, S.; Khakbiz, M.; Atai, M. Effect of nanoparticles on surface characteristics of dental nanocomposite. Med. Devices Sens. 2020, 3, e10081. [Google Scholar] [CrossRef]
- Faris, T.M.; Abdulrahim, R.H.; Mahmood, M.A.; Mhammed Dalloo, G.A.; Gul, S.S. In vitro evaluation of dental color stability using various aesthetic restorative materials after immersion in different drinks. BMC Oral Health 2023, 23, 49. [Google Scholar] [CrossRef]
- Allccahuaman-Avalos, R.; Medina-Sánchez, R.; Castro-Ramirez, L.; Ladera-Castañeda, M.; Cervantes-Ganoza, L.; Martínez-Campos, R.; Cayo-Rojas, C. In Vitro Color Stability Evaluation of Three Polished and Unpolished Nanohybrid Resin Composites Immersed in a 0.12% Chlorhexidine-Based Mouthwash at Different Times. Polymers 2023, 15, 1339. [Google Scholar] [CrossRef]
- Nuaimi, H.O.; Ragab, H.M. Effect of aggressive beverage on the color stability of different nano-hybrid resin based composite. Eur. J. Gen. Dent. 2014, 3, 190–193. [Google Scholar] [CrossRef]
- Al-Shami, A.M.; Alshami, M.A.; Al-Kholani, A.I. Color stability of nanohybrid and microhybrid composites after immersion in common coloring beverages at different times: A laboratory study. BDJ Open 2023, 9, 39. [Google Scholar] [CrossRef]
- Meshki, R.; Rashidi, M. Effect of natural and commercially produced juices on colour stability of microhybrid and nanohybrid composites. BDJ Open 2022, 8, 11. [Google Scholar] [CrossRef]
- Nasim, I.; Neelakantan, P.; Sujeer, R.; Subbarao, C.V. Color stability ofmicrofilled, microhybrid and nanocomposite resins—An in vitro study. J. Dent. 2010, 38, e137–e142. [Google Scholar] [CrossRef] [PubMed]
- Heintze, S.D.; Forjanic, M.; Ohmiti, K.; Rousson, V. Surface deterioration of dental materials after simulated toothbrushing in relation to brushing time and load. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2010, 26, 306–319. [Google Scholar] [CrossRef] [PubMed]
- Lainović, T.; Blažić, L.; Kukuruzović, D.; Vilotic, M.; Ivanisevic, A.; Kakaš, D. Effect of diamond paste finishing on surface topography and roughness of dental nanohybrid composites–AFM analysis. Procedia Eng. 2014, 69, 945–951. [Google Scholar] [CrossRef]
- Takahashi, R.; Jin, J.; Nikaido, T.; Tagami, J.; Hickel, R.; Kunzelmann, K.-H. Surface characterization of current composites after toothbrush abrasion. Dent. Mater. J. 2013, 32, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Aytac, F.; Karaarslan, E.S.; Agaccioglu, M.; Tastan, E.; Buldur, M.; Kuyucu, E. Effects of novel finishing and polishing systems on surface roughness and morphology of nanocomposites. J. Esthet. Restor. Dent. 2016, 28, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Gallo, S.; Poggio, C.; Ricaldone, V.; Arciola, C.R.; Scribante, A. New Resin-Based Bulk-Fill Composites: In vitro Evaluation of Micro-Hardness and Depth of Cure as Infection Risk Indexes. Materials 2020, 13, 1308. [Google Scholar] [CrossRef]
- Peker, O.; Bolgul, B. Evaluation of surface roughness and color changes of restorative materials used with different polishing procedures in pediatric dentistry. J. Clin. Pediatr. Dent. 2023, 47, 72–79. [Google Scholar] [CrossRef]

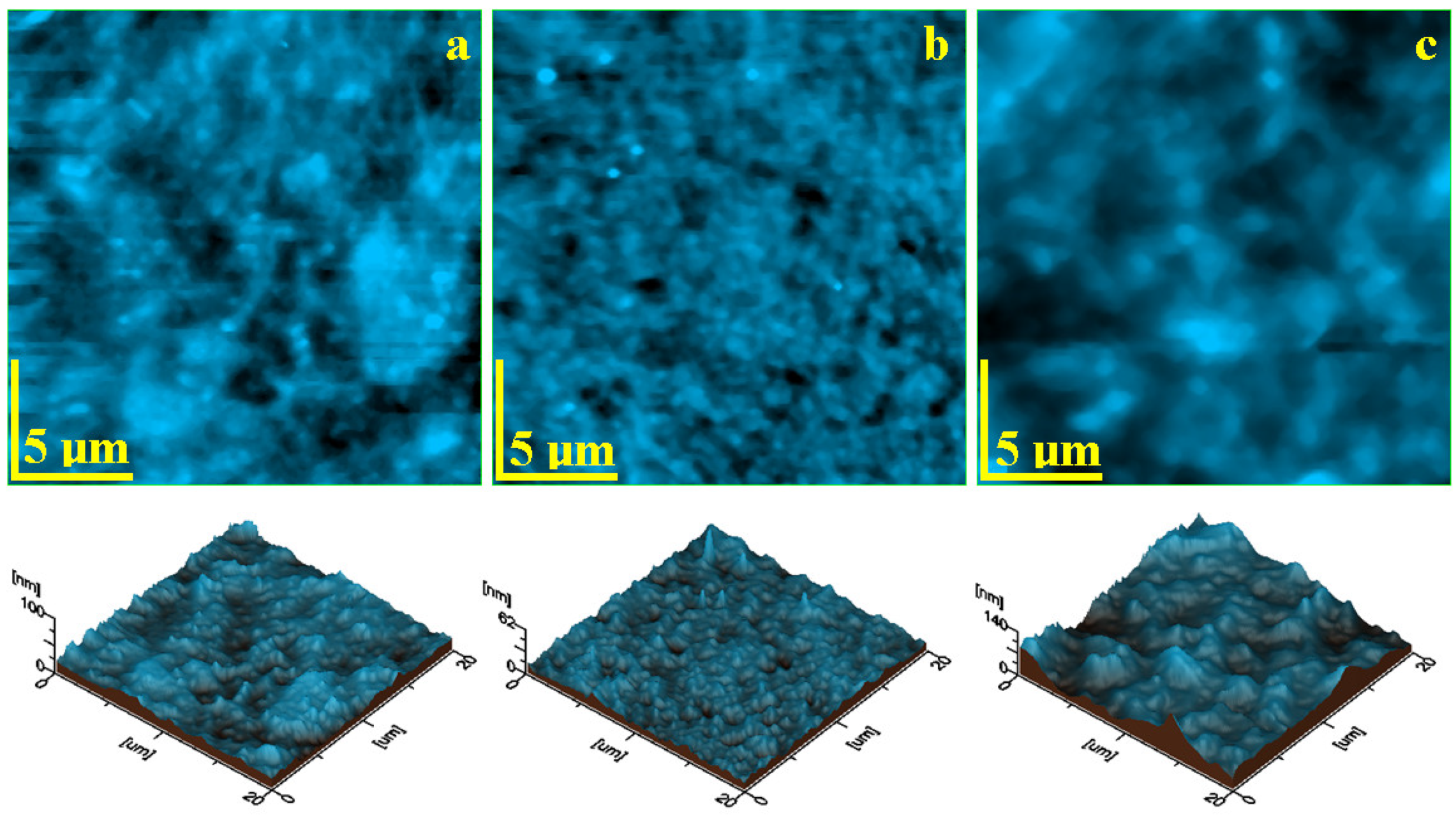
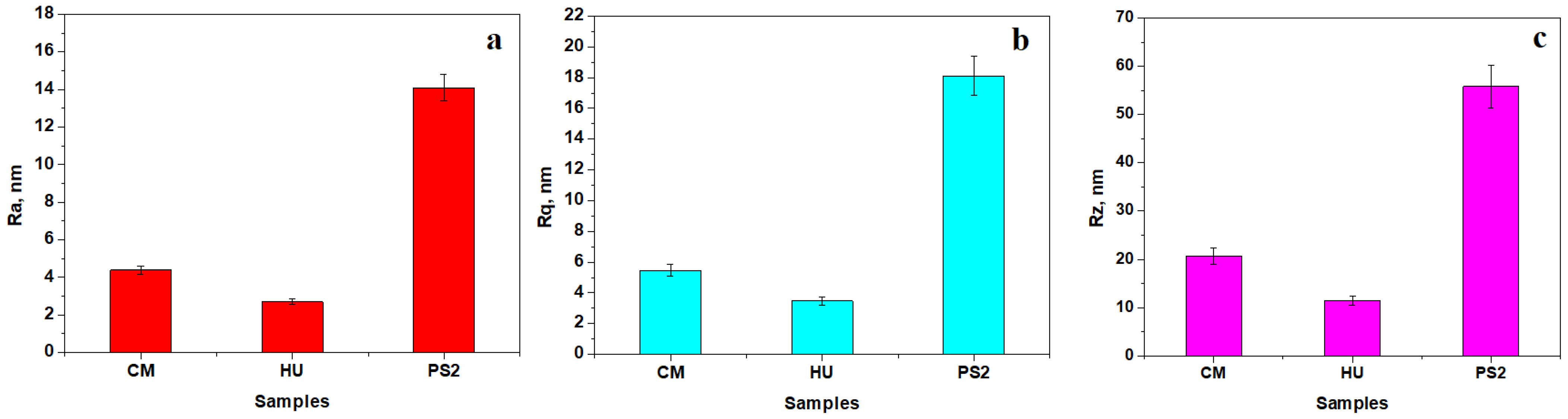
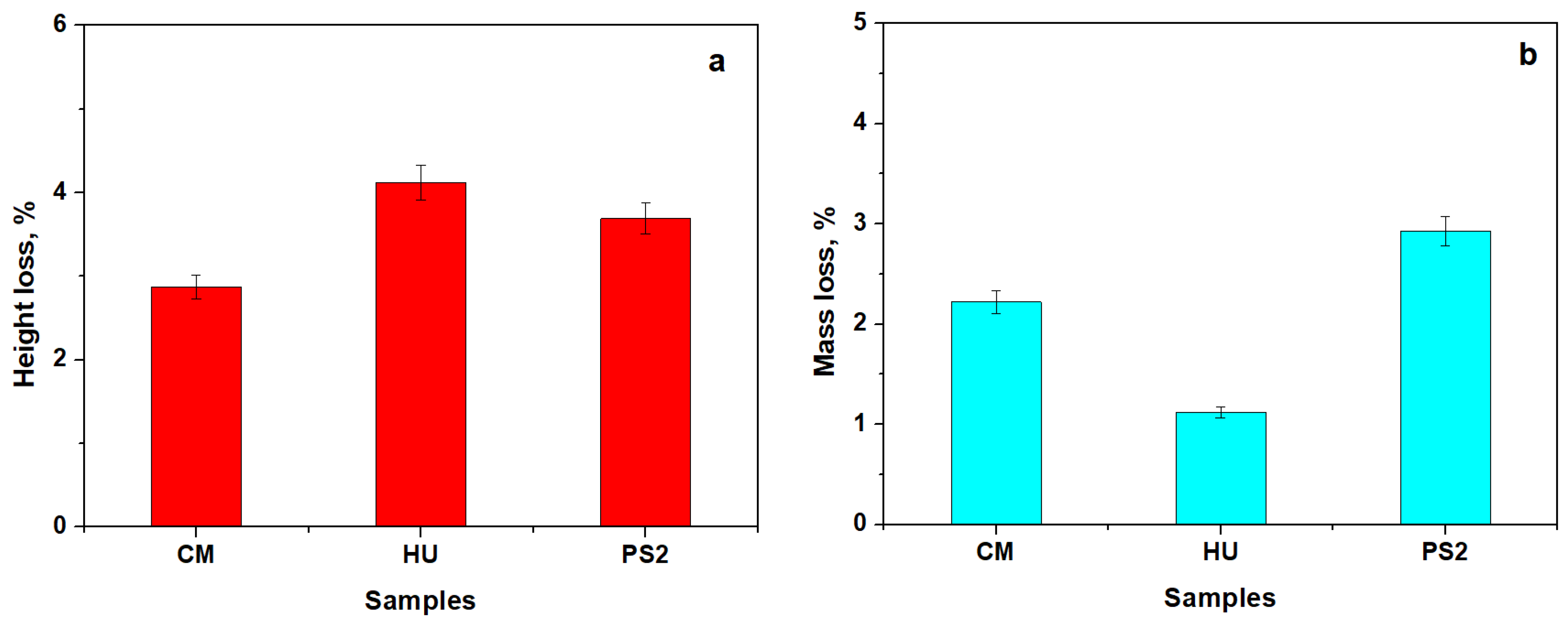
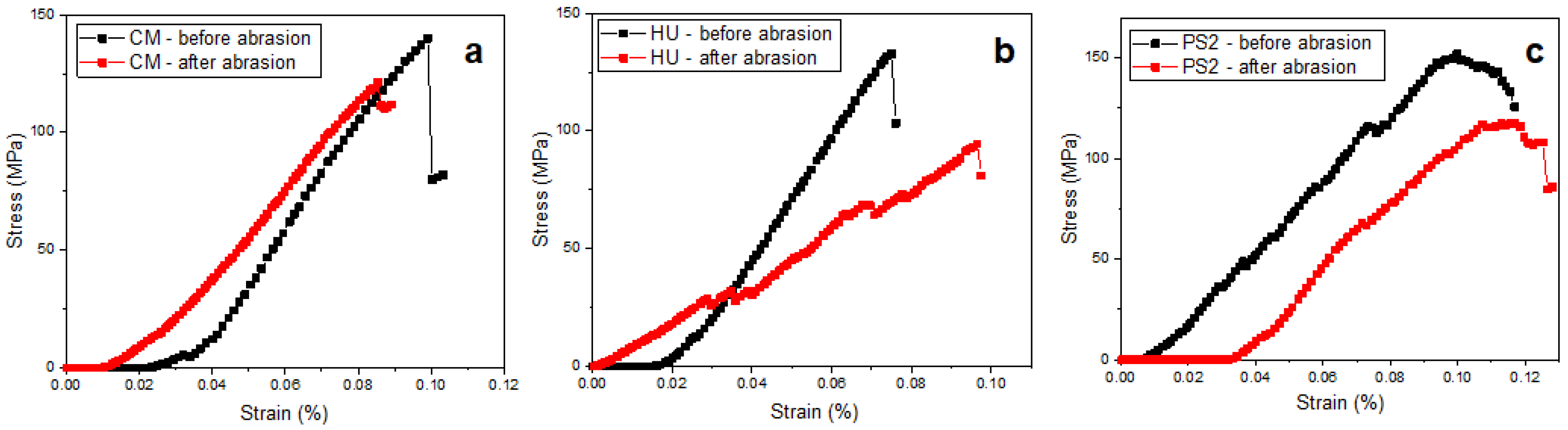

| Code | Material | Type | Matrix | Filler |
|---|---|---|---|---|
| HU | Harmonize Universal (Kerr Ltd., Uxbridge, UK) | Nanohybrid spherical | Bis-GMA, TEGDMA, Bis-EMA | 5–400 nm particle size. Spherical zirconia and silica nanoparticles, rheological modifier. |
| CM | Clear Fil Majesty ES-2 (Kuraray Noritake Dental Inc., Nagoya, Japan) | Nanohybrid | Bis-GMA, hydrophobic aromatic DMA, and hydrophobic aliphatic DMA, dl-camphorquinone | Silanated barium glass (particle size 0.37–1.5 μm) and pre-polymerized organic filler. |
| PS2 | Experimental composite resin | Nanohybrid | Bis-GMA, UDMA, TEGDMA | 40–60 nm particle size. HA-Zn, colloidal silica, barium glasses, lantan glasses |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Păstrav, M.; Păstrav, O.; Chisnoiu, A.M.; Chisnoiu, R.M.; Cuc, S.; Petean, I.; Saroși, C.; Feștilă, D. Properties of Nanohybrid Dental Composites—A Comparative In Vitro Study. Biomedicines 2024, 12, 243. https://doi.org/10.3390/biomedicines12010243
Păstrav M, Păstrav O, Chisnoiu AM, Chisnoiu RM, Cuc S, Petean I, Saroși C, Feștilă D. Properties of Nanohybrid Dental Composites—A Comparative In Vitro Study. Biomedicines. 2024; 12(1):243. https://doi.org/10.3390/biomedicines12010243
Chicago/Turabian StylePăstrav, Mihaela, Ovidiu Păstrav, Andrea Maria Chisnoiu, Radu Marcel Chisnoiu, Stanca Cuc, Ioan Petean, Codruța Saroși, and Dana Feștilă. 2024. "Properties of Nanohybrid Dental Composites—A Comparative In Vitro Study" Biomedicines 12, no. 1: 243. https://doi.org/10.3390/biomedicines12010243
APA StylePăstrav, M., Păstrav, O., Chisnoiu, A. M., Chisnoiu, R. M., Cuc, S., Petean, I., Saroși, C., & Feștilă, D. (2024). Properties of Nanohybrid Dental Composites—A Comparative In Vitro Study. Biomedicines, 12(1), 243. https://doi.org/10.3390/biomedicines12010243









