Nab-Paclitaxel in the Treatment of Gastrointestinal Cancers—Improvements in Clinical Efficacy and Safety
Abstract
1. Introduction
2. Pharmacokinetics of Nab-Paclitaxel
3. Nab-Paclitaxel in Pancreatic Cancer
4. Nab-Paclitaxel in Esophageal Cancer
5. Nab-Paclitaxel in Gastric Cancer
6. Nab-Paclitaxel in Cholangiocarcinoma
7. Nab-Paclitaxel in Colorectal Cancer
8. Future Prospective and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Menzin, A.W.; King, S.A.; Aikins, J.K.; Mikuta, J.J.; Rubin, S.C. Taxol (paclitaxel) was approved by FDA for the treatment of patients with recurrent ovarian cancer. Gynecol. Oncol. 1994, 54, 103. [Google Scholar]
- Tannock, I.F.; de Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Théodore, C.; James, N.D.; Turesson, I.; et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef]
- Sparreboom, A.; van Zuylen, L.; Brouwer, E.; Loos, W.J.; de Bruijn, P.; Gelderblom, H.; Pillay, M.; Nooter, K.; Stoter, G.; Verweij, J. Cremophor EL-mediated alteration of paclitaxel distribution in human blood: Clinical pharmacokinetic implications. Cancer Res. 1999, 59, 1454–1457. [Google Scholar] [PubMed]
- ten Tije, A.J.; Verweij, J.; Loos, W.J.; Sparreboom, A. Pharmacological effects of formulation vehicles: Implications for cancer chemotherapy. Clin. Pharmacokinet. 2003, 42, 665–685. [Google Scholar] [CrossRef] [PubMed]
- Gallo, J.M.; Li, S.; Guo, P.; Reed, K.; Ma, J. The effect of P-glycoprotein on paclitaxel brain and brain tumor distribution in mice. Cancer Res. 2003, 63, 5114–5117. [Google Scholar] [PubMed]
- Hendrikx, J.J.; Lagas, J.S.; Rosing, H.; Schellens, J.H.; Beijnen, J.H.; Schinkel, A.H. P-glycoprotein and cytochrome P450 3A act together in restricting the oral bioavailability of paclitaxel. Int. J. Cancer 2013, 132, 2439–2447. [Google Scholar] [CrossRef] [PubMed]
- Stinchcombe, T.E. Nanoparticle albumin-bound paclitaxel: A novel Cremphor-EL-free formulation of paclitaxel. Nanomedicine 2007, 2, 415–423. [Google Scholar] [CrossRef]
- Gradishar, W.J. Albumin-bound paclitaxel: A next-generation taxane. Expert. Opin. Pharmacother. 2006, 7, 1041–1053. [Google Scholar] [CrossRef]
- Desai, N.; Trieu, V.; Yao, Z.; Louie, L.; Ci, S.; Yang, A.; Tao, C.; De, T.; Beals, B.; Dykes, D.; et al. Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin. Cancer Res. 2006, 12, 1317–1324. [Google Scholar] [CrossRef]
- Ibrahim, N.K.; Desai, N.; Legha, S.; Soon-Shiong, P.; Theriault, R.L.; Rivera, E.; Esmaeli, B.; Ring, S.E.; Bedikian, A.; Hortobagyi, G.N.; et al. Phase I and pharmacokinetic study of ABI-007, a Cremophor-free, protein-stabilized, nanoparticle formulation of paclitaxel. Clin. Cancer Res. 2002, 8, 1038–1044. [Google Scholar]
- Borgå, O.; Henriksson, R.; Bjermo, H.; Lilienberg, E.; Heldring, N.; Loman, N. Maximum Tolerated Dose and Pharmacokinetics of Paclitaxel Micellar in Patients with Recurrent Malignant Solid Tumours: A Dose-Escalation Study. Adv. Ther. 2019, 36, 1150–1163. [Google Scholar] [CrossRef]
- FDA Approves Abraxane for Metastatic Breast Cancer. Oncology NEWS International; MJH Life Sciences: Cranbury, NJ, USA, 2005. [Google Scholar]
- Levin, J. FDA Approves ABRAXANE® for the First-Line Treatment of Advanced Non-Small Cell Lung Cancer; Fierce Pharma: Washington, DC, USA, 2012. [Google Scholar]
- Kundranda, M.N.; Niu, J. Albumin-bound paclitaxel in solid tumors: Clinical development and future directions. Drug Des. Dev. Ther. 2015, 9, 3767–3777. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.A. Nanostructure-mediated drug delivery. Nanomedicine 2005, 1, 22–30. [Google Scholar] [CrossRef]
- Foote, M. Using nanotechnology to improve the characteristics of antineoplastic drugs: Improved characteristics of nab-paclitaxel compared with solvent-based paclitaxel. Biotechnol. Annu. Rev. 2007, 13, 345–357. [Google Scholar] [CrossRef]
- Desai, N.P.; Trieu, V.; Hwang, L.Y.; Wu, R.; Soon-Shiong, P.; Gradishar, W.J. Improved effectiveness of nanoparticle albumin-bound (nab) paclitaxel versus polysorbate-based docetaxel in multiple xenografts as a function of HER2 and SPARC status. Anticancer Drugs 2008, 19, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Nagaraju, G.P.; El-Rayes, B.F. SPARC and DNA methylation: Possible diagnostic and therapeutic implications in gastrointestinal cancers. Cancer Lett. 2013, 328, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Trieu, V.; Hwang, J.; Desai, N. Nanoparticle Albumin-bound (nab) technology may enhance antitumor activity via targeting of SPARC protein. Abstract 53; New Targets and Delivery System for Cancer Diagnosis and Treatment (SKCC): San Diego, CA, USA.
- Gardner, E.R.; Dahut, W.L.; Scripture, C.D.; Jones, J.; Aragon-Ching, J.B.; Desai, N.; Hawkins, M.J.; Sparreboom, A.; Figg, W.D. Randomized crossover pharmacokinetic study of solvent-based paclitaxel and nab-paclitaxel. Clin. Cancer Res. 2008, 14, 4200–4205. [Google Scholar] [CrossRef] [PubMed]
- Sparreboom, A.; Scripture, C.D.; Trieu, V.; Williams, P.J.; De, T.; Yang, A.; Beals, B.; Figg, W.D.; Hawkins, M.; Desai, N. Comparative preclinical and clinical pharmacokinetics of a cremophor-free, nanoparticle albumin-bound paclitaxel (ABI-007) and paclitaxel formulated in Cremophor (Taxol). Clin. Cancer Res. 2005, 11, 4136–4143. [Google Scholar] [CrossRef]
- Ueno, H.; Ikeda, M.; Ueno, M.; Mizuno, N.; Ioka, T.; Omuro, Y.; Nakajima, T.E.; Furuse, J. Phase I/II study of nab-paclitaxel plus gemcitabine for chemotherapy-naive Japanese patients with metastatic pancreatic cancer. Cancer Chemother. Pharmacol. 2016, 77, 595–603. [Google Scholar] [CrossRef]
- Cohen, S.J.; O’Neil, B.H.; Berlin, J.; Ames, P.; McKinley, M.; Horan, J.; Catalano, P.M.; Davies, A.; Weekes, C.D.; Leichman, L. A phase 1b study of erlotinib in combination with gemcitabine and nab-paclitaxel in patients with previously untreated advanced pancreatic cancer: An Academic Oncology GI Cancer Consortium study. Cancer Chemother. Pharmacol. 2016, 77, 693–701. [Google Scholar] [CrossRef]
- Nakayama, N.; Ishido, K.; Chin, K.; Nishimura, K.; Azuma, M.; Matsusaka, S.; Inokuchi, Y.; Tanabe, S.; Kumekawa, Y.; Koizumi, W. A phase I study of S-1 in combination with nab-paclitaxel in patients with unresectable or recurrent gastric cancer. Gastric Cancer 2017, 20, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A., 3rd; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Reni, M.; Cordio, S.; Milandri, C.; Passoni, P.; Bonetto, E.; Oliani, C.; Luppi, G.; Nicoletti, R.; Galli, L.; Bordonaro, R.; et al. Gemcitabine versus cisplatin, epirubicin, fluorouracil, and gemcitabine in advanced pancreatic cancer: A randomised controlled multicentre phase III trial. Lancet Oncol. 2005, 6, 369–376. [Google Scholar] [CrossRef]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef]
- Okada, S.; Sakata, Y.; Matsuno, S.; Kurihara, M.; Sasaki, Y.; Ohashi, Y.; Taguchi, T. Phase II study of docetaxel in patients with metastatic pancreatic cancer: A Japanese cooperative study. Cooperative Group of Docetaxel for Pancreatic Cancer in Japan. Br. J. Cancer 1999, 80, 438–443. [Google Scholar] [CrossRef]
- Rougier, P.; Adenis, A.; Ducreux, M.; de Forni, M.; Bonneterre, J.; Dembak, M.; Clouet, P.; Lebecq, A.; Baille, P.; Lefresne-Soulas, F.; et al. A phase II study: Docetaxel as first-line chemotherapy for advanced pancreatic adenocarcinoma. Eur. J. Cancer 2000, 36, 1016–1025. [Google Scholar] [CrossRef]
- Cereda, S.; Reni, M. Weekly docetaxel as salvage therapy in patients with gemcitabine-refractory metastatic pancreatic cancer. J. Chemother. 2008, 20, 509–512. [Google Scholar] [CrossRef]
- Saif, M.W.; Syrigos, K.; Penney, R.; Kaley, K. Docetaxel second-line therapy in patients with advanced pancreatic cancer: A retrospective study. Anticancer Res. 2010, 30, 2905–2909. [Google Scholar]
- Schneider, B.P.; Ganjoo, K.N.; Seitz, D.E.; Picus, J.; Fata, F.; Stoner, C.; Calley, C.; Loehrer, P.J. Phase II study of gemcitabine plus docetaxel in advanced pancreatic cancer: A Hoosier Oncology Group study. Oncology 2003, 65, 218–223. [Google Scholar] [CrossRef]
- Lutz, M.P.; Van Cutsem, E.; Wagener, T.; Van Laethem, J.L.; Vanhoefer, U.; Wils, J.A.; Gamelin, E.; Koehne, C.H.; Arnaud, J.P.; Mitry, E.; et al. Docetaxel plus gemcitabine or docetaxel plus cisplatin in advanced pancreatic carcinoma: Randomized phase II study 40984 of the European Organisation for Research and Treatment of Cancer Gastrointestinal Group. J. Clin. Oncol. 2005, 23, 9250–9256. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.P.; Kulke, M.H.; Fuchs, C.S.; Grossbard, M.L.; Grossman, S.R.; Morgan, J.A.; Earle, C.C.; Shivdasani, R.; Kim, H.; Mayer, R.J.; et al. A Phase II study of gemcitabine and docetaxel in patients with metastatic pancreatic carcinoma. Cancer 2002, 94, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, N.; Zhang, C.; Schwarz, A.M.; Hinz, S.; Wang, C.; Williams, N.S.; Schwarz, M.A.; Schwarz, R.E. Comparative benefits of Nab-paclitaxel over gemcitabine or polysorbate-based docetaxel in experimental pancreatic cancer. Carcinogenesis 2013, 34, 2361–2369. [Google Scholar] [CrossRef]
- Rajeshkumar, N.V.; Yabuuchi, S.; Pai, S.G.; Tong, Z.; Hou, S.; Bateman, S.; Pierce, D.W.; Heise, C.; Von Hoff, D.D.; Maitra, A.; et al. Superior therapeutic efficacy of nab-paclitaxel over cremophor-based paclitaxel in locally advanced and metastatic models of human pancreatic cancer. Br. J. Cancer 2016, 115, 442–453. [Google Scholar] [CrossRef]
- Mahtani, R.L.; Parisi, M.; Glück, S.; Ni, Q.; Park, S.; Pelletier, C.; Faria, C.; Braiteh, F. Comparative effectiveness of early-line nab-paclitaxel vs. paclitaxel in patients with metastatic breast cancer: A US community-based real-world analysis. Cancer. Manag. Res. 2018, 10, 249–256. [Google Scholar] [CrossRef]
- Von Hoff, D.D.; Ramanathan, R.K.; Borad, M.J.; Laheru, D.A.; Smith, L.S.; Wood, T.E.; Korn, R.L.; Desai, N.; Trieu, V.; Iglesias, J.L.; et al. Gemcitabine plus nab-paclitaxel is an active regimen in patients with advanced pancreatic cancer: A phase I/II trial. J. Clin. Oncol. 2011, 29, 4548–4554. [Google Scholar] [CrossRef]
- Hosein, P.J.; de Lima Lopes, G., Jr.; Pastorini, V.H.; Gomez, C.; Macintyre, J.; Zayas, G.; Reis, I.; Montero, A.J.; Merchan, J.R.; Rocha Lima, C.M. A phase II trial of nab-Paclitaxel as second-line therapy in patients with advanced pancreatic cancer. Am. J. Clin. Oncol. 2013, 36, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef]
- Bachet, J.B.; Hammel, P.; Desramé, J.; Meurisse, A.; Chibaudel, B.; André, T.; Debourdeau, P.; Dauba, J.; Lecomte, T.; Seitz, J.F.; et al. Nab-paclitaxel plus either gemcitabine or simplified leucovorin and fluorouracil as first-line therapy for metastatic pancreatic adenocarcinoma (AFUGEM GERCOR): A non-comparative, multicentre, open-label, randomised phase 2 trial. Lancet Gastroenterol. Hepatol. 2017, 2, 337–346. [Google Scholar] [CrossRef]
- Ko, A.H.; Murphy, P.B.; Peyton, J.D.; Shipley, D.L.; Al-Hazzouri, A.; Rodriguez, F.A.; Womack, M.S.t.; Xiong, H.Q.; Waterhouse, D.M.; Tempero, M.A.; et al. A Randomized, Double-Blinded, Phase II Trial of Gemcitabine and Nab-Paclitaxel Plus Apatorsen or Placebo in Patients with Metastatic Pancreatic Cancer: The RAINIER Trial. Oncologist 2017, 22, 1427-e1129. [Google Scholar] [CrossRef]
- Weiss, G.J.; Blaydorn, L.; Beck, J.; Bornemann-Kolatzki, K.; Urnovitz, H.; Schütz, E.; Khemka, V. Phase Ib/II study of gemcitabine, nab-paclitaxel, and pembrolizumab in metastatic pancreatic adenocarcinoma. Investig. New Drugs 2018, 36, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Hingorani, S.R.; Zheng, L.; Bullock, A.J.; Seery, T.E.; Harris, W.P.; Sigal, D.S.; Braiteh, F.; Ritch, P.S.; Zalupski, M.M.; Bahary, N.; et al. HALO 202: Randomized Phase II Study of PEGPH20 Plus Nab-Paclitaxel/Gemcitabine Versus Nab-Paclitaxel/Gemcitabine in Patients With Untreated, Metastatic Pancreatic Ductal Adenocarcinoma. J. Clin. Oncol. 2018, 36, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Tempero, M.A.; Sigal, D.; Oh, D.Y.; Fazio, N.; Macarulla, T.; Hitre, E.; Hammel, P.; Hendifar, A.E.; Bates, S.E.; et al. Randomized Phase III Trial of Pegvorhyaluronidase Alfa With Nab-Paclitaxel Plus Gemcitabine for Patients With Hyaluronan-High Metastatic Pancreatic Adenocarcinoma. J. Clin. Oncol. 2020, 38, 3185–3194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Du, C.; Sun, Y.; Yang, L.; Cui, C.; Jiang, Z.; Wang, C.; Wang, J.; Zhou, A. Nab-paclitaxel plus S-1 as first-line followed by S-1 maintenance for advanced pancreatic adenocarcinoma: A single-arm phase II trial. Cancer Chemother. Pharmacol. 2018, 82, 655–660. [Google Scholar] [CrossRef]
- Reni, M.; Zanon, S.; Peretti, U.; Chiaravalli, M.; Barone, D.; Pircher, C.; Balzano, G.; Macchini, M.; Romi, S.; Gritti, E.; et al. Nab-paclitaxel plus gemcitabine with or without capecitabine and cisplatin in metastatic pancreatic adenocarcinoma (PACT-19): A randomised phase 2 trial. Lancet Gastroenterol. Hepatol. 2018, 3, 691–697. [Google Scholar] [CrossRef]
- Mahipal, A.; Tella, S.H.; Kommalapati, A.; Goyal, G.; Soares, H.; Neuger, A.; Copolla, D.; Kim, J.; Kim, R. Phase 1 trial of enzalutamide in combination with gemcitabine and nab-paclitaxel for the treatment of advanced pancreatic cancer. Investig. New Drugs 2019, 37, 473–481. [Google Scholar] [CrossRef]
- Karasic, T.B.; O’Hara, M.H.; Loaiza-Bonilla, A.; Reiss, K.A.; Teitelbaum, U.R.; Borazanci, E.; De Jesus-Acosta, A.; Redlinger, C.; Burrell, J.A.; Laheru, D.A.; et al. Effect of Gemcitabine and nab-Paclitaxel With or Without Hydroxychloroquine on Patients With Advanced Pancreatic Cancer: A Phase 2 Randomized Clinical Trial. JAMA Oncol. 2019, 5, 993–998. [Google Scholar] [CrossRef]
- Zeh, H.J.; Bahary, N.; Boone, B.A.; Singhi, A.D.; Miller-Ocuin, J.L.; Normolle, D.P.; Zureikat, A.H.; Hogg, M.E.; Bartlett, D.L.; Lee, K.K.; et al. A Randomized Phase II Preoperative Study of Autophagy Inhibition with High-Dose Hydroxychloroquine and Gemcitabine/Nab-Paclitaxel in Pancreatic Cancer Patients. Clin. Cancer Res. 2020, 26, 3126–3134. [Google Scholar] [CrossRef]
- Hu, Z.I.; Bendell, J.C.; Bullock, A.; LoConte, N.K.; Hatoum, H.; Ritch, P.; Hool, H.; Leach, J.W.; Sanchez, J.; Sohal, D.P.S.; et al. A randomized phase II trial of nab-paclitaxel and gemcitabine with tarextumab or placebo in patients with untreated metastatic pancreatic cancer. Cancer Med. 2019, 8, 5148–5157. [Google Scholar] [CrossRef]
- Kundranda, M.; Gracian, A.C.; Zafar, S.F.; Meiri, E.; Bendell, J.; Algül, H.; Rivera, F.; Ahn, E.R.; Watkins, D.; Pelzer, U.; et al. Randomized, double-blind, placebo-controlled phase II study of istiratumab (MM-141) plus nab-paclitaxel and gemcitabine versus nab-paclitaxel and gemcitabine in front-line metastatic pancreatic cancer (CARRIE). Ann. Oncol. 2020, 31, 79–87. [Google Scholar] [CrossRef]
- De Jesus-Acosta, A.; Sugar, E.A.; O’Dwyer, P.J.; Ramanathan, R.K.; Von Hoff, D.D.; Rasheed, Z.; Zheng, L.; Begum, A.; Anders, R.; Maitra, A.; et al. Phase 2 study of vismodegib, a hedgehog inhibitor, combined with gemcitabine and nab-paclitaxel in patients with untreated metastatic pancreatic adenocarcinoma. Br. J. Cancer 2020, 122, 498–505. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, E.M.; Barone, D.; Mahalingam, D.; Bekaii-Saab, T.; Shao, S.H.; Wolf, J.; Rosano, M.; Krause, S.; Richards, D.A.; Yu, K.H.; et al. Randomised phase II trial of gemcitabine and nab-paclitaxel with necuparanib or placebo in untreated metastatic pancreas ductal adenocarcinoma. Eur. J. Cancer 2020, 132, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.P.; Samra, J.S.; Haghighi, K.S.; Donoghoe, M.W.; Burge, M.; Harris, M.T.; Chua, Y.J.; Mitchell, J.; O’Rourke, N.; Chan, H.; et al. The AGITG GAP Study: A Phase II Study of Perioperative Gemcitabine and Nab-Paclitaxel for Resectable Pancreas Cancer. Ann. Surg. Oncol. 2020, 27, 2506–2515. [Google Scholar] [CrossRef] [PubMed]
- Safran, H.; Charpentier, K.P.; Perez, K.; Mantripragada, K.; Miner, T.; DiPetrillo, T.; Kuritzky, B.; Apor, E.; Bishop, K.; Luppe, D.; et al. FOLFOX+Nab-Paclitaxel (FOLFOX-A) for Advanced Pancreatic Cancer: A Brown University Oncology Research Group Phase I Study. Am. J. Clin. Oncol. 2016, 39, 619–622. [Google Scholar] [CrossRef]
- Raufi, A.G.; Breakstone, R.; Leonard, K.; Charpentier, K.; Beard, R.; Renaud, J.; Cavanaugh, L.; Sturtevant, A.; MacKinnon, K.; Almhanna, K.; et al. Adjuvant FOLFOX+Nab-Paclitaxel (FOLFOX-A) for Pancreatic Cancer: A Brown University Oncology Research Group Phase II Study (BrUOG295). Am. J. Clin. Oncol. 2020, 43, 857–860. [Google Scholar] [CrossRef]
- Wainberg, Z.A.; Hochster, H.S.; Kim, E.J.; George, B.; Kaylan, A.; Chiorean, E.G.; Waterhouse, D.M.; Guiterrez, M.; Parikh, A.; Jain, R.; et al. Open-label, Phase I Study of Nivolumab Combined with nab-Paclitaxel Plus Gemcitabine in Advanced Pancreatic Cancer. Clin. Cancer Res. 2020, 26, 4814–4822. [Google Scholar] [CrossRef]
- O’Hara, M.H.; O’Reilly, E.M.; Varadhachary, G.; Wolff, R.A.; Wainberg, Z.A.; Ko, A.H.; Fisher, G.; Rahma, O.; Lyman, J.P.; Cabanski, C.R.; et al. CD40 agonistic monoclonal antibody APX005M (sotigalimab) and chemotherapy, with or without nivolumab, for the treatment of metastatic pancreatic adenocarcinoma: An open-label, multicentre, phase 1b study. Lancet Oncol. 2021, 22, 118–131. [Google Scholar] [CrossRef]
- Bloomston, M.; Zervos, E.E.; Rosemurgy, A.S., 2nd. Matrix metalloproteinases and their role in pancreatic cancer: A review of preclinical studies and clinical trials. Ann. Surg Oncol. 2002, 9, 668–674. [Google Scholar] [CrossRef]
- Awasthi, N.; Mikels-Vigdal, A.J.; Stefanutti, E.; Schwarz, M.A.; Monahan, S.; Smith, V.; Schwarz, R.E. Therapeutic efficacy of anti-MMP9 antibody in combination with nab-paclitaxel-based chemotherapy in pre-clinical models of pancreatic cancer. J. Cell Mol. Med. 2019, 23, 3878–3887. [Google Scholar] [CrossRef]
- Bendell, J.; Sharma, S.; Patel, M.R.; Windsor, K.S.; Wainberg, Z.A.; Gordon, M.; Chaves, J.; Berlin, J.; Brachmann, C.B.; Zavodovskaya, M.; et al. Safety and Efficacy of Andecaliximab (GS-5745) Plus Gemcitabine and Nab-Paclitaxel in Patients with Advanced Pancreatic Adenocarcinoma: Results from a Phase I Study. Oncologist 2020, 25, 954–962. [Google Scholar] [CrossRef]
- Kunzmann, V.; Siveke, J.T.; Algül, H.; Goekkurt, E.; Siegler, G.; Martens, U.; Waldschmidt, D.; Pelzer, U.; Fuchs, M.; Kullmann, F.; et al. Nab-paclitaxel plus gemcitabine versus nab-paclitaxel plus gemcitabine followed by FOLFIRINOX induction chemotherapy in locally advanced pancreatic cancer (NEOLAP-AIO-PAK-0113): A multicentre, randomised, phase 2 trial. Lancet Gastroenterol. Hepatol. 2021, 6, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Tempero, M.; Oh, D.Y.; Tabernero, J.; Reni, M.; Van Cutsem, E.; Hendifar, A.; Waldschmidt, D.T.; Starling, N.; Bachet, J.B.; Chang, H.M.; et al. Ibrutinib in combination with nab-paclitaxel and gemcitabine for first-line treatment of patients with metastatic pancreatic adenocarcinoma: Phase III RESOLVE study. Ann. Oncol. 2021, 32, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Giommoni, E.; Maiello, E.; Vaccaro, V.; Rondini, E.; Vivaldi, C.; Tortora, G.; Toppo, L.; Giordano, G.; Latiano, T.P.; Lamperini, C.; et al. Activity and Safety of NAB-FOLFIRI and NAB-FOLFOX as First-Line Treatment for metastatic Pancreatic Cancer (NabucCO Study). Curr. Oncol. 2021, 28, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Uemura, K.; Sudo, T.; Hashimoto, Y.; Sumiyoshi, T.; Okada, K.; Seo, S.; Otsuka, H.; Murakami, Y.; Takahashi, S. A phase II study of gemcitabine/nab-paclitaxel/S-1 combination neoadjuvant chemotherapy for patients with borderline resectable pancreatic cancer with arterial contact. Eur. J. Cancer 2021, 159, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, D.B.; Nissen, N.; Hatoum, H.; Musher, B.; Seng, J.; Coveler, A.L.; Al-Rajabi, R.; Yeo, C.J.; Leiby, B.; Banks, J.; et al. A Phase 3 Randomized Clinical Trial of Chemotherapy With or Without Algenpantucel-L (HyperAcute-Pancreas) Immunotherapy in Subjects With Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer. Ann. Surg. 2022, 275, 45–53. [Google Scholar] [CrossRef]
- Babiker, H.; Schlegel, P.J.; Hicks, L.G.; Bullock, A.J.; Burhani, N.; Mahadevan, D.; Elquza, E.; Borad, M.J.; Benaim, E.; Peterson, C.; et al. A multicenter phase 1/2 study investigating the safety, pharmacokinetics, pharmacodynamics and efficacy of a small molecule antimetabolite, RX-3117, plus nab-paclitaxel in pancreatic adenocarcinoma. Investig. New Drugs 2022, 40, 81–90. [Google Scholar] [CrossRef]
- Padrón, L.J.; Maurer, D.M.; O’Hara, M.H.; O’Reilly, E.M.; Wolff, R.A.; Wainberg, Z.A.; Ko, A.H.; Fisher, G.; Rahma, O.; Lyman, J.P.; et al. Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: Clinical and immunologic analyses from the randomized phase 2 PRINCE trial. Nat. Med. 2022, 28, 1167–1177. [Google Scholar] [CrossRef]
- Renouf, D.J.; Loree, J.M.; Knox, J.J.; Topham, J.T.; Kavan, P.; Jonker, D.; Welch, S.; Couture, F.; Lemay, F.; Tehfe, M.; et al. The CCTG PA.7 phase II trial of gemcitabine and nab-paclitaxel with or without durvalumab and tremelimumab as initial therapy in metastatic pancreatic ductal adenocarcinoma. Nat. Commun. 2022, 13, 5020. [Google Scholar] [CrossRef] [PubMed]
- Dean, A.; Gill, S.; McGregor, M.; Broadbridge, V.; Järveläinen, H.A.; Price, T. Dual αV-integrin and neuropilin-1 targeting peptide CEND-1 plus nab-paclitaxel and gemcitabine for the treatment of metastatic pancreatic ductal adenocarcinoma: A first-in-human, open-label, multicentre, phase 1 study. Lancet Gastroenterol. Hepatol. 2022, 7, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Ikenaga, N.; Miyasaka, Y.; Ohtsuka, T.; Nakata, K.; Adachi, T.; Eguchi, S.; Nishihara, K.; Inomata, M.; Kurahara, H.; Hisaka, T.; et al. A Prospective Multicenter Phase II Trial of Neoadjuvant Chemotherapy with Gemcitabine Plus Nab-Paclitaxel for Borderline Resectable Pancreatic Cancer with Arterial Involvement. Ann. Surg. Oncol. 2023, 30, 193–202. [Google Scholar] [CrossRef]
- Grierson, P.M.; Tan, B.; Pedersen, K.S.; Park, H.; Suresh, R.; Amin, M.A.; Trikalinos, N.A.; Knoerzer, D.; Kreider, B.; Reddy, A.; et al. Phase Ib Study of Ulixertinib Plus Gemcitabine and Nab-Paclitaxel in Patients with Metastatic Pancreatic Adenocarcinoma. Oncologist 2023, 28, e115–e123. [Google Scholar] [CrossRef] [PubMed]
- Ozaka, M.; Nakachi, K.; Kobayashi, S.; Ohba, A.; Imaoka, H.; Terashima, T.; Ishii, H.; Mizusawa, J.; Katayama, H.; Kataoka, T.; et al. A randomised phase II study of modified FOLFIRINOX versus gemcitabine plus nab-paclitaxel for locally advanced pancreatic cancer (JCOG1407). Eur. J. Cancer 2023, 181, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Feig, C.; Gopinathan, A.; Neesse, A.; Chan, D.S.; Cook, N.; Tuveson, D.A. The pancreas cancer microenvironment. Clin. Cancer Res. 2012, 18, 4266–4276. [Google Scholar] [CrossRef] [PubMed]
- Mantoni, T.S.; Schendel, R.R.; Rödel, F.; Niedobitek, G.; Al-Assar, O.; Masamune, A.; Brunner, T.B. Stromal SPARC expression and patient survival after chemoradiation for non-resectable pancreatic adenocarcinoma. Cancer Biol. Ther. 2008, 7, 1806–1815. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, P.P.; Cuevas, C.; Chang, A.E.; Goel, V.K.; Von Hoff, D.D.; Hingorani, S.R. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 2012, 21, 418–429. [Google Scholar] [CrossRef]
- Jacobetz, M.A.; Chan, D.S.; Neesse, A.; Bapiro, T.E.; Cook, N.; Frese, K.K.; Feig, C.; Nakagawa, T.; Caldwell, M.E.; Zecchini, H.I.; et al. Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer. Gut 2013, 62, 112–120. [Google Scholar] [CrossRef]
- Du, J.; Gu, J.; Li, J. Mechanisms of drug resistance of pancreatic ductal adenocarcinoma at different levels. Biosci. Rep. 2020, 40. [Google Scholar] [CrossRef]
- Erkan, M.; Kurtoglu, M.; Kleeff, J. The role of hypoxia in pancreatic cancer: A potential therapeutic target? Expert. Rev. Gastroenterol. Hepatol. 2016, 10, 301–316. [Google Scholar] [CrossRef]
- Sparano, J.A. Defining a role and predicting benefit from platinum-based therapy in breast cancer: An evolving story. J. Clin. Oncol. 2015, 33, 1–3. [Google Scholar] [CrossRef]
- Catenacci, D.V.; Junttila, M.R.; Karrison, T.; Bahary, N.; Horiba, M.N.; Nattam, S.R.; Marsh, R.; Wallace, J.; Kozloff, M.; Rajdev, L.; et al. Randomized Phase Ib/II Study of Gemcitabine Plus Placebo or Vismodegib, a Hedgehog Pathway Inhibitor, in Patients With Metastatic Pancreatic Cancer. J. Clin. Oncol. 2015, 33, 4284–4292. [Google Scholar] [CrossRef]
- Chan, J.S.K.; Sng, M.K.; Teo, Z.Q.; Chong, H.C.; Twang, J.S.; Tan, N.S. Targeting nuclear receptors in cancer-associated fibroblasts as concurrent therapy to inhibit development of chemoresistant tumors. Oncogene 2018, 37, 160–173. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhao, N.; Cui, J.; Wu, H.; Xiong, J.; Peng, T. Exosomes derived from cancer stem cells of gemcitabine-resistant pancreatic cancer cells enhance drug resistance by delivering miR-210. Cell Oncol. 2020, 43, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Mackey, J.R.; Mani, R.S.; Selner, M.; Mowles, D.; Young, J.D.; Belt, J.A.; Crawford, C.R.; Cass, C.E. Functional nucleoside transporters are required for gemcitabine influx and manifestation of toxicity in cancer cell lines. Cancer Res. 1998, 58, 4349–4357. [Google Scholar]
- Amrutkar, M.; Gladhaug, I.P. Pancreatic Cancer Chemoresistance to Gemcitabine. Cancers 2017, 9, 157. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, T.; Ramachandran, V.; Fournier, K.F.; Wang, H.; Marquis, L.; Abbruzzese, J.L.; Gallick, G.E.; Logsdon, C.D.; McConkey, D.J.; Choi, W. Epithelial to mesenchymal transition contributes to drug resistance in pancreatic cancer. Cancer Res. 2009, 69, 5820–5828. [Google Scholar] [CrossRef] [PubMed]
- Maloney, S.M.; Hoover, C.A.; Morejon-Lasso, L.V.; Prosperi, J.R. Mechanisms of Taxane Resistance. Cancers 2020, 12, 3323. [Google Scholar] [CrossRef]
- El Hassouni, B.; Li Petri, G.; Liu, D.S.K.; Cascioferro, S.; Parrino, B.; Hassan, W.; Diana, P.; Ali, A.; Frampton, A.E.; Giovannetti, E. Pharmacogenetics of treatments for pancreatic cancer. Expert. Opin. Drug Metab. Toxicol. 2019, 15, 437–447. [Google Scholar] [CrossRef]
- McCarroll, J.A.; Sharbeen, G.; Liu, J.; Youkhana, J.; Goldstein, D.; McCarthy, N.; Limbri, L.F.; Dischl, D.; Ceyhan, G.O.; Erkan, M.; et al. βIII-tubulin: A novel mediator of chemoresistance and metastases in pancreatic cancer. Oncotarget 2015, 6, 2235–2249. [Google Scholar] [CrossRef]
- Guo, F.; Li, J.; Qi, Y.; Hou, J.; Chen, H.; Jiang, S.W. HE4 overexpression decreases pancreatic cancer Capan-1 cell sensitivity to paclitaxel via cell cycle regulation. Cancer Cell Int. 2020, 20, 163. [Google Scholar] [CrossRef]
- Parasido, E.; Avetian, G.S.; Naeem, A.; Graham, G.; Pishvaian, M.; Glasgow, E.; Mudambi, S.; Lee, Y.; Ihemelandu, C.; Choudhry, M.; et al. The Sustained Induction of c-MYC Drives Nab-Paclitaxel Resistance in Primary Pancreatic Ductal Carcinoma Cells. Mol. Cancer Res. 2019, 17, 1815–1827. [Google Scholar] [CrossRef]
- Comandatore, A.; Immordino, B.; Balsano, R.; Capula, M.; Garajovà, I.; Ciccolini, J.; Giovannetti, E.; Morelli, L. Potential Role of Exosomes in the Chemoresistance to Gemcitabine and Nab-Paclitaxel in Pancreatic Cancer. Diagnostics 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Pandilla, R.; Kotapalli, V.; Gowrishankar, S.; Chigurupati, M.; Patnaik, S.; Uppin, S.; Rao, S.; Kalidindi, N.; Regulagadda, S.; Sundaram, C.; et al. Distinct genetic aberrations in oesophageal adeno and squamous carcinoma. Eur. J. Clin. Investig. 2013, 43, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Hassan, M.S.; Awasthi, N.; Li, J.; Williams, F.; Schwarz, M.A.; Schwarz, R.E.; von Holzen, U. Superior Therapeutic Efficacy of Nanoparticle Albumin Bound Paclitaxel Over Cremophor-Bound Paclitaxel in Experimental Esophageal Adenocarcinoma. Transl. Oncol. 2018, 11, 426–435. [Google Scholar] [CrossRef]
- Wang, H.Y.; Yao, Z.H.; Tang, H.; Zhao, Y.; Zhang, X.S.; Yao, S.N.; Yang, S.J.; Liu, Y.Y. Weekly nanoparticle albumin-bound paclitaxel in combination with cisplatin versus weekly solvent-based paclitaxel plus cisplatin as first-line therapy in Chinese patients with advanced esophageal squamous cell carcinoma. Onco-Targets Ther. 2016, 9, 5663–5669. [Google Scholar] [CrossRef]
- Shi, Y.; Qin, R.; Wang, Z.K.; Dai, G.H. Nanoparticle albumin-bound paclitaxel combined with cisplatin as the first-line treatment for metastatic esophageal squamous cell carcinoma. Onco-Targets Ther. 2013, 6, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Liao, F.X.; Rong, Y.M.; Yang, Q.; Yin, C.X.; He, W.Z.; Cai, X.Y.; Guo, G.F.; Qiu, H.J.; Chen, X.X.; et al. Efficacy of taxane-based regimens in a first-line setting for recurrent and/or metastatic Chinese patients with esophageal cancer. Asian Pac. J. Cancer Prev. 2014, 15, 5493–5498. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, W.; Qian, D.; Guan, Y.; Chen, X.; Zhang, H.; Wang, J.; Pang, Q. Efficacy and safety of weekly nab-paclitaxel plus cisplatin with concurrent intensity-modulated radiotherapy in patients with inoperable, locally advanced esophageal cancer: A pilot trial. Onco Targets Ther. 2018, 11, 6333–6338. [Google Scholar] [CrossRef]
- Huang, R.; Qiu, Z.; Zheng, C.; Zeng, R.; Chen, W.; Wang, S.; Li, E.; Xu, Y. Neoadjuvant Therapy for Locally Advanced Esophageal Cancers. Front. Oncol. 2022, 12, 734581. [Google Scholar] [CrossRef]
- Fan, Y.; Jiang, Y.; Zhou, X.; Chen, Q.; Huang, Z.; Xu, Y.; Gong, L.; Yu, H.; Yang, H.; Liu, J.; et al. Phase II study of neoadjuvant therapy with nab-paclitaxel and cisplatin followed by surgery in patients with locally advanced esophageal squamous cell carcinoma. Oncotarget 2016, 7, 50624–50634. [Google Scholar] [CrossRef]
- Tang, H.R.; Ma, H.F.; An, S.M.; Badakhshi, H.; Deng, J.Y.; Zhang, J.H.; Chen, Y.; Zhang, Z.; Guo, X.M.; Jiang, G.L.; et al. A Phase II Study of Concurrent Chemoradiotherapy With Paclitaxel and Cisplatin for Inoperable Esophageal Squamous Cell Carcinoma. Am. J. Clin. Oncol. 2016, 39, 350–354. [Google Scholar] [CrossRef]
- Yang, G.; Su, X.; Yang, H.; Luo, G.; Gao, C.; Zheng, Y.; Xie, W.; Huang, M.; Bei, T.; Bai, Y.; et al. Neoadjuvant programmed death-1 blockade plus chemotherapy in locally advanced esophageal squamous cell carcinoma. Ann. Transl. Med. 2021, 9, 1254. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, Y.; Liu, Z.; Fu, X.; Cai, X.; Li, H.; Zhu, L.; Shen, Y.; Zhang, H.; Sun, Y.; et al. Multicenter, single-arm, phase II trial of camrelizumab and chemotherapy as neoadjuvant treatment for locally advanced esophageal squamous cell carcinoma. J. Immunother. Cancer 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Duan, H.; Ni, Y.; Zhou, Y.; Wang, X.; Qi, H.; Gong, L.; Liu, H.; Tian, F.; Lu, Q.; et al. Tislelizumab combined with chemotherapy as neoadjuvant therapy for surgically resectable esophageal cancer: A prospective, single-arm, phase II study (TD-NICE). Int. J. Surg. 2022, 103, 106680. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, J.; Lin, W.; Shao, D.; Depypere, L.; Zhang, Z.; Li, Z.; Cui, F.; Du, Z.; Zeng, Y.; et al. Neoadjuvant camrelizumab plus chemotherapy for resectable, locally advanced esophageal squamous cell carcinoma (NIC-ESCC2019): A multicenter, phase 2 study. Int. J. Cancer 2022, 151, 128–137. [Google Scholar] [CrossRef]
- Yang, Y.; Li, H.; Chen, X.; Qin, J.; Li, Y.; Shen, Y.; Zhang, R.; Kang, X.; Wang, Z.; Zheng, Q.; et al. Comparison of neoadjuvant nab-paclitaxel plus immunotherapy versus paclitaxel plus immunotherapy for esophageal squamous cell carcinoma. Thorac. Cancer 2023. [Google Scholar] [CrossRef]
- Yang, W.; Xing, X.; Yeung, S.J.; Wang, S.; Chen, W.; Bao, Y.; Wang, F.; Feng, S.; Peng, F.; Wang, X.; et al. Neoadjuvant programmed cell death 1 blockade combined with chemotherapy for resectable esophageal squamous cell carcinoma. J. Immunother. Cancer 2022, 10. [Google Scholar] [CrossRef]
- Sasaki, Y.; Nishina, T.; Yasui, H.; Goto, M.; Muro, K.; Tsuji, A.; Koizumi, W.; Toh, Y.; Hara, T.; Miyata, Y. Phase II trial of nanoparticle albumin-bound paclitaxel as second-line chemotherapy for unresectable or recurrent gastric cancer. Cancer Sci. 2014, 105, 812–817. [Google Scholar] [CrossRef]
- Bando, H.; Shimodaira, H.; Fujitani, K.; Takashima, A.; Yamaguchi, K.; Nakayama, N.; Takahashi, T.; Oki, E.; Azuma, M.; Nishina, T.; et al. A phase II study of nab-paclitaxel in combination with ramucirumab in patients with previously treated advanced gastric cancer. Eur. J. Cancer 2018, 91, 86–91. [Google Scholar] [CrossRef]
- Sato, S.; Kunisaki, C.; Tanaka, Y.; Sato, K.; Miyamoto, H.; Yukawa, N.; Fujii, Y.; Kimura, J.; Takagawa, R.; Takahashi, M.; et al. A Phase II Study of Tri-weekly Low-dose Nab-paclitaxel Chemotherapy for Patients with Advanced Gastric Cancer. Anticancer Res. 2018, 38, 6911–6917. [Google Scholar] [CrossRef]
- Takashima, A.; Shitara, K.; Fujitani, K.; Koeda, K.; Hara, H.; Nakayama, N.; Hironaka, S.; Nishikawa, K.; Kimura, Y.; Amagai, K.; et al. Peritoneal metastasis as a predictive factor for nab-paclitaxel in patients with pretreated advanced gastric cancer: An exploratory analysis of the phase III ABSOLUTE trial. Gastric Cancer 2019, 22, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, D.; Mochizuki, Y.; Torii, K.; Takeda, S.; Kawase, Y.; Ishigure, K.; Teramoto, H.; Ando, M.; Kodera, Y. Phase II multi-institutional prospective trial of nab-paclitaxel as second-line chemotherapy for advanced gastric cancer refractory to fluoropyrimidine with modified dose reduction criteria (CCOG1303). Int. J. Clin. Oncol. 2020, 25, 1793–1799. [Google Scholar] [CrossRef] [PubMed]
- Tamura, S.; Taniguchi, H.; Nishikawa, K.; Imamura, H.; Fujita, J.; Takeno, A.; Matsuyama, J.; Kimura, Y.; Kawada, J.; Hirao, M.; et al. A phase II trial of dose-reduced nab-paclitaxel for patients with previously treated, advanced or recurrent gastric cancer (OGSG 1302). Int. J. Clin. Oncol. 2020, 25, 2035–2043. [Google Scholar] [CrossRef]
- Sahai, V.; Catalano, P.J.; Zalupski, M.M.; Lubner, S.J.; Menge, M.R.; Nimeiri, H.S.; Munshi, H.G.; Benson, A.B., 3rd; O’Dwyer, P.J. Nab-Paclitaxel and Gemcitabine as First-line Treatment of Advanced or Metastatic Cholangiocarcinoma: A Phase 2 Clinical Trial. JAMA Oncol. 2018, 4, 1707–1712. [Google Scholar] [CrossRef]
- Shroff, R.T.; Javle, M.M.; Xiao, L.; Kaseb, A.O.; Varadhachary, G.R.; Wolff, R.A.; Raghav, K.P.S.; Iwasaki, M.; Masci, P.; Ramanathan, R.K.; et al. Gemcitabine, Cisplatin, and nab-Paclitaxel for the Treatment of Advanced Biliary Tract Cancers: A Phase 2 Clinical Trial. JAMA Oncol. 2019, 5, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Ducreux, M.; Bennouna, J.; Adenis, A.; Conroy, T.; Lièvre, A.; Portales, F.; Jeanes, J.; Li, L.; Romano, A. Efficacy and safety of nab-paclitaxel in patients with previously treated metastatic colorectal cancer: A phase II COLO-001 trial. Cancer Chemother. Pharmacol. 2017, 79, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Overman, M.J.; Adam, L.; Raghav, K.; Wang, J.; Kee, B.; Fogelman, D.; Eng, C.; Vilar, E.; Shroff, R.; Dasari, A.; et al. Phase II study of nab-paclitaxel in refractory small bowel adenocarcinoma and CpG island methylator phenotype (CIMP)-high colorectal cancer. Ann. Oncol. 2018, 29, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.D.; Syn, N.L.; Moehler, M.; Grothe, W.; Yong, W.P.; Tai, B.C.; Ho, J.; Unverzagt, S. Chemotherapy for advanced gastric cancer. Cochrane Database Syst. Rev. 2017, 8, Cd004064. [Google Scholar] [CrossRef]
- Park, S.R.; Chun, J.H.; Kim, Y.W.; Lee, J.H.; Choi, I.J.; Kim, C.G.; Lee, J.S.; Bae, J.M.; Kim, H.K. Phase II study of low-dose docetaxel/fluorouracil/cisplatin in metastatic gastric carcinoma. Am. J. Clin. Oncol. 2005, 28, 433–438. [Google Scholar] [CrossRef]
- Sun, D.S.; Jeon, E.K.; Won, H.S.; Park, J.C.; Shim, B.Y.; Park, S.Y.; Hong, Y.S.; Kim, H.K.; Ko, Y.H. Outcomes in elderly patients treated with a single-agent or combination regimen as first-line chemotherapy for recurrent or metastatic gastric cancer. Gastric Cancer 2015, 18, 644–652. [Google Scholar] [CrossRef]
- Narahara, H.; Iishi, H.; Imamura, H.; Tsuburaya, A.; Chin, K.; Imamoto, H.; Esaki, T.; Furukawa, H.; Hamada, C.; Sakata, Y. Randomized phase III study comparing the efficacy and safety of irinotecan plus S-1 with S-1 alone as first-line treatment for advanced gastric cancer (study GC0301/TOP-002). Gastric Cancer 2011, 14, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Al-Batran, S.E.; Hofheinz, R.D.; Pauligk, C.; Kopp, H.G.; Haag, G.M.; Luley, K.B.; Meiler, J.; Homann, N.; Lorenzen, S.; Schmalenberg, H.; et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): Results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016, 17, 1697–1708. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, H.J.; Kim, S.Y.; Kim, T.Y.; Lee, K.W.; Baek, S.K.; Kim, T.Y.; Ryu, M.H.; Nam, B.H.; Zang, D.Y. Second-line chemotherapy versus supportive cancer treatment in advanced gastric cancer: A meta-analysis. Ann. Oncol. 2013, 24, 2850–2854. [Google Scholar] [CrossRef] [PubMed]
- Iacovelli, R.; Pietrantonio, F.; Farcomeni, A.; Maggi, C.; Palazzo, A.; Ricchini, F.; de Braud, F.; Di Bartolomeo, M. Chemotherapy or targeted therapy as second-line treatment of advanced gastric cancer. A systematic review and meta-analysis of published studies. PLoS ONE 2014, 9, e108940. [Google Scholar] [CrossRef] [PubMed]
- Ghosn, M.; Tabchi, S.; Kourie, H.R.; Tehfe, M. Metastatic gastric cancer treatment: Second line and beyond. World J. Gastroenterol. 2016, 22, 3069–3077. [Google Scholar] [CrossRef]
- Sym, S.J.; Hong, J.; Park, J.; Cho, E.K.; Lee, J.H.; Park, Y.H.; Lee, W.K.; Chung, M.; Kim, H.S.; Park, S.H.; et al. A randomized phase II study of biweekly irinotecan monotherapy or a combination of irinotecan plus 5-fluorouracil/leucovorin (mFOLFIRI) in patients with metastatic gastric adenocarcinoma refractory to or progressive after first-line chemotherapy. Cancer Chemother. Pharmacol. 2013, 71, 481–488. [Google Scholar] [CrossRef]
- Ford, H.E.; Marshall, A.; Bridgewater, J.A.; Janowitz, T.; Coxon, F.Y.; Wadsley, J.; Mansoor, W.; Fyfe, D.; Madhusudan, S.; Middleton, G.W.; et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): An open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014, 15, 78–86. [Google Scholar] [CrossRef]
- Awasthi, N.; Schwarz, M.A.; Zhang, C.; Klinz, S.G.; Meyer-Losic, F.; Beaufils, B.; Thiagalingam, A.; Schwarz, R.E. Augmenting Experimental Gastric Cancer Activity of Irinotecan through Liposomal Formulation and Antiangiogenic Combination Therapy. Mol. Cancer Ther. 2022, 21, 1149–1159. [Google Scholar] [CrossRef]
- Grojean, M.; Schwarz, M.A.; Schwarz, J.R.; Hassan, S.; von Holzen, U.; Zhang, C.; Schwarz, R.E.; Awasthi, N. Targeted dual inhibition of c-Met/VEGFR2 signalling by foretinib improves antitumour effects of nanoparticle paclitaxel in gastric cancer models. J. Cell Mol. Med. 2021, 25, 4950–4961. [Google Scholar] [CrossRef]
- Crawford, K.; Bontrager, E.; Schwarz, M.A.; Chaturvedi, A.; Lee, D.D.; Md Sazzad, H.; von Holzen, U.; Zhang, C.; Schwarz, R.E.; Awasthi, N. Targeted FGFR/VEGFR/PDGFR inhibition with dovitinib enhances the effects of nab-paclitaxel in preclinical gastric cancer models. Cancer Biol. Ther. 2021, 22, 619–629. [Google Scholar] [CrossRef]
- Awasthi, N.; Schwarz, M.A.; Zhang, C.; Schwarz, R.E. Augmentation of Nab-Paclitaxel Chemotherapy Response by Mechanistically Diverse Antiangiogenic Agents in Preclinical Gastric Cancer Models. Mol. Cancer Ther. 2018, 17, 2353–2364. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Awasthi, N.; Schwarz, M.A.; Schwarz, R.E. The dual PI3K/mTOR inhibitor NVP-BEZ235 enhances nab-paclitaxel antitumor response in experimental gastric cancer. Int. J. Oncol. 2013, 43, 1627–1635. [Google Scholar] [CrossRef]
- Shi, W.J.; Gao, J.B. Molecular mechanisms of chemoresistance in gastric cancer. World J. Gastrointest. Oncol. 2016, 8, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.; de la Fouchardière, C.; Kim, S.; Cohen, R.; Bachet, J.B.; Tournigand, C.; Ferraz, J.M.; Lefevre, M.; Colin, D.; Svrcek, M.; et al. Oxaliplatin, 5-Fluorouracil and Nab-paclitaxel as perioperative regimen in patients with resectable gastric adenocarcinoma: A GERCOR phase II study (FOXAGAST). Eur. J. Cancer 2019, 107, 46–52. [Google Scholar] [CrossRef]
- Blechacz, B.; Gores, G.J. Cholangiocarcinoma: Advances in pathogenesis, diagnosis, and treatment. Hepatology 2008, 48, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Macias, R.I. Cholangiocarcinoma: Biology, Clinical Management, and Pharmacological Perspectives. ISRN Hepatol. 2014, 2014, 828074. [Google Scholar] [CrossRef]
- Rizvi, S.; Gores, G.J. Pathogenesis, diagnosis, and management of cholangiocarcinoma. Gastroenterology 2013, 145, 1215–1229. [Google Scholar] [CrossRef]
- Gusani, N.J.; Balaa, F.K.; Steel, J.L.; Geller, D.A.; Marsh, J.W.; Zajko, A.B.; Carr, B.I.; Gamblin, T.C. Treatment of unresectable cholangiocarcinoma with gemcitabine-based transcatheter arterial chemoembolization (TACE): A single-institution experience. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract. 2008, 12, 129–137. [Google Scholar] [CrossRef]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef]
- Woodford, R.; Brungs, D.; Leighton, C.; Grimison, P.; Sjoquist, K.M.; Becker, T.; Robinson, S.; Gebski, V.; Wilson, K.; Chantrill, L.; et al. Combination chemotherapy with NAB(®) -paclitaxel and capecitabine for patients with advanced biliary tract cancer (NAP-CAPABIL Pilot Study). Asia Pac. J. Clin. Oncol. 2022, 18, e220–e226. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Barsouk, A. Epidemiology of colorectal cancer: Incidence, mortality, survival, and risk factors. Prz. Gastroenterol. 2019, 14, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Einzig, A.I.; Neuberg, D.; Wiernik, P.H.; Grochow, L.B.; Ramirez, G.; O’Dwyer, P.J.; Petrelli, N.J. Phase II Trial of Paclitaxel in Patients with Advanced Colon Cancer Previously Untreated with Cytotoxic Chemotherapy: An Eastern Cooperative Oncology Group Trial (PA286). Am. J. Ther. 1996, 3, 750–754. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, R.Z.; Duan, Z.; Lamendola, D.E.; Penson, R.T.; Seiden, M.V. Paclitaxel resistance: Molecular mechanisms and pharmacologic manipulation. Curr. Cancer Drug Targets 2003, 3, 1–19. [Google Scholar] [CrossRef]
- Cordon-Cardo, C.; O’Brien, J.P.; Boccia, J.; Casals, D.; Bertino, J.R.; Melamed, M.R. Expression of the multidrug resistance gene product (P-glycoprotein) in human normal and tumor tissues. J. Histochem. Cytochem. 1990, 38, 1277–1287. [Google Scholar] [CrossRef]
- Jang, S.H.; Wientjes, M.G.; Au, J.L. Kinetics of P-glycoprotein-mediated efflux of paclitaxel. J. Pharmacol. Exp. Ther. 2001, 298, 1236–1242. [Google Scholar] [PubMed]
- Koziara, J.M.; Whisman, T.R.; Tseng, M.T.; Mumper, R.J. In-vivo efficacy of novel paclitaxel nanoparticles in paclitaxel-resistant human colorectal tumors. J. Control Release 2006, 112, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Swanton, C.; Tomlinson, I.; Downward, J. Chromosomal instability, colorectal cancer and taxane resistance. Cell Cycle 2006, 5, 818–823. [Google Scholar] [CrossRef]
- Hartley, C.; Rowan, D.; Chen, X.; Gomez-Arellano, L.; West, A.M.; Oshima, K.; Mackinnon, A.C. Increased SPARC expression is associated with neoadjuvant therapy in resectable pancreatic ductal adenocarcinoma. Pract. Lab. Med. 2020, 21, e00171. [Google Scholar] [CrossRef]
- Hidalgo, M.; Plaza, C.; Musteanu, M.; Illei, P.; Brachmann, C.B.; Heise, C.; Pierce, D.; Lopez-Casas, P.P.; Menendez, C.; Tabernero, J.; et al. SPARC Expression Did Not Predict Efficacy of nab-Paclitaxel plus Gemcitabine or Gemcitabine Alone for Metastatic Pancreatic Cancer in an Exploratory Analysis of the Phase III MPACT Trial. Clin. Cancer Res. 2015, 21, 4811–4818. [Google Scholar] [CrossRef]
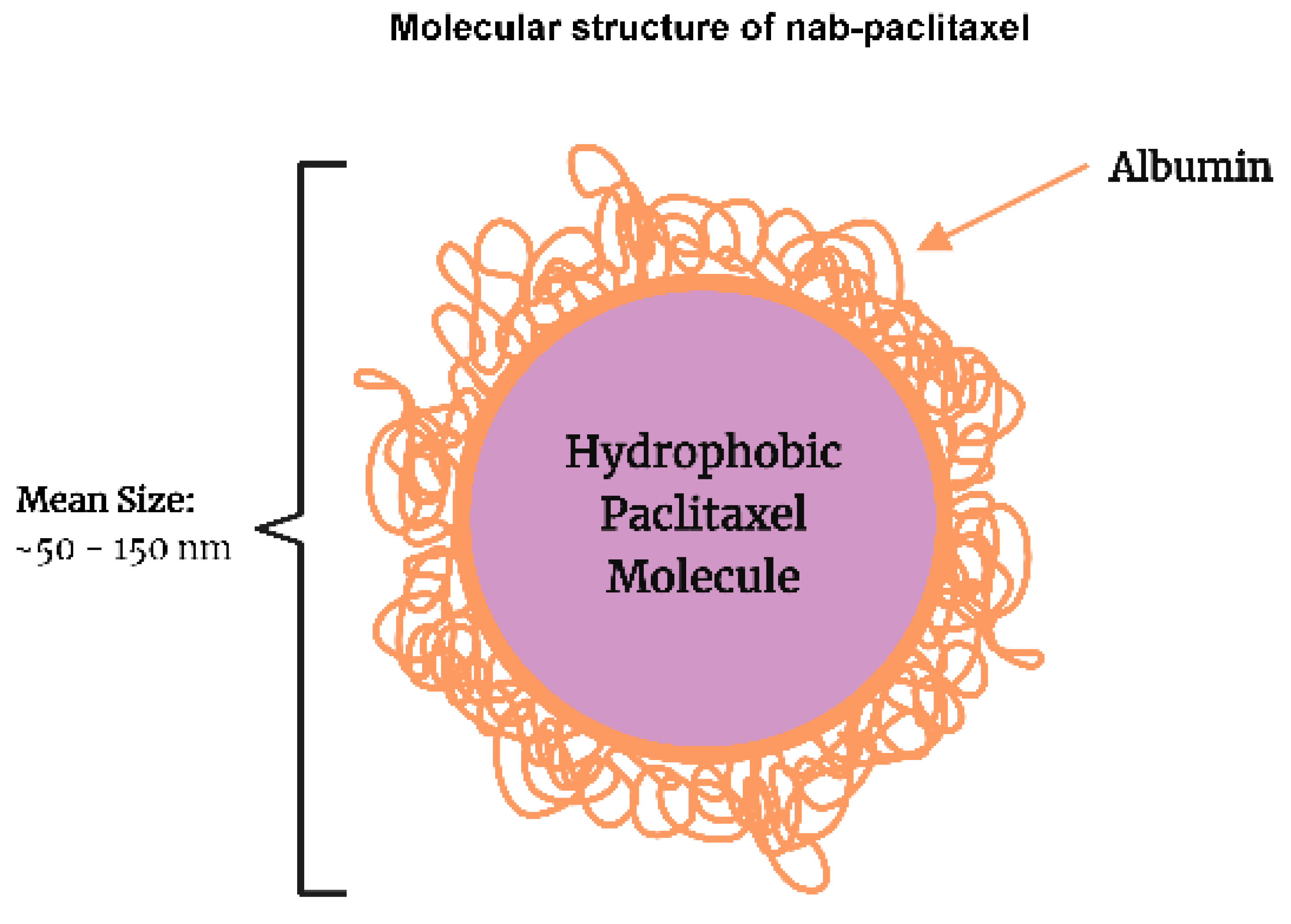
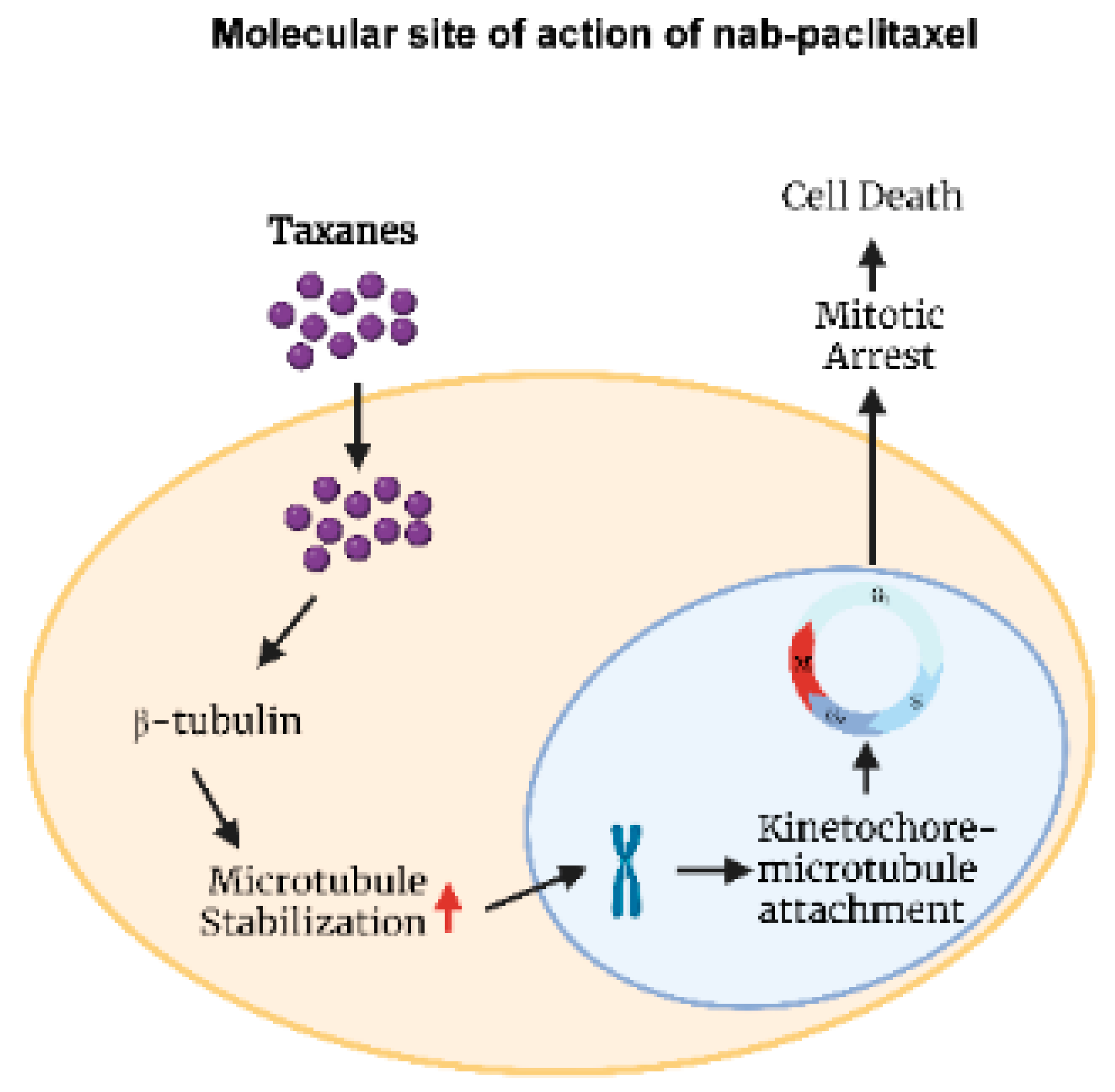
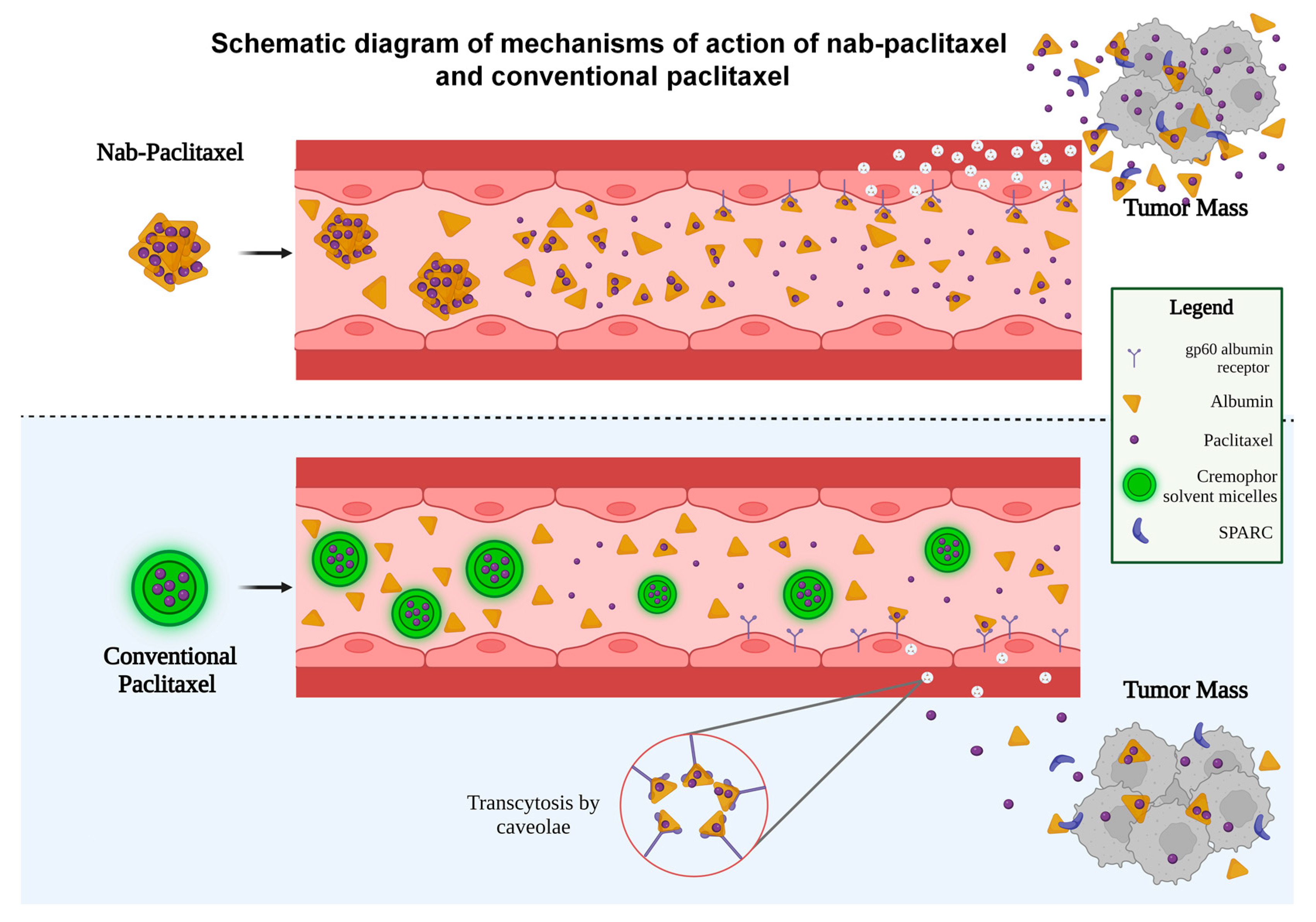
| Authors, | Patients | Therapeutic | ORR | Median | Median | Common |
|---|---|---|---|---|---|---|
| Year | Regimen | (%) | PFS (Mo) | OS (Mo) | Adverse Events % | |
| Hematologic (Grade ≥ 3) | ||||||
| Von Hoff et al., 2011 [39] | Untreated advanced | NG | 48 | 7.9 | 12.2 | neutropenia 67 leukopenia 44 Thrombocytopenia 23 |
| Hosein et al., 2013 [40] | Gemcitabine refractory | nab-pac | 58 | 1.7 | 7.3 | neutropenia 32 anemia 11 |
| Von Hoff et al., 2013 [41] | Untreated metastatic | NG vs. gemcitabine | 23 vs. 7 | 5.5 vs. 3.7 | 8.5 vs. 6.7 | neutropenia 38 vs. 27 leukopenia 31 vs. 16 thrombocytopenia 13 vs. 9 anemia 13 vs. 12 |
| Bachet et al., 2017 [42]. | untreated metastatic | NG vs nab-pac+ leucovorin/5-FU | at 4 m PFS rate 54% vs. 56% | neutropenia 32 vs. 23 thrombocytopenia 18 (NG anemia 13 (NG) | ||
| Ko et al., 2017 [43]. | Untreated metastatic | NG+apatorsen vs NG | 18 for both | 2.7 vs. 3.8 | 5.3 vs. 6.9 | leukopenia 0 (both) thrombocytopenia 0 (both) |
| Weiss et al., 2018 [44]. | untreated metastatic | NG+ pembrolizumab | 9.1 | 15 | neutropenia 47 thrombocytopenia 20 | |
| Hingorani et al., 2018 [45] | untreated metastatic | NG+PEGPH20 vs NG | In high hyaluronan patients | neutropenia 29 vs. 18 | ||
| 45 vs. 31 | 9.2 vs. 5.2 | 11.5 vs. 8.5 | thrombocytopenia 16 vs. 9 | |||
| Cutsem et al., 2020 [46]. | untreated Metastatic High hyaluronan | NG+PEGPH20 vs. NG | 47 vs. 36 | 7.1 vs. 7.1 | 11.2 vs. 11.5 | thrombocytopenia 21 vs. 16 |
| Zhang et al., 2018 [47]. | untreated advanced | nab-pac+ S1 then S1 maintenance | 53.1 | 6.2 | 13.6 | neutropenia 27.6 |
| Reni et al., 2018 [48]. | untreated metastatic | NG+cisplatin+ capecitabine vs. NG | at 6 months DFR 74 vs. 46% | neutropenia 41 vs. 39 anemia 21 vs. 22 | ||
| Karasic et al., 2019 [50]. | untreated advanced | NG+HCQ vs NG | 38.2 vs. 21 | 5.7 vs. 6.4 | 11.1 vs. 12.1 | neutropenia 42.6 vs. 22.6 anemia 3.7 vs. 17 |
| Zeh et al., 2020 [51]. | Untreated resectable | preoperative NG+ HCQ vs. NG | recurrence-free survival 16.6 vs. 13.5 | 36 vs. 32 | all grade ≥ 3 adverse events 62.5 vs. 60.5 | |
| Hu et al., 2019 [52]. | Untreated metastatic | NG+ Tarextumab vs NG | 20.2 vs. 31.8 | 3.7 vs. 5.5 | 6.4 vs. 7.9 | neutropenia 9 vs. 18 thrombocytopenia 49 vs. 25 anemia 29 vs. 26 |
| Kundranda et al, 2020 [53]. | untreated metastatic | NG+ Istiratumab vs NG | 39.5 vs. 51.2 | high IGF-1 3.6 vs. 7.3 high IGF-1/HRG+ 4.1 vs. 7.3 | 8.9 vs. 11.7 | neutropenia 30 vs. 34 anemia 18.6 vs. 18.2 thrombocytopenia 16 vs. 7 |
| De Jesus-Acosta et al, 2020 [54]. | untreated metastatic | NG+ vismodegib | 40 | 5.4 | 9.8 | No data about neutropenia, anemia or thrombocytopenia |
| O’Reilly et al., 2020 [55]. | Untreated metastatic | NG+ Necuparanib vs NG | 5.5 vs. 6.9 | 10.7 vs. 9.99 | neutropenia 33 vs. 34 thrombocytopenia 27 vs. 5 anemia 22 vs. 11 | |
| Barbour et al., 2020 [56]. | resectable | Perioperative NG | 12.3 | 23.5 | neutropenia 40 anemia 10 thrombocytopenia 5 | |
| Raufi et al., 2020 [58]. | resected | adjuvant FOLFOX plus nab-pac | 19.7 | 53.5 | neutropenia 26 | |
| Kunzmann et al, 2021 [64]. | locally advanced resectable | NG vs. NG followed by FOLFIRINOX | surgical conversion rate 35.9% vs. 43.9% | 18.5 vs. 20.7 | neutropenia 28 vs. 24 | |
| Tempero et al, 2021 [65]. | untreated metastatic | NG+ibrutinib vs. NG | 29 vs. 42 | 5.3 vs. 6 | 9.7 vs. 10.8 | neutropenia 24 vs. 35 anemia 16 vs. 17 |
| Giommoni et al., 2021 [66]. | untreated metastatic | nab-pac+FOLFIRI vs nab-pac+FOLFOX | 31 for both | 6 vs. 5.6 | 10.2 vs. 10.4 | neutropenia 19 vs. 29 febrile neutropenia 12 vs. 1 thrombocytopenia 2.4 vs. 0 anemia 7 vs. 10 |
| Kondo et al., 2021 [67]. | borderlineresectable arterial contact | Neoadjuvant NG+S-1 | 43 | 24.2 | 41 | neutropenia 25 leukopenia 19 anemia 2 |
| Hewitt et al, 2022 [68]. | Borderline resectable or locally advancedunresectable | soc [(FOLFIRINOX or NG) plus chemoradition] plus algenpantucel vs soc + placebo | 12.4 vs. 13.4 | 14.3 vs. 14.9 | all grade ≥ 3 adverse events 81 vs. 75 | |
| Babiker et al., 2022 [69]. | untreated metastatic | nab-pac plus RX-3117 | 23.1 | 5.6 | neutropenia 20 anemia 22 | |
| Padron et al., 2022 [70]. | untreated metastatic | NG+nivolumabNG+sotigalimab NG+sotiga+nivo | 50 vs. 33 vs 31 | 6.4 vs. 7.3 vs. 6.7 | 16.7 vs. 11.4 vs. 10.1 | neutropenia 33, 54, 57 thrombocytopenia 11, 16, 60 anemia 33, 24, 51 |
| Renouf et al., 2022 [71]. | untreated metastatic | NG+ durvalumab +tremelimumabvs NG | 30.3 vs. 23 | 5.5 vs. 5.4 | 9.8 vs. 8.8 | neutropenia 49 vs. 44 thrombocytopenia 11 vs. 16 anemia 22 vs. 32 lymphopenia 38 vs. 20 |
| Ikenaga et al., 2023 [73]. | borderline resectable with arterial involvement | Neoadjuvant NG | 24.9 | neutropenia 68 leukopenia 39 thrombocytopenia 7 anemia 4 | ||
| Ozaka et al., 2023 [75]. | untreated locally Advanced | mFOLFIRINOXvs NG | 30.9 vs. 42.1 | 11.2 vs. 9.4 | 23 vs. 21.3 | neutropenia 60 vs. 79 leukopenia 23 vs. 44 anemia 11 vs. 19 |
| Authors, | Patients | Therapeutic | ORR | Median | Median | Common |
|---|---|---|---|---|---|---|
| Year | Regimen | (%) | PFS (Mo) | OS (Mo) | Adverse Events % | |
| Hematologic (Grade ≥ 3) | ||||||
| Yun Fan et al., 2016 [103]. | neoadjuvantlocally advancedESCC | Nab-paclitaxel + cisplatin | ORR 65.7% | 34.7 | 37.8 | neutropenia 11.5 anemia 8.6 thrombocytopenia 5 |
| Guozhen Yang et al, 2021 [105]. | neoadjuvant locally advancedESCC | Nab-paclitaxel + camrelizumab + S1 | 33.33% (cPR) | no surgicaldelay | no preoperativedeath | neutropenia 0 anemia 0 thrombocytopenia 0 |
| Jun Liuet al, 2022 [106]. | neoadjuvantlocally advancedESCC | Nab-paclitaxel+ carboplatin+ camrelizumab | 39.2% (cPR) | no surgicaldelay | no preoperativedeath | neutropenia 50 anemia 6.7 thrombocytopenia 6.7 |
| Yafan Yang et al., 2023 [109]. | neoadjuvant locally resectable ESCCICIs+ paclitaxel + cisplatin | ICIs+ Nab-paclitaxel+ cisplatin vs. | 36.7% (cPR) vs. 21.4% (cPR) | PFS not yet reached | OS not yet reached | not yet reached |
| YasutsunaSasaki et al., 2014 [111]. | previously treated resectable or recurrent GC | Nab-paclitaxel | ORR 27.8% | 2.9 | 9.2 | neutropenia 49.1 anemia 7.3 thrombocytopenia 0 |
| Hideaki Bonda et al., 2018 [112]. | previouslytreated advanced GC | Nab-paclitaxel+ ramucirumab | ORR 54.8% | 7.6 | not yet reached | neutropenia 76.7 anemia 11.6 |
| Sho Sato et al., 2018 [113]. | previously treatedunresectable or recurrent GC | Nab-paclitaxel | ORR 5.9% | 2.4 | 9.2 | neutropenia 5.9 anemia 8.8 |
| Atsuo Takashima et al., 2019 [114]. | pretreated advancedPM GC | Nab-paclitaxel vs. sb-paclitaxel | ------- | 4.0 vs. 2.6 | 7.6 vs. 4.9 | -------- |
| DaisukeKobayashi et al., 2020 [115]. | previously treated advanced GC | Nab-paclitaxel | ORR 16% | 3.5 | 9.0 | neutropenia 49 anemia 2 thrombocytopenia 0 |
| ShegeyukiTamura et al, 2020 [116]. | previouslytreatedunresectable or recurrent GC | Nab-paclitaxel (low dose) | ORR 3.1% | 2.2 | 6.3 | neutropenia 37.5 anemia 12.5 thrombocytopenia 3.1 |
| VaibhavSahaiet al, 2018 [117]. | First line therapyadvanced or metastatic CCA | Nab-paclitaxel+ gemcitabine | ORR 30% | 7.7 | 12.4 | neutropenia 43 anemia 15 thrombocytopenia 16 |
| Rachna Shroff et al., 2019 [118]. | First line therapy advanced BTCs | Nab-paclitaxel + gemcitabine + cisplatin | PRR 45% | 11.8 | 19.2 | neutropenia 33 anemia 16 thrombocytopenia 13 |
| Michel Ducreux et al., 2017 [119]. | pretreatedmCRC | Nab-paclitaxel | ORR 0% | 8.1 weeks | -------- | neutropenia 22 anemia 5 |
| Overman et al., 2018 [120]. | pretreated refractory SBAvsCIMP high CRC | Nab-paclitaxel | ORR 20% vs. ORR 0% | 3.2 vs. 2.1 | not yet reached | neutropenia 9 anemia 0 thrombocytopenia 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassan, M.S.; Awasthi, N.; Ponna, S.; von Holzen, U. Nab-Paclitaxel in the Treatment of Gastrointestinal Cancers—Improvements in Clinical Efficacy and Safety. Biomedicines 2023, 11, 2000. https://doi.org/10.3390/biomedicines11072000
Hassan MS, Awasthi N, Ponna S, von Holzen U. Nab-Paclitaxel in the Treatment of Gastrointestinal Cancers—Improvements in Clinical Efficacy and Safety. Biomedicines. 2023; 11(7):2000. https://doi.org/10.3390/biomedicines11072000
Chicago/Turabian StyleHassan, Md Sazzad, Niranjan Awasthi, Saisantosh Ponna, and Urs von Holzen. 2023. "Nab-Paclitaxel in the Treatment of Gastrointestinal Cancers—Improvements in Clinical Efficacy and Safety" Biomedicines 11, no. 7: 2000. https://doi.org/10.3390/biomedicines11072000
APA StyleHassan, M. S., Awasthi, N., Ponna, S., & von Holzen, U. (2023). Nab-Paclitaxel in the Treatment of Gastrointestinal Cancers—Improvements in Clinical Efficacy and Safety. Biomedicines, 11(7), 2000. https://doi.org/10.3390/biomedicines11072000





