Immunosuppression in Sepsis: Biomarkers and Specialized Pro-Resolving Mediators
Abstract
1. Introduction
2. Methods
2.1. Search Term Strategy
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Data Extraction
2.5. Data Analysis
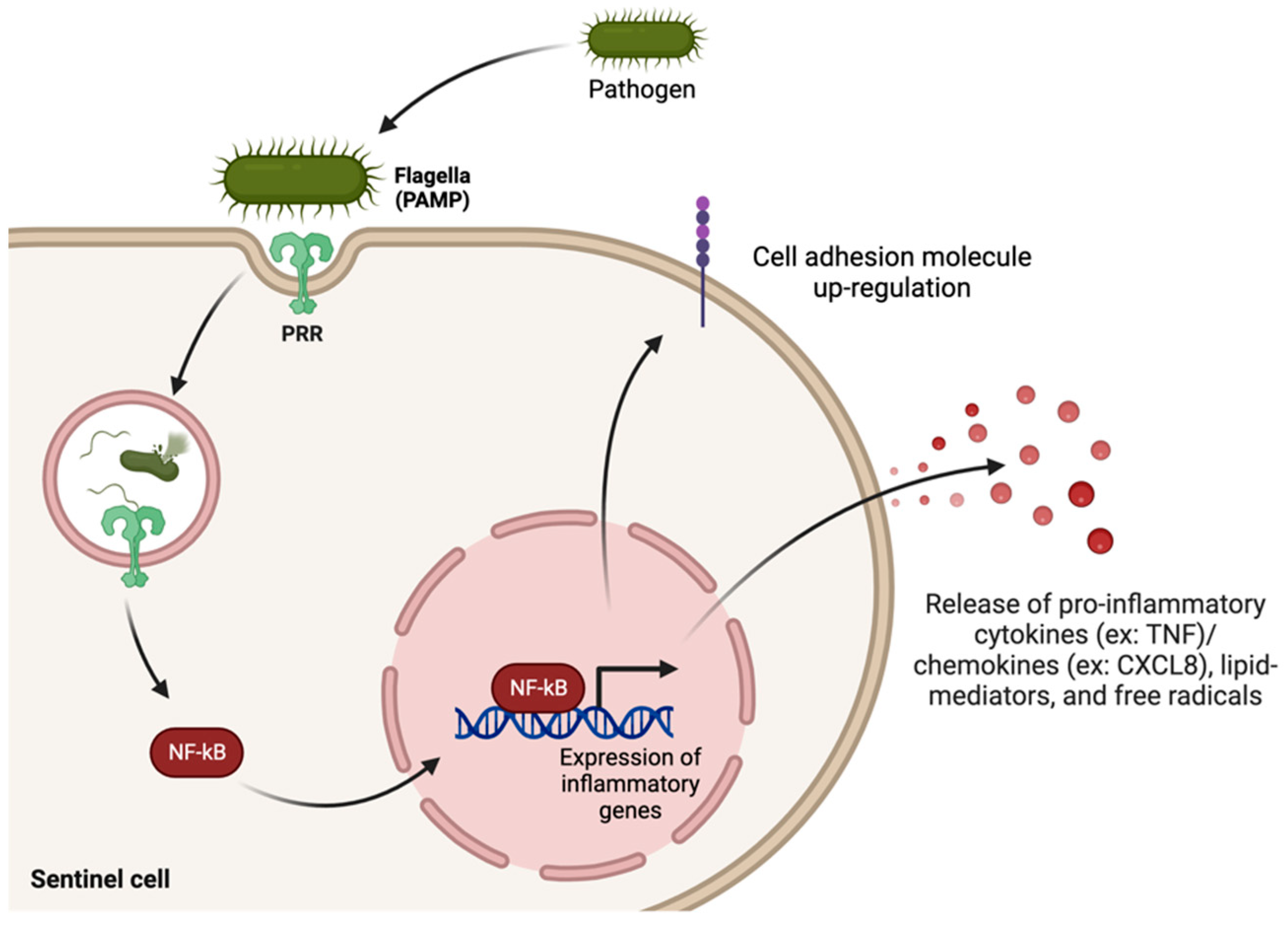
3. The Innate Immune System and Inflammatory Response in Infection
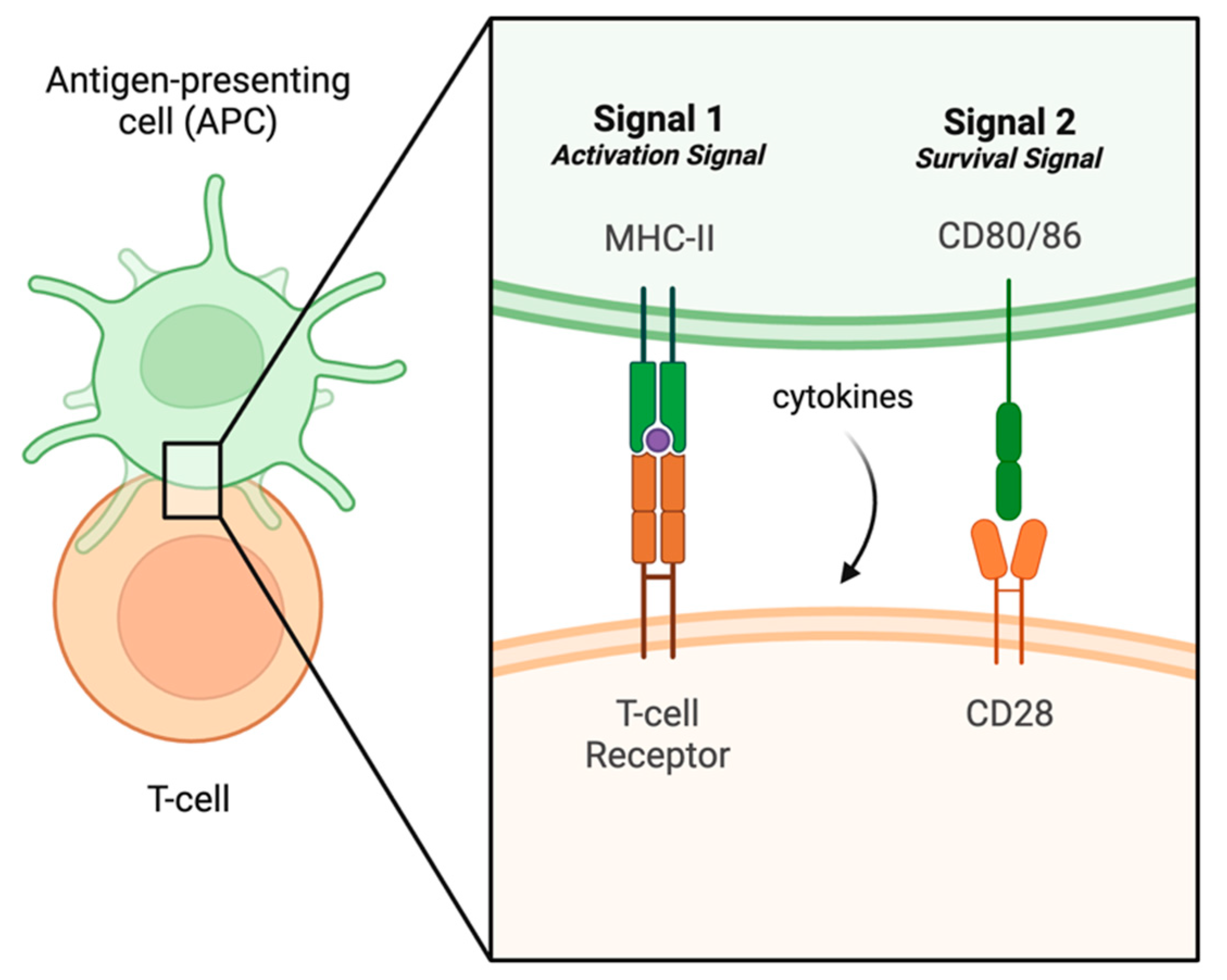
4. The Adaptive Immune System and Response to Infection
5. Inflammation Resolution
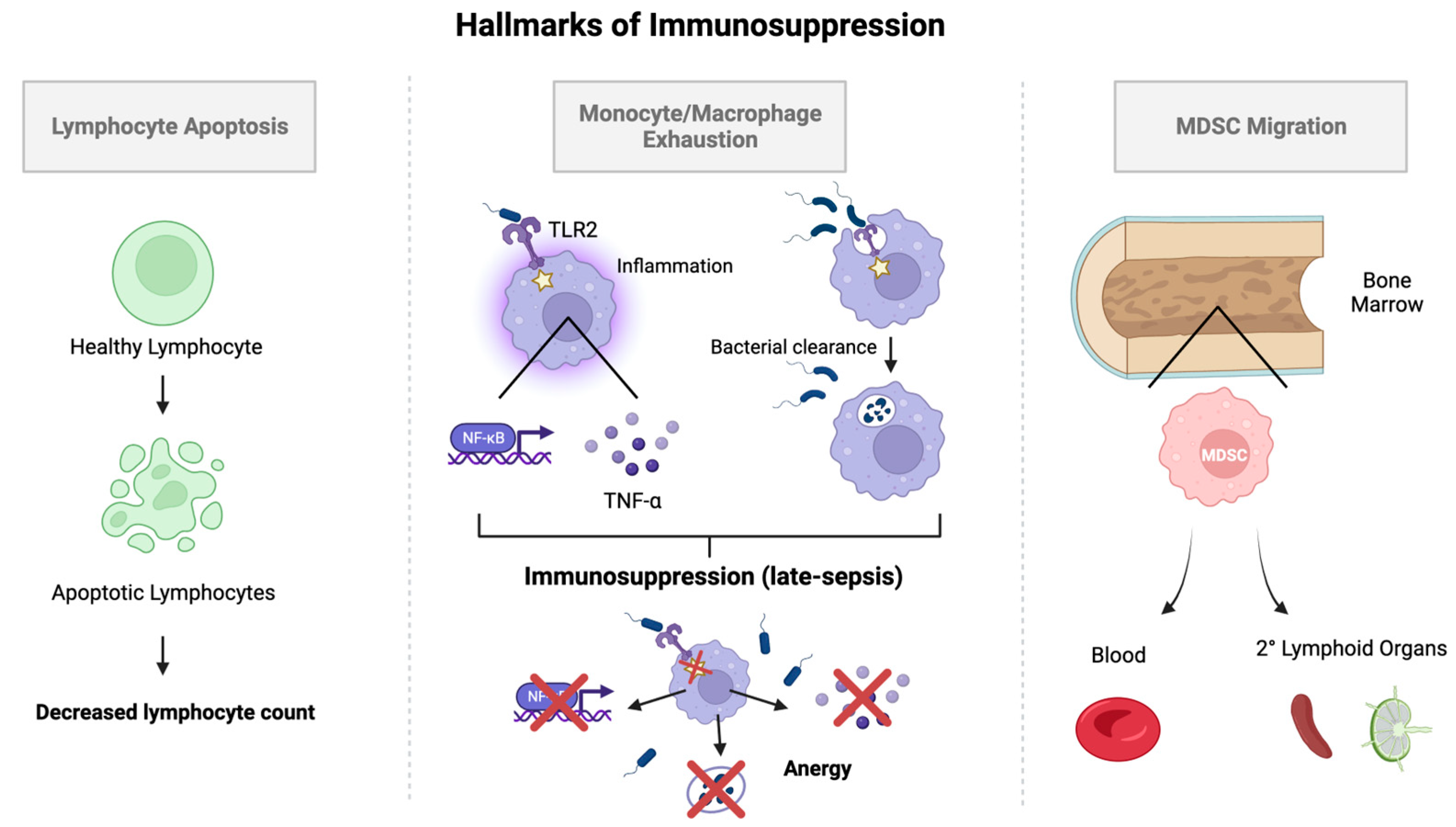
6. Mechanisms of Immunosuppression in Sepsis
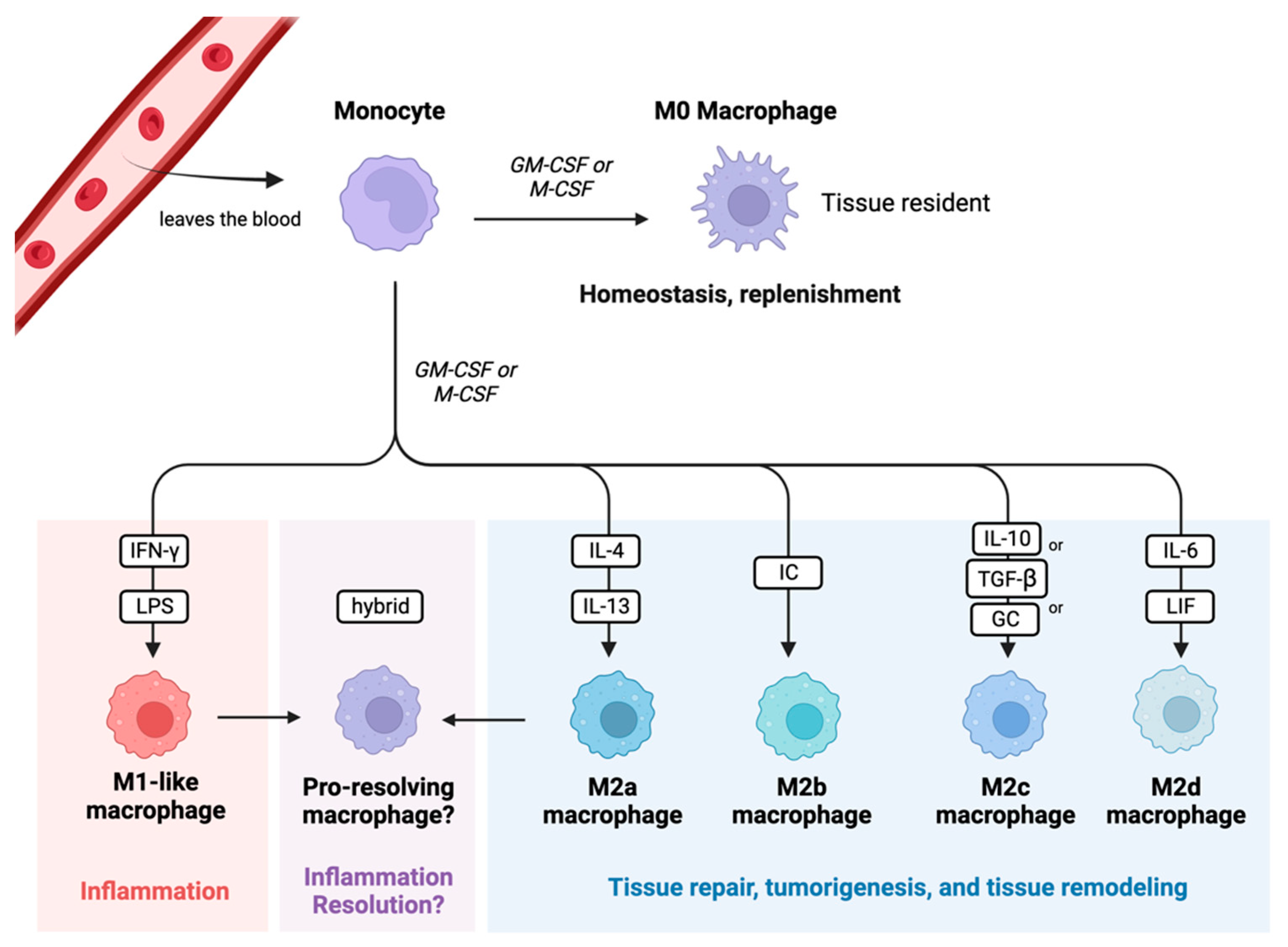
6.1. Monocyte/Macrophage Exhaustion
6.1.1. PD-1 and PDL-1
6.1.2. Exhausted Monocytes/Macrophages
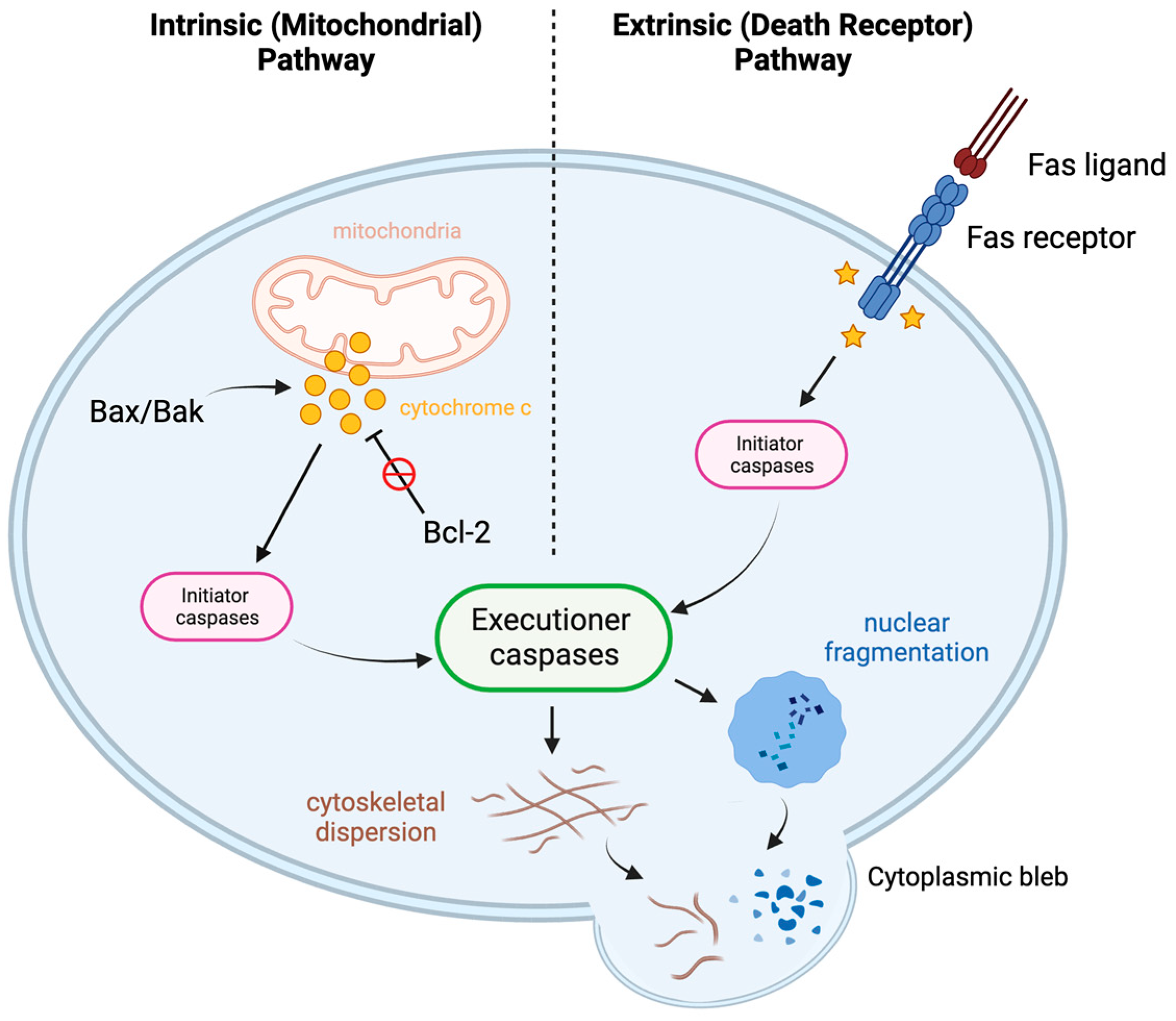
6.2. Lymphocyte Apoptosis
6.3. Myeloid-Derived Suppressor Cells
7. Biomarkers
7.1. IL-10
7.2. GPR18
7.3. HLA-DR
| Biomarker | Role and Correlation with Outcomes Due to Late Sepsis | References |
|---|---|---|
| Decreased lymphocyte count | Late-septic patients are reported to have a decreased lymphocyte count compared with non-septic patients. The decrease is thought to be contributed to inappropriate activation of apoptosis pathways. This biomarker is correlated with a higher likelihood of developing secondary infections. | [3,51,89,91,92,96,97] |
| Increased MDSC production/ migration | Late-septic patients have higher levels of MDSCs in the blood and secondary lymphoid organs compared with non-septic patients. While the purpose of increased MDSC production remains controversial, these cells are thought to contribute to worse outcomes due to sepsis due to their immunosuppressive and anti-inflammatory nature. | [3,98,103,104,106,107,108,109,110,111,112,136,137,138] |
| Increased IL-10 | Late-septic patients have elevated levels of IL-10 compared with non-septic patients. Elevated anti-inflammatory responses triggered by IL-10 suggests that the body will not be able to mount an inflammatory response if the patient does develop a secondary infection. This biomarker is correlated with worse severity and worse clinical outcomes due to sepsis. | [3,127,128] |
| GPR18 expression | Late-septic patients have changes in GPR18 expression on their immune cells compared with non-septic patients. Changes to GPR18 expression suggests that there is decreased specialized pro-resolving mediator (SPM) bioavailability and uncoupled inflammation resolution circuits. Increased GPR18 expression on monocytes and decreased GPR18 expression on neutrophils is correlated with an increase in mortality due to sepsis. | [130,131,132] |
| Decreased HLA-DR expression | Late-septic patients have 70% less HLA-DR expression compared with non-septic patients. Decreased HLA-DR expression suggests that the immune system will be unable to activate T-cell-mediated adaptive immune responses if the patient develops a secondary infection. This biomarker is correlated with worse sequential organ failure assessment (SOFA) scores due to sepsis. | [3,70,134,135] |
8. Specialized Pro-Resolving Mediators (SPMs) and Immunosuppression in Sepsis
9. Summary and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SPMs | specialized pro-resolving mediators |
| MDSCs | myeloid-derived suppressor cells |
| CRS | cytokine release syndrome |
| ILCs | innate lymphoid cells |
| PAMPs | pathogen-associated molecular patterns |
| PRRs | pattern recognition receptors |
| iPRRs | inhibitory pattern recognition receptors |
| LPS | lipopolysaccharide |
| NF-κB | nuclear factor kappa light chain enhancer of activated B cells |
| APC | antigen-presenting cell |
| TCR | T-cell receptor |
| IFN-γ | interferon gamma |
| TNF-α | tumor necrosis factor alpha |
| TLR2 | Toll-like receptor 2 |
| IL-4 | interleukin 4 |
| IL-6 | interleukin 6 |
| IL-7 | interleukin 7 |
| IL-8 | interleukin 8 |
| IL-10 | interleukin 10 |
| IL-13 | interleukin 13 |
| GM-CSF | granulocyte macrophage colony-stimulating factor |
| M-CSF | macrophage colony-stimulating factor |
| ICs | immune complexes |
| GCs | glucocorticoids |
| LIF | leukemia inhibitory factor |
| CTLA-4 | cytotoxic T-lymphocyte-associated protein 4 |
| PD-1 | programmed cell death protein 1 |
| PD-L1 | programmed death ligand 1 |
| CLP | cecal ligation and puncture |
| HLA-DR | human leukocyte antigen DR isotype |
| ET | endotoxin tolerance |
| HMGB1 | high mobility group box 1 |
| ROS | reactive oxygen species |
| Bcl-2 | B cell lymphoma 2 |
| FasL | Fas ligand |
| Bax | Bcl-2-associated X protein |
| Bak | Bcl-2 antagonist killer 1 |
| LTx | lethal toxin |
| TUNEL | transferase-mediated dUTP nick-end labeling |
| G-CSF | granulocyte colony-stimulating factor |
| CXCL2 | chemokine ligand 2 |
| ICU | intensive care unit |
| M-MDSCs | monocytic MDSCs |
| PMN-MDSCs | polymorphic MDSCs |
| GPR18 | G-protein-coupled receptor 18 |
| RvD1 | resolvin D1 |
| RvD2 | resolvin D2 |
| PTX3 | pentraxin 3 |
| HLA | human leukocyte antigen |
| MHC | major histocompatibility complex |
| APC | antigen-presenting cell |
| SOFA | sequential organ failure assessment |
| AA | arachidonic acid |
| EPA | eicosapentaenoic acid |
| DHA | docosahexaenoic acid |
References
- Athale, J.; Busch, L.M.; O’Grady, N.P. Cytokine Release Syndrome and Sepsis. Infect. Dis. Clin. N. Am. 2022, 36, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Huang, S.-Y.; Sun, J.-H.; Zhang, H.-C.; Cai, Q.-L.; Gao, C.; Li, L.; Cao, J.; Xu, F.; Zhou, Y.; et al. Sepsis-Induced Immunosuppression: Mechanisms, Diagnosis and Current Treatment Options. Mil. Med. Res. 2022, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.C. Why Have Clinical Trials in Sepsis Failed? Trends Mol. Med. 2014, 20, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Esposito, R. Tools to Study Adaptive and Innate Immune Response 2021. Available online: https://www.enzolifesciences.com/science-center/technotes/2021/may/tools-to-study-adaptive-and-innate-immune-response/ (accessed on 20 December 2023).
- Chaplin, D.D. Overview of the Immune Response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef]
- Mazzurana, L.; Rao, A.; Van Acker, A.; Mjösberg, J. The Roles for Innate Lymphoid Cells in the Human Immune System. Semin. Immunopathol. 2018, 40, 407–419. [Google Scholar] [CrossRef]
- Janeway, C.J.; Travers, P.; Walport, M. Principles of Innate and Adaptive Immunity. In Immunobiology: The Immune System in Health and Disease; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Zigterman, B.G.R.; Dubois, L. Inflammation and Infection: Cellular and Biochemical Processes. Ned. Tijdschr. Voor Tandheelkd. 2022, 129, 125–129. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204–7218. [Google Scholar] [CrossRef]
- Nedeva, C. Inflammation and Cell Death of the Innate and Adaptive Immune System during Sepsis. Biomolecules 2021, 11, 1011. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Horwitz, D.A.; Fahmy, T.M.; Piccirillo, C.A.; La Cava, A. Rebalancing Immune Homeostasis to Treat Autoimmune Diseases. Trends Immunol. 2019, 40, 888–908. [Google Scholar] [CrossRef] [PubMed]
- Kourtzelis, I.; Hajishengallis, G.; Chavakis, T. Phagocytosis of Apoptotic Cells in Resolution of Inflammation. Front. Immunol. 2020, 11, 553. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Savill, J. Resolution of Inflammation: The Beginning Programs the End. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Rumpret, M.; von Richthofen, H.J.; Peperzak, V.; Meyaard, L. Inhibitory Pattern Recognition Receptors. J. Exp. Med. 2022, 219, e20211463. [Google Scholar] [CrossRef] [PubMed]
- Jendro, M.; Goronzy, J.J.; Weyand, C.M. Structural and Functional Characterization of Hla-Dr Molecules Circulating in the Serum. Autoimmunity 1991, 8, 289–296. [Google Scholar] [CrossRef]
- Cheadle, W.G. The Human Leukocyte Antigens and Their Relationship to Infection. Am. J. Surg. 1993, 165, 75S–81S. [Google Scholar] [CrossRef]
- Petersone, L.; Edner, N.M.; Ovcinnikovs, V.; Heuts, F.; Ross, E.M.; Ntavli, E.; Wang, C.J.; Walker, L.S.K. T Cell/B Cell Collaboration and Autoimmunity: An Intimate Relationship. Front. Immunol. 2018, 9, 1941. [Google Scholar] [CrossRef]
- Xing, Y.; Hogquist, K.A. T-Cell Tolerance: Central and Peripheral. Cold Spring Harb. Perspect. Biol. 2012, 4, a006957. [Google Scholar] [CrossRef]
- Tai, Y.; Wang, Q.; Korner, H.; Zhang, L.; Wei, W. Molecular Mechanisms of T Cells Activation by Dendritic Cells in Autoimmune Diseases. Front. Pharmacol. 2018, 9, 642. [Google Scholar] [CrossRef]
- Sharma, P.; Allison, J.P. The Future of Immune Checkpoint Therapy. Science 2015, 348, 56–61. [Google Scholar] [CrossRef]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Barnig, C.; Bezema, T.; Calder, P.C.; Charloux, A.; Frossard, N.; Garssen, J.; Haworth, O.; Dilevskaya, K.; Levi-Schaffer, F.; Lonsdorfer, E.; et al. Activation of Resolution Pathways to Prevent and Fight Chronic Inflammation: Lessons From Asthma and Inflammatory Bowel Disease. Front. Immunol. 2019, 10, 1699. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Clish, C.B.; Brannon, J.; Colgan, S.P.; Chiang, N.; Gronert, K. Novel Functional Sets of Lipid-Derived Mediators with Antiinflammatory Actions Generated from Omega-3 Fatty Acids via Cyclooxygenase 2–Nonsteroidal Antiinflammatory Drugs and Transcellular Processing. J. Exp. Med. 2000, 192, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, L.; Yu, C.; Yang, X.-F.; Wang, H. Monocyte and Macrophage Differentiation: Circulation Inflammatory Monocyte as Biomarker for Inflammatory Diseases. Biomark. Res. 2014, 2, 1. [Google Scholar] [CrossRef]
- Lam, R.S.; O’Brien-Simpson, N.M.; Holden, J.A.; Lenzo, J.C.; Fong, S.B.; Reynolds, E.C. Unprimed, M1 and M2 Macrophages Differentially Interact with Porphyromonas Gingivalis. PLoS ONE 2016, 11, e0158629. [Google Scholar] [CrossRef] [PubMed]
- Rőszer, T. Understanding the Mysterious M2 Macrophage through Activation Markers and Effector Mechanisms. Mediat. Inflamm. 2015, 2015, 816460. [Google Scholar] [CrossRef]
- Strizova, Z.; Benesova, I.; Bartolini, R.; Novysedlak, R.; Cecrdlova, E.; Foley, L.K.; Striz, I. M1/M2 Macrophages and Their Overlaps—Myth or Reality? Clin. Sci. 2023, 137, 1067–1093. [Google Scholar] [CrossRef]
- Watanabe, S.; Alexander, M.; Misharin, A.V.; Budinger, G.R.S. The Role of Macrophages in the Resolution of Inflammation. J. Clin. Investig. 2019, 129, 2619–2628. [Google Scholar] [CrossRef]
- Sica, A.; Mantovani, A. Macrophage Plasticity and Polarization: In Vivo Veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, X.-H.; Jin, L. Macrophage Polarization in Physiological and Pathological Pregnancy. Front. Immunol. 2019, 10, 792. [Google Scholar] [CrossRef]
- Zhang, Q.; Sioud, M. Tumor-Associated Macrophage Subsets: Shaping Polarization and Targeting. Int. J. Mol. Sci. 2023, 24, 7493. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Mildner, A.; Yona, S. Developmental and Functional Heterogeneity of Monocytes. Immunity 2018, 49, 595–613. [Google Scholar] [CrossRef] [PubMed]
- Nahrendorf, M.; Swirski, F.K. Abandoning M1/M2 for a Network Model of Macrophage Function. Circ. Res. 2016, 119, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Schmidt, S.V.; Sander, J.; Draffehn, A.; Krebs, W.; Quester, I.; De Nardo, D.; Gohel, T.D.; Emde, M.; Schmidleithner, L.; et al. Transcriptome-Based Network Analysis Reveals a Spectrum Model of Human Macrophage Activation. Immunity 2014, 40, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Saas, P.; Chagué, C.; Maraux, M.; Cherrier, T. Toward the Characterization of Human Pro-Resolving Macrophages? Front. Immunol. 2020, 11, 593300. [Google Scholar] [CrossRef] [PubMed]
- Vannella, K.M.; Wynn, T.A. Mechanisms of Organ Injury and Repair by Macrophages. Annu. Rev. Physiol. 2017, 79, 593–617. [Google Scholar] [CrossRef] [PubMed]
- Bah, A.; Vergne, I. Macrophage Autophagy and Bacterial Infections. Front. Immunol. 2017, 8, 1483. [Google Scholar] [CrossRef]
- Cronan, M.R.; Beerman, R.W.; Rosenberg, A.F.; Saelens, J.W.; Johnson, M.G.; Oehlers, S.H.; Sisk, D.M.; Jurcic Smith, K.L.; Medvitz, N.A.; Miller, S.E.; et al. Macrophage Epithelial Reprogramming Underlies Mycobacterial Granuloma Formation and Promotes Infection. Immunity 2016, 45, 861–876. [Google Scholar] [CrossRef]
- Linke, M.; Pham, H.T.T.; Katholnig, K.; Schnöller, T.; Miller, A.; Demel, F.; Schütz, B.; Rosner, M.; Kovacic, B.; Sukhbaatar, N.; et al. Chronic Signaling via the Metabolic Checkpoint Kinase mTORC1 Induces Macrophage Granuloma Formation and Marks Sarcoidosis Progression. Nat. Immunol. 2017, 18, 293–302. [Google Scholar] [CrossRef]
- Zhang, T.; Yu-Jing, L.; Ma, T. Role of Regulation of PD-1 and PD-L1 Expression in Sepsis. Front. Immunol. 2023, 14, 1029438. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, J.; Deng, X.; Xiong, F.; Ge, J.; Xiang, B.; Wu, X.; Ma, J.; Zhou, M.; Li, X.; et al. Role of the Tumor Microenvironment in PD-L1/PD-1-Mediated Tumor Immune Escape. Mol. Cancer 2019, 18, 10. [Google Scholar] [CrossRef] [PubMed]
- Alsaab, H.O.; Sau, S.; Alzhrani, R.; Tatiparti, K.; Bhise, K.; Kashaw, S.K.; Iyer, A.K. PD-1 and PD-L1 Checkpoint Signaling Inhibition for Cancer Immunotherapy: Mechanism, Combinations, and Clinical Outcome. Front. Pharmacol. 2017, 8, 561. [Google Scholar] [CrossRef] [PubMed]
- Karwacz, K.; Bricogne, C.; MacDonald, D.; Arce, F.; Bennett, C.L.; Collins, M.; Escors, D. PD-L1 Co-stimulation Contributes to Ligand-induced T Cell Receptor Down-modulation on CD8 + T Cells. EMBO Mol. Med. 2011, 3, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Yuan, P.; Wu, H.; Chen, J.; Fu, J.; Li, P.; Lu, J.; Wei, W. Dendritic Cells with an Increased PD-L1 by TGF-β Induce T Cell Anergy for the Cytotoxicity of Hepatocellular Carcinoma Cells. Int. Immunopharmacol. 2014, 20, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhou, Y.; Ding, Q.; Jiao, Z.; Lu, L.; Yang, N.; Ma, Y.; Chou, K.-Y. High Level Expression of B7H1 Molecules by Keratinocytes Suppresses Xeno- and Allo-Reactions by Inducing Type I Regulatory T Cells. Transplant. Immunol. 2009, 21, 192–197. [Google Scholar] [CrossRef]
- Han, Y.; Liu, D.; Li, L. PD-1/PD-L1 Pathway: Current Researches in Cancer. Am. J. Cancer Res. 2020, 10, 727–742. [Google Scholar]
- Pardoll, D.M. The Blockade of Immune Checkpoints in Cancer Immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Colston, E.; Yende, S.; Crouser, E.D.; Martin, G.S.; Albertson, T.; Bartz, R.R.; Brakenridge, S.C.; Delano, M.J.; Park, P.K.; et al. Immune Checkpoint Inhibition in Sepsis: A Phase 1b Randomized Study to Evaluate the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of Nivolumab. Intensive Care Med. 2019, 45, 1360–1371. [Google Scholar] [CrossRef]
- Yang, L.; Gao, Q.; Li, Q.; Guo, S. PD-L1 Blockade Improves Survival in Sepsis by Reversing Monocyte Dysfunction and Immune Disorder. Inflammation 2023, 1–15. [Google Scholar] [CrossRef]
- Ruan, W.-S.; Feng, M.-X.; Xu, J.; Xu, Y.-G.; Song, C.-Y.; Lin, L.-Y.; Li, L.; Lu, Y.-Q. Early Activation of Myeloid-Derived Suppressor Cells Participate in Sepsis-Induced Immune Suppression via PD-L1/PD-1 Axis. Front. Immunol. 2020, 11, 1299. [Google Scholar] [CrossRef]
- Patera, A.C.; Drewry, A.M.; Chang, K.; Beiter, E.R.; Osborne, D.; Hotchkiss, R.S. Frontline Science: Defects in Immune Function in Patients with Sepsis Are Associated with PD-1 or PD-L1 Expression and Can Be Restored by Antibodies Targeting PD-1 or PD-L1. J. Leukoc. Biol. 2016, 100, 1239–1254. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.K.; Zhao, Y.; Singer, M.; Spencer, J.; Shankar-Hari, M. Lymphocyte Subset Expression and Serum Concentrations of PD-1/PD-L1 in Sepsis—Pilot Study. Crit. Care 2018, 22, 95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, J.; Lou, J.; Zhou, Y.; Bo, L.; Zhu, J.; Zhu, K.; Wan, X.; Cai, Z.; Deng, X. Upregulation of Programmed Death-1 on T Cells and Programmed Death Ligand-1 on Monocytes in Septic Shock Patients. Crit. Care 2011, 15, R70. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K.; Lopez-Collazo, E. Endotoxin Tolerance: New Mechanisms, Molecules and Clinical Significance. Trends Immunol. 2009, 30, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Cavaillon, J.-M.; Adib-Conquy, M. Bench-to-Bedside Review: Endotoxin Tolerance as a Model of Leukocyte Reprogramming in Sepsis. Crit. Care 2006, 10, 233. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoppstädter, J.; Dembek, A.; Linnenberger, R.; Dahlem, C.; Barghash, A.; Fecher-Trost, C.; Fuhrmann, G.; Koch, M.; Kraegeloh, A.; Huwer, H.; et al. Toll-Like Receptor 2 Release by Macrophages: An Anti-Inflammatory Program Induced by Glucocorticoids and Lipopolysaccharide. Front. Immunol. 2019, 10, 1634. [Google Scholar] [CrossRef]
- Pradhan, K.; Yi, Z.; Geng, S.; Li, L. Development of Exhausted Memory Monocytes and Underlying Mechanisms. Front. Immunol. 2021, 12, 778830. [Google Scholar] [CrossRef]
- Cavaillon, J.M.; Adib-Conquy, M.; Cloëz-Tayarani, I.; Fitting, C. Immunodepression in Sepsis and SIRS Assessed by Ex Vivo Cytokine Production Is Not a Generalized Phenomenon: A Review. J. Endotoxin Res. 2001, 7, 85–93. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Monneret, G.; Payen, D. Sepsis-Induced Immunosuppression: From Cellular Dysfunctions to Immunotherapy. Nat. Rev. Immunol. 2013, 13, 862–874. [Google Scholar] [CrossRef]
- Bick, A.; Buys, W.; Engler, A.; Madel, R.; Atia, M.; Faro, F.; Westendorf, A.M.; Limmer, A.; Buer, J.; Herbstreit, F.; et al. Immune Hyporeactivity to Bacteria and Multiple TLR-Ligands, yet No Response to Checkpoint Inhibition in Patients Just after Meeting Sepsis-3 Criteria. PLoS ONE 2022, 17, e0273247. [Google Scholar] [CrossRef]
- Boomer, J.S.; To, K.; Chang, K.C.; Takasu, O.; Osborne, D.F.; Walton, A.H.; Bricker, T.L.; Jarman, S.D.; Kreisel, D.; Krupnick, A.S.; et al. Immunosuppression in Patients Who Die of Sepsis and Multiple Organ Failure. JAMA 2011, 306, 2594. [Google Scholar] [CrossRef]
- Blackwell, T.S.; Blackwell, T.R.; Christman, J.W. Induction of Endotoxin Tolerance Depletes Nuclear Factor-κB and Suppresses Its Activation in Rat Alveolar Macrophages. J. Leukoc. Biol. 1997, 62, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.; Li, L.; McCall, C.E.; Yoza, B.K. Endotoxin Tolerance Disrupts Chromatin Remodeling and NF-κB Transactivation at the IL-1β Promoter. J. Immunol. 2005, 175, 461–468. [Google Scholar] [CrossRef]
- Del Fresno, C.; García-Rio, F.; Gómez-Piña, V.; Soares-Schanoski, A.; Fernández-Ruíz, I.; Jurado, T.; Kajiji, T.; Shu, C.; Marín, E.; Gutierrez Del Arroyo, A.; et al. Potent Phagocytic Activity with Impaired Antigen Presentation Identifying Lipopolysaccharide-Tolerant Human Monocytes: Demonstration in Isolated Monocytes from Cystic Fibrosis Patients. J. Immunol. 2009, 182, 6494–6507. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Maitra, U.; Morris, M.; Li, L. Molecular Mechanism Responsible for the Priming of Macrophage Activation. J. Biol. Chem. 2013, 288, 3897–3906. [Google Scholar] [CrossRef]
- Dobrovolskaia, M.A.; Vogel, S.N. Toll Receptors, CD14, and Macrophage Activation and Deactivation by LPS. Microbes Infect. 2002, 4, 903–914. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.L.; Medzhitov, R. Gene-Specific Control of the TLR-Induced Inflammatory Response. Clin. Immunol. 2009, 130, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Hoogendijk, A.J.; Garcia-Laorden, M.I.; Van Vught, L.A.; Wiewel, M.A.; Belkasim-Bohoudi, H.; Duitman, J.; Horn, J.; Schultz, M.J.; Scicluna, B.P.; Van ‘T Veer, C.; et al. Sepsis Patients Display a Reduced Capacity to Activate Nuclear Factor-κB in Multiple Cell Types*. Crit. Care Med. 2017, 45, e524–e531. [Google Scholar] [CrossRef]
- Huang, X.; Venet, F.; Wang, Y.L.; Lepape, A.; Yuan, Z.; Chen, Y.; Swan, R.; Kherouf, H.; Monneret, G.; Chung, C.-S.; et al. PD-1 Expression by Macrophages Plays a Pathologic Role in Altering Microbial Clearance and the Innate Inflammatory Response to Sepsis. Proc. Natl. Acad. Sci. USA 2009, 106, 6303–6308. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Aziz, M.; Yen, H.-T.; Ma, G.; Murao, A.; Wang, P. Extracellular CIRP Dysregulates Macrophage Bacterial Phagocytosis in Sepsis. Cell Mol. Immunol. 2022, 20, 80–93. [Google Scholar] [CrossRef]
- Chen, W.; Qiang, X.; Wang, Y.; Zhu, S.; Li, J.; Babaev, A.; Yang, H.; Gong, J.; Becker, L.; Wang, P.; et al. Identification of Tetranectin-Targeting Monoclonal Antibodies to Treat Potentially Lethal Sepsis. Sci. Transl. Med. 2020, 12, eaaz3833. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zhang, Q.; Tan, J.; Xiong, Y.; Liang, Y.; Yan, J.; Gu, F.; Xu, Y. HMGB1 Induces Macrophage Pyroptosis in Chronic Endometritis. Int. Immunopharmacol. 2023, 123, 110706. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jiang, H.; Wu, G.; Huang, P.; Wang, H.; An, H.; Liu, S.; Zhang, W. The Pathogenesis and Potential Therapeutic Targets in Sepsis. MedComm 2023, 4, e418. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, W.; Xu, Y.; Wu, D.; Gao, Z.; Zhou, J.; Qian, H.; He, B.; Wang, G. Extracellular HMGB1 Impairs Macrophage-Mediated Efferocytosis by Suppressing the Rab43-Controlled Cell Surface Transport of CD91. Front. Immunol. 2022, 13, 767630. [Google Scholar] [CrossRef] [PubMed]
- Parker, K.H.; Sinha, P.; Horn, L.A.; Clements, V.K.; Yang, H.; Li, J.; Tracey, K.J.; Ostrand-Rosenberg, S. HMGB1 Enhances Immune Suppression by Facilitating the Differentiation and Suppressive Activity of Myeloid-Derived Suppressor Cells. Cancer Res. 2014, 74, 5723–5733. [Google Scholar] [CrossRef] [PubMed]
- Döcke, W.-D.; Randow, F.; Syrbe, U.; Krausch, D.; Asadullah, K.; Reinke, P.; Volk, H.-D.; Kox, W. Monocyte Deactivation in Septic Patients: Restoration by IFN-γ Treatment. Nat. Med. 1997, 3, 678–681. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Ner-Gaon, H.; Varon, J.; Cullen, A.M.; Guo, J.; Choi, J.; Barragan-Bradford, D.; Higuera, A.; Pinilla-Vera, M.; Short, S.A.P.; et al. Post-Sepsis Immunosuppression Depends on NKT Cell Regulation of mTOR/IFN-γ in NK Cells. J. Clin. Investig. 2020, 130, 3238–3252. [Google Scholar] [CrossRef] [PubMed]
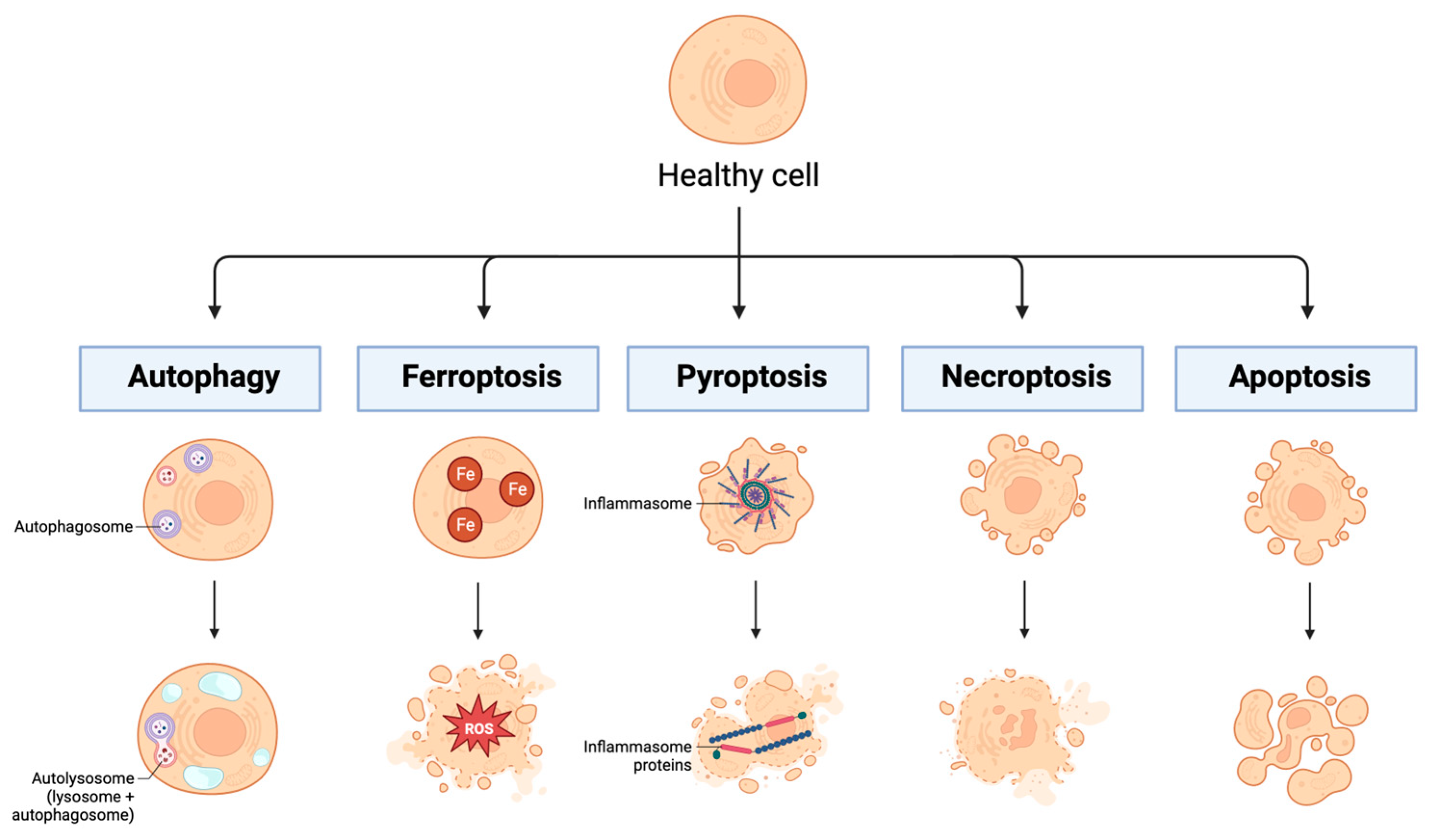
- Shen, S.; Shao, Y.; Li, C. Different Types of Cell Death and Their Shift in Shaping Disease. Cell Death Discov. 2023, 9, 284. [Google Scholar] [CrossRef]
- Gao, W.; Wang, X.; Zhou, Y.; Wang, X.; Yu, Y. Autophagy, Ferroptosis, Pyroptosis, and Necroptosis in Tumor Immunotherapy. Sig Transduct. Target. Ther. 2022, 7, 196. [Google Scholar] [CrossRef]
- Bertheloot, D.; Latz, E.; Franklin, B.S. Necroptosis, Pyroptosis and Apoptosis: An Intricate Game of Cell Death. Cell Mol. Immunol. 2021, 18, 1106–1121. [Google Scholar] [CrossRef]
- Tang, D.; Kang, R.; Berghe, T.V.; Vandenabeele, P.; Kroemer, G. The Molecular Machinery of Regulated Cell Death. Cell Res. 2019, 29, 347–364. [Google Scholar] [CrossRef]
- Rathmell, J.C.; Thompson, C.B. Pathways of Apoptosis in Lymphocyte Development, Homeostasis, and Disease. Cell 2002, 109, S97–S107. [Google Scholar] [CrossRef] [PubMed]
- Alberts, B. (Ed.) Molecular Biology of the Cell, 4th ed.; Garland: New York, NY, USA, 2002; ISBN 978-0-8153-3218-3. [Google Scholar]
- Tsujimoto, Y. Role of Bcl-2 Family Proteins in Apoptosis: Apoptosomes or Mitochondria?: Role of Bcl-2 Family Proteins in Apoptosis. Genes. Cells 1998, 3, 697–707. [Google Scholar] [CrossRef]
- Peña-Blanco, A.; García-Sáez, A.J. Bax, Bak and beyond—Mitochondrial Performance in Apoptosis. FEBS J. 2018, 285, 416–431. [Google Scholar] [CrossRef]
- Sheikh Motahar Vahedi, H.; Bagheri, A.; Jahanshir, A.; Seyedhosseini, J.; Vahidi, E. Association of Lymphopenia with Short Term Outcomes of Sepsis Patients; a Brief Report. Arch. Acad. Emerg. Med. 2019, 7, e14. [Google Scholar]
- Carrero, J.; Unanue, E. Lymphocyte Apoptosis as an Immune Subversion Strategy of Microbial Pathogens. Trends Immunol. 2006, 27, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Van Hauwermeiren, F.; Van Opdenbosch, N.; Van Gorp, H.; De Vasconcelos, N.; Van Loo, G.; Vandenabeele, P.; Kanneganti, T.-D.; Lamkanfi, M. Bacillus Anthracis Induces NLRP3 Inflammasome Activation and Caspase-8–Mediated Apoptosis of Macrophages to Promote Lethal Anthrax. Proc. Natl. Acad. Sci. USA 2022, 119, e2116415119. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, R.S.; Swanson, P.E.; Knudson, C.M.; Chang, K.C.; Cobb, J.P.; Osborne, D.F.; Zollner, K.M.; Buchman, T.G.; Korsmeyer, S.J.; Karl, I.E. Overexpression of Bcl-2 in Transgenic Mice Decreases Apoptosis and Improves Survival in Sepsis. J. Immunol. 1999, 162, 4148–4156. [Google Scholar] [CrossRef]
- Wang, S.D.; Huang, K.J.; Lin, Y.S.; Lei, H.Y. Sepsis-Induced Apoptosis of the Thymocytes in Mice. J. Immunol. 1994, 152, 5014–5021. [Google Scholar] [CrossRef]
- Luan, Y.; Yao, Y.; Xiao, X.; Sheng, Z. Insights into the Apoptotic Death of Immune Cells in Sepsis. J. Interferon Cytokine Res. 2015, 35, 17–22. [Google Scholar] [CrossRef]
- Luan, Y.-Y.; Dong, N.; Xie, M.; Xiao, X.-Z.; Yao, Y.-M. The Significance and Regulatory Mechanisms of Innate Immune Cells in the Development of Sepsis. J. Interferon Cytokine Res. 2014, 34, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.; Teramae, H.; Futatsugi, M.; Takano, K.; Yamamoto, S.; Tomita, K.; Suzuki, T.; Yokoo, H.; Koike, K.; Hattori, Y. Up-Regulation of Histamine H 4 Receptors Contributes to Splenic Apoptosis in Septic Mice: Counteraction of the Antiapoptotic Action of Nuclear Factor-κB. J. Pharmacol. Exp. Ther. 2010, 332, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Bo, L.; Wang, F.; Zhu, J.; Li, J.; Deng, X. Granulocyte-Colony Stimulating Factor (G-CSF) and Granulocyte-Macrophage Colony Stimulating Factor (GM-CSF) for Sepsis: A Meta-Analysis. Crit. Care 2011, 15, R58. [Google Scholar] [CrossRef] [PubMed]
- Francois, B.; Jeannet, R.; Daix, T.; Walton, A.H.; Shotwell, M.S.; Unsinger, J.; Monneret, G.; Rimmelé, T.; Blood, T.; Morre, M.; et al. Interleukin-7 Restores Lymphocytes in Septic Shock: The IRIS-7 Randomized Clinical Trial. JCI Insight 2018, 3, e98960. [Google Scholar] [CrossRef]
- Zhang, W.; Fang, X.; Gao, C.; Song, C.; He, Y.; Zhou, T.; Yang, X.; Shang, Y.; Xu, J. MDSCs in Sepsis-Induced Immunosuppression and Its Potential Therapeutic Targets. Cytokine Growth Factor. Rev. 2023, 69, 90–103. [Google Scholar] [CrossRef]
- Veglia, F.; Sanseviero, E.; Gabrilovich, D.I. Myeloid-Derived Suppressor Cells in the Era of Increasing Myeloid Cell Diversity. Nat. Rev. Immunol. 2021, 21, 485–498. [Google Scholar] [CrossRef]
- Goldmann, O.; Beineke, A.; Medina, E. Identification of a Novel Subset of Myeloid-Derived Suppressor Cells During Chronic Staphylococcal Infection That Resembles Immature Eosinophils. J. Infect. Dis. 2017, 216, 1444–1451. [Google Scholar] [CrossRef]
- Bronte, V.; Brandau, S.; Chen, S.-H.; Colombo, M.P.; Frey, A.B.; Greten, T.F.; Mandruzzato, S.; Murray, P.J.; Ochoa, A.; Ostrand-Rosenberg, S.; et al. Recommendations for Myeloid-Derived Suppressor Cell Nomenclature and Characterization Standards. Nat. Commun. 2016, 7, 12150. [Google Scholar] [CrossRef]
- Talmadge, J.E.; Gabrilovich, D.I. History of Myeloid-Derived Suppressor Cells. Nat. Rev. Cancer 2013, 13, 739–752. [Google Scholar] [CrossRef]
- Brudecki, L.; Ferguson, D.A.; McCall, C.E.; El Gazzar, M. Myeloid-Derived Suppressor Cells Evolve during Sepsis and Can Enhance or Attenuate the Systemic Inflammatory Response. Infect. Immun. 2012, 80, 2026–2034. [Google Scholar] [CrossRef]
- Cuenca, A.G.; Delano, M.J.; Kelly-Scumpia, K.M.; Moreno, C.; Scumpia, P.O.; LaFace, D.M.; Heyworth, P.G.; Efron, P.A.; Moldawer, L.L. A Paradoxical Role for Myeloid-Derived Suppressor Cells in Sepsis and Trauma. Mol. Med. 2011, 17, 281–292. [Google Scholar] [CrossRef]
- Delano, M.J.; Scumpia, P.O.; Weinstein, J.S.; Coco, D.; Nagaraj, S.; Kelly-Scumpia, K.M.; O’Malley, K.A.; Wynn, J.L.; Antonenko, S.; Al-Quran, S.Z.; et al. MyD88-Dependent Expansion of an Immature GR-1+CD11b+ Population Induces T Cell Suppression and Th2 Polarization in Sepsis. J. Exp. Med. 2007, 204, 1463–1474. [Google Scholar] [CrossRef] [PubMed]
- Mathias, B.; Delmas, A.L.; Ozrazgat-Baslanti, T.; Vanzant, E.L.; Szpila, B.E.; Mohr, A.M.; Moore, F.A.; Brakenridge, S.C.; Brumback, B.A.; Moldawer, L.L.; et al. Human Myeloid-Derived Suppressor Cells Are Associated with Chronic Immune Suppression After Severe Sepsis/Septic Shock. Ann. Surg. 2017, 265, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Landoni, V.I.; Martire-Greco, D.; Rodriguez-Rodrigues, N.; Chiarella, P.; Schierloh, P.; Isturiz, M.A.; Fernández, G.C. Immature Myeloid Gr-1+ CD11b+ Cells from Lipopolysaccharide-Immunosuppressed Mice Acquire Inhibitory Activity in the Bone Marrow and Migrate to Lymph Nodes to Exert Their Suppressive Function. Clin. Sci. 2016, 130, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Li, H.; Sun, Z. Targeting Myeloid-Derived Suppressor Cells for Cancer Therapy. Cancer Biol. Med. 2021, 18, 992–1009. [Google Scholar] [CrossRef]
- Uhel, F.; Azzaoui, I.; Grégoire, M.; Pangault, C.; Dulong, J.; Tadié, J.-M.; Gacouin, A.; Camus, C.; Cynober, L.; Fest, T.; et al. Early Expansion of Circulating Granulocytic Myeloid-Derived Suppressor Cells Predicts Development of Nosocomial Infections in Patients with Sepsis. Am. J. Respir. Crit. Care Med. 2017, 196, 315–327. [Google Scholar] [CrossRef]
- Arocena, A.R.; Onofrio, L.I.; Pellegrini, A.V.; Carrera Silva, A.E.; Paroli, A.; Cano, R.C.; Aoki, M.P.; Gea, S. Myeloid-derived Suppressor Cells Are Key Players in the Resolution of Inflammation during a Model of Acute Infection. Eur. J. Immunol. 2014, 44, 184–194. [Google Scholar] [CrossRef]
- Sander, L.E.; Sackett, S.D.; Dierssen, U.; Beraza, N.; Linke, R.P.; Müller, M.; Blander, J.M.; Tacke, F.; Trautwein, C. Hepatic Acute-Phase Proteins Control Innate Immune Responses during Infection by Promoting Myeloid-Derived Suppressor Cell Function. J. Exp. Med. 2010, 207, 1453–1464. [Google Scholar] [CrossRef]
- Schrijver, I.T.; Karakike, E.; Théroude, C.; Baumgartner, P.; Harari, A.; Giamarellos-Bourboulis, E.J.; Calandra, T.; Roger, T. High Levels of Monocytic Myeloid-Derived Suppressor Cells Are Associated with Favorable Outcome in Patients with Pneumonia and Sepsis with Multi-Organ Failure. Intensive Care Med. Exp. 2022, 10, 5. [Google Scholar] [CrossRef]
- Hollen, M.K.; Stortz, J.A.; Darden, D.; Dirain, M.L.; Nacionales, D.C.; Hawkins, R.B.; Cox, M.C.; Lopez, M.-C.; Rincon, J.C.; Ungaro, R.; et al. Myeloid-Derived Suppressor Cell Function and Epigenetic Expression Evolves over Time after Surgical Sepsis. Crit. Care 2019, 23, 355. [Google Scholar] [CrossRef]
- Mayr, F.B.; Yende, S.; Angus, D.C. Epidemiology of Severe Sepsis. Virulence 2014, 5, 4–11. [Google Scholar] [CrossRef]
- Rangel-Frausto, M.S. THE EPIDEMIOLOGY OF BACTERIAL SEPSIS. Infect. Dis. Clin. North Am. 1999, 13, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Tindal, E.W.; Armstead, B.E.; Monaghan, S.F.; Heffernan, D.S.; Ayala, A. Emerging Therapeutic Targets for Sepsis. Expert. Opin. Ther. Targets 2021, 25, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Barichello, T.; Generoso, J.S.; Singer, M.; Dal-Pizzol, F. Biomarkers for Sepsis: More than Just Fever and Leukocytosis—A Narrative Review. Crit. Care 2022, 26, 14. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801. [Google Scholar] [CrossRef]
- Andersen, L.W.; Mackenhauer, J.; Roberts, J.C.; Berg, K.M.; Cocchi, M.N.; Donnino, M.W. Etiology and Therapeutic Approach to Elevated Lactate Levels. Mayo Clin. Proc. 2013, 88, 1127–1140. [Google Scholar] [CrossRef]
- Chen, H.; Li, T.; Yan, S.; Liu, M.; Liu, K.; Zhang, H.; Gao, M.; Xiao, X. Pentraxin-3 Is a Strong Biomarker of Sepsis Severity Identification and Predictor of 90-Day Mortality in Intensive Care Units via Sepsis 3.0 Definitions. Diagnostics 2021, 11, 1906. [Google Scholar] [CrossRef]
- Hamed, S.; Behnes, M.; Pauly, D.; Lepiorz, D.; Barre, M.; Becher, T.; Lang, S.; Akin, I.; Borggrefe, M.; Bertsch, T.; et al. Diagnostic Value of Pentraxin-3 in Patients with Sepsis and Septic Shock in Accordance with Latest Sepsis-3 Definitions. BMC Infect. Dis. 2017, 17, 554. [Google Scholar] [CrossRef]
- Jie, H.; Li, Y.; Pu, X.; Ye, J. Pentraxin 3, a Predicator for 28-Day Mortality in Patients with Septic Shock. Am. J. Med. Sci. 2017, 353, 242–246. [Google Scholar] [CrossRef]
- Lee, Y.T.; Gong, M.; Chau, A.; Wong, W.T.; Bazoukis, G.; Wong, S.H.; Lampropoulos, K.; Xia, Y.; Li, G.; Wong, M.C.S.; et al. Pentraxin-3 as a Marker of Sepsis Severity and Predictor of Mortality Outcomes: A Systematic Review and Meta-Analysis. J. Infect. 2018, 76, 1–10. [Google Scholar] [CrossRef]
- Davoudian, S.; Piovani, D.; Desai, A.; Mapelli, S.N.; Leone, R.; Sironi, M.; Valentino, S.; Silva-Gomes, R.; Stravalaci, M.; Asgari, F.; et al. A Cytokine/PTX3 Prognostic Index as a Predictor of Mortality in Sepsis. Front. Immunol. 2022, 13, 979232. [Google Scholar] [CrossRef]
- Caironi, P.; Masson, S.; Mauri, T.; Bottazzi, B.; Leone, R.; Magnoli, M.; Barlera, S.; Mamprin, F.; Fedele, A.; Mantovani, A.; et al. Pentraxin 3 in Patients with Severe Sepsis or Shock: The ALBIOS Trial. Eur. J. Clin. Investig. 2017, 47, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Jiang, C.; Fang, J.; Li, Z.; Cai, H. Pentraxin-3 as a Predictive Marker of Mortality in Sepsis: An Updated Systematic Review and Meta-Analysis. Crit. Care 2022, 26, 167. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-P.; Chen, C.-K.; Chung, K.; Tseng, J.-C.; Hua, C.-C.; Liu, Y.-C.; Chuang, D.-Y.; Yang, C.-H. Serial Cytokine Levels in Patients with Severe Sepsis. Inflamm. Res. 2009, 58, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Gogos, C.A.; Drosou, E.; Bassaris, H.P.; Skoutelis, A. Pro- versus Anti-inflammatory Cytokine Profile in Patients with Severe Sepsis: A Marker for Prognosis and Future Therapeutic Options. J. Infect. Dis. 2000, 181, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.S.; Cheng, G. Role of Interleukin 10 Transcriptional Regulation in Inflammation and Autoimmune Disease. Crit. Rev. Immunol. 2012, 32, 23–63. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zheng, Z.; Yin, Z.; Zhang, J.; Qin, J.; Wan, J.; Wang, M. Resolvin D2 and Its Receptor GPR18 in Cardiovascular and Metabolic Diseases: A Promising Biomarker and Therapeutic Target. Pharmacol. Res. 2023, 195, 106832. [Google Scholar] [CrossRef]
- Jundi, B.; Lee, D.-H.; Jeon, H.; Duvall, M.G.; Nijmeh, J.; Abdulnour, R.-E.E.; Pinilla-Vera, M.; Baron, R.M.; Han, J.; Voldman, J.; et al. Inflammation Resolution Circuits Are Uncoupled in Acute Sepsis and Correlate with Clinical Severity. JCI Insight 2021, 6, e148866. [Google Scholar] [CrossRef]
- Zhang, L.; Qiu, C.; Yang, L.; Zhang, Z.; Zhang, Q.; Wang, B.; Wang, X. GPR18 Expression on PMNs as Biomarker for Outcome in Patient with Sepsis. Life Sci. 2019, 217, 49–56. [Google Scholar] [CrossRef]
- Wang, E.; Adams, S.; Marincola, F.M.; Stroncek, D.F. Human Leukocyte and Granulocyte Antigens and Antibodies: The HLA and HNA Systems. In Blood Banking and Transfusion Medicine; Elsevier: Amsterdam, The Netherlands, 2007; pp. 129–156. ISBN 978-0-443-06981-9. [Google Scholar]
- Berry, P.A.; Antoniades, C.G.; Carey, I.; McPhail, M.J.W.; Hussain, M.J.; Davies, E.T.; Wendon, J.A.; Vergani, D. Severity of the Compensatory Anti-Inflammatory Response Determined by Monocyte HLA-DR Expression May Assist Outcome Prediction in Cirrhosis. Intensive Care Med. 2011, 37, 453–460. [Google Scholar] [CrossRef]
- Venet, F.; Monneret, G. Advances in the Understanding and Treatment of Sepsis-Induced Immunosuppression. Nat. Rev. Nephrol. 2018, 14, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, E.; Wenthe, J.; Irenaeus, S.; Loskog, A.; Ullenhag, G. Gemcitabine Reduces MDSCs, Tregs and TGFβ-1 While Restoring the Teff/Treg Ratio in Patients with Pancreatic Cancer. J. Transl. Med. 2016, 14, 282. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Monneret, G.; Payen, D. Immunosuppression in Sepsis: A Novel Understanding of the Disorder and a New Therapeutic Approach. Lancet Infect. Dis. 2013, 13, 260–268. [Google Scholar] [CrossRef]
- Law, A.M.K.; Valdes-Mora, F.; Gallego-Ortega, D. Myeloid-Derived Suppressor Cells as a Therapeutic Target for Cancer. Cells 2020, 9, 561. [Google Scholar] [CrossRef] [PubMed]
- Abdulnour, R.E.; Sham, H.P.; Douda, D.N.; Colas, R.A.; Dalli, J.; Bai, Y.; Ai, X.; Serhan, C.N.; Levy, B.D. Aspirin-Triggered Resolvin D1 Is Produced during Self-Resolving Gram-Negative Bacterial Pneumonia and Regulates Host Immune Responses for the Resolution of Lung Inflammation. Mucosal Immunol. 2016, 9, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Basil, M.C.; Levy, B.D. Specialized Pro-Resolving Mediators: Endogenous Regulators of Infection and Inflammation. Nat. Rev. Immunol. 2016, 16, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Purvis, G.S.D.; Collotta, D.; Al Zoubi, S.; Sugimoto, M.A.; Cacace, A.; Martin, L.; Colas, R.A.; Collino, M.; Dalli, J.; et al. RvE1 Attenuates Polymicrobial Sepsis-Induced Cardiac Dysfunction and Enhances Bacterial Clearance. Front. Immunol. 2020, 11, 2080. [Google Scholar] [CrossRef]
- Chiang, N.; de la Rosa, X.; Libreros, S.; Serhan, C.N. Novel Resolvin D2 Receptor Axis in Infectious Inflammation. J. Immunol. 2017, 198, 842–851. [Google Scholar] [CrossRef]
- El Kebir, D.; Gjorstrup, P.; Filep, J.G. Resolvin E1 Promotes Phagocytosis-Induced Neutrophil Apoptosis and Accelerates Resolution of Pulmonary Inflammation. Proc. Natl. Acad. Sci. USA 2012, 109, 14983–14988. [Google Scholar] [CrossRef]
- Seki, H.; Fukunaga, K.; Arita, M.; Arai, H.; Nakanishi, H.; Taguchi, R.; Miyasho, T.; Takamiya, R.; Asano, K.; Ishizaka, A.; et al. The Anti-Inflammatory and Proresolving Mediator Resolvin E1 Protects Mice from Bacterial Pneumonia and Acute Lung Injury. J. Immunol. 2010, 184, 836–843. [Google Scholar] [CrossRef]
- Spite, M.; Norling, L.V.; Summers, L.; Yang, R.; Cooper, D.; Petasis, N.A.; Flower, R.J.; Perretti, M.; Serhan, C.N. Resolvin D2 Is a Potent Regulator of Leukocytes and Controls Microbial Sepsis. Nature 2009, 461, 1287–1291. [Google Scholar] [CrossRef] [PubMed]
- Winkler, J.W.; Orr, S.K.; Dalli, J.; Cheng, C.-Y.C.; Sanger, J.M.; Chiang, N.; Petasis, N.A.; Serhan, C.N. Resolvin D4 Stereoassignment and Its Novel Actions in Host Protection and Bacterial Clearance. Sci. Rep. 2016, 6, 18972. [Google Scholar] [CrossRef]
- Gao, J.; Su, Y.; Wang, Z. Lung Inflammation Resolution by RvD1 and RvD2 in a Receptor-Dependent Manner. Pharmaceutics 2023, 15, 1527. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Levy, B.D. Resolvins in Inflammation: Emergence of the pro-Resolving Superfamily of Mediators. J. Clin. Investig. 2018, 128, 2657–2669. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Dichter, E.; Lacorte, G.; Kerner, D.; Spur, B.; Rodriguez, A.; Yin, K. Lipoxin A4 Increases Survival by Decreasing Systemic Inflammation and Bacterial Load in Sepsis. Shock 2011, 36, 410–416. [Google Scholar] [CrossRef]
- Dalli, J.; Colas, R.A.; Quintana, C.; Barragan-Bradford, D.; Hurwitz, S.; Levy, B.D.; Choi, A.M.; Serhan, C.N.; Baron, R.M. Human Sepsis Eicosanoid and Proresolving Lipid Mediator Temporal Profiles: Correlations with Survival and Clinical Outcomes. Crit. Care Med. 2017, 45, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Muenzer, J.T.; Davis, C.G.; Chang, K.; Schmidt, R.E.; Dunne, W.M.; Coopersmith, C.M.; Hotchkiss, R.S. Characterization and Modulation of the Immunosuppressive Phase of Sepsis. Infect. Immun. 2010, 78, 1582–1592. [Google Scholar] [CrossRef] [PubMed]
- Sundarasivarao, P.Y.K.; Walker, J.M.; Rodriguez, A.; Spur, B.W.; Yin, K. Resolvin D2 Induces Anti-Microbial Mechanisms in a Model of Infectious Peritonitis and Secondary Lung Infection. Front. Immunol. 2022, 13, 1011944. [Google Scholar] [CrossRef]
- Walker, J.M.; Sundarasivarao, P.Y.K.; Thornton, J.M.; Sochacki, K.; Rodriguez, A.; Spur, B.W.; Acharya, N.K.; Yin, K. Resolvin D2 Promotes Host Defense in a 2—Hit Model of Sepsis with Secondary Lung Infection. Prostaglandins Other Lipid Mediat. 2022, 159, 106617. [Google Scholar] [CrossRef]
- Chiang, N.; Fredman, G.; Bäckhed, F.; Oh, S.F.; Vickery, T.; Schmidt, B.A.; Serhan, C.N. Infection Regulates Pro-Resolving Mediators That Lower Antibiotic Requirements. Nature 2012, 484, 524–528. [Google Scholar] [CrossRef]
- Thornton, J.M.; Padovani, C.M.; Rodriguez, A.; Spur, B.W.; Yin, K. Lipoxin A4 Promotes Antibiotic and Monocyte Bacterial Killing in Established Pseudomonas Aeruginosa Biofilm Formed under Hydrodynamic Conditions. FASEB J. 2023, 37, e23098. [Google Scholar] [CrossRef] [PubMed]
- Thornton, J.M.; Walker, J.M.; Sundarasivarao, P.Y.K.; Spur, B.W.; Rodriguez, A.; Yin, K. Lipoxin A4 Promotes Reduction and Antibiotic Efficacy against Pseudomonas Aeruginosa Biofilm. Prostaglandins Other Lipid Mediat. 2021, 152, 106505. [Google Scholar] [CrossRef] [PubMed]
- Codagnone, M.; Cianci, E.; Lamolinara, A.; Mari, V.C.; Nespoli, A.; Isopi, E.; Mattoscio, D.; Arita, M.; Bragonzi, A.; Iezzi, M.; et al. Resolvin D1 Enhances the Resolution of Lung Inflammation Caused by Long-Term Pseudomonas Aeruginosa Infection. Mucosal Immunol. 2018, 11, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Isopi, E.; Mattoscio, D.; Codagnone, M.; Mari, V.C.; Lamolinara, A.; Patruno, S.; D’Aurora, M.; Cianci, E.; Nespoli, A.; Franchi, S.; et al. Resolvin D1 Reduces Lung Infection and Inflammation Activating Resolution in Cystic Fibrosis. Front. Immunol. 2020, 11, 581. [Google Scholar] [CrossRef]

| Innate Immune System | Adaptive Immune System | |
|---|---|---|
| Response Speed? | Quick | Slow upon first exposure |
| Nonspecific or Specific? | Nonspecific | Specific |
| Development of Traditional Immunologic Memory? | No | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padovani, C.M.; Yin, K. Immunosuppression in Sepsis: Biomarkers and Specialized Pro-Resolving Mediators. Biomedicines 2024, 12, 175. https://doi.org/10.3390/biomedicines12010175
Padovani CM, Yin K. Immunosuppression in Sepsis: Biomarkers and Specialized Pro-Resolving Mediators. Biomedicines. 2024; 12(1):175. https://doi.org/10.3390/biomedicines12010175
Chicago/Turabian StylePadovani, Cristina M., and Kingsley Yin. 2024. "Immunosuppression in Sepsis: Biomarkers and Specialized Pro-Resolving Mediators" Biomedicines 12, no. 1: 175. https://doi.org/10.3390/biomedicines12010175
APA StylePadovani, C. M., & Yin, K. (2024). Immunosuppression in Sepsis: Biomarkers and Specialized Pro-Resolving Mediators. Biomedicines, 12(1), 175. https://doi.org/10.3390/biomedicines12010175






