Effect of Single High Dose Vitamin D Substitution in Hospitalized COVID-19 Patients with Vitamin D Deficiency on Length of Hospital Stay
Abstract
1. Introduction
2. Materials and Methods
2.1. Outcomes
2.2. Data Collection and Management
2.3. Statistical Analysis
3. Results
3.1. Clinical Characteristics at Time of Randomization
3.2. Primary Outcome
Post Hoc Analysis
3.3. Secondary Outcomes
4. Discussion
Strengths and Limitations
5. Conclusions
6. Participant Privacy and Confidentiality
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tazerji, S.S.; Shahabinejad, F.; Tokasi, M.; Rad, M.A.; Khan, M.S.; Safdar, M.; Filipiak, K.J.; Szarpak, L.; Dzieciatkowski, T.; Jurgiel, J.; et al. Global data analysis and risk factors associated with morbidity and mortality of COVID-19. Gene Rep. 2022, 26, 101505. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Ma, S.; Yu, L.; Wang, W.; Yin, Z. Modeling the Global Dynamic Contagion of COVID-19. Front. Public Health 2022, 9, 809987. [Google Scholar] [CrossRef]
- Zhou, H.; Yang, J.; Zhou, C.; Chen, B.; Fang, H.; Chen, S.; Zhang, X.; Wang, L.; Zhang, L. A Review of SARS-CoV2: Compared With SARS-CoV and MERS-CoV. Front. Med. 2021, 8, 628370. [Google Scholar] [CrossRef] [PubMed]
- Cava, E.; Neri, B.; Carbonelli, M.G.; Riso, S.; Carbone, S. Obesity pandemic during COVID-19 outbreak: Narrative review and future considerations. Clin. Nutr. Edinb. Scotl. 2021, 40, 1637–1643. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Ding, M.; Dong, X.; Zhang, J.; Kursat Azkur, A.; Azkur, D.; Gan, H.; Sun, Y.; Fu, W.; Li, W.; et al. Risk factors for severe and critically ill COVID-19 patients: A review. Allergy 2021, 76, 428–455. [Google Scholar] [CrossRef]
- Wilk-Sledziewska, K.; Sielatycki, P.J.; Uscinska, N.; Bujno, E.; Rosolowski, M.; Kakareko, K.; Sledziewski, R.; Rydzewska-Rosolowska, A.; Hryszko, T.; Zbroch, E. The Impact of Cardiovascular Risk Factors on the Course of COVID-19. J. Clin. Med. 2022, 11, 2250. [Google Scholar] [CrossRef]
- Parohan, M.; Yaghoubi, S.; Seraji, A.; Javanbakht, M.H.; Sarraf, P.; Djalali, M. Risk factors for mortality in patients with Coronavirus disease 2019 (COVID-19) infection: A systematic review and meta-analysis of observational studies. Aging Male 2020, 23, 1416–1424. [Google Scholar] [CrossRef]
- Dessie, Z.G.; Zewotir, T. Mortality-related risk factors of COVID-19: A systematic review and meta-analysis of 42 studies and 423,117 patients. BMC Infect. Dis. 2021, 21, 855. [Google Scholar] [CrossRef]
- Jordan, T.; Siuka, D.; Rotovnik, N.K.; Pfeifer, M. COVID-19 and Vitamin D- a Systematic Review. Zdr. Varst. 2022, 61, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Cannell, J.J.; Vieth, R.; Umhau, J.C.; Holick, M.F.; Grant, W.B.; Madronich, S.; Garland, C.F.; Giovannucci, E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Griffiths, C.J.; Martineau, A.R. Vitamin D in the prevention of acute respiratory infection: Systematic review of clinical studies. J. Steroid Biochem. Mol. Biol. 2013, 136, 321–329. [Google Scholar] [CrossRef]
- Hansdottir, S.; Monick, M.M.; Hinde, S.L.; Lovan, N.; Look, D.C.; Hunninghake, G.W. Respiratory epithelial cells convert inactive vitamin D to its active form: Potential effects on host defense. J. Immunol. 2008, 181, 7090–7099. [Google Scholar] [CrossRef]
- Olliver, M.; Spelmink, L.; Hiew, J.; Meyer-Hoffert, U.; Henriques-Normark, B.; Bergman, P. Immunomodulatory Effects of Vitamin D on Innate and Adaptive Immune Responses to Streptococcus pneumoniae. J. Infect. Dis. 2013, 208, 1474–1481. [Google Scholar] [CrossRef] [PubMed]
- Greiller, C.L.; Martineau, A.R. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients 2015, 7, 4240–4270. [Google Scholar] [CrossRef]
- Park, S.; Lee, M.G.; Hong, S.-B.; Lim, C.-M.; Koh, Y.; Huh, J.W. Effect of vitamin D deficiency in Korean patients with acute respiratory distress syndrome. Korean J. Intern. Med. 2018, 33, 1129–1136. [Google Scholar] [CrossRef]
- Sabico, S.; Enani, M.A.; Sheshah, E.; Aljohani, N.J.; Aldisi, D.A.; Alotaibi, N.H.; Alshingetti, N.; Alomar, S.Y.; Alnaami, A.M.; Amer, O.E.; et al. Effects of a 2-Week 5000 IU versus 1000 IU Vitamin D3 Supplementation on Recovery of Symptoms in Patients with Mild to Moderate COVID-19: A Randomized Clinical Trial. Nutrients 2021, 13, 2170. [Google Scholar] [CrossRef]
- Maghbooli, Z.; Sahraian, M.A.; Jamalimoghadamsiahkali, S.; Asadi, A.; Zarei, A.; Zendehdel, A.; Varzandi, T.; Mohammadnabi, S.; Alijani, N.; Karimi, M.; et al. Treatment with 25-Hydroxyvitamin D(3) (Calcifediol) Is Associated With a Reduction in the Blood Neutrophil-to-Lymphocyte Ratio Marker of Disease Severity in Hospitalized Patients With COVID-19: A Pilot Multicenter, Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2021, 27, 1242–1251. [Google Scholar] [CrossRef]
- Karonova, T.L.; Andreeva, A.T.; Golovatuk, K.A.; Bykova, E.S.; Simanenkova, A.V.; Vashukova, M.A.; Grant, W.B.; Shlyakhto, E.V. Low 25(OH)D Level Is Associated with Severe Course and Poor Prognosis in COVID-19. Nutrients 2021, 13, 3021. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Bhansali, A.; Khare, N.; Suri, V.; Yaddanapudi, N.; Sachdeva, N.; Puri, G.D.; Malhotra, P. Short term, high-dose vitamin D supplementation for COVID-19 disease: A randomised, placebo-controlled, study (SHADE study). Postgrad. Med. J. 2022, 98, 87. [Google Scholar] [CrossRef]
- Sulli, A.; Gotelli, E.; Casabella, A.; Paolino, S.; Pizzorni, C.; Alessandri, E.; Grosso, M.; Ferone, D.; Smith, V.; Cutolo, M. Vitamin D and Lung Outcomes in Elderly COVID-19 Patients. Nutrients 2021, 13, 717. [Google Scholar] [CrossRef]
- Murai, I.H.; Fernandes, A.L.; Sales, L.P.; Pinto, A.J.; Goessler, K.F.; Duran, C.S.C.; Silva, C.B.R.; Franco, A.S.; Macedo, M.B.; Dalmolin, H.H.H.; et al. Effect of a Single High Dose of Vitamin D3 on Hospital Length of Stay in Patients With Moderate to Severe COVID-19: A Randomized Clinical Trial. JAMA 2021, 325, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Elamir, Y.M.; Amir, H.; Lim, S.; Rana, Y.P.; Lopez, C.G.; Feliciano, N.V.; Omar, A.; Grist, W.P.; Via, M.A. A randomized pilot study using calcitriol in hospitalized COVID-19 patients. Bone 2022, 154, 116175. [Google Scholar] [CrossRef]
- Cannata-Andía, J.B.; Díaz-Sottolano, A.; Fernández, P.; Palomo-Antequera, C.; Herrero-Puente, P.; Mouzo, R.; Carrillo-López, N.; Panizo, S.; Ibañez, G.H.; Cusumano, C.A.; et al. A single-oral bolus of 100,000 IU of cholecalciferol at hospital admission did not improve outcomes in the COVID-19 disease: The COVID-VIT-D-a randomised multicentre international clinical trial. BMC Med. 2022, 20, 83. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Xie, J.; Li, C.; Liu, L.; Yang, Y. High Dose Vitamin D3 Supplementation Is Not Associated With Lower Mortality in Critically Ill Patients: A Meta-Analysis of Randomized Control Trials. Front. Nutr. 2022, 9, 762316. [Google Scholar] [CrossRef]
- Malihi, Z.; Lawes, C.M.M.; Wu, Z.; Huang, Y.; Waayer, D.; Toop, L.; Khaw, K.-T.; Camargo, C.A., Jr.; Scragg, R. Monthly high-dose vitamin D supplementation does not increase kidney stone risk or serum calcium: Results from a randomized controlled trial. Am. J. Clin. Nutr. 2019, 109, 1578. [Google Scholar] [CrossRef]
- Amrein, K.; Sourij, H.; Wagner, G.; Holl, A.; Pieber, T.R.; Smolle, K.H.; Stojakovic, T.; Schnedl, C.; Dobnig, H. Short-term effects of high-dose oral vitamin D3 in critically ill vitamin D deficient patients: A randomized, double-blind, placebo-controlled pilot study. Crit. Care 2011, 15, R104. [Google Scholar] [CrossRef] [PubMed]
- Apaydin, M.; Can, A.G.; Kizilgul, M.; Beysel, S.; Kan, S.; Caliskan, M.; Demirci, T.; Ozcelik, O.; Ozbek, M.; Cakal, E. The effects of single high-dose or daily low-dosage oral colecalciferol treatment on vitamin D levels and muscle strength in postmenopausal women. BMC Endocr. Disord. 2018, 18, 48. [Google Scholar] [CrossRef] [PubMed]
- Ganmaa, D.; Munkhzul, B.; Fawzi, W.; Spiegelman, D.; Willett, W.C.; Bayasgalan, P.; Baasansuren, E.; Buyankhishig, B.; Oyun-Erdene, S.; Jolliffe, D.A.; et al. High-Dose Vitamin D(3) during Tuberculosis Treatment in Mongolia. A Randomized Controlled Trial. Am. J. Respir. Crit. Care Med. 2017, 196, 628–637. [Google Scholar] [CrossRef]
- Ataide, F.L.; Carvalho Bastos, L.M.; Vicente Matias, M.F.; Skare, T.L.; Freire de Carvalho, J. Safety and effectiveness of vitamin D mega-dose: A systematic review. Clin. Nutr. ESPEN 2021, 46, 115–120. [Google Scholar] [CrossRef]
- Ramasamy, I. Vitamin D Metabolism and Guidelines for Vitamin D Supplementation. Clin. Biochem. Rev. 2020, 41, 103–126. [Google Scholar] [CrossRef]
- ICHH Guideline. International Conference on Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Integrated addendum to ICH E6 (R1): Guideline for good clinical practice E6 (R2). Curr. Step 2015, 2, 1–60. [Google Scholar]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Jaun, F.; Boesing, M.; Lüthi-Corridori, G.; Abig, K.; Makhdoomi, A.; Bloch, N.; Lins, C.; Raess, A.; Grillmayr, V.; Haas, P.; et al. High-dose vitamin D substitution in patients with COVID-19: Study protocol for a randomized, double-blind, placebo-controlled, multi-center study—VitCov Trial. Trials 2022, 23, 114. [Google Scholar] [CrossRef]
- Mariani, J.; Antonietti, L.; Tajer, C.; Ferder, L.; Inserra, F.; Sanchez Cunto, M.; Brosio, D.; Ross, F.; Zylberman, M.; López, D.E.; et al. High-dose vitamin D versus placebo to prevent complications in COVID-19 patients: Multicentre randomized controlled clinical trial. PLoS ONE 2022, 17, e0267918. [Google Scholar] [CrossRef] [PubMed]
- De Niet, S.; Trémège, M.; Coffiner, M.; Rousseau, A.-F.; Calmes, D.; Frix, A.-N.; Gester, F.; Delvaux, M.; Dive, A.-F.; Guglielmi, E.; et al. Positive Effects of Vitamin D Supplementation in Patients Hospitalized for COVID-19: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2022, 14, 3048. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Alcalá Díaz, J.F.; López Miranda, J.; Bouillon, R.; Quesada Gomez, J.M. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubée, V.; Legrand, E.; Sacco, G.; Annweiler, C. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef] [PubMed]
- Asghar, M.S.; Yasmin, F.; Dapke, K.; Shah, S.M.I.; Zafar, M.D.B.; Khan, A.A.; Mohiuddin, O.; Surani, S. Evaluation of Vitamin-D Status and Its Association with Clinical Outcomes Among COVID-19 Patients in Pakistan. Am. J. Trop. Med. Hyg. 2022, 106, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Karonova, T.L.; Golovatyuk, K.A.; Kudryavtsev, I.V.; Chernikova, A.T.; Mikhaylova, A.A.; Aquino, A.D.; Lagutina, D.I.; Zaikova, E.K.; Kalinina, O.V.; Golovkin, A.S.; et al. Effect of Cholecalciferol Supplementation on the Clinical Features and Inflammatory Markers in Hospitalized COVID-19 Patients: A Randomized, Open-Label, Single-Center Study. Nutrients 2022, 14, 2602. [Google Scholar] [CrossRef]
- Torres, M.; Casado, G.; Vigón, L.; Rodríguez-Mora, S.; Mateos, E.; Ramos-Martín, F.; López-Wolf, D.; Sanz-Moreno, J.; Ryan-Murua, P.; Taboada-Martínez, M.L.; et al. Changes in the immune response against SARS-CoV-2 in individuals with severe COVID-19 treated with high dose of vitamin D. Biomed. Pharmacother. 2022, 150, 112965. [Google Scholar] [CrossRef]
- Cervero, M.; López-Wolf, D.; Casado, G.; Novella-Mena, M.; Ryan-Murua, P.; Taboada-Martínez, M.L.; Rodríguez-Mora, S.; Vigón, L.; Coiras, M.; Torres, M. Beneficial Effect of Short-Term Supplementation of High Dose of Vitamin D3 in Hospitalized Patients With COVID-19: A Multicenter, Single-Blinded, Prospective Randomized Pilot Clinical Trial. Front. Pharmacol. 2022, 13, 863587. [Google Scholar] [CrossRef]
- Alsufiani, H.M.; AlGhamdi, S.A.; AlShaibi, H.F.; Khoja, S.O.; Saif, S.F.; Carlberg, C. A Single Vitamin D3 Bolus Supplementation Improves Vitamin D Status and Reduces Proinflammatory Cytokines in Healthy Females. Nutrients 2022, 14, 3963. [Google Scholar] [CrossRef]
- Martineau, A.R.; MacLaughlin, B.D.; Hooper, R.L.; Barnes, N.C.; Jolliffe, D.A.; Greiller, C.L.; Kilpin, K.; McLaughlin, D.; Fletcher, G.; Mein, C.A.; et al. Double-blind randomised placebo-controlled trial of bolus-dose vitamin D3 supplementation in adults with asthma (ViDiAs). Thorax 2015, 70, 451. [Google Scholar] [CrossRef]
- Hiremath, G.; Cettomai, D.; Baynes, M.; Ratchford, J.; Newsome, S.; Harrison, D.; Kerr, D.; Greenberg, B.; Calabresi, P. Vitamin D status and effect of low-dose cholecalciferol and high-dose ergocalciferol supplementation in multiple sclerosis. Mult. Scler. J. 2009, 15, 735–740. [Google Scholar] [CrossRef]
- Carlberg, C.; Haq, A. The concept of the personal vitamin D response index. Vitam. Defic. Hum. Health 2018, 175, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-F.; Huang, Y.-H.; Ju, L.-Y.; Weng, S.-C.; Lee, Y.-S.; Chou, Y.-Y.; Lin, C.-S.; Lin, S.-Y. Health-Related Quality of Life Measured by EQ-5D in Relation to Hospital Stay and Readmission in Elderly Patients Hospitalized for Acute Illness. Int. J. Environ. Res. Public. Health 2020, 17, 5333. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.-M.; Kang, H.G.; Kim, B.J. Syncope after Influenza Virus Infection. J. Korean Med. Sci. 2020, 35, 1145989. [Google Scholar] [CrossRef]
- Marando, M.; Fusi-Schmidhauser, T.; Tamburello, A.; Grazioli Gauthier, L.; Rigamonti, E.; Argentieri, G.; Puligheddu, C.; Pagnamenta, A.; Valenti, A.; Pons, M.; et al. 1-year radiological, functional and quality-of-life outcomes in patients with SARS-CoV-2 pneumonia—A prospective observational study. NPJ Prim. Care Respir. Med. 2022, 32, 8. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Beaudenon, M.; Simon, R.; Guenet, M.; Otekpo, M.; Célarier, T.; Gautier, J. GERIA-COVID study group Vitamin D supplementation prior to or during COVID-19 associated with better 3-month survival in geriatric patients: Extension phase of the GERIA-COVID study. J. Steroid Biochem. Mol. Biol. 2021, 213, 105958. [Google Scholar] [CrossRef]
- AlSafar, H.; Grant, W.B.; Hijazi, R.; Uddin, M.; Alkaabi, N.; Tay, G.; Mahboub, B.; Al Anouti, F. COVID-19 Disease Severity and Death in Relation to Vitamin D Status among SARS-CoV-2-Positive UAE Residents. Nutrients 2021, 13, 1714. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Beaudenon, M.; Gautier, J.; Gonsard, J.; Boucher, S.; Chapelet, G.; Darsonval, A.; Fougère, B.; Guérin, O.; Houvet, M.; et al. High-dose versus standard-dose vitamin D supplementation in older adults with COVID-19 (COVIT-TRIAL): A multicenter, open-label, randomized controlled superiority trial. PLoS Med. 2022, 19, e1003999. [Google Scholar] [CrossRef]
- Saponaro, F.; Franzini, M.; Okoye, C.; Antognoli, R.; Campi, B.; Scalese, M.; Neri, T.; Carrozzi, L.; Monzani, F.; Zucchi, R.; et al. Is There a Crucial Link Between Vitamin D Status and Inflammatory Response in Patients with COVID-19? Front. Immunol. 2022, 12, 745713. [Google Scholar] [CrossRef]
- Gregoriano, C.; Koch, D.; Haubitz, S.; Conen, A.; Fux, C.A.; Mueller, B.; Bernasconi, L.; Hammerer-Lercher, A.; Oberle, M.; Burgermeister, S. Characteristics, predictors and outcomes among 99 patients hospitalised with COVID-19 in a tertiary care centre in Switzerland: An observational analysis. Swiss Med. Wkly. 2020, 150, w20316. [Google Scholar] [CrossRef]
- Tahir, N.; Zahra, F. Neutrophilia. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Rosales, C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types? Front. Physiol. 2018, 9, 113. [Google Scholar] [CrossRef]
- Mayadas, T.N.; Cullere, X.; Lowell, C.A. The Multifaceted Functions of Neutrophils. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 181–218. [Google Scholar] [CrossRef]
- Brass, D.; Mckay, P.; Scott, F. Investigating an incidental finding of lymphopenia. BMJ 2014, 348, g1721. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Viswanathan, S.; Aghoram, R. Idiopathic CD4 lymphocytopenia: Current insights. Immunotargets Ther. 2020, 9, 79. [Google Scholar] [CrossRef]
- Hall, P.; Cash, J. What is the real function of the liver ‘function’tests? Ulster Med. J. 2012, 81, 30. [Google Scholar] [PubMed]
- Luo, M.; Ballester, M.P.; Soffientini, U.; Jalan, R.; Mehta, G. SARS-CoV-2 infection and liver involvement. Hepatol. Int. 2022, 16, 755–774. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.; Waters, B.; Stanfill, A.G. Elevated liver function tests in COVID-19: Causes, clinical evidence, and potential treatments. Nurse Pract. 2021, 46, 21–26. [Google Scholar] [CrossRef]
- Ali, N. Elevated level of C-reactive protein may be an early marker to predict risk for severity of COVID-19. J. Med. Virol. 2020, 92, 2409–2411. [Google Scholar] [CrossRef]
- Luan, Y.; Yin, C.; Yao, Y. Update Advances on C-Reactive Protein in COVID-19 and Other Viral Infections. Front. Immunol. 2021, 12, 720363. [Google Scholar] [CrossRef] [PubMed]
- Al-Jarallah, M.; Rajan, R.; Dashti, R.; Al Saber, A.; Pan, J.; Zhanna, K.D.; Abdelnaby, H.; Aboelhassan, W.; Almutairi, F.; Abdullah, M.; et al. In-hospital mortality in SARS-CoV-2 stratified by serum 25-hydroxy-vitamin D levels: A retrospective study. J. Med. Virol. 2021, 93, 5880–5885. [Google Scholar] [CrossRef]
- Davoudi, A.; Najafi, N.; Aarabi, M.; Tayebi, A.; Nikaeen, R.; Izadyar, H.; Salar, Z.; Delavarian, L.; Vaseghi, N.; Daftarian, Z.; et al. Lack of association between vitamin D insufficiency and clinical outcomes of patients with COVID-19 infection. BMC Infect. Dis. 2021, 21, 450. [Google Scholar] [CrossRef] [PubMed]
- da Rocha, A.P.; Atallah, A.N.; Aldrighi, J.M.; Pires, A.L.R.; Dos Santos Puga, M.E.; Pinto, A.C.P.N. Insufficient evidence for vitamin D use in COVID-19: A rapid systematic review. Int. J. Clin. Pract. 2021, 75, e14649. [Google Scholar] [CrossRef] [PubMed]
- Malinverni, S.; Ochogavia, Q.; Lecrenier, S.; Scorpinniti, M.; Preiser, J.-C.; Cotton, F.; Mols, P.; Bartiaux, M. Severe vitamin D deficiency in patients admitted to the emergency department with severe sepsis is associated with an increased 90-day mortality. Emerg. Med. J. 2022, 40, 36–41. [Google Scholar] [CrossRef]

| Inclusion Criteria |
| Informed consent as documented by signature |
| >18 years old |
| Ongoing COVID-19 infection (PCR confirmed) |
| Hospitalized in one of the participating study centers |
| Laboratory-confirmed serum 25(OH)D3 < 50 nmol/L (<20 ng/mL) |
| Exclusion criteria |
| Known hypersensitivity against one of the used vitamin D3 products |
| Active malignancy |
| Hypercalcemia defined as serum calcium >2.2 mmol/L |
| Granulomatous disease (e.g., sarcoidosis) |
| History of renal stones in the past 12 months |
| Pregnancy or breastfeeding |
| Admission to ICU before inclusion or at day of inclusion |
| Baseline Characteristics | N a (Intervention/Control) | Intervention | Control | p-Value | |
|---|---|---|---|---|---|
| Age | years | 39/39 | 60.49 ± 13.84 | 61.38 ± 15.29 | 0.787 |
| Sex | female, % (n) | 39/39 | 35.9 (14) | 17.9 (7) | 0.125 ‡ |
| BMI | kg/m2 | 39/38 | 28.55 ± 5.09 | 28.44 ± 5.68 | 0.951 † |
| RR | /min | 33/34 | 24 ± 5 | 23 ± 6 | 0.362 † |
| SpO2 | % | 37/37 | 92 ± 2.5 | 93 ± 2.5 | 0.119 |
| Patient with dyspnea b | % | 6/13 | 92 ± 2.5 | 92 ± 3 | 0.487 |
| Oxygen supplementation requirement | % (n) | 38/39 | 37.8 (14) | 62.2 (23) | 0.084 |
| Patients with dyspnea b | % (n) | 6/13 | 50 (3) | 71.4 (10) | 0.393 |
| O2 -supplementation | L/min | 36/35 | 1.5 (±2) | 2 (±2) | 0.071 † |
| Patients with dyspnea b | L/min | 6/13 | 2 ± 2.5 | 3 ± 3 | 0.610 |
| Symptom onset to hospitalization | days | 39/39 | 8 ± 4 | 7 ± 4 | 0.335 |
| Radiological evidence | % (n) | 39/39 | 100 (39) | 94.9 (37) | 0.152 |
| Quality of life c | 26/20 | ||||
| SF-12 physical score | 31.39 ± 10.61 | 27.85 ± 11.75 | 0.352 † | ||
| SF-12 mental score | 41.76 ± 15.51 | 51.66 ± 8.04 | 0.018 † | ||
| Hematology | |||||
| Hemoglobin | g/L | 30/34 | 129.67 ± 15.89 | 134.68 ± 15.78 | 0.211 |
| RBC | 106/µL | 31/34 | 4.35 ± 0.43 | 4.353 ± 0.74 | 0.440 † |
| Leukocytes | 103/µL | 31/34 | 7.11 ± 2.46 | 7.647 ± 3.23 | 0.451 |
| Thrombocytes | 103/µL | 30/34 | 244.90 ± 81.02 | 238.56 ± 101.88 | 0.783 |
| Blood chemistry | |||||
| 25(OH)D3 | nmol/L | 37/39 | 31.46 ± 10.95 | 28.54 ± 10.13 | 0.232 |
| PTH | pmol/L | 7/6 | 3.14 ± 1.34 | 4.160 ± 3.93 | 0.943 † |
| Calcium | mmol/L | 9/13 | 2.15 ± 0.06 | 2.09 ± 0.34 | 0.282 † |
| Phosphate | mmol/L | 7/12 | 1.00 ± 0.29 | 0.86 ± 0.27 | 0.570 |
| CRP d | mg/L | 28/31 | 96.50 ± 57.07 | 66.67 ± 61.23 | 0.015 † |
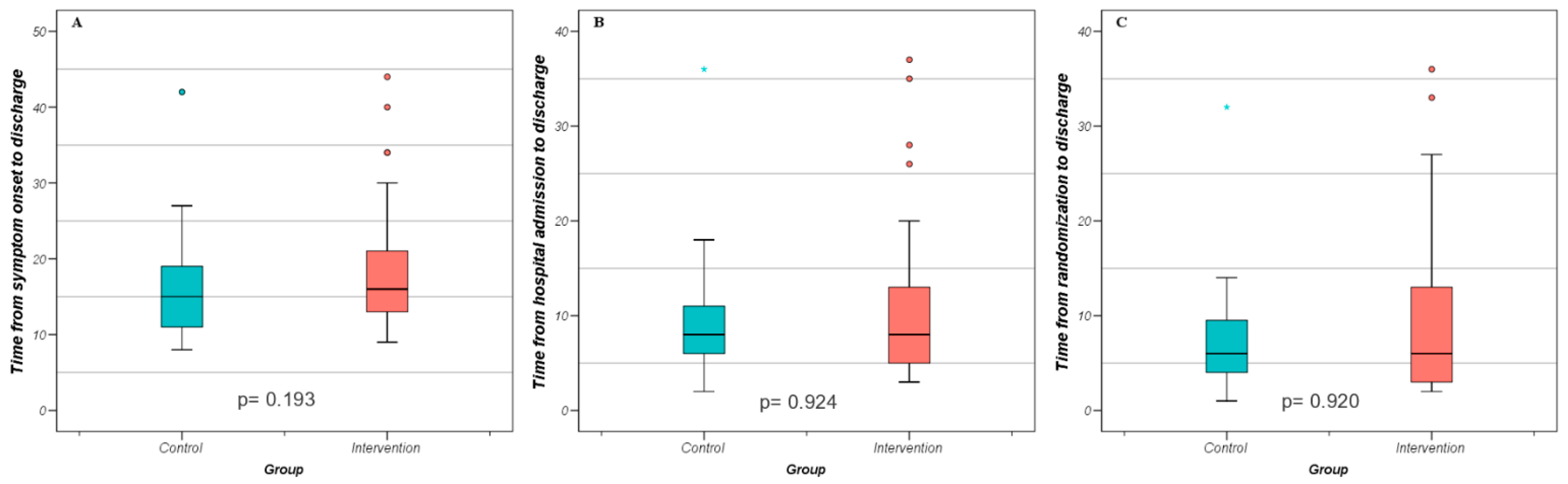
| Length of Stay in Days | Intervention, n = 39 | Control, n = 39 | p-Value |
|---|---|---|---|
| Median (IQR) | Median (IQR) | ||
| Time from symptom onset to discharge | 16 (9) | 15 (8) | 0.193 † |
| Time from hospital admission to discharge | 8 (8) | 8 (6) | 0.924 † |
| Time from randomization to discharge | 6 (10) | 6 (6) | 0.920 † |
| Length of Stay (Days) When Serum 25(OH) D Level <25 nmol /L, n = 25 | Intervention, n = 12 | Control, n = 13 | p-Value |
|---|---|---|---|
| Median (IQR) | Median (IQR) | ||
| Time from symptom onset to discharge | 15 (11) | 15 (9) | 0.764 † |
| Time from hospital admission to discharge | 5.5 (13) | 9 (6) | 0.299 † |
| Time from randomization to discharge | 4.5 (13) | 7 (5) | 0.444 † |
| COVID-19-Related Complications | Odds Ratio | 95% Confidence Interval | p-Value | |
|---|---|---|---|---|
| Lower Level | Upper Level | |||
| ICU Admission | 1.371 | 0.232 | 4.319 | 1.00 |
| ICU Admission COVID-19-related a | 1.371 | 0.286 | 6.576 | 0.693 |
| Intubation | 4.343 | 0.463 | 40.749 | 0.199 |
| Other complications | 1.542 | 0.243 | 9.776 | 0.646 |
| Death | 0.487 | 0.042 | 5.601 | 0.564 |
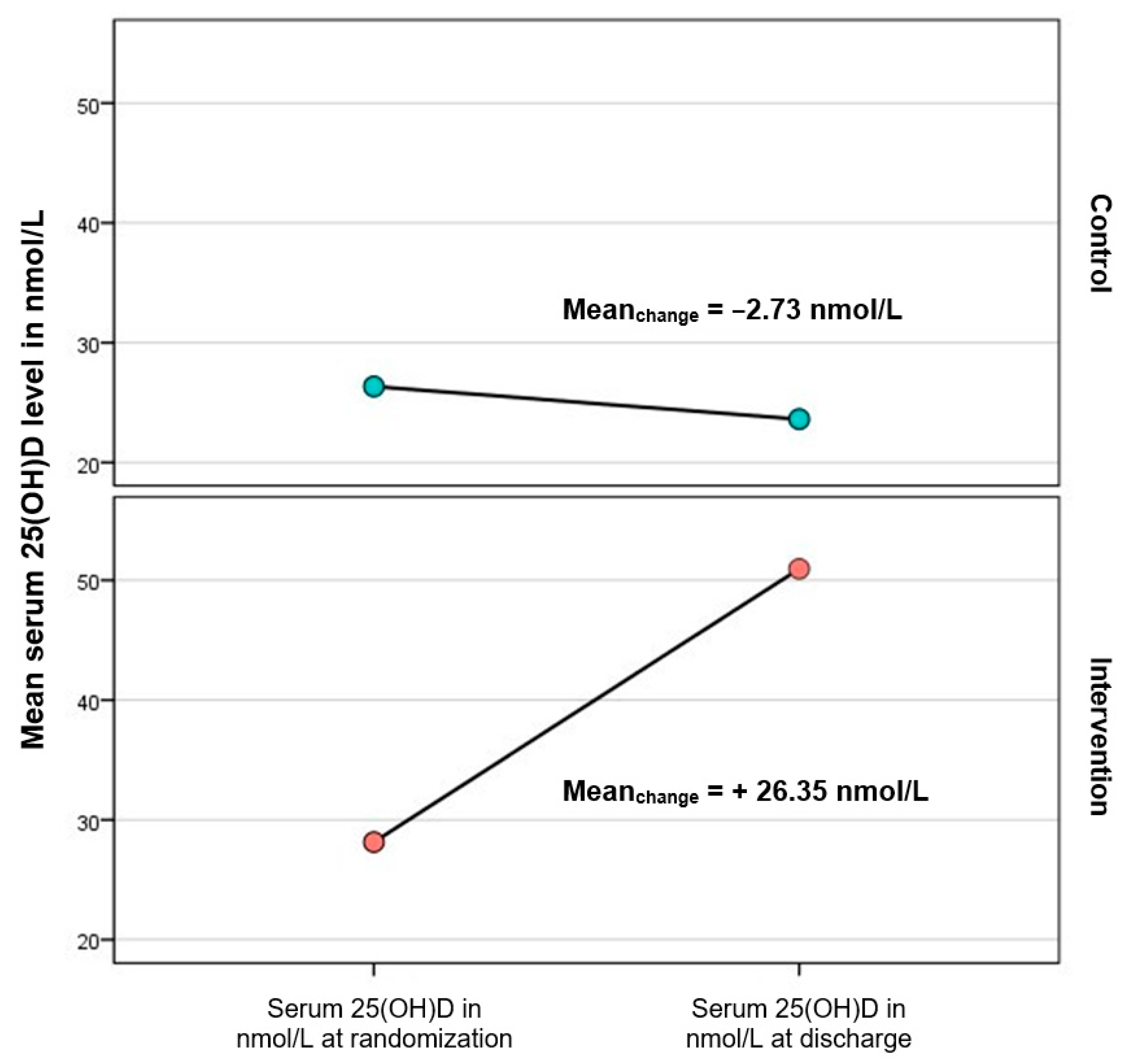
| Serum 25(OH)D Level in nmol/L | N Available (Intervention/Control) a | Intervention | Control | p-Value |
|---|---|---|---|---|
| 25(OH)D level at randomization | 37/39 | 31.46 ± 10.948 [11–44] | 28.54 ± 10.13 [9–45] | 0.232 |
| 25(OH)D level at discharge | 21/26 | 50.95 ± 16.98 [2–83] | 23.62 ± 8.88 [2–61] | <0.001 *** |
| Change 25(OH)D level | 21/26 | 26.35 ± 8.88 [9–45] | −2.73 ± 10.23 [−33–16] | <0.001 *** |
| Patients with 25(OH)D level >50 nmol at discharge | 21/26 | 23.1 (9) | 2.6 (1) | 0.003 ‡ ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaun, F.; Boesing, M.; Luethi-Corridori, G.; Abig, K.; Bloch, N.; Giezendanner, S.; Grillmayr, V.; Haas, P.; Leuppi-Taegtmeyer, A.B.; Muser, J.; et al. Effect of Single High Dose Vitamin D Substitution in Hospitalized COVID-19 Patients with Vitamin D Deficiency on Length of Hospital Stay. Biomedicines 2023, 11, 1277. https://doi.org/10.3390/biomedicines11051277
Jaun F, Boesing M, Luethi-Corridori G, Abig K, Bloch N, Giezendanner S, Grillmayr V, Haas P, Leuppi-Taegtmeyer AB, Muser J, et al. Effect of Single High Dose Vitamin D Substitution in Hospitalized COVID-19 Patients with Vitamin D Deficiency on Length of Hospital Stay. Biomedicines. 2023; 11(5):1277. https://doi.org/10.3390/biomedicines11051277
Chicago/Turabian StyleJaun, Fabienne, Maria Boesing, Giorgia Luethi-Corridori, Kristin Abig, Nando Bloch, Stéphanie Giezendanner, Victoria Grillmayr, Philippe Haas, Anne B. Leuppi-Taegtmeyer, Jürgen Muser, and et al. 2023. "Effect of Single High Dose Vitamin D Substitution in Hospitalized COVID-19 Patients with Vitamin D Deficiency on Length of Hospital Stay" Biomedicines 11, no. 5: 1277. https://doi.org/10.3390/biomedicines11051277
APA StyleJaun, F., Boesing, M., Luethi-Corridori, G., Abig, K., Bloch, N., Giezendanner, S., Grillmayr, V., Haas, P., Leuppi-Taegtmeyer, A. B., Muser, J., Raess, A., Schuetz, P., Brändle, M., & Leuppi, J. D. (2023). Effect of Single High Dose Vitamin D Substitution in Hospitalized COVID-19 Patients with Vitamin D Deficiency on Length of Hospital Stay. Biomedicines, 11(5), 1277. https://doi.org/10.3390/biomedicines11051277






