Valproate-Induced Metabolic Syndrome
Abstract
1. Introduction
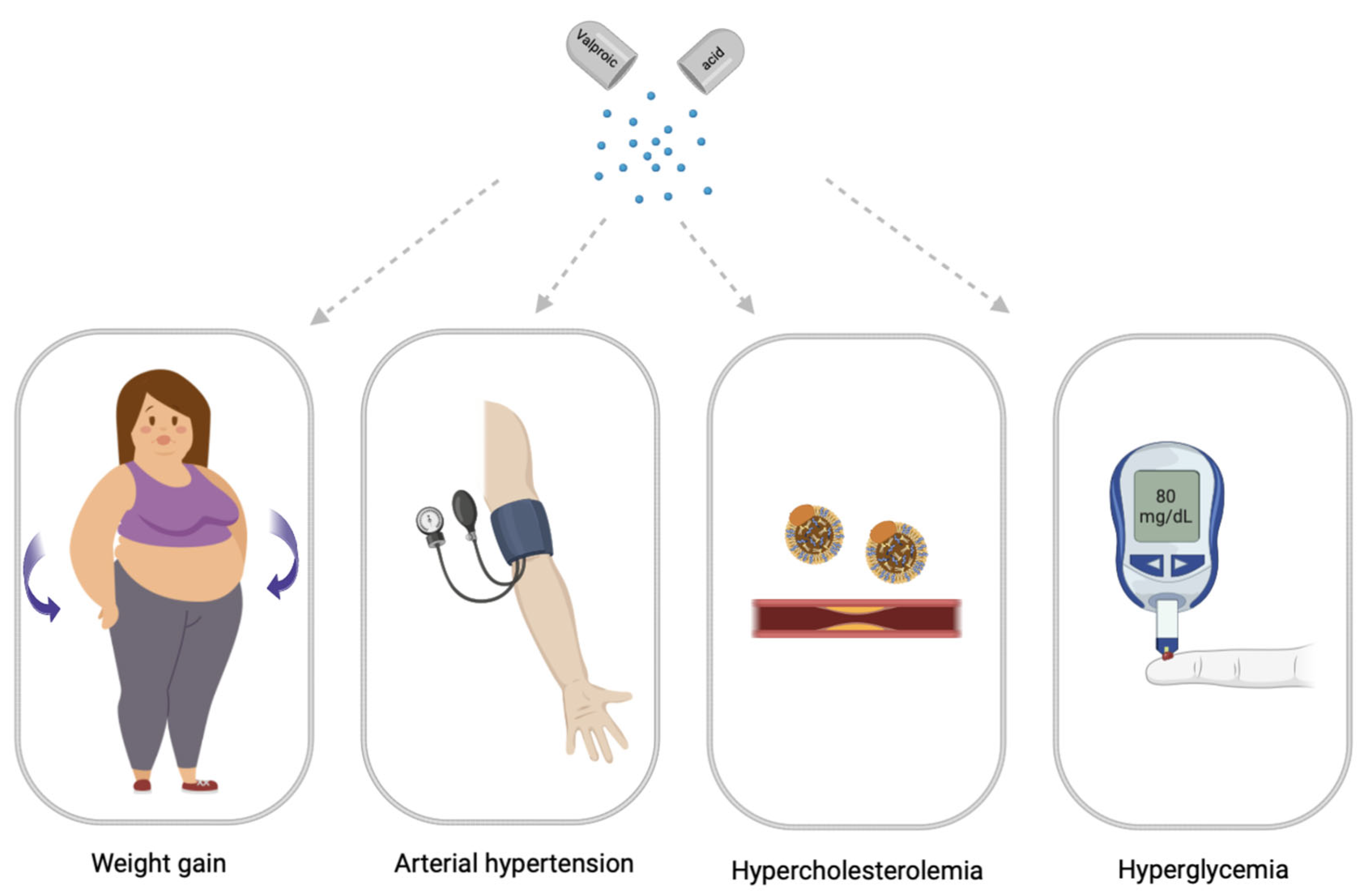
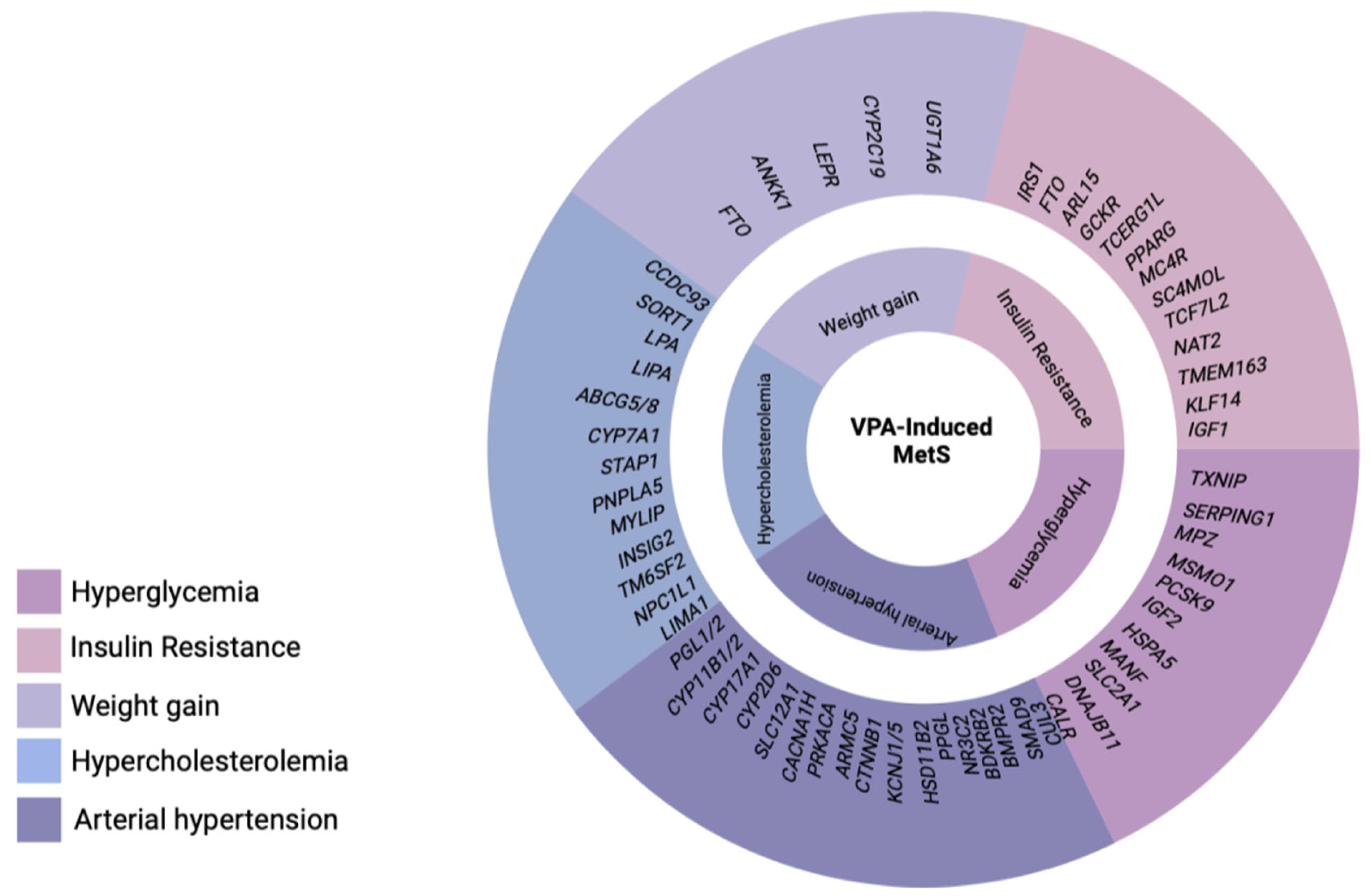
2. Main Clinical Symptoms of Valproate-Induced Metabolic Syndrome
2.1. Valproate-Induced Weight Gain
- −
- An increase in body weight, body mass index (BMI), and waist circumference;
- −
- An increase in the amount of fat in the abdomen, measured by the waist circumference or the ratio of waist to hips;
- −
- Insulin resistance, which can contribute to the development of abdominal obesity.
2.2. Valproate-Induced Insulin Resistance
2.3. Valproate-Induced Arterial Hypertension
2.4. Valproate-Induced Hypercholesterolemia
2.5. Valproate-Induced Hyperglycemia
3. Blood Biomarkers (Serum and Plasma) of Valproate-Induced Metabolic Syndrome
3.1. Carbohydrates
Glucose
3.2. Acids
Uric Acid
3.3. Hormones
3.3.1. Insulin
3.3.2. Adiponectin
3.3.3. Chemerin
3.3.4. Ghrelin
3.3.5. Leptin
3.3.6. Omentin
3.3.7. Testosterone
3.3.8. Parathyroid Hormone
3.3.9. Thyroid Stimulating Hormone
3.4. Other Organic Compounds
Direct and Total Bilirubin
3.5. Proteins
3.5.1. Adipocyte Fatty Acid Binding Protein
3.5.2. C-Peptide
3.5.3. Cystatin-C
3.5.4. Ferritin
3.5.5. Fibrinogen
3.5.6. Fibroblast Growth Factor 21
3.5.7. Monocyte Chemoattractant Protein-1
3.5.8. Plasminogen Activator Inhibitor-1
3.5.9. Retinol-Binding Protein 4
3.5.10. Tumor Necrosis Factor Alpha
3.5.11. Neuropeptide Y
3.6. Lipids
3.6.1. Apolipoprotein A1
3.6.2. Apolipoprotein B
3.6.3. Free Fatty Acids
3.6.4. Oxidized Low Density Lipoprotein
3.6.5. Cholesterol
3.6.6. High Density Lipoprotein
3.7. Enzymes
3.7.1. Superoxide Dismutase
3.7.2. Gamma-Glutamyl Transferase
3.7.3. Lipoprotein-Associated Phospholipase A
3.7.4. Amylase
3.8. Vitamins
3.8.1. 25-Hydroxyvitamin D
3.8.2. Vitamin E
3.9. Other
CD40 Ligand
4. Urinary Biomarkers of Valproate-Induced Metabolic Syndrome
4.1. Carbohydrates
4.1.1. Glucose
4.1.2. Maltitol
4.2. Amino Acids
4.2.1. Aromatic Amino Acids
4.2.2. Histidine
4.3. Organic Acids
4-Hydroxyphenylpyruvic Acid
5. Discussion
6. Limitation
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kazamel, M.; Stino, A.M.; Smith, A.G. Metabolic syndrome and peripheral neuropathy. Muscle Nerve 2021, 63, 285–293. [Google Scholar] [CrossRef]
- Oye-Somefun, A.; Kuk, J.L.; Ardern, C.I. Associations between elevated kidney and liver biomarker ratios, metabolic syndrome and all-cause and coronary heart disease (CHD) mortality: Analysis of the U.S. National Health and Nutrition Examination Survey (NHANES). BMC Cardiovasc. Disord. 2021, 21, 352. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Pan, Y.; Jing, J.; Zhao, X.; Liu, L.; Meng, X.; Wang, Y.; Wang, Y. Recurrent Stroke in Minor Ischemic Stroke or Transient Ischemic Attack with Metabolic Syndrome and/or Diabetes Mellitus. J. Am. Heart Assoc. 2017, 6, e005446. [Google Scholar] [CrossRef] [PubMed]
- Penninx, B.W.J.H.; Lange, S.M.M. Metabolic syndrome in psychiatric patients: Overview, mechanisms, and implications. Dialogues Clin. Neurosci. 2018, 20, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Khasanova, A.K.; Dobrodeeva, V.S.; Shnayder, N.A.; Petrova, M.M.; Pronina, E.A.; Bochanova, E.N.; Lareva, N.V.; Garganeeva, N.P.; Smirnova, D.A.; Nasyrova, R.F. Blood and Urinary Biomarkers of Antipsychotic-Induced Metabolic Syndrome. Metabolites 2022, 12, 726. [Google Scholar] [CrossRef]
- Available online: cadiresearch.org/topic/metabolic-syndrome/metabolic-syndrome-global/ms-criteria (accessed on 21 March 2023).
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48. [Google Scholar] [CrossRef]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497. [Google Scholar] [CrossRef]
- Suplotova, L.A.; Smetanina, S.A.; Novakovskaya, N.A. Prevalence of metabolic syndrome and its components in women in different ethnic groups. Obes. Metab. 2011, 8, 48–51. [Google Scholar] [CrossRef]
- Borisov, I.V.; Bondar, V.A.; Petrova, M.V.; Kuzovlev, A.N.; Ohlopkov, V.A.; Kanarski, M.M.; Nekrasova, J.J. Metabolic syndrome: Definition, pathogenesis and rehabilitation. Bull. All-Russ. Soc. Spec. Med. Soc. Expert. Rehabil. Rehabil. Ind. 2020, 4, 114–125. [Google Scholar] [CrossRef]
- Chen, D.C.; Du, X.D.; Yin, G.Z.; Yang, K.B.; Nie, Y.; Wang, N.; Li, Y.L.; Xiu, M.H.; He, S.C.; Yang, F.D.; et al. Impaired glucose tolerance in first-episode drug-naïve patients with schizophrenia: Relationships with clinical phenotypes and cognitive deficits. Psychol. Med. 2016, 46, 3219–3230. [Google Scholar] [CrossRef]
- Wofford, M.R.; King, D.S.; Harrell, T.K. Drug-induced metabolic syndrome. J. Clin. Hypertens. 2006, 8, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Sychev, D.A.; Ostroumova, O.D.; Pereverzev, A.P. Drug-induced diseases: Approaches to diagnosis, correction and prevention Pharmacovigilance. Pharmateka 2020, 27, 113–126. [Google Scholar] [CrossRef]
- Shnayder, N.A.; Grechkina, V.V.; Khasanova, A.K.; Bochanova, E.N.; Dontceva, E.A.; Petrova, M.M.; Asadullin, A.R.; Shipulin, G.A.; Altynbekov, K.S.; Al-Zamil, M.; et al. Therapeutic and Toxic Effects of Valproic Acid Metabolites. Metabolites 2023, 13, 134. [Google Scholar] [CrossRef]
- Zuo, S.; Fries, B.E.; Szafara, K.; Regal, R. Valproic Acid as a potentiator of metabolic syndrome in institutionalized residents on concomitant antipsychotics: Fat chance, or slim to none? Pharm. Ther. 2015, 40, 126–132. [Google Scholar]
- Shnaider, N.A.; Dmitrenko, D.V. Chronic valproic acid intoxication in epileptology: Diagnosis and treatment. Neurol. Neuropsychiatry Psychosom. 2016, 8, 94–99. [Google Scholar] [CrossRef]
- Fang, J.; Chen, S.; Tong, N.; Chen, L.; An, D.; Mu, J.; Zhou, D. Metabolic syndrome among Chinese obese patients with epilepsy on sodium valproate. Seizure 2012, 21, 578–582. [Google Scholar] [CrossRef] [PubMed]
- George, L.J.; Singh, P.; Aneja, S.; Singh, R.; Solanki, R.S.; Seth, A. Insulin Resistance in children on Sodium Valproate—A hospital based cross-sectional study in Indian children. Trop. Doct. 2023, 53, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Lee, Y.H.; Lee, Y.J.; Kim, K.J.; Kim, S.G. Weight Gain Predicts Metabolic Syndrome among North Korean Refugees in South Korea. Int. J. Environ. Res. Public Health 2021, 18, 8479. [Google Scholar] [CrossRef]
- Belcastro, V.; D’Egidio, C.; Striano, P.; Verrotti, A. Metabolic and endocrine effects of valproic acid chronic treatment. Epilepsy Res. 2013, 107, 1–8. [Google Scholar] [CrossRef]
- Romoli, M.; Mazzocchetti, P.; D’Alonzo, R.; Siliquini, S.; Rinaldi, V.E.; Verrotti, A.; Calabresi, P.; Costa, C. Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences. Curr. Neuropharmacol. 2019, 17, 926–946. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, P.; Tang, H.L.; Yan, H.J.; Jiang, W.; Shi, H.; Chen, S.Y.; Gao, M.M.; Zeng, X.D.; Long, Y.S. Valproate-Induced Epigenetic Upregulation of Hypothalamic Fto Expression Potentially Linked with Weight Gain. Cell. Mol. Neurobiol. 2021, 41, 1257–1269. [Google Scholar] [CrossRef] [PubMed]
- Rehman, T.; Sachan, D.; Chitkara, A. Serum Insulin and Leptin Levels in Children with Epilepsy on Valproate-associated Obesity. J. Pediatr. Neurosci. 2017, 12, 135–137. [Google Scholar] [CrossRef] [PubMed]
- Münzberg, H.; Björnholm, M.; Bates, S.H.; Myers, M.G., Jr. Leptin receptor action and mechanisms of leptin resistance. Cell Mol. Life Sci. 2005, 62, 642–652. [Google Scholar] [CrossRef]
- Rauchenzauner, M.; Laimer, M.; Luef, G.; Kaser, S.; Engl, J.; Tatarczyk, T.; Ciardi, C.; Tschoner, A.; Lechleitner, M.; Patsch, J.; et al. Adiponectin receptor R1 is upregulated by valproic acid but not by topiramate in human hepatoma cell line, HepG2. Seizure 2008, 17, 723–726. [Google Scholar] [CrossRef]
- Qiao, L.; Schaack, J.; Shao, J. Suppression of adiponectin gene expression by histone deacetylase inhibitor valproic acid. Endocrinology 2006, 147, 865–874. [Google Scholar] [CrossRef]
- Brown, A.E.; Walker, M. Genetics of Insulin Resistance and the Metabolic Syndrome. Curr. Cardiol. Rep. 2016, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef]
- Aly, R.H.; Amr, N.H.; Saad, W.E.; Megahed, A.A. Insulin resistance in patients on valproic acid: Relation to adiponectin. Acta Neurol. Scand. 2015, 131, 169–175. [Google Scholar] [CrossRef]
- Jian, J.; Li, L.G.; Zhao, P.J.; Zheng, R.J.; Dong, X.W.; Zhao, Y.H.; Yin, B.Q.; Cheng, H.; Li, H.L.; Li, E.Y. TCHis mitigate oxidative stress and improve abnormal behavior in a prenatal valproic acid-exposed rat model of autism. Physiol. Genom. 2022, 54, 325–336. [Google Scholar] [CrossRef]
- Brown, R.; Imran, S.A.; Ur, E.; Wilkinson, M. Valproic acid and CEBPalpha-mediated regulation of adipokine gene expression in hypothalamic neurons and 3T3-L1 adipocytes. Neuroendocrinology 2008, 88, 25–34. [Google Scholar] [CrossRef]
- Khan, S.; Kumar, S.; Jena, G. Valproic acid reduces insulin-resistance, fat deposition and FOXO1-mediated gluconeogenesis in type-2 diabetic rat. Biochimie 2016, 125, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Rauchenzauner, M.; Laimer, M.; Wiedmann, M.; Tschoner, A.; Salzmann, K.; Sturm, W.; Sandhofer, A.; Walser, G.; Luef, G.; Ebenbichler, C.F. The novel insulin resistance parameters RBP4 and GLP-1 in patients treated with valproic acid: Just a sidestep? Epilepsy Res. 2013, 104, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomstrӧm-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Russ. J. Cardiol. 2021, 26, 4701. (In Russian) [Google Scholar] [CrossRef]
- Aune, D.; Huang, W.; Nie, J.; Wang, Y. Hypertension and the Risk of All-Cause and Cause-Specific Mortality: An Outcome-Wide Association Study of 67 Causes of Death in the National Health Interview Survey. Biomed Res. Int. 2021, 2021, 9376134. [Google Scholar] [CrossRef]
- Abaseynejad, F.; Akrami, R.; Mohebbati, R.; Sehab Negah, S.; Mohammad-Zadeh, M. The Effect of Sodium Valproate on Cardiovascular Responses in Pentylenetetrazol Kindling Model of Epilepsy. Biomed. J. Sci. Tech. Res. 2022, 42, 33592–33596. [Google Scholar] [CrossRef]
- Sousa-Lopes, A.; de Freitas, R.A.; Carneiro, F.S.; Nunes, K.P.; Allahdadi, K.J.; Webb, R.C.; Tostes, R.C.; Giachini, F.R.; Lima, V.V. Angiotensin (1-7) Inhibits Ang II-mediated ERK1/2 Activation by Stimulating MKP-1 Activation in Vascular Smooth Muscle Cells. Int. J. Mol. Cell. Med. 2020, 9, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xing, B.; Dang, Y.H.; Qu, C.L.; Zhu, F.; Yan, C.X. Microinjection of valproic acid into the ventrolateral orbital cortex enhances stress-related memory formation. PLoS ONE 2013, 8, e52698. [Google Scholar] [CrossRef] [PubMed]
- Rajeshwari, T.; Raja, B.; Manivannan, J.; Silambarasan, T. Valproic acid attenuates blood pressure, vascular remodeling and modulates ET-1 expression in L-NAME induced hypertensive rats. Biomed. Prev. Nutr. 2014, 4, 195–202. [Google Scholar]
- Sivananthan, M.; Mohiuddin, S. Valproate Induced Hypertensive Urgency. Case Rep. Psychiatry 2016, 2016, 1458548. [Google Scholar] [CrossRef]
- Zárate, A.; Manuel-Apolinar, L.; Saucedo, R.; Hernández-Valencia, M.; Basurto, L. Hypercholesterolemia as a Risk Factor for Cardiovascular Disease: Current Controversial Therapeutic Management. Arch. Med. Res. 2016, 47, 491–495. [Google Scholar] [CrossRef]
- Peters, S.A.; Singhateh, Y.; Mackay, D.; Huxley, R.R.; Woodward, M. Total cholesterol as a risk factor for coronary heart disease and stroke in women compared with men: A systematic review and meta-analysis. Atherosclerosis 2016, 248, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Kusumastuti, K.; Jaeri, S. The effect of long-term valproic acid treatment in the level of total cholesterol among adult. Indian J. Pharmacol. 2020, 52, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Hamed, S.A. Atherosclerosis in epilepsy: Its causes and implications. Epilepsy Behav. 2014, 41, 290–296. [Google Scholar] [CrossRef]
- Aziz, R.S.; Saeed, U.; Ali, L.; Arshad, M.; Abbas, R.; Mushtaq, S.; Asif Shahzad, A.; Shaukat, A. Effect on lipid profile due to prolong Valproic acid intake. Pak. J. Med. Health Sci. 2021, 15, 1497–1499. [Google Scholar] [CrossRef]
- Guo, H.L.; Jing, X.; Sun, J.Y.; Hu, Y.H.; Xu, Z.J.; Ni, M.M.; Chen, F.; Lu, X.P.; Qiu, J.C.; Wang, T. Valproic Acid and the Liver Injury in Patients with Epilepsy: An Update. Curr. Pharm. Des. 2019, 25, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Verrotti, A.; Scardapane, A.; Franzoni, E.; Manco, R.; Chiarelli, F. Increased oxidative stress in epileptic children treated with valproic acid. Epilepsy Res. 2008, 78, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Verrotti, A.; la Torre, R.; Trotta, D.; Mohn, A.; Chiarelli, F. Valproate-induced insulin resistance and obesity in children. Horm. Res. 2009, 71, 125–131. [Google Scholar] [CrossRef]
- Fathallah, N.; Slim, R.; Larif, S.; Hmouda, H.; Ben Salem, C. Drug-Induced Hyperglycaemia and Diabetes. Drug Saf. 2015, 38, 1153–1168. [Google Scholar] [CrossRef]
- Felisbino, M.B.; Ziemann, M.; Khurana, I.; Okabe, J.; Al-Hasani, K.; Maxwell, S.; Harikrishnan, K.N.; de Oliveira, C.B.M.; Mello, M.L.S.; El-Osta, A. Valproic acid influences the expression of genes implicated with hyperglycaemia-induced complement and coagulation pathways. Sci. Rep. 2021, 11, 2163. [Google Scholar] [CrossRef]
- Mansoub, S.; Chan, M.K.; Adeli, K. Gap analysis of pediatric reference intervals for risk biomarkers of cardiovascular disease and the metabolic syndrome. Clin. Biochem. 2006, 39, 569–587. [Google Scholar] [CrossRef]
- Rezzani, R.; Franco, C. Liver, Oxidative Stress and Metabolic Syndromes. Nutrients 2021, 13, 301. [Google Scholar] [CrossRef] [PubMed]
- Son, D.H.; Lee, H.S.; Lee, Y.J.; Lee, J.H.; Han, J.H. Comparison of triglyceride-glucose index and HOMA-IR for predicting prevalence and incidence of metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Gluvic, Z.; Zaric, B.; Resanovic, I.; Obradovic, M.; Mitrovic, A.; Radak, D.; Isenovic, E.R. Link between Metabolic Syndrome and Insulin Resistance. Curr. Vasc. Pharmacol. 2017, 15, 30–39. [Google Scholar] [CrossRef]
- Tune, J.D.; Goodwill, A.G.; Sassoon, D.J.; Mather, K.J. Cardiovascular consequences of metabolic syndrome. Transl. Res. 2017, 183, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Rakitin, A.; Kõks, S.; Haldre, S. Valproate modulates glucose metabolism in patients with epilepsy after first exposure. Epilepsia 2015, 56, e172–e175. [Google Scholar] [CrossRef]
- Wishart, D.S.; Guo, A.; Oler, E.; Wang, F.; Anjum, A.; Peters, H.; Dizon, R.; Sayeeda, Z.; Tian, S.; Lee, B.L.; et al. HMDB 5.0: The Human Metabolome Database for 2022. Nucleic Acids Res. 2022, 50, D622–D631. Available online: https://hmdb.ca/ (accessed on 16 March 2023). [CrossRef] [PubMed]
- Wang, Q.; Wen, X.; Kong, J. Recent Progress on Uric Acid Detection: A Review. Crit. Rev. Anal. Chem. 2020, 50, 359–375. [Google Scholar] [CrossRef]
- Fathallah-Shaykh, S.A.; Cramer, M.T. Uric acid and the kidney. Pediatr. Nephrol. 2014, 29, 999–1008. [Google Scholar] [CrossRef]
- Sanchez-Lozada, L.G.; Rodriguez-Iturbe, B.; Kelley, E.E.; Nakagawa, T.; Madero, M.; Feig, D.I.; Borghi, C.; Piani, F.; Cara-Fuentes, G.; Bjornstad, P.; et al. Uric Acid and Hypertension: An Update with Recommendations. Am. J. Hypertens. 2020, 33, 583–594. [Google Scholar] [CrossRef]
- Spatola, L.; Ferraro, P.M.; Gambaro, G.; Badalamenti, S.; Dauriz, M. Metabolic syndrome and uric acid nephrolithiasis: Insulin resistance in focus. Metabolism 2018, 83, 225–233. [Google Scholar] [CrossRef]
- Attilakos, A.; Voudris, K.A.; Garoufi, A.; Mastroyianni, S.; Dimou, S.; Prassouli, A.; Katsarou, E. Effect of sodium valproate monotherapy on serum uric acid concentrations in ambulatory epileptic children: A prospective long-term study. Eur. J. Paediatr. Neurol. 2006, 10, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Qin, X.; Liang, X.; Liu, M.; Zhang, X. Lipidomic characteristics and clinical findings of epileptic patients treated with valproic acid. J. Cell. Mol. Med. 2019, 23, 6017–6023. [Google Scholar] [CrossRef] [PubMed]
- Thevis, M.; Thomas, A.; Schänzer, W. Insulin. In Doping in Sports: Biochemical Principles, Effects and Analysis; Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2010; pp. 209–226. [Google Scholar] [CrossRef]
- Heise, T. The future of insulin therapy. Diabetes Res. Clin. Pract. 2021, 175, 108820. [Google Scholar] [CrossRef]
- Quianzon, C.C.; Cheikh, I. History of insulin. J. Community Hosp. Intern. Med. Perspect. 2012, 2, 18701. [Google Scholar] [CrossRef] [PubMed]
- Titchenell, P.M.; Lazar, M.A.; Birnbaum, M.J. Unraveling the Regulation of Hepatic Metabolism by Insulin. Trends Endocrinol. Metab. 2017, 28, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Yaryari, A.M.; Mazdeh, M.; Mohammadi, M.; Haghi, A.R.; Ghiasian, M.; Mehrpooya, M. Evaluation of serum levels of asprosin and other metabolic profiles in patients with idiopathic tonic-clonic generalized epilepsy on treatment with valproic acid. Eur. J. Clin. Pharmacol. 2022, 78, 393–403. [Google Scholar] [CrossRef]
- Çiçek, N.P.; Kamaşak, T.; Serin, M.; Okten, A.; Alver, A.; Cansu, A. The effects of valproate and topiramate use on serum insulin, leptin, neuropeptide Y and ghrelin levels in epileptic children. Seizure 2018, 58, 90–95. [Google Scholar] [CrossRef]
- Chen, J.; Tan, B.; Karteris, E.; Zervou, S.; Digby, J.; Hillhouse, E.W.; Vatish, M.; Randeva, H.S. Secretion of adiponectin by human placenta: Differential modulation of adiponectin and its receptors by cytokines. Diabetologia 2006, 49, 1292–1302. [Google Scholar] [CrossRef]
- Maeda, K.; Okubo, K.; Shimomura, I.; Funahashi, T.; Matsuzawa, Y.; Matsubara, K. cDNA cloning and expression of a novel adipose specific collagen-like factor, apM1 (AdiPose Most abundant Gene transcript 1). Biochem. Biophys. Res. Commun. 1996, 221, 286–289. [Google Scholar] [CrossRef]
- Chen, D.; Zhang, Y.; Yidilisi, A.; Xu, Y.; Dong, Q.; Jiang, J. Causal Associations Between Circulating Adipokines and Cardiovascular Disease: A Mendelian Randomization Study. J. Clin. Endocrinol. Metab. 2022, 107, e2572–e2580. [Google Scholar] [CrossRef]
- Ouchi, N.; Walsh, K. Adiponectin as an anti-inflammatory factor. Clin. Chim. Acta 2007, 380, 24–30. [Google Scholar] [CrossRef]
- Sonmez, F.M.; Zaman, D.; Aksoy, A.; Deger, O.; Aliyazicioglu, R.; Karaguzel, G.; Fazlioglu, K. The effects of topiramate and valproate therapy on insulin, c-peptide, leptin, neuropeptide Y, adiponectin, visfatin, and resistin levels in children with epilepsy. Seizure 2013, 22, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.F.; Beck-Sickinger, A.G. Chemerin—Exploring a versatile adipokine. Biol. Chem. 2022, 403, 625–642. [Google Scholar] [CrossRef] [PubMed]
- Afify, A.A.; Fathy, G.; Elzawahry, M.; Taha, S.I. Assessment of serum chemerin levels in acanthosis nigricans: A case-control study. J. Cosmet. Dermatol. 2022, 21, 6414–6421. [Google Scholar] [CrossRef]
- Buckinx, A.; De Bundel, D.; Kooijman, R.; Smolders, I. Targeting the Ghrelin Receptor as a Novel Therapeutic Option for Epilepsy. Biomedicines 2021, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Dobrodeeva, V.S.; Shnayder, N.A.; Mironov, K.O.; Nasyrova, R.F. Pharmacogenetic markers of antipsychotic-induced weight gain: Leptin and neuroepeptide Y. V.M. Bekhterev Rev. Psychiatry Med. Psychol. 2021, 55, 3–10. [Google Scholar] [CrossRef]
- Wu, J.T.; Kral, J.G. Ghrelin: Integrative neuroendocrine peptide in health and disease. Ann. Surg. 2004, 239, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, F.; Kojima, M.; Hosoda, H.; Nakazato, M.; Kangawa, K. Ghrelin: A novel peptide for growth hormone release and feeding regulation. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 391–395. [Google Scholar] [CrossRef]
- Vasyukova, O.V.; Vitebskaya, A.V. Grelin: Biological significance and prospects for use in endocrinology. Probl. Endokrinol. 2006, 52, 3–7. (In Russian) [Google Scholar] [CrossRef]
- Li, E.; Chung, H.; Kim, Y.; Kim, D.H.; Ryu, J.H.; Sato, T.; Kojima, M.; Park, S. Ghrelin directly stimulates adult hippocampal neurogenesis: Implications for learning and memory. Endocr. J. 2013, 60, 781–789. [Google Scholar] [CrossRef]
- Beck, B.; Pourié, G. Ghrelin, neuropeptide Y, and other feeding-regulatory peptides active in the hippocampus: Role in learning and memory. Nutr. Rev. 2013, 71, 541–561. [Google Scholar] [CrossRef] [PubMed]
- Schalla, M.A.; Stengel, A. The Role of Ghrelin in Anorexia Nervosa. Int. J. Mol. Sci. 2018, 19, 2117. [Google Scholar] [CrossRef] [PubMed]
- Haqq, A.M.; Farooqi, I.S.; O’Rahilly, S.; Stadler, D.D.; Rosenfeld, R.G.; Pratt, K.L.; LaFranchi, S.H.; Purnell, J.Q. Serum ghrelin levels are inversely correlated with body mass index, age, and insulin concentrations in normal children and are markedly increased in Prader-Willi syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Heshmat, R.; Shafiee, G.; Qorbani, M.; Azizi-Soleiman, F.; Djalalinia, S.; Esmaeil Motlagh, M.; Ardalan, G.; Ahadi, Z.; Safari, O.; Safiri, S.; et al. Association of ghrelin with cardiometabolic risk factors in Iranian adolescents: The CASPIAN-III study. J. Cardiovasc. Thorac. Res. 2016, 8, 107–112. [Google Scholar] [CrossRef]
- Dobrodeeva, V.S.; Shnayder, N.A.; Novitsky, M.A.; Asadullin, A.R.; Vaiman, E.E.; Petrova, M.M.; Limankin, O.V.; Neznanov, N.G.; Garganeeva, N.P.; Nasyrova, R.F. Association of a Single-Nucleotide Variant rs11100494 of the NPY5R Gene with Antipsychotic-Induced Metabolic Disorders. Pharmaceutics 2022, 14, 222. [Google Scholar] [CrossRef]
- Qadir, M.I.; Ahmed, Z. lep Expression and Its Role in Obesity and Type-2 Diabetes. Crit. Rev. Eukaryot. Gene Expr. 2017, 27, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Van Swieten, M.M.; Pandit, R.; Adan, R.A.; van der Plasse, G. The neuroanatomical function of leptin in the hypothalamus. J. Chem. Neuroanat. 2014, 61–62, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Yan, E.; Chen, S.; Hong, K.; Kim, W.S.; Bajpai, A.; Treyzon, L.; Gratton, L.; Elashoff, R.; Wang, H.J.; Li, Z.; et al. Insulin, hs-CRP, leptin, and adiponectin. An analysis of their relationship to the metabolic syndrome in an obese population with an elevated waist circumference. Metab. Syndr. Relat. Disord. 2008, 6, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Vatier, C.; Jéru, I.; Fellahi, S.; Capeau, J.; Bastard, J.P.; Vigouroux, C.; groupe de travail RIHN Adipokines. Leptine, adiponectine, syndromes lipodystrophiques et d’insulino-résistance sévère [Leptin, adiponectin, lipodystrophic and severe insulin resistance syndromes]. Ann. Biol. Clin. 2020, 78, 261–264. (In French) [Google Scholar] [CrossRef]
- Zhao, S.; Kusminski, C.M.; Scherer, P.E. Adiponectin, Leptin and Cardiovascular Disorders. Circ. Res. 2021, 128, 136–149. [Google Scholar] [CrossRef]
- Sun, X.; Li, T.; Tian, Y.; Ren, S.; Li, L.; Li, P. Omentin as an Independent Predictor of Metabolic Syndrome and Obesity Among Adolescents in Northeast China. Diabetes Metab. Syndr. Obes. 2022, 15, 3913–3922. [Google Scholar] [CrossRef] [PubMed]
- Chyra, M.; Roczniak, W.; Świętochowska, E.; Dudzińska, M.; Oświęcimska, J. The Effect of the Ketogenic Diet on Adiponectin, Omentin and Vaspin in Children with Drug-Resistant Epilepsy. Nutrients 2022, 14, 479. [Google Scholar] [CrossRef] [PubMed]
- Salam, R.; Kshetrimayum, A.S.; Keisam, R. Testosterone and metabolic syndrome: The link. Indian J. Endocrinol. Metab. 2012, 16 (Suppl. 1), S12–S19. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, G.R. Testosterone and metabolic syndrome. Asian J. Androl. 2015, 17, 192–196. [Google Scholar] [CrossRef]
- Stephen, L.J.; Kwan, P.; Shapiro, D.; Dominiczak, M.; Brodie, M.J. Hormone profiles in young adults with epilepsy treated with sodium valproate or lamotrigine monotherapy. Epilepsia 2001, 42, 1002–1006. [Google Scholar] [CrossRef]
- Smagulova, A.; Uakhit, R.; Kiyan, V. First Record of Alternaria alternata causing necrosis of Thuja (Thuja occidentalis) in Kazakhstan. Plant Dis. 2022, 106, 2773–3007. [Google Scholar] [CrossRef]
- Røste, L.S.; Taubøll, E.; Mørkrid, L.; Bjørnenak, T.; Saetre, E.R.; Mørland, T.; Gjerstad, L. Antiepileptic drugs alter reproductive endocrine hormones in men with epilepsy. Eur. J. Neurol. 2005, 12, 118–124. [Google Scholar] [CrossRef]
- Khan, M.; Jose, A.; Sharma, S. Physiology, Parathyroid Hormone. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Guo, Y.; Lin, Z.; Huang, Y.; Yu, L. Effects of valproate, lamotrigine, and levetiracetam monotherapy on bone health in newly diagnosed adult patients with epilepsy. Epilepsy Behav. 2020, 113, 107489. [Google Scholar] [CrossRef]
- Vasilchenko, O.A. Effect of use of anticonvulsants on bone tissue. Mod. Sci. Bull. 2016, 10, 69–72. Available online: https://elibrary.ru/item.asp?id=26944816 (accessed on 12 March 2023).
- Dontseva, E.A.; Pilipenko, P.I.; Shnayder, N.A.; Petrova, M.M.; Nasyrova, R.F. Prevalence of anticonvulsant-induced vitamin D deficiency. Epilepsy Paroxysmal Cond. 2022, 14, 304–315. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Shen, C.H.; Lai, Q.L.; Fang, G.L.; Ming, W.J.; Lu, R.Y.; Ding, M.P. Effects of antiepileptic drug on thyroid hormones in patients with epilepsy: A meta-analysis. Seizure 2016, 35, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Longhi, S.; Radetti, G. Thyroid function and obesity. J. Clin. Res. Pediatr. Endocrinol. 2013, 5 (Suppl. 1), 40–44. [Google Scholar] [CrossRef] [PubMed]
- Güngör, O.; Özkaya, A.K.; Temiz, F. The effect of antiepileptic drugs on thyroid hormonal function: Valproic acid and phenobarbital. Acta Neurol. Belg. 2020, 120, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Cansu, A.; Serdaroğlu, A.; Camurdan, O.; Hirfanoğlu, T.; Bideci, A.; Gücüyener, K. The evaluation of thyroid functions, thyroid antibodies, and thyroid volumes in children with epilepsy during short-term administration of oxcarbazepine and valproate. Epilepsia 2006, 47, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Attilakos, A.; Katsarou, E.; Prassouli, A.; Mastroyianni, S.; Voudris, K.; Fotinou, A.; Garoufi, A. Thyroid function in children with epilepsy treated with sodium valproate monotherapy: A prospective study. Clin. Neuropharmacol. 2009, 32, 32–34. [Google Scholar] [CrossRef]
- Fevery, J. Bilirubin in clinical practice: A review. Liver Int. 2008, 28, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Vogel, M.E.; Idelman, G.; Konaniah, E.S.; Zucker, S.D. Bilirubin Prevents Atherosclerotic Lesion Formation in Low-Density Lipoprotein Receptor-Deficient Mice by Inhibiting Endothelial VCAM-1 and ICAM-1 Signaling. J. Am. Heart Assoc. 2017, 6, e004820. [Google Scholar] [CrossRef]
- Meseguer, E.S.; Elizalde, M.U.; Borobia, A.M.; Ramírez, E. Valproic Acid-Induced Liver Injury: A Case-Control Study from a Prospective Pharmacovigilance Program in a Tertiary Hospital. J. Clin. Med. 2021, 10, 1153. [Google Scholar] [CrossRef]
- Dikalova, A.E.; Itani, H.A.; Nazarewicz, R.R.; McMaster, W.G.; Flynn, C.R.; Uzhachenko, R.; Fessel, J.P.; Gamboa, J.L.; Harrison, D.G.; Dikalov, S.I. Sirt3 Impairment and SOD2 Hyperacetylation in Vascular Oxidative Stress and Hypertension. Circ. Res. 2017, 121, 564–574. [Google Scholar] [CrossRef]
- Harjumäki, R.; Pridgeon, C.S.; Ingelman-Sundberg, M. CYP2E1 in Alcoholic and Non-Alcoholic Liver Injury. Roles of ROS, Reactive Intermediates and Lipid Overload. Int. J. Mol. Sci. 2021, 22, 8221. [Google Scholar] [CrossRef]
- Yu, H.Y.; Shen, Y.Z. Displacement effect of valproate on bilirubin-albumin binding in human plasma. J. Formos. Med. Assoc. 1999, 98, 201–204. [Google Scholar] [PubMed]
- Karpisek, M.; Stejskal, D.; Kotolova, H.; Kollar, P.; Janoutova, G.; Ochmanova, R.; Cizek, L.; Horakova, D.; Yahia, R.B.; Lichnovska, R.; et al. Treatment with atorvastatin reduces serum adipocyte-fatty acid binding protein value in patients with hyperlipidaemia. Eur. J. Clin. Investig. 2007, 37, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Masetti, M.; Bianchi, G.; Gianotti, G.; Giovagnoli, M.; Vizioli, L.; Zorzi, V.; Rossi, V.; Forti, P.; Zoli, M. Adipocyte-fatty acid binding protein and non-alcoholic fatty liver disease in the elderly. Aging Clin. Exp. Res. 2014, 26, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Li, H.L.; Wu, X.; Xu, A.; Hoo, R.L. A-FABP in Metabolic Diseases and the Therapeutic Implications: An Update. Int. J. Mol. Sci. 2021, 22, 9386. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Yang, K.; Wang, S.; Xie, Y.; Zhang, L.; Tian, X. PXR-mediated expression of FABP4 promotes valproate-induced lipid accumulation in HepG2 cells. Toxicol. Lett. 2021, 346, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Vitins, A.P.; Kienhuis, A.S.; Speksnijder, E.N.; Roodbergen, M.; Luijten, M.; van der Ven, L.T. Mechanisms of amiodarone and valproic acid induced liver steatosis in mouse in vivo act as a template for other hepatotoxicity models. Arch. Toxicol. 2014, 88, 1573–1588. [Google Scholar] [CrossRef]
- Vejrazkova, D.; Vankova, M.; Lukasova, P.; Vcelak, J.; Bendlova, B. Insights into the physiology of C-peptide. Physiol. Res. 2020, 69 (Suppl. 2), S237–S243. [Google Scholar] [CrossRef]
- Gonzalez-Mejia, M.E.; Porchia, L.M.; Torres-Rasgado, E.; Ruiz-Vivanco, G.; Pulido-Pérez, P.; Báez-Duarte, B.G.; Pérez-Fuentes, R. C-Peptide Is a Sensitive Indicator for the Diagnosis of Metabolic Syndrome in Subjects from Central Mexico. Metab. Syndr. Relat. Disord. 2016, 14, 210–216. [Google Scholar] [CrossRef]
- Pujia, R.; Maurotti, S.; Coppola, A.; Romeo, S.; Pujia, A.; Montalcini, T. The Potential Role of C-peptide in Sexual and Reproductive Functions in Type 1 Diabetes Mellitus: An Update. Curr. Diabetes Rev. 2022, 18, e051021196983. [Google Scholar] [CrossRef]
- Barbaro, G. Highly active antiretroviral therapy-associated metabolic syndrome: Pathogenesis and cardiovascular risk. Am. J. Ther. 2006, 13, 248–260. [Google Scholar] [CrossRef]
- Xu, Y.; Ding, Y.; Li, X.; Wu, X. Cystatin C is a disease-associated protein subject to multiple regulation. Immunol. Cell Biol. 2015, 93, 442–451. [Google Scholar] [CrossRef]
- Van der Laan, S.W.; Fall, T.; Soumaré, A.; Teumer, A.; Sedaghat, S.; Baumert, J.; Zabaneh, D.; van Setten, J.; Isgum, I.; Galesloot, T.E.; et al. Cystatin C and Cardiovascular Disease: A Mendelian Randomization Study. J. Am. Coll. Cardiol. 2016, 68, 934–945. [Google Scholar] [CrossRef]
- Ferguson, T.W.; Komenda, P.; Tangri, N. Cystatin C as a biomarker for estimating glomerular filtration rate. Curr. Opin. Nephrol. Hypertens. 2015, 24, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Naruse, H.; Ishii, J.; Kawai, T.; Hattori, K.; Ishikawa, M.; Okumura, M.; Kan, S.; Nakano, T.; Matsui, S.; Nomura, M.; et al. Cystatin C in acute heart failure without advanced renal impairment. Am. J. Med. 2009, 122, 566–573. [Google Scholar] [CrossRef]
- Ishigaki, K.; Kato, I.; Murakami, T.; Sato, T.; Shichiji, M.; Ishiguro, K.; Ishizuka, K.; Funatsuka, M.; Saito, K.; Osawa, M.; et al. Renal dysfunction is rare in Fukuyama congenital muscular dystrophy. Brain Dev. 2019, 41, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Knovich, M.A.; Coffman, L.G.; Torti, F.M.; Torti, S.V. Serum ferritin: Past, present and future. Biochim. Biophys. Acta 2010, 1800, 760–769. [Google Scholar] [CrossRef]
- Gautam, S.; Alam, F.; Moin, S.; Noor, N.; Arif, S.H. Role of ferritin and oxidative stress index in gestational diabetes mellitus. J. Diabetes Metab. Disord. 2021, 20, 1615–1619. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Y.; Cheng, C.-M.; Chung, M.-S. Anemia Secondary to Valproic Acid in a Female Patient with Schizoaffective Disorder. Taiwan. J. Psychiatry 2015, 29, 61–62. [Google Scholar]
- Kaczorowska-Hac, B.; Matheisel, A.; Maciejka-Kapuscinska, L.; Wisniewski, J.; Alska, A.; Adamkiewicz-Drozynska, E.; Balcerska, A.; Reszczynska, I. Anemia secondary to valproic acid therapy in a 13-year-old boy: A case report. J. Med. Case Rep. 2012, 6, 239. [Google Scholar] [CrossRef]
- Yang, W.J.; Chen, Z.H.; Zheng, Y.N. Valproic acid induced aplastic crisis and Stevens-Johnson syndrome in a single pediatric patient. Heliyon 2022, 9, e12461. [Google Scholar] [CrossRef]
- Valproic Acid and Serum Ferritin Increased—A Phase IV Clinical Study of FDA Data. Available online: https://www.ehealthme.com/ds/valproic-acid/serum-ferritin-increased/ (accessed on 12 March 2023).
- Chen, H.F.; Xu, L.P.; Luo, Z.Y.; Yu, Z.Q.; Li, Z.Y.; Cui, Q.Y.; Qin, L.M.; Ren, Y.Y.; Shen, H.S.; Tang, J.Q.; et al. Valproic acid-associated low fibrinogen and delayed intracranial hemorrhage: Case report and mini literature review. Drug Des. Dev. Ther. 2013, 7, 767–770. [Google Scholar] [CrossRef]
- Surma, S.; Banach, M. Fibrinogen and Atherosclerotic Cardiovascular Diseases—Review of the Literature and Clinical Studies. Int. J. Mol. Sci. 2022, 23, 193. [Google Scholar] [CrossRef] [PubMed]
- Onat, A.; Ozhan, H.; Erbilen, E.; Albayrak, S.; Küçükdurmaz, Z.; Can, G.; Keleş, I.; Hergenç, G. Independent prediction of metabolic syndrome by plasma fibrinogen in men, and predictors of elevated levels. Int. J. Cardiol. 2009, 135, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Hasanoǧlu, C.; Karalök, Z.S.; Kalenderoǧlu, M.D.; Haspolat, Ş. A Rare Cause of Hemorrhage from Valproic Acid—Induced Hypofibrinogenemia and Review of Literature. Ann. Clin. Case Rep. 2021, 6, 1910. [Google Scholar]
- Kharitonenkov, A.; Shiyanova, T.L.; Koester, A.; Ford, A.M.; Micanovic, R.; Galbreath, E.J.; Sandusky, G.E.; Hammond, L.J.; Moyers, J.S.; Owens, R.A.; et al. FGF-21 as a novel metabolic regulator. J. Clin. Investig. 2005, 115, 1627–1635. [Google Scholar] [CrossRef]
- Keipert, S.; Ost, M. Stress-induced FGF21 and GDF15 in obesity and obesity resistance. Trends Endocrinol. Metab. 2021, 32, 904–915. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Guo, C.; Liang, X.; Li, R.; Chen, J. Potential biomarker of fibroblast growth factor 21 in valproic acid-treated livers. Biofactors 2019, 45, 740–749. [Google Scholar] [CrossRef]
- Chang, H.H.; Chen, P.S.; Cheng, Y.W.; Wang, T.Y.; Yang, Y.K.; Lu, R.B. FGF21 Is Associated with Metabolic Effects and Treatment Response in Depressed Bipolar II Disorder Patients Treated with Valproate. Int. J. Neuropsychopharmacol. 2018, 21, 319–324. [Google Scholar] [CrossRef]
- Leng, Y.; Wang, J.; Wang, Z.; Liao, H.M.; Wei, M.; Leeds, P.; Chuang, D.M. Valproic Acid and Other HDAC Inhibitors Upregulate FGF21 Gene Expression and Promote Process Elongation in Glia by Inhibiting HDAC2 and 3. Int. J. Neuropsychopharmacol. 2016, 19, pyw035. [Google Scholar] [CrossRef]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interferon Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef]
- Panee, J. Monocyte Chemoattractant Protein 1 (MCP-1) in obesity and diabetes. Cytokine 2012, 60, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.; Donham, P.; van Rooijen, N.; Wood, J.G.; Gonzalez, N.C. Monocyte chemoattractant protein-1 released from alveolar macrophages mediates the systemic inflammation of acute alveolar hypoxia. Am. J. Respir. Cell. Mol. Biol. 2011, 45, 53–61. [Google Scholar] [CrossRef]
- Verrotti, A.; Basciani, F.; Trotta, D.; Greco, R.; Morgese, G.; Chiarelli, F. Effect of anticonvulsant drugs on interleukins-1, -2 and -6 and monocyte chemoattractant protein-1. Clin. Exp. Med. 2001, 1, 133–136. [Google Scholar] [CrossRef]
- Shao, L.; Wu, B.; Liu, C.; Chong, W. Valproic acid inhibits classical monocyte-derived tissue factor and alleviates hemorrhagic shock-induced acute lung injury in rats. Shock 2023, 59, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Altalhi, R.; Pechlivani, N.; Ajjan, R.A. PAI-1 in Diabetes: Pathophysiology and Role as a Therapeutic Target. Int. J. Mol. Sci. 2021, 22, 3170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Gao, B.; Xu, B. Association between plasminogen activator inhibitor-1 (PAI-1) 4G/5G polymorphism and risk of Alzheimer’s disease, metabolic syndrome, and female infertility: A meta-analysis. Medicine 2020, 99, e23660. [Google Scholar] [CrossRef]
- Martinez de Lizarrondo, S.; Gauberti, M.; Vivien, D. Valproic acid: A relevant thromboprophylactic strategy? J. Thromb. Haemost. 2016, 14, 2493–2495. [Google Scholar] [CrossRef] [PubMed]
- Saluveer, O.; Larsson, P.; Ridderstråle, W.; Hrafnkelsdóttir, T.J.; Jern, S.; Bergh, N. Profibrinolytic effect of the epigenetic modifier valproic acid in man. PLoS ONE 2014, 9, e107582. [Google Scholar] [CrossRef]
- Larsson, P.; Alwis, I.; Niego, B.; Sashindranath, M.; Fogelstrand, P.; Wu, M.C.; Glise, L.; Magnusson, M.; Daglas, M.; Bergh, N.; et al. Valproic acid selectively increases vascular endothelial tissue-type plasminogen activator production and reduces thrombus formation in the mouse. J. Thromb. Haemost. 2016, 14, 2496–2508. [Google Scholar] [CrossRef]
- Larsson, P.; Ulfhammer, E.; Magnusson, M.; Bergh, N.; Lunke, S.; El-Osta, A.; Medcalf, R.L.; Svensson, P.A.; Karlsson, L.; Jern, S. Role of histone acetylation in the stimulatory effect of valproic acid on vascular endothelial tissue-type plasminogen activator expression. PLoS ONE 2012, 7, e31573. [Google Scholar] [CrossRef]
- Christou, G.A.; Tselepis, A.D.; Kiortsis, D.N. The metabolic role of retinol binding protein 4: An update. Horm. Metab. Res. 2012, 44, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Jialal, I.; Devaraj, S. Subcutaneous adipose tissue biology in metabolic syndrome. Horm. Mol. Biol. Clin. Investig. 2018, 33, 20170074. [Google Scholar] [CrossRef] [PubMed]
- Chuang, C.M.; Chang, C.H.; Wang, H.E.; Chen, K.C.; Peng, C.C.; Hsieh, C.L.; Peng, R.Y. Valproic acid downregulates RBP4 and elicits hypervitaminosis A-teratogenesis--a kinetic analysis on retinol/retinoic acid homeostatic system. PLoS ONE 2012, 7, e43692. [Google Scholar] [CrossRef] [PubMed]
- Weiner, F.R.; Shah, A.; Smith, P.J.; Rubin, C.S.; Zern, M.A. Regulation of collagen gene expression in 3T3-L1 cells. Effects of adipocyte differentiation and tumor necrosis factor alpha. Biochemistry 1989, 28, 4094–4099. [Google Scholar] [CrossRef] [PubMed]
- Tzanavari, T.; Giannogonas, P.; Karalis, K.P. TNF-alpha and obesity. Curr. Dir. Autoimmun. 2010, 11, 145–156. [Google Scholar] [CrossRef]
- McCormick, M. Letter to the editor: Obesity and inflammation: Change in adiponectin, C-reactive protein, tumour necrosis factor-alpha and interleukin-6 after bariatric surgery. Obes. Surg. 2013, 23, 111. [Google Scholar] [CrossRef] [PubMed]
- Ichiyama, T.; Okada, K.; Lipton, J.M.; Matsubara, T.; Hayashi, T.; Furukawa, S. Sodium valproate inhibits production of TNF-alpha and IL-6 and activation of NF-kappaB. Brain Res. 2000, 857, 246–251. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Z.Y.; Fauser, U.; Schluesener, H.J. Valproic acid attenuates inflammation in experimental autoimmune neuritis. Cell. Mol. Life Sci. 2008, 65, 4055–4065. [Google Scholar] [CrossRef]
- Wang, C.; Luan, Z.; Yang, Y.; Wang, Z.; Cui, Y.; Gu, G. Valproic acid induces apoptosis in differentiating hippocampal neurons by the release of tumor necrosis factor-α from activated astrocytes. Neurosci. Lett. 2011, 497, 122–127. [Google Scholar] [CrossRef]
- Farrelly, L.A.; Savage, N.T.; O’Callaghan, C.; Toulouse, A.; Yilmazer-Hanke, D.M. Therapeutic concentrations of valproate but not amitriptyline increase neuropeptide Y (NPY) expression in the human SH-SY5Y neuroblastoma cell line. Regul. Pept. 2013, 186, 123–130. [Google Scholar] [CrossRef]
- Brill, J.; Lee, M.; Zhao, S.; Fernald, R.D.; Huguenard, J.R. Chronic valproic acid treatment triggers increased neuropeptide y expression and signaling in rat nucleus reticularis thalami. J. Neurosci. 2006, 26, 6813–6822. [Google Scholar] [CrossRef] [PubMed]
- Jong, R.J. The Role of NPY in Seizure Control and Weight Gain when Valpoic Acid Is Used by Epilepsy Patients. Available online: https://studenttheses.uu.nl/handle/20.500.12932/17789 (accessed on 2 September 2012).
- Van der Vorst, E.P.C. High-Density Lipoproteins and Apolipoprotein A1. In Vertebrate and Invertebrate Respiratory Proteins, Lipoproteins and other Body Fluid Proteins; Subcellular Biochemistry; Springer: Berlin/Heidelberg, Germany, 2020; Volume 94, pp. 399–420. [Google Scholar] [CrossRef]
- Koseki, M.; Yamashita, S.; Ogura, M.; Ishigaki, Y.; Ono, K.; Tsukamoto, K.; Hori, M.; Matsuki, K.; Yokoyama, S.; Harada-Shiba, M. Current Diagnosis and Management of Tangier Disease. J. Atheroscler. Thromb. 2021, 28, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, Z.Y.; Guo, X.L.; Tu, M. Monocyte to High-Density lipoprotein and Apolipoprotein A1 Ratios: Novel Indicators for Metabolic Syndrome in Chinese Newly Diagnosed Type 2 Diabetes. Front. Endocrinol. 2022, 13, 935776. [Google Scholar] [CrossRef] [PubMed]
- Morita, S.Y. Metabolism and Modification of Apolipoprotein B-Containing Lipoproteins Involved in Dyslipidemia and Atherosclerosis. Biol. Pharm. Bull. 2016, 39, 1–24. [Google Scholar] [CrossRef]
- Marston, N.A.; Giugliano, R.P.; Melloni, G.E.M.; Park, J.G.; Morrill, V.; Blazing, M.A.; Ference, B.; Stein, E.; Stroes, E.S.; Braunwald, E.; et al. Association of Apolipoprotein B-Containing Lipoproteins and Risk of Myocardial Infarction in Individuals With and Without Atherosclerosis: Distinguishing Between Particle Concentration, Type, and Content. JAMA Cardiol. 2022, 7, 250–256. [Google Scholar] [CrossRef]
- Elhadad, M.A.; Wilson, R.; Zaghlool, S.B.; Huth, C.; Gieger, C.; Grallert, H.; Graumann, J.; Rathmann, W.; Koenig, W.; Sinner, M.F.; et al. Metabolic syndrome and the plasma proteome: From association to causation. Cardiovasc. Diabetol. 2021, 20, 111. [Google Scholar] [CrossRef]
- Nosadini, R.; Tonolo, G. Role of oxidized low density lipoproteins and free fatty acids in the pathogenesis of glomerulopathy and tubulointerstitial lesions in type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 79–85. [Google Scholar] [CrossRef]
- Sobczak, A.I.S.; Blindauer, C.A.; Stewart, A.J. Changes in Plasma Free Fatty Acids Associated with Type-2 Diabetes. Nutrients 2019, 11, 2022. [Google Scholar] [CrossRef]
- Ghosh, A.; Gao, L.; Thakur, A.; Siu, P.M.; Lai, C.W.K. Role of free fatty acids in endothelial dysfunction. J. Biomed. Sci. 2017, 24, 50. [Google Scholar] [CrossRef]
- Silva, M.F.; Aires, C.C.; Luis, P.B.; Ruiter, J.P.; IJlst, L.; Duran, M.; Wanders, R.J.; Tavares de Almeida, I. Valproic acid metabolism and its effects on mitochondrial fatty acid oxidation: A review. J. Inherit. Metab. Dis. 2008, 31, 205–216. [Google Scholar] [CrossRef]
- Chang, R.; Chou, M.C.; Hung, L.Y.; Wang, M.E.; Hsu, M.C.; Chiu, C.H. Study of Valproic Acid-Enhanced Hepatocyte Steatosis. Biomed Res. Int. 2016, 2016, 9576503. [Google Scholar] [CrossRef]
- Johno, I.; Huang, M.Y.; Levy, R.H. Systemic interaction between valproic acid and free fatty acids in rhesus monkeys. Epilepsia 1982, 23, 649–656. [Google Scholar] [CrossRef]
- Zhang, L.F.; Liu, L.S.; Chu, X.M.; Xie, H.; Cao, L.J.; Guo, C.; A, J.Y.; Cao, B.; Li, M.J.; Wang, G.J.; et al. Combined effects of a high-fat diet and chronic valproic acid treatment on hepatic steatosis and hepatotoxicity in rats. Acta Pharmacol. Sin. 2014, 35, 363–372. [Google Scholar] [CrossRef]
- Parthasarathy, S.; Raghavamenon, A.; Garelnabi, M.O.; Santanam, N. Oxidized low-density lipoprotein. Methods Mol. Biol. 2010, 610, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Jaeri, S. The long term effect of valproic acid in lipid profiles among adult. J. Neurol. Sci. 2019, 405S, 116543. [Google Scholar] [CrossRef]
- Schade, D.S.; Shey, L.; Eaton, R.P. Cholesterol Review: A Metabolically Important Molecule. Endocr. Pract. 2020, 26, 1514–1523. [Google Scholar] [CrossRef]
- Stadler, J.T.; Marsche, G. Obesity-Related Changes in High-Density Lipoprotein Metabolism and Function. Int. J. Mol. Sci. 2020, 21, 8985. [Google Scholar] [CrossRef]
- Delacrétaz, A.; Glatard, A.; Dubath, C.; Gholam, M.; Gamma, F.; von Gunten, A.; Conus, P.; Eap, C.B. Valproate is associated with early decrease of high-density lipoprotein cholesterol levels in the psychiatric population. Basic Clin. Pharmacol. Toxicol. 2021, 129, 26–35. [Google Scholar] [CrossRef]
- Cengiz, M.; Yüksel, A.; Seven, M. The effects of carbamazepine and valproic acid on the erythrocyte glutathione, glutathione peroxidase, superoxide dismutase and serum lipid peroxidation in epileptic children. Pharmacol. Res. 2000, 41, 423–425. [Google Scholar] [CrossRef]
- Dzięgielewska-Gęsiak, S.; Wyszomirska, K.; Fatyga, E.; Wysocka, E.; Muc-Wierzgoń, M. The role of oxidant-antioxidant markers and resistin in metabolic syndrome elderly individuals. Sci. Prog. 2021, 104, 368504211006510. [Google Scholar] [CrossRef] [PubMed]
- Lucas, S.; Hansen, J. 281—Valproic acid inhibits superoxide dismutase activity in mouse P19 cells. Free Radic. Biol. Med. 2018, 128, S118. [Google Scholar] [CrossRef]
- Hadzagic-Catibusic, F.; Hasanbegovic, E.; Melunovic, M.; Zubcevic, S.; Uzicanin, S. Effects of Carbamazepine and Valproate on Serum Aspartate Aminotransferase, Alanine Aminotransferase and Gamma—Glutamyltransferase in Children. Med. Arch. 2017, 71, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Doi, H.; Ko, T.; Fukuma, T.; Kadono, T.; Asaeda, K.; Kobayashi, R.; Nakano, T.; Doi, T.; Nakatsugawa, Y.; et al. Frequently abnormal serum gamma-glutamyl transferase activity is associated with future development of fatty liver: A retrospective cohort study. BMC Gastroenterol. 2020, 20, 217. [Google Scholar] [CrossRef]
- Ndrepepa, G.; Kastrati, A. Gamma-glutamyl transferase and cardiovascular disease. Ann. Transl. Med. 2016, 4, 481. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Bhattarai, B.; Kafle, P.; Khalid, M.; Jonnadula, S.K.; Lamicchane, J.; Kanth, R.; Gayam, V. Elevated Liver Enzymes in Patients with Type 2 Diabetes Mellitus and Non-alcoholic Fatty Liver Disease. Cureus 2018, 10, e3626. [Google Scholar] [CrossRef] [PubMed]
- Carlquist, J.F.; Muhlestein, J.B.; Anderson, J.L. Lipoprotein-associated phospholipase A2: A new biomarker for cardiovascular risk assessment and potential therapeutic target. Expert Rev. Mol. Diagn. 2007, 7, 511–517. [Google Scholar] [CrossRef]
- Reynolds, T.M. Amylase. Br. J. Hosp. Med. 2009, 70, M8–M9. [Google Scholar] [CrossRef] [PubMed]
- Akinfemiwa, O.; Zubair, M.; Muniraj, T. Amylase. In StatPearls [Internet]; NBK557738; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Voudris, K.; Attilakos, A.; Katsarou, E.; Mastroyianni, S.; Dimou, S.; Skardoutsou, A.; Prassouli, A.; Garoufi, A. Serum total amylase, pancreatic amylase and lipase activities in epileptic children treated with sodium valproate monotherapy. Brain Dev. 2006, 28, 572–575. [Google Scholar] [CrossRef]
- Reid, I.R.; Bolland, M.J.; Grey, A. Effects of vitamin D supplements on bone mineral density: A systematic review and meta-analysis. Lancet 2014, 383, 146–155. [Google Scholar] [CrossRef]
- Qiu, J.; Guo, H.; Li, L.; Xu, Z.; Xu, Z.; Jing, X.; Hu, Y.; Wen, X.; Chen, F.; Lu, X. Valproic acid therapy decreases serum 25-hydroxyvitamin D level in female infants and toddlers with epilepsy—A pilot longitudinal study. J. Biomed. Res. 2020, 35, 61–67. [Google Scholar] [CrossRef]
- Xu, Z.; Jing, X.; Li, G.; Sun, J.; Guo, H.; Hu, Y.; Sun, F.; Wen, X.; Chen, F.; Wang, T.; et al. Valproate decreases vitamin D levels in pediatric patients with epilepsy. Seizure 2019, 71, 60–65. [Google Scholar] [CrossRef]
- Durá-Travé, T.; Gallinas-Victoriano, F.; Malumbres-Chacón, M.; Moreno-Gónzalez, P.; Aguilera-Albesa, S.; Yoldi-Petri, M.E. Vitamin D deficiency in children with epilepsy taking valproate and levetiracetam as monotherapy. Epilepsy Res. 2018, 139, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Alsemeh, A.E.; Ahmed, M.M.; Fawzy, A.; Samy, W.; Tharwat, M.; Rezq, S. Vitamin E rescues valproic acid-induced testicular injury in rats: Role of autophagy. Life Sci. 2022, 296, 120434. [Google Scholar] [CrossRef]
- Abdella, E.M.; Galaly, S.R.; Mohammed, H.M.; Khadrawy, S.M. Protective role of vitamin E against valproic acid-induced cytogenotoxicity and hepatotoxicity in mice. J. Basic Appl. Zool. 2014, 67, 127–139. [Google Scholar] [CrossRef]
- Bosmans, L.A.; Bosch, L.; Kusters, P.J.H.; Lutgens, E.; Seijkens, T.T.P. The CD40-CD40L Dyad as Immunotherapeutic Target in Cardiovascular Disease. J. Cardiovasc. Transl. Res. 2021, 14, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Daub, S.; Lutgens, E.; Münzel, T.; Daiber, A. CD40/CD40L and Related Signaling Pathways in Cardiovascular Health and Disease-The Pros and Cons for Cardioprotection. Int. J. Mol. Sci. 2020, 21, 8533. [Google Scholar] [CrossRef]
- Davidson, D.C.; Hirschman, M.P.; Spinelli, S.L.; Morrell, C.N.; Schifitto, G.; Phipps, R.P.; Maggirwar, S.B. Antiplatelet activity of valproic acid contributes to decreased soluble CD40 ligand production in HIV type 1-infected individuals. J. Immunol. 2011, 186, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Bruzzone, C.; Gil-Redondo, R.; Seco, M.; Barragán, R.; de la Cruz, L.; Cannet, C.; Schäfer, H.; Fang, F.; Diercks, T.; Bizkarguenaga, M.; et al. A molecular signature for the metabolic syndrome by urine metabolomics. Cardiovasc. Diabetol. 2021, 20, 155. [Google Scholar] [CrossRef]
- Malla, P.; Kumar, R.; Mahapatra, M.K.; Kumar, M. Ramping glucosuria for management of type 2 diabetes mellitus: An emerging cynosure. Med. Res. Rev. 2014, 34, 1146–1167. [Google Scholar] [CrossRef]
- Sturov, N.V.; Popov, S.V.; Mamporia, N.K.; Mager, A.A. Urinary tract infections in patients with type 2 diabetes mellitus with pharmacological glucosuria. Ter. Arkh. 2020, 92, 106–109. [Google Scholar] [CrossRef]
- Tomoeda, K.; Awata, H.; Matsuura, T.; Matsuda, I.; Ploechl, E.; Milovac, T.; Boneh, A.; Scott, C.R.; Danks, D.M.; Endo, F. Mutations in the 4-hydroxyphenylpyruvic acid dioxygenase gene are responsible for tyrosinemia type III and hawkinsinuria. Mol. Genet. Metab. 2000, 71, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.; Du, D.; Chen, L. Metabolomics Provides Novel Insights into Epilepsy Diagnosis and Treatment: A Review. Neurochem Res. 2022, 47, 844–859. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.snpedia.com/index.php/SNPedia (accessed on 21 April 2023).
- Rossi, G.; Ceolotto, G.; Caroccia, B.; Lenzini, L. Genetic screening in arterial hypertension. Nat. Rev. Endocrinol. 2017, 13, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.ncbi.nlm.nih.gov (accessed on 22 April 2023).
- Available online: https://www.ensembl.org/index.html (accessed on 23 April 2023).




| Biomarker | Reference Values | Change in MetS | Symptom of MetS | References |
|---|---|---|---|---|
| A. Carbohydrates | ||||
| Glucose | 7.19 ± 0.94 µmol/L | Controversial | Insulin resistance | [53,54,55,56,57] |
| B. Acids | ||||
| Uric acid | 335.67 ± 94.77 µmol/L | Controversial | CVD | [57,58,59,60,61,62,63] |
| C. Hormones | ||||
| Adiponectin | 13,086.6–14,196.7 ng/mL | Low | Insulin resistance | [57,70,71,72,73,74] |
| Chemerin | 78.12–112.10 ng/mL | High | CHD | [57,75,76] |
| Ghrelin | 110.2 ± 41.23 ng/mL | High | Obesity | [57,77,78,79,80,81,82,83,84,85,86] |
| Insulin | 0.85–1.7 µmol/L | High | Insulin resistance | [57,64,65,66,67,68,69] |
| Leptin | 8.54–14.4 ng/mL | High | Insulin resistance Leptin resistance | [74,87,88,89,90,91,92] |
| Omentin | 140.19 ± 7.35 ng/mL | Normal | CVD | [57,93,94] |
| Parathyroid hormone | 15–65 ng/mL | High | CVD | [57,100,101,102,103] |
| Testosterone | M 270–1070 ng/dL F 15–70 ng/dL | Controversial | Obesity | [57,95,96,97,98,99] |
| Thyroid stimulating hormone | 0.4–5.5 µIU/mL | Controversial | CVD | [57,104,105,106,107,108] |
| D. Other organic compounds | ||||
| Bilirubin direct and total | 2.5–9 µmol/L | High | CVD | [57,109,110,111,112,113,114] |
| E. Proteins | ||||
| Adipocyte fatty acid-binding protein | 20.2–32.3 microg/L | High | Obesity cardiometabolic disorders | [57,115,116,117,118] |
| C-peptide | 1.1–0.9 ng/mL | Controversial | Insulin-related diseases | [57,120,121,122,123] |
| Cystatin C | 0.5–0.89 ± 0.23 mg/L | High | Cardiometabolic disorders | [57,124,125,126,127,128] |
| Ferritin | M 20–250 µg/L, F 10–120 µg/L | Controversial | Oxidative stress Cardiometabolic disorders | [57,129,130] |
| Fibrinogen | 0.53–1.6 g/L | Low | Bleeding | [57,135,136,137,138] |
| Fibroblast Growth Factor 21 | 107.44 ± 18.07–204.19 ± 53.57 ng/mL | High | Obesity CHD | [57,139,140,141,142] |
| Monocyte chemoattractant protein-1 | 300– 500 pg/mL | Controversial | CHD | [57,144,145,146,147,148] |
| Plasminogen activator inhibitor-1 | 1–24 E/mL | Low | Profibrinolytic effect | [57,149,150,151,152,153,154] |
| Retinol-binding protein 4 | 6–400.0 ng/mL | Low | CVD | [57,155,156,157] |
| Tumor necrosis factor-a | <8.1 ng/mL | Controversial | CHD | [57,158,159,160,161,162,163] |
| Neuropeptide Y | 16 nM | High | Hyperinsulinemia | [57,164,165] |
| F. Lipids | ||||
| Oxidized low density lipoprotein | 26–117 IU/L | Controversial | CVD | [57,180,181] |
| Cholesterol | 3–6 mmol/L | Controversial | CVD | [57,182] |
| Apolipoprotein A1 | 0.99 ± 0.29 g/L | Low | CVD | [57,167,168,169] |
| Apolipoprotein B | 0.97–0.09 g/L | Low | CVD | [57,170,171,172] |
| Free fatty acids | M 8.3–10.9 ng/mL, F 11.4–13.6 ng/mL | High | Insulin resistance | [57,173,174,175,176,177,178,179] |
| High density lipoprotein | 2.89–0.23 mmol/L | Low | Insulin resistance | [57,183,184] |
| Low-density lipoprotein cholesterol | 0.94 ± 0.23 mmol/L | Low | CVD | [57] |
| Triglycerides | 3.19–0.21 mmol/L | High | Dyslipidemia Obesity | [57] |
| G. Enzymes | ||||
| Superoxide Dismutase | 1200–2000 U/g | Low | Oxidative stress Inflammation | [57,185,186,187] |
| Gamma-glutamyl transferase | M 10–71 U/L, F 6–42 U/L | High | Oxidative stress Inflammation | [57,188,189,190] |
| Lipoprotein-associated phospholipase A | <200 ng/mL | Controversial | CVD | [57,192] |
| Amylase | 28–85 U/L | Controversial | Oxidative stress Inflammation | [57,193,194,195] |
| H. Vitamins | ||||
| 25-Hydroxyvitamin D | 30–100 ng/mL | Low | CVD Inflammation | [57,194,195,196,197,198,199] |
| Vitamin E | 5.00–18.00 µg/mL | Low | Oxidative stress Inflammation | [57,200,201] |
| I. Other | ||||
| Ligand CD40 | N/A | High | CVD Diabetes mellitus | [202,203,204] |
| Biomarker | Reference Values | Change in MetS | Symptom of MetS | References |
|---|---|---|---|---|
| A. Carbohydrates | ||||
| Glucose | 0–0.8 mmol/L | High | Insulin resistance | [57,205,206,207] |
| Maltitol | None | High | Insulin resistance | [57,205] |
| B. Amino acids | ||||
| Aromatic amino acids | None | High | DM 2 | [57,205] |
| Histidine | 52–162 µmol/mmol | Low | AH | [57,205] |
| Tryptophan | 0.4–1.4 mg | High | CVD | [57] |
| C. Acids | ||||
| 4-hydroxyphenylpyruvic acid (4-HPPA) | None | High | Insulin resistance | [57,205,206,207,208,209] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shnayder, N.A.; Grechkina, V.V.; Trefilova, V.V.; Efremov, I.S.; Dontceva, E.A.; Narodova, E.A.; Petrova, M.M.; Soloveva, I.A.; Tepnadze, L.E.; Reznichenko, P.A.; et al. Valproate-Induced Metabolic Syndrome. Biomedicines 2023, 11, 1499. https://doi.org/10.3390/biomedicines11051499
Shnayder NA, Grechkina VV, Trefilova VV, Efremov IS, Dontceva EA, Narodova EA, Petrova MM, Soloveva IA, Tepnadze LE, Reznichenko PA, et al. Valproate-Induced Metabolic Syndrome. Biomedicines. 2023; 11(5):1499. https://doi.org/10.3390/biomedicines11051499
Chicago/Turabian StyleShnayder, Natalia A., Violetta V. Grechkina, Vera V. Trefilova, Ilya S. Efremov, Evgenia A. Dontceva, Ekaterina A. Narodova, Marina M. Petrova, Irina A. Soloveva, Liia E. Tepnadze, Polina A. Reznichenko, and et al. 2023. "Valproate-Induced Metabolic Syndrome" Biomedicines 11, no. 5: 1499. https://doi.org/10.3390/biomedicines11051499
APA StyleShnayder, N. A., Grechkina, V. V., Trefilova, V. V., Efremov, I. S., Dontceva, E. A., Narodova, E. A., Petrova, M. M., Soloveva, I. A., Tepnadze, L. E., Reznichenko, P. A., Al-Zamil, M., Altynbekova, G. I., Strelnik, A. I., & Nasyrova, R. F. (2023). Valproate-Induced Metabolic Syndrome. Biomedicines, 11(5), 1499. https://doi.org/10.3390/biomedicines11051499








