GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting
Abstract
1. Introduction
2. Materials and Methods
2.1. Approval of Animal Studies
2.2. Carbon Tetrachloride Liver Fibrosis Model
2.3. Skeletal Muscle Injury Model
2.4. Gene Expression Analysis
2.5. Protein Quantification
2.6. Histology and Immunohistochemistry
2.7. Human Liver Sample Collection
2.8. ActRIIA-Fc and ActRIIB-Fc Proteins and Antibodies
2.9. Body Composition
2.10. Cell Culture and In Vitro Assays
2.11. Data Analysis and Presentation
3. Results
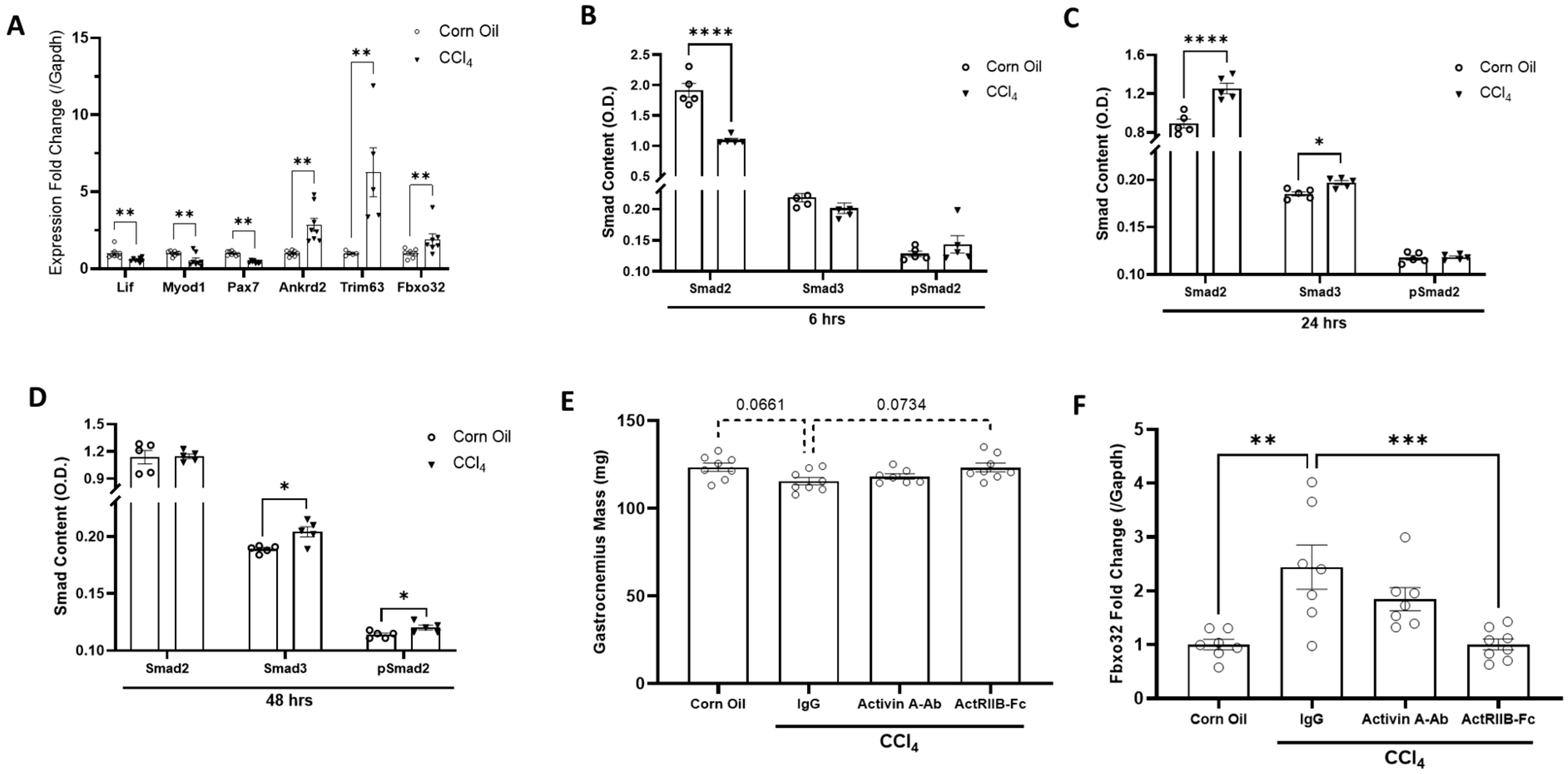
3.1. Acute Liver Injury Induces Rapid and Negative Responses in Skeletal Muscle
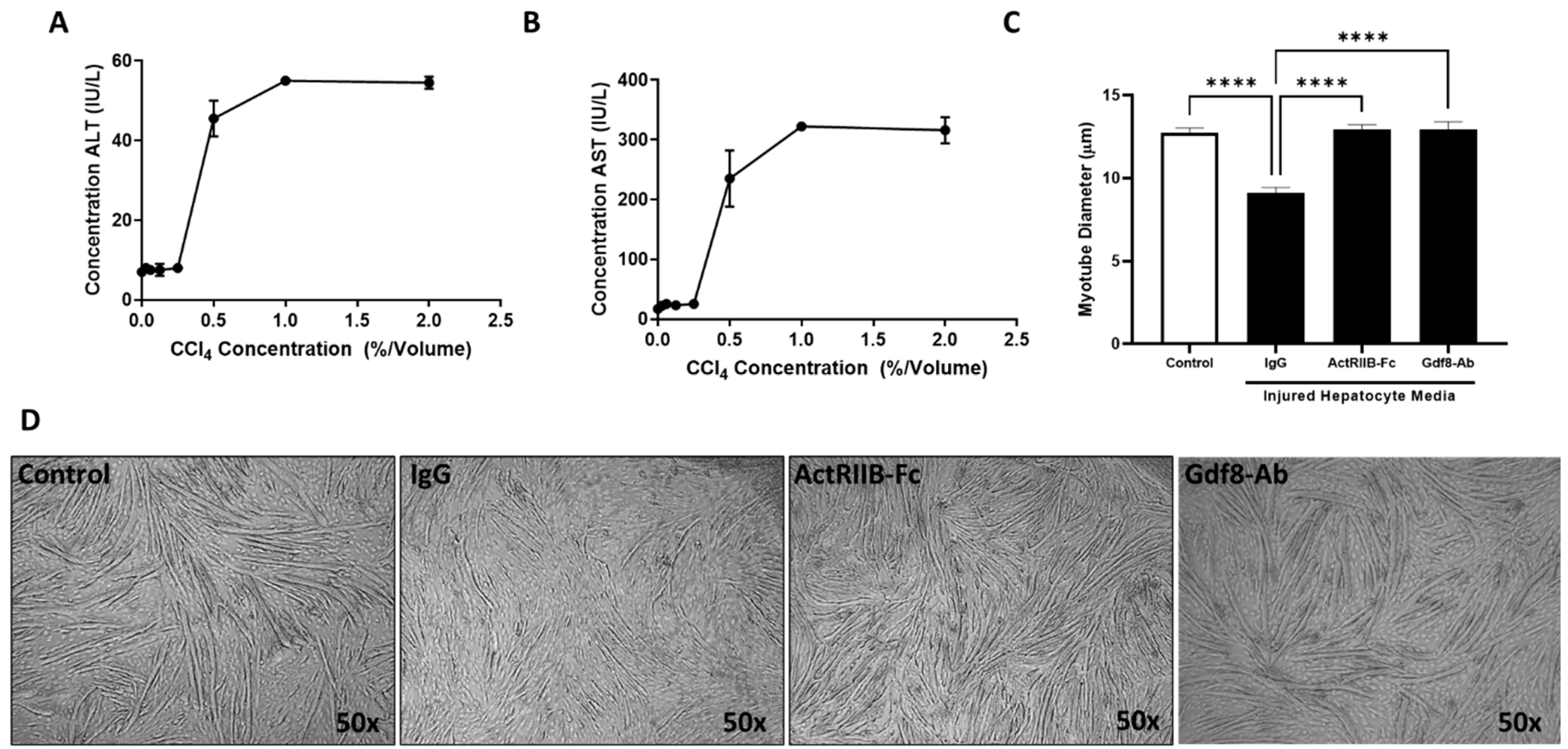
3.2. Neutralization of ActRIIB Ligands but Not Activin A May Prevent the Initiation of Liver Injury-Induced Muscle Atrophy
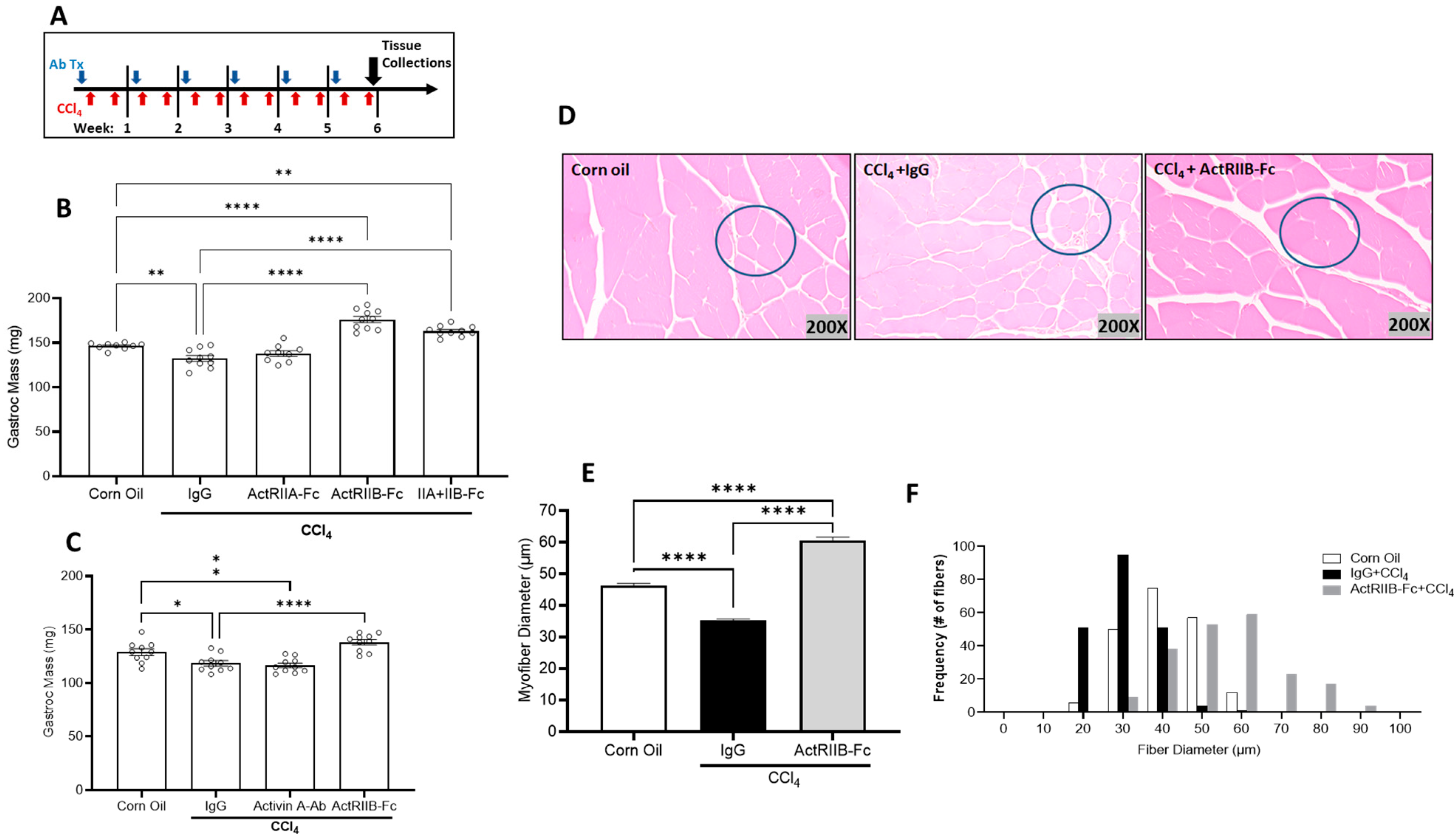
3.3. Neutralization of ActRIIB Ligands Prevents Chronic Liver Injury-Induced Muscle Atrophy Independent of Gender
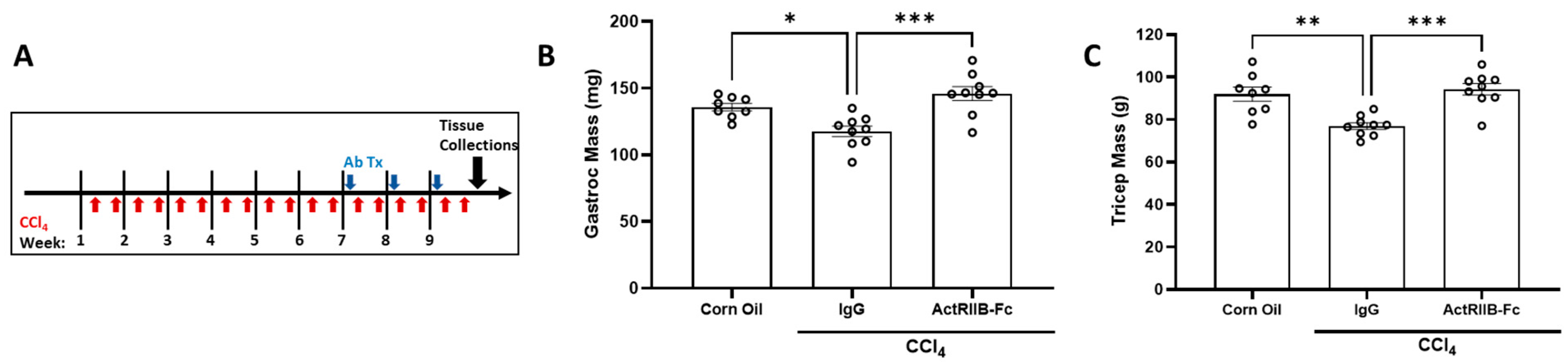
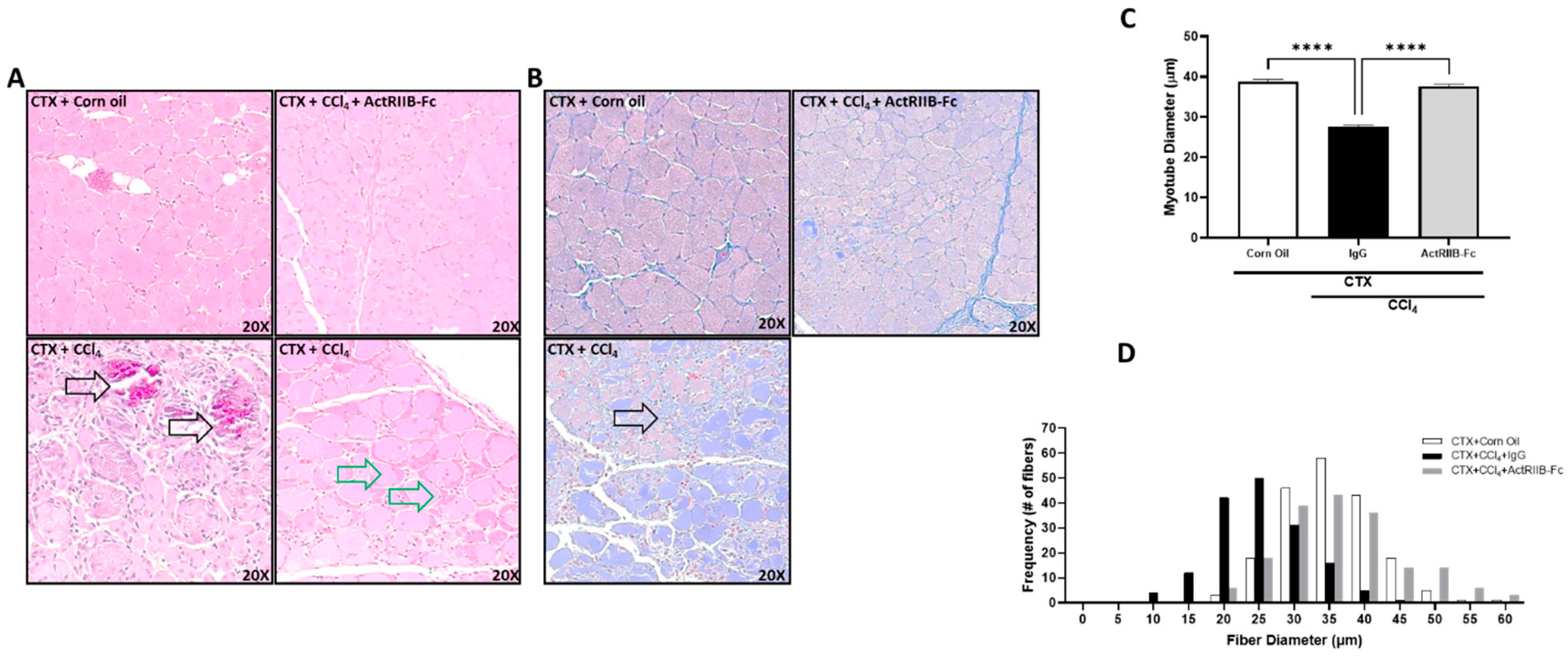
3.4. Neutralization of ActRIIB Ligands Reverses Chronic Liver Injury-Induced Muscle Atrophy
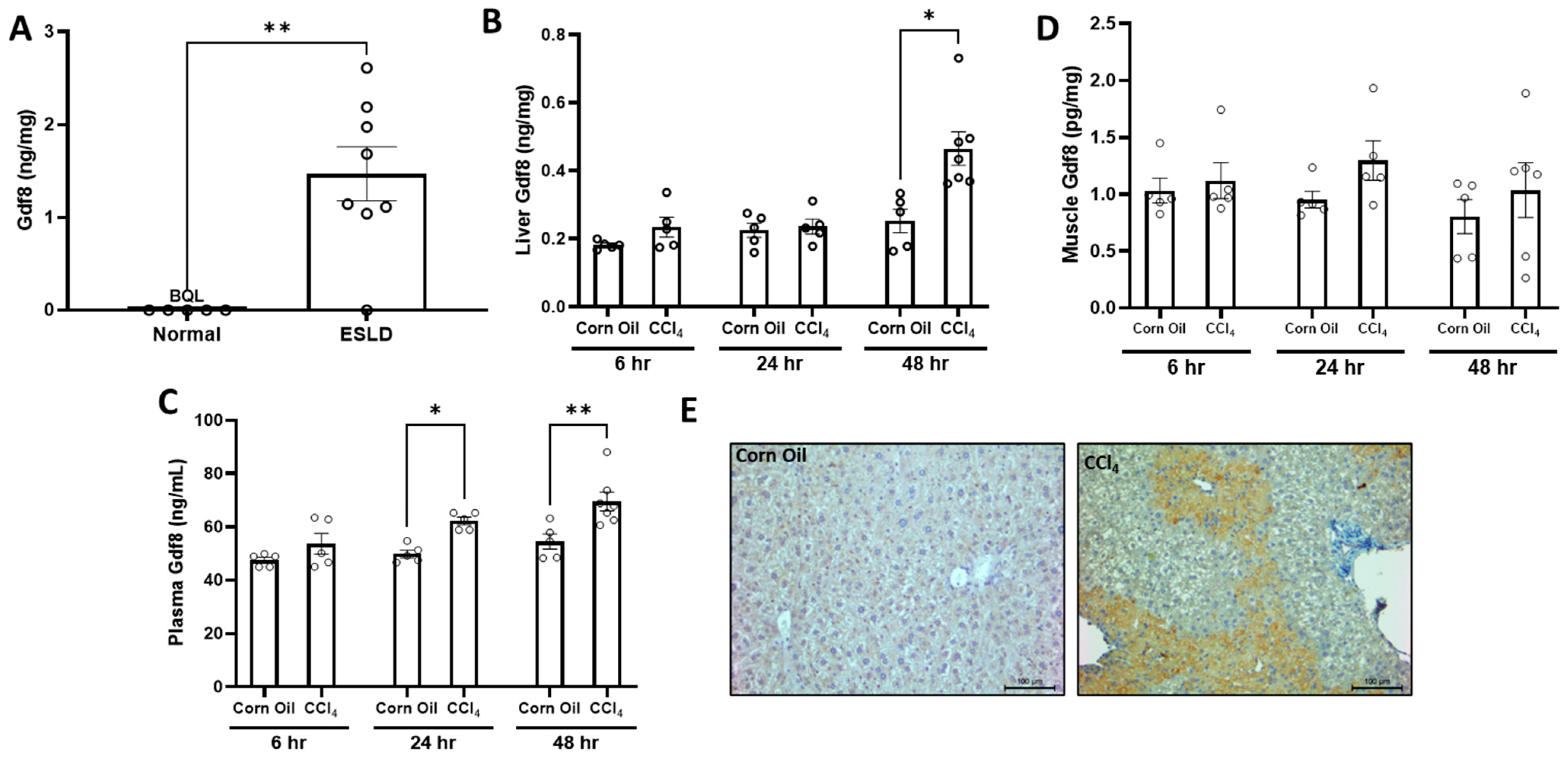
3.5. Injured Liver Produces Gdf8 in Both Humans and Mice
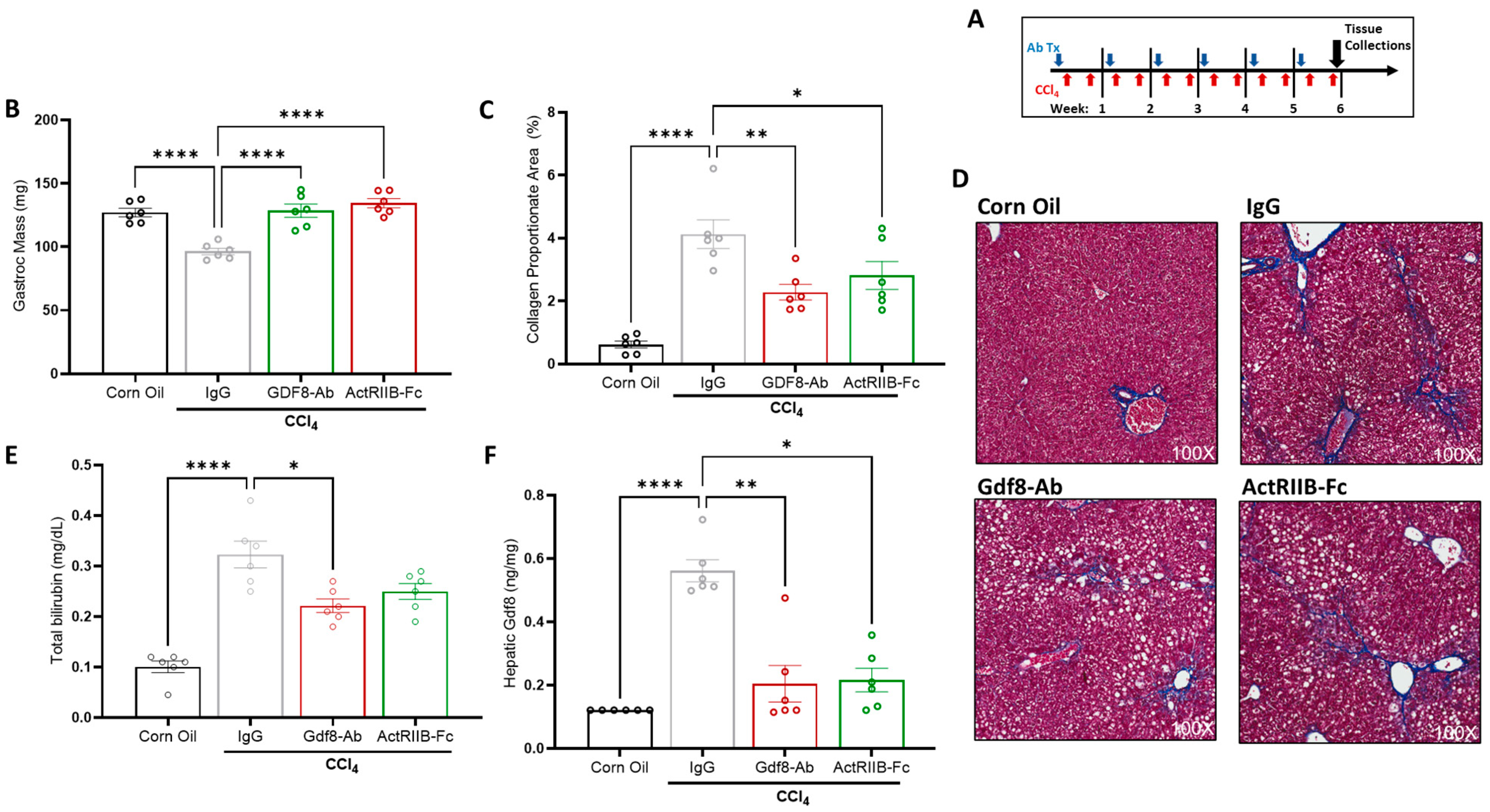
3.6. Neutralization of Gdf8 Prevents Chronic Liver Injury and Concomitant Muscle Mass Loss
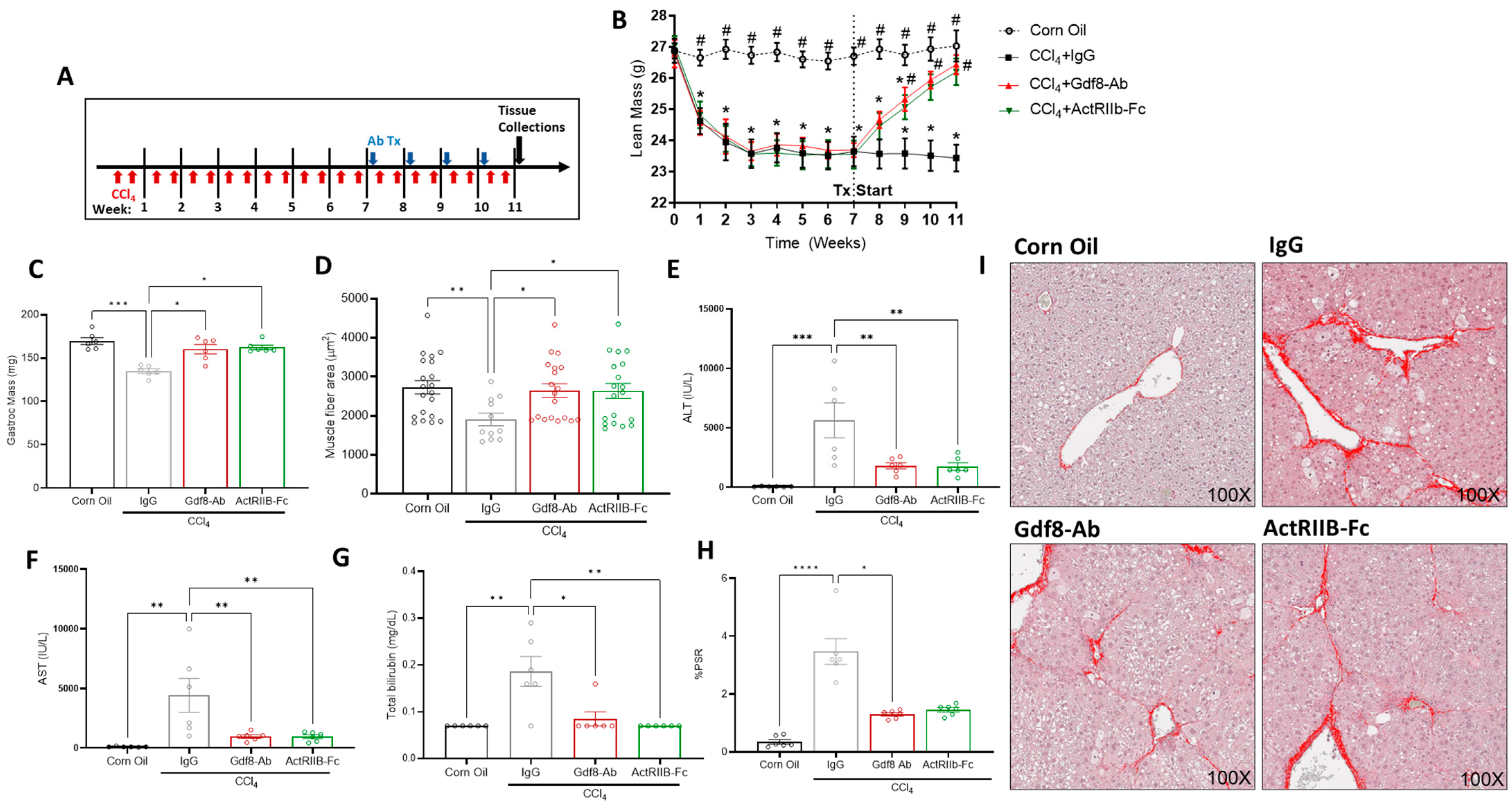
3.7. Neutralization of Gdf8 Reverses Chronic Liver Injury and Concomitant Muscle Mass Loss
3.8. Liver Injury Negatively Affects Muscle Regeneration, Which Is Prohibited by Neutralization of ActRIIB Ligands
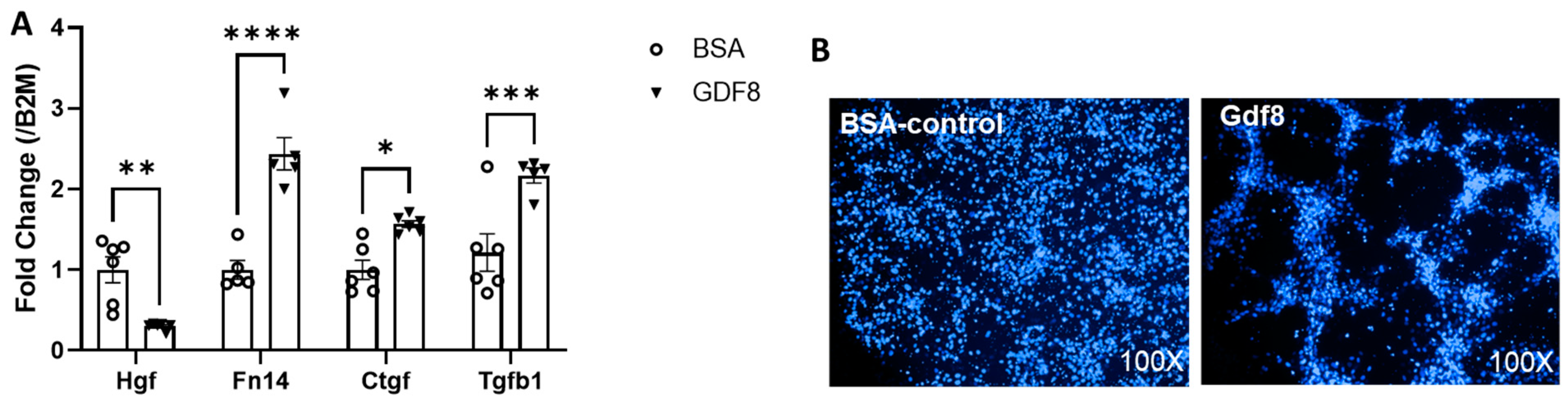
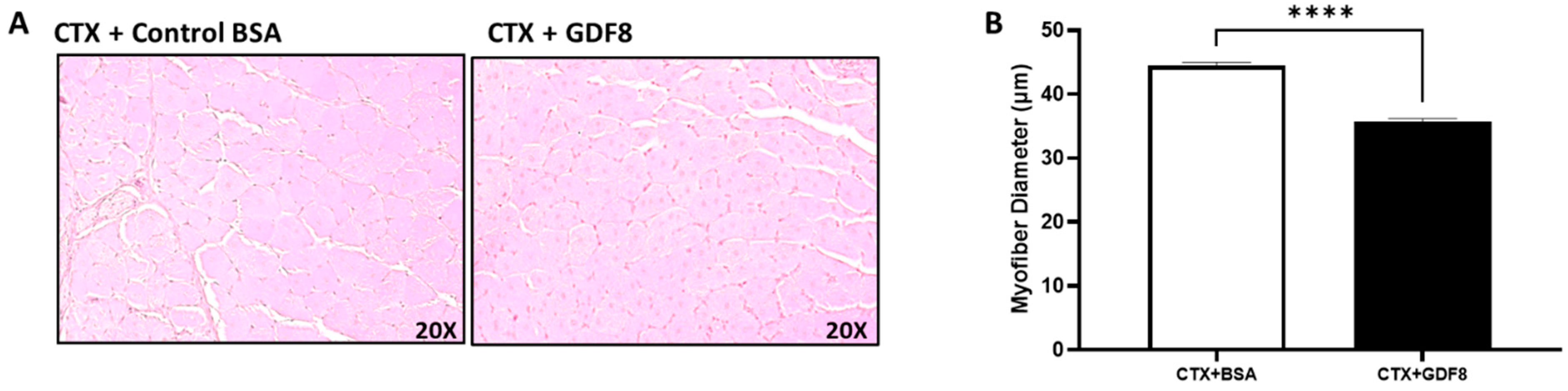
3.9. Exogenous Gdf8 Disrupts Muscle Regeneration, Mimicking Liver Injury
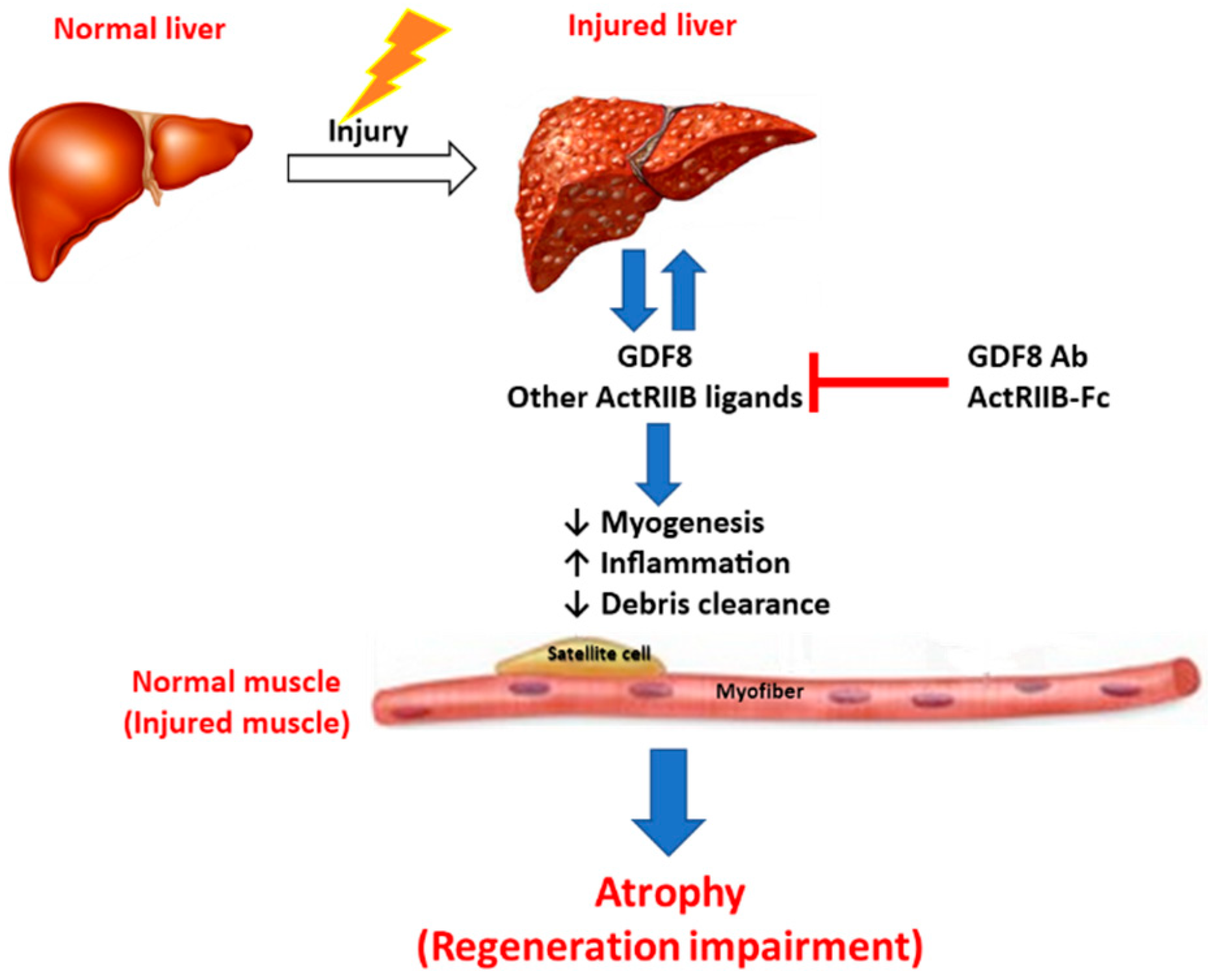
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kruszynska, Y.; Williams, N.; Perry, M.; Home, P. The relationship between insulin sensitivity and skeletal muscle enzyme activities in hepatic cirrhosis. Hepatology 1988, 6, 1615–1619. [Google Scholar] [CrossRef]
- Hsu, C.S.; Kao, J.H. Sarcopenia and chronic liver diseases. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 1229–1244. [Google Scholar] [CrossRef] [PubMed]
- Bhanji, R.A.; Narayanan, P.; Allen, A.M.; Malhi, H.; Watt, K.D. Sarcopenia in hiding: The risk and consequence of underestimating muscle dysfunction in nonalcoholic steatohepatitis. Hepatology 2017, 66, 2055–2065. [Google Scholar] [CrossRef]
- Koo, B.K.; Kim, D.; Joo, S.K.; Kim, J.H.; Chang, M.S.; Kim, B.G.; Lee, K.L.; Kim, W. Sarcopenia is an independent risk factor for non-alcoholic steatohepatitis and significant fibrosis. J. Hepatol. 2017, 66, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Kim, S.U.; Song, K.; Park, J.Y.; Kim, D.Y.; Ahn, S.H.; Lee, B.W.; Kang, E.S.; Cha, B.S.; Han, K.H. Sarcopenia is associated with significant liver fibrosis independently of obesity and insulin resistance in nonalcoholic fatty liver disease: Nationwide surveys (KNHANES 2008–2011). Hepatology 2016, 63, 776–786. [Google Scholar] [CrossRef]
- Petta, S.; Ciminnisi, S.; Di Marco, V.; Cabibi, D.; Camma, C.; Licata, A.; Marchesini, G.; Craxi, A. Sarcopenia is associated with severe liver fibrosis in patients with non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2017, 45, 510–518. [Google Scholar] [CrossRef]
- Alberino, F.; Gatta, A.; Amodio, P.; Merkel, C.; Di Pascoli, L.; Boffo, G.; Caregaro, L. Nutrition and survival in patients with liver cirrhosis. Nutrition 2001, 17, 445–450. [Google Scholar] [CrossRef]
- Merli, M.; Romiti, A.; Riggio, O.; Capocaccia, L. Optimal nutritional indexes in chronic liver disease. JPEN J. Parenter. Enter. Nutr. 1987, 11 (Suppl. 5), 130S–134S. [Google Scholar] [CrossRef]
- Merli, M.; Riggio, O.; Dally, L. Does malnutrition affect survival in cirrhosis? PINC (Policentrica Italiana Nutrizione Cirrosi). Hepatology 1996, 23, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.C.; Hwang, S.Y.; Choi, H.Y.; Yoo, H.J.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, D.S.; Choi, K.M. Relationship between sarcopenia and nonalcoholic fatty liver disease: The Korean Sarcopenic Obesity Study. Hepatology 2014, 59, 1772–1778. [Google Scholar] [CrossRef]
- Choudhary, N.S.; Saigal, S.; Saraf, N.; Mohanka, R.; Rastogi, A.; Goja, S.; Menon, P.B.; Mishra, S.; Mittal, A.; Soin, A.S. Sarcopenic obesity with metabolic syndrome: A newly recognized entity following living donor liver transplantation. Clin. Transplant. 2015, 29, 211–215. [Google Scholar] [CrossRef]
- Ooi, P.H.; Hager, A.; Mazurak, V.C.; Dajani, K.; Bhargava, R.; Gilmour, S.M.; Mager, D.R. Sarcopenia in Chronic Liver Disease: Impact on Outcomes. Liver Transpl. 2019, 25, 1422–1438. [Google Scholar] [CrossRef]
- Montano-Loza, A.J. Skeletal muscle abnormalities and outcomes after liver transplantation. Liver Transpl. 2014, 20, 1293–1295. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.; Lord, S.R.; Studenski, S.A.; Warden, S.J.; Fielding, R.A.; Recknor, C.P.; Hochberg, M.C.; Ferrari, S.L.; Blain, H.; Binder, E.F.; et al. Myostatin antibody (LY2495655) in older weak fallers: A proof-of-concept, randomised, phase 2 trial. Lancet Diabetes Endocrinol. 2015, 3, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Rooks, D.; Praestgaard, J.; Hariry, S.; Laurent, D.; Petricoul, O.; Perry, R.G.; Lach-Trifilieff, E.; Roubenoff, R. Treatment of Sarcopenia with Bimagrumab: Results from a Phase II, Randomized, Controlled, Proof-of-Concept Study. J. Am. Geriatr. Soc. 2017, 65, 1988–1995. [Google Scholar] [CrossRef] [PubMed]
- Attie, K.M.; Borgstein, N.G.; Yang, Y.; Condon, C.H.; Wilson, D.M.; Pearsall, A.E.; Kumar, R.; Willins, D.A.; Seehra, J.S.; Sherman, M.L. A single ascending-dose study of muscle regulator ACE-031 in healthy volunteers. Muscle Nerve 2013, 47, 416–423. [Google Scholar] [CrossRef]
- Dasarathy, S.; Merli, M. Sarcopenia from mechanism to diagnosis and treatment in liver disease. J. Hepatol. 2016, 65, 1232–1244. [Google Scholar] [CrossRef]
- Liedtke, C.; Luedde, T.; Sauerbruch, T.; Scholten, D.; Streetz, K.; Tacke, F.; Tolba, R.; Trautwein, C.; Trebicka, J.; Weiskirchen, R. Experimental liver fibrosis research: Update on animal models, legal issues and translational aspects. Fibrogenesis Tissue Repair 2013, 6, 19. [Google Scholar] [CrossRef]
- Slater, T.F.; Cheeseman, K.H.; Ingold, K.U. Carbon tetrachloride toxicity as a model for studying free-radical mediated liver injury. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1985, 311, 633–645. [Google Scholar] [CrossRef]
- Heindryckx, F.; Colle, I.; Van Vlierberghe, H. Experimental mouse models for hepatocellular carcinoma research. Int. J. Exp. Pathol. 2009, 90, 367–386. [Google Scholar] [CrossRef]
- Nevzorova, Y.A.; Bangen, J.M.; Hu, W.; Haas, U.; Weiskirchen, R.; Gassler, N.; Huss, S.; Tacke, F.; Sicinski, P.; Trautwein, C.; et al. Cyclin E1 controls proliferation of hepatic stellate cells and is essential for liver fibrogenesis in mice. Hepatology 2012, 56, 1140–1149. [Google Scholar] [CrossRef]
- Ikushima, H.; Miyazono, K. Biology of transforming growth factor-beta signaling. Curr. Pharm. Biotechnol. 2011, 12, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Ikushima, H.; Miyazono, K. TGF-beta signal transduction spreading to a wider field: A broad variety of mechanisms for context-dependent effects of TGF-beta. Cell Tissue Res. 2012, 347, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, K. Activins, myostatin and related TGF-beta family members as novel therapeutic targets for endocrine, metabolic and immune disorders. Curr. Drug Targets Immune Endocr. Metab. Disord. 2004, 4, 157–166. [Google Scholar] [CrossRef]
- Fabregat, I.; Moreno-Caceres, J.; Sanchez, A.; Dooley, S.; Dewidar, B.; Giannelli, G.; Ten Dijke, P.; Consortium, I.-L. TGF-beta signalling and liver disease. FEBS J. 2016, 283, 2219–2232. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Colgan, T.D.; Walton, K.L.; Gregorevic, P.; Harrison, C.A. The TGF-beta Signalling Network in Muscle Development, Adaptation and Disease. Adv. Exp. Med. Biol. 2016, 900, 97–131. [Google Scholar] [CrossRef]
- Lotinun, S.; Pearsall, R.S.; Horne, W.C.; Baron, R. Activin receptor signaling: A potential therapeutic target for osteoporosis. Curr. Mol. Pharmacol. 2012, 5, 195–204. [Google Scholar] [CrossRef]
- Wang, Y.; Hamang, M.; Culver, A.; Jiang, H.; Yanum, J.; Garcia, V.; Lee, J.; White, E.; Kusumanchi, P.; Chalasani, N.; et al. Activin B promotes the initiation and progression of liver fibrosis. Hepatol. Commun. 2022, 6, 2812–2826. [Google Scholar] [CrossRef]
- Garry, G.A.; Antony, M.L.; Garry, D.J. Cardiotoxin Induced Injury and Skeletal Muscle Regeneration. Methods Mol. Biol. 2016, 1460, 61–71. [Google Scholar] [CrossRef]
- Smith, R.C.; Cramer, M.S.; Mitchell, P.J.; Capen, A.; Huber, L.; Wang, R.; Myers, L.; Jones, B.E.; Eastwood, B.J.; Ballard, D.; et al. Myostatin Neutralization Results in Preservation of Muscle Mass and Strength in Preclinical Models of Tumor-Induced Muscle Wasting. Mol. Cancer Ther. 2015, 14, 1661–1670. [Google Scholar] [CrossRef]
- Smith, R.C.; Cramer, M.S.; Mitchell, P.J.; Lucchesi, J.; Ortega, A.M.; Livingston, E.W.; Ballard, D.; Zhang, L.; Hanson, J.; Barton, K.; et al. Inhibition of myostatin prevents microgravity-induced loss of skeletal muscle mass and strength. PLoS ONE 2020, 15, e0230818. [Google Scholar] [CrossRef]
- Ingawale, D.K.; Mandlik, S.K.; Naik, S.R. Models of hepatotoxicity and the underlying cellular, biochemical and immunological mechanism(s): A critical discussion. Environ. Toxicol. Pharmacol. 2014, 37, 118–133. [Google Scholar] [CrossRef]
- Giusto, M.; Barberi, L.; Di Sario, F.; Rizzuto, E.; Nicoletti, C.; Ascenzi, F.; Renzi, A.; Caporaso, N.; D’Argenio, G.; Gaudio, E.; et al. Skeletal muscle myopenia in mice model of bile duct ligation and carbon tetrachloride-induced liver cirrhosis. Physiol. Rep. 2017, 5, e13153. [Google Scholar] [CrossRef] [PubMed]
- Moresi, V.; Williams, A.H.; Meadows, E.; Flynn, J.M.; Potthoff, M.J.; McAnally, J.; Shelton, J.M.; Backs, J.; Klein, W.H.; Richardson, J.A.; et al. Myogenin and class II HDACs control neurogenic muscle atrophy by inducing E3 ubiquitin ligases. Cell 2010, 143, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Latres, E.; Mastaitis, J.; Fury, W.; Miloscio, L.; Trejos, J.; Pangilinan, J.; Okamoto, H.; Cavino, K.; Na, E.; Papatheodorou, A.; et al. Activin A more prominently regulates muscle mass in primates than does GDF8. Nat. Commun. 2017, 8, 15153. [Google Scholar] [CrossRef] [PubMed]
- Rebbapragada, A.; Benchabane, H.; Wrana, J.L.; Celeste, A.J.; Attisano, L. Myostatin signals through a transforming growth factor beta-like signaling pathway to block adipogenesis. Mol. Cell. Biol. 2003, 23, 7230–7242. [Google Scholar] [CrossRef] [PubMed]
- Sako, D.; Grinberg, A.V.; Liu, J.; Davies, M.V.; Castonguay, R.; Maniatis, S.; Andreucci, A.J.; Pobre, E.G.; Tomkinson, K.N.; Monnell, T.E.; et al. Characterization of the ligand binding functionality of the extracellular domain of activin receptor type IIb. J. Biol. Chem. 2010, 285, 21037–21048. [Google Scholar] [CrossRef]
- Langley, B.; Thomas, M.; Bishop, A.; Sharma, M.; Gilmour, S.; Kambadur, R. Myostatin inhibits myoblast differentiation by down-regulating MyoD expression. J. Biol. Chem. 2002, 277, 49831–49840. [Google Scholar] [CrossRef]
- McFarlane, C.; Hennebry, A.; Thomas, M.; Plummer, E.; Ling, N.; Sharma, M.; Kambadur, R. Myostatin signals through Pax7 to regulate satellite cell self-renewal. Exp. Cell Res. 2008, 314, 317–329. [Google Scholar] [CrossRef]
- Qiu, J.; Thapaliya, S.; Runkana, A.; Yang, Y.; Tsien, C.; Mohan, M.L.; Narayanan, A.; Eghtesad, B.; Mozdziak, P.E.; McDonald, C.; et al. Hyperammonemia in cirrhosis induces transcriptional regulation of myostatin by an NF-kappaB-mediated mechanism. Proc. Natl. Acad. Sci. USA 2013, 110, 18162–18167. [Google Scholar] [CrossRef]
- Nishikawa, H.; Enomoto, H.; Ishii, A.; Iwata, Y.; Miyamoto, Y.; Ishii, N.; Yuri, Y.; Hasegawa, K.; Nakano, C.; Nishimura, T.; et al. Elevated serum myostatin level is associated with worse survival in patients with liver cirrhosis. J. Cachexia Sarcopenia Muscle 2017, 8, 915–925. [Google Scholar] [CrossRef]
- Delogu, W.; Caligiuri, A.; Provenzano, A.; Rosso, C.; Bugianesi, E.; Coratti, A.; Macias-Barragan, J.; Galastri, S.; Di Maira, G.; Marra, F. Myostatin regulates the fibrogenic phenotype of hepatic stellate cells via c-jun N-terminal kinase activation. Dig. Liver Dis. Off. J. Ital. Soc. Gastroenterol. Ital. Assoc. Study Liver 2019, 51, 1400–1408. [Google Scholar] [CrossRef]
- Yoshio, S.; Shimagaki, T.; Hashida, R.; Kawaguchi, T.; Tsutsui, Y.; Sakamoto, Y.; Yoshida, Y.; Kawai, H.; Yoshikawa, S.; Yamazoe, T.; et al. Myostatin as a fibroblast-activating factor impacts on postoperative outcome in patients with hepatocellular carcinoma. Hepatol. Res. 2021, 51, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Yaden, B.C.; Wang, Y.X.; Wilson, J.M.; Culver, A.E.; Milner, A.; Datta-Mannan, A.; Shetler, P.; Croy, J.E.; Dai, G.; Krishnan, V. Inhibition of activin A ameliorates skeletal muscle injury and rescues contractile properties by inducing efficient remodeling in female mice. Am. J. Pathol. 2014, 184, 1152–1166. [Google Scholar] [CrossRef] [PubMed]
- Jindal, A.; Jagdish, R.K. Sarcopenia: Ammonia metabolism and hepatic encephalopathy. Clin. Mol. Hepatol. 2019, 25, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Tsien, C.; Thapalaya, S.; Narayanan, A.; Weihl, C.C.; Ching, J.K.; Eghtesad, B.; Singh, K.; Fu, X.; Dubyak, G.; et al. Hyperammonemia-mediated autophagy in skeletal muscle contributes to sarcopenia of cirrhosis. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E983–E993. [Google Scholar] [CrossRef]
- Mowat, N.A.; Edwards, C.R.; Fisher, R.; McNeilly, A.S.; Green, J.R.; Dawson, A.M. Hypothalamic-pituitary-gonadal function in men with cirrhosis of the liver. Gut 1976, 17, 345–350. [Google Scholar] [CrossRef]
- Dasarathy, S.; Mullen, K.D.; Dodig, M.; Donofrio, B.; McCullough, A.J. Inhibition of aromatase improves nutritional status following portacaval anastomosis in male rats. J. Hepatol. 2006, 45, 214–220. [Google Scholar] [CrossRef]
- Bucuvalas, J.C.; Cutfield, W.; Horn, J.; Sperling, M.A.; Heubi, J.E.; Campaigne, B.; Chernausek, S.D. Resistance to the growth-promoting and metabolic effects of growth hormone in children with chronic liver disease. J. Pediatr. 1990, 117, 397–402. [Google Scholar] [CrossRef]
- Moller, S.; Becker, U.; Gronbaek, M.; Juul, A.; Winkler, K.; Skakkebaek, N.E. Short-term effect of recombinant human growth hormone in patients with alcoholic cirrhosis. J. Hepatol. 1994, 21, 710–717. [Google Scholar] [CrossRef]
- Shangraw, R.E.; Jahoor, F. Effect of liver disease and transplantation on urea synthesis in humans: Relationship to acid-base status. Am. J. Physiol. 1999, 276, G1145–G1152. [Google Scholar] [CrossRef] [PubMed]
- Olde Damink, S.W.; Jalan, R.; Dejong, C.H. Interorgan ammonia trafficking in liver disease. Metab. Brain Dis. 2009, 24, 169–181. [Google Scholar] [CrossRef]
- Liu, W.; Thomas, S.G.; Asa, S.L.; Gonzalez-Cadavid, N.; Bhasin, S.; Ezzat, S. Myostatin is a skeletal muscle target of growth hormone anabolic action. J. Clin. Endocrinol. Metab. 2003, 88, 5490–5496. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.S.; Cabbabe, A.; Kambadur, R.; Nicholas, G.; Csete, M. Brief-reports: Elevated myostatin levels in patients with liver disease: A potential contributor to skeletal muscle wasting. Anesth. Analg. 2010, 111, 707–709. [Google Scholar] [CrossRef]
- Merli, M.; Giusto, M.; Molfino, A.; Bonetto, A.; Rossi, M.; Ginanni Corradini, S.; Baccino, F.M.; Rossi Fanelli, F.; Costelli, P.; Muscaritoli, M. MuRF-1 and p-GSK3beta expression in muscle atrophy of cirrhosis. Liver Int. 2013, 33, 714–721. [Google Scholar] [CrossRef]
- Ravenscroft, G.; Jackaman, C.; Sewry, C.A.; McNamara, E.; Squire, S.E.; Potter, A.C.; Papadimitriou, J.; Griffiths, L.M.; Bakker, A.J.; Davies, K.E.; et al. Actin nemaline myopathy mouse reproduces disease, suggests other actin disease phenotypes and provides cautionary note on muscle transgene expression. PLoS ONE 2011, 6, e28699. [Google Scholar] [CrossRef] [PubMed]
- Weber, F.L., Jr.; Macechko, P.T.; Kelson, S.R.; Karajiannis, E.; Hassan, M.O. Increased muscle protein catabolism caused by carbon tetrachloride hepatic injury in rats. Gastroenterology 1992, 102, 1700–1706. [Google Scholar] [CrossRef]
- De Feritas Oliveira, C.; da Silva Lopes, S.; Mendes, M.M.; Homsi-Brandeburgo, M.I.; Hamaguchi, A.; de Alcantara, T.M.; Clissa, P.B.; de Melo Rodrigues, V. Insights of local tissue damage and regeneration induced by BnSP-7, a myotoxin isolated from Bothrops (neuwiedi) pauloensis snake venom. Toxicon 2009, 53, 560–569. [Google Scholar] [CrossRef]
- Manfredi, L.H.; Paula-Gomes, S.; Zanon, N.M.; Kettelhut, I.C. Myostatin promotes distinct responses on protein metabolism of skeletal and cardiac muscle fibers of rodents. Braz. J. Med. Biol. Res. 2017, 50, e6733. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Wang, F.; Jin, C.; Wu, X.; Chan, W.K.; McKeehan, W.L. Increased carbon tetrachloride-induced liver injury and fibrosis in FGFR4-deficient mice. Am. J. Pathol. 2002, 161, 2003–2010. [Google Scholar] [CrossRef]
- Li, Z.B.; Kollias, H.D.; Wagner, K.R. Myostatin directly regulates skeletal muscle fibrosis. J. Biol. Chem. 2008, 283, 19371–19378. [Google Scholar] [CrossRef] [PubMed]
- Kovanecz, I.; Masouminia, M.; Gelfand, R.; Vernet, D.; Rajfer, J.; Gonzalez-Cadavid, N.F. Myostatin, a profibrotic factor and the main inhibitor of striated muscle mass, is present in the penile and vascular smooth muscle. Int. J. Impot. Res. 2017, 29, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Lach-Trifilieff, E.; Minetti, G.C.; Sheppard, K.; Ibebunjo, C.; Feige, J.N.; Hartmann, S.; Brachat, S.; Rivet, H.; Koelbing, C.; Morvan, F.; et al. An antibody blocking activin type II receptors induces strong skeletal muscle hypertrophy and protects from atrophy. Mol. Cell. Biol. 2014, 34, 606–618. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; Coleman, L.A.; Miller, R.; Rooks, D.S.; Laurent, D.; Petricoul, O.; Praestgaard, J.; Swan, T.; Wade, T.; Perry, R.G.; et al. Effect of Bimagrumab vs Placebo on Body Fat Mass Among Adults With Type 2 Diabetes and Obesity: A Phase 2 Randomized Clinical Trial. JAMA Netw. Open 2021, 4, e2033457. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Culver, A.; Hamang, M.; Wang, Y.; Jiang, H.; Yanum, J.; White, E.; Gawrieh, S.; Vuppalanchi, R.K.; Chalasani, N.P.; Dai, G.; et al. GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting. Biomedicines 2023, 11, 1909. https://doi.org/10.3390/biomedicines11071909
Culver A, Hamang M, Wang Y, Jiang H, Yanum J, White E, Gawrieh S, Vuppalanchi RK, Chalasani NP, Dai G, et al. GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting. Biomedicines. 2023; 11(7):1909. https://doi.org/10.3390/biomedicines11071909
Chicago/Turabian StyleCulver, Alexander, Matthew Hamang, Yan Wang, Huaizhou Jiang, Jennifer Yanum, Emily White, Samer Gawrieh, Raj K. Vuppalanchi, Naga P. Chalasani, Guoli Dai, and et al. 2023. "GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting" Biomedicines 11, no. 7: 1909. https://doi.org/10.3390/biomedicines11071909
APA StyleCulver, A., Hamang, M., Wang, Y., Jiang, H., Yanum, J., White, E., Gawrieh, S., Vuppalanchi, R. K., Chalasani, N. P., Dai, G., & Yaden, B. C. (2023). GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting. Biomedicines, 11(7), 1909. https://doi.org/10.3390/biomedicines11071909




