The Multifaceted Role of Osteopontin in Prostate Pathologies
Abstract
:1. Introduction
2. Osteopontin: Structure, Splice Variants, and Roles in Non-Prostatic Tissues
3. Benign Prostate Disease
3.1. Benign Prostatic Hyperplasia Tissue
3.2. Animal Models of Prostatic Inflammation
3.3. Animal Models of BPH
4. Cell-Specific Roles of OPN in the Prostate
4.1. Resident Prostate Cells
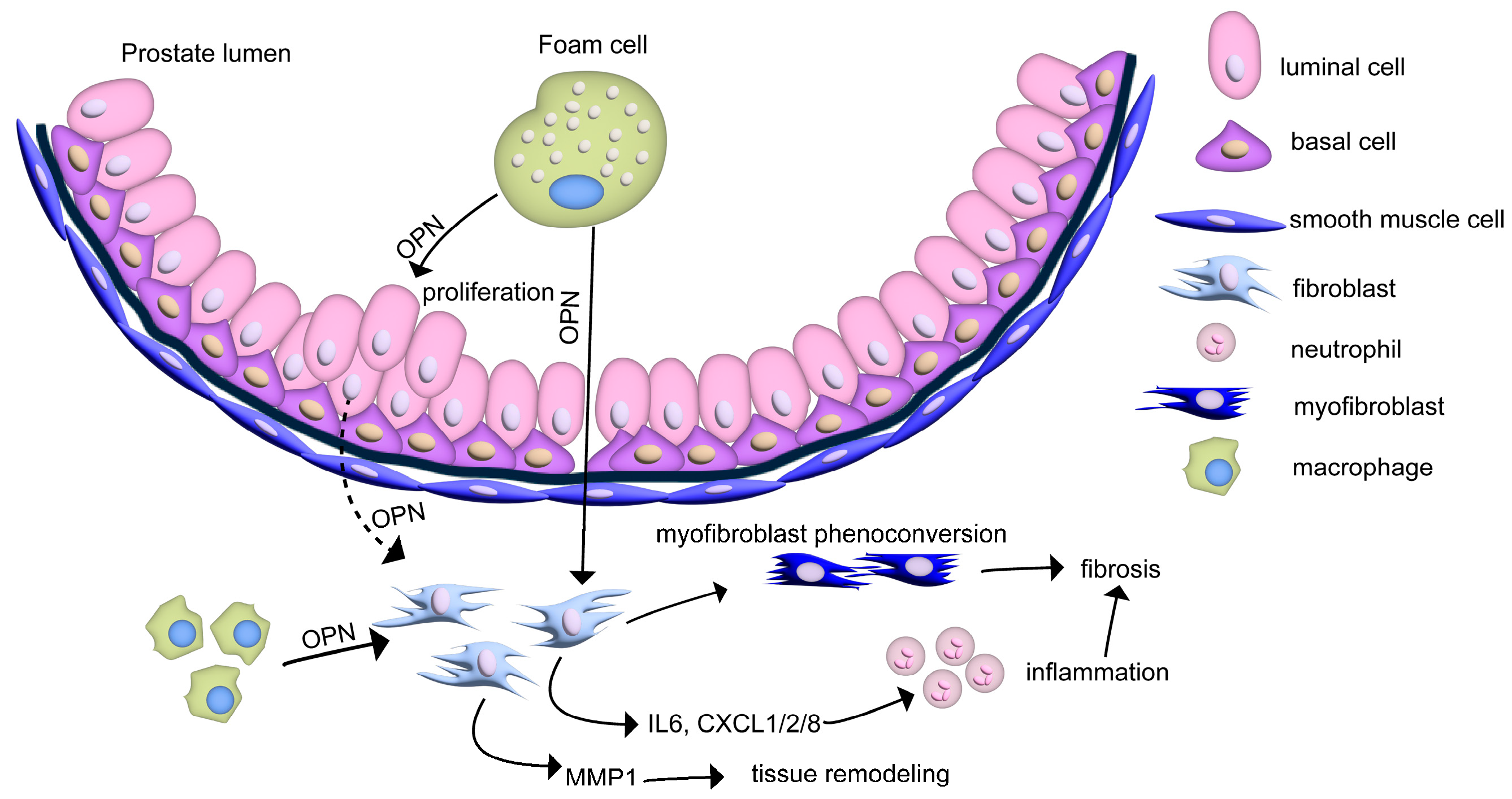
4.2. Macrophages and Foam Cells
4.3. Other Immune Cells
5. Speculating the Role of Luminal OPN in Prostate Disease
6. OPN and Prostate Cancer
7. Conclusions
Funding
Conflicts of Interest
References
- Lilja, H. Biology of prostate-specific antigen. Urology 2003, 62 (Suppl. S1), 27–33. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, F.; Rampin, O. Neural control of erection. Physiol. Behav. 2004, 83, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Krieger, J.N.; Nyberg, L., Jr.; Nickel, J.C. NIH consensus definition and classification of prostatitis. JAMA 1999, 282, 236–237. [Google Scholar] [CrossRef]
- Lepor, H. Pathophysiology of lower urinary tract symptoms in the aging male population. Rev. Urol. 2005, 7 (Suppl. S7), S3–S11. [Google Scholar]
- Rodriguez-Nieves, J.A.; Macoska, J.A. Prostatic fibrosis, lower urinary tract symptoms, and BPH. Nat. Rev. Urol. 2013, 10, 546–550. [Google Scholar] [CrossRef]
- McVary, K.T. BPH: Epidemiology and comorbidities. Am. J. Manag. Care 2006, 12 (Suppl. S5), S122–S128. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Ng, K.L. The Etiology of Prostate Cancer. In Prostate Cancer; Bott, S.R.J., Ng, K.L., Eds.; Exon Publications: Brisbane, Australia, 2021. [Google Scholar]
- Vellky, J.E.; Ricke, W.A. Development and prevalence of castration-resistant prostate cancer subtypes. Neoplasia 2020, 22, 566–575. [Google Scholar] [CrossRef]
- Dai, X.; Fang, X.; Ma, Y.; Xianyu, J. Benign Prostatic Hyperplasia and the Risk of Prostate Cancer and Bladder Cancer: A Meta-Analysis of Observational Studies. Medicine 2016, 95, e3493. [Google Scholar] [CrossRef]
- McNeal, J.E. Origin and evolution of benign prostatic enlargement. Investig. Urol. 1978, 15, 340–345. [Google Scholar]
- De Marzo, A.M.; Platz, E.A.; Sutcliffe, S.; Xu, J.; Gronberg, H.; Drake, C.G.; Nakai, Y.; Isaacs, W.B.; Nelson, W.G. Inflammation in prostate carcinogenesis. Nat. Rev. Cancer 2007, 7, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Agnholt, J.; Kelsen, J.; Schack, L.; Hvas, C.L.; Dahlerup, J.F.; Sorensen, E.S. Osteopontin, a protein with cytokine-like properties, is associated with inflammation in Crohn’s disease. Scand. J. Immunol. 2007, 65, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Sennels, H.; Sorensen, S.; Ostergaard, M.; Knudsen, L.; Hansen, M.; Skjodt, H.; Peters, N.; Colic, A.; Grau, K.; Jacobsen, S. Circulating levels of osteopontin, osteoprotegerin, total soluble receptor activator of nuclear factor-kappa B ligand, and high-sensitivity C-reactive protein in patients with active rheumatoid arthritis randomized to etanercept alone or in combination with methotrexate. Scand. J. Rheumatol. 2008, 37, 241–247. [Google Scholar] [CrossRef]
- Komine-Aizawa, S.; Masuda, H.; Mazaki, T.; Shiono, M.; Hayakawa, S.; Takayama, T. Plasma osteopontin predicts inflammatory bowel disease activities. Int. Surg. 2015, 100, 38–43. [Google Scholar] [CrossRef]
- Leung, T.M.; Wang, X.; Kitamura, N.; Fiel, M.I.; Nieto, N. Osteopontin delays resolution of liver fibrosis. Lab. Investig. 2013, 93, 1082–1089. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.; Gibson, K.; Cisneros, J.; Richards, T.J.; Yang, Y.; Becerril, C.; Yousem, S.; Herrera, I.; Ruiz, V.; Selman, M.; et al. Up-regulation and profibrotic role of osteopontin in human idiopathic pulmonary fibrosis. PLoS Med. 2005, 2, e251. [Google Scholar] [CrossRef]
- Nagao, T.; Okura, T.; Irita, J.; Jotoku, M.; Enomoto, D.; Desilva, V.R.; Miyoshi, K.; Kurata, M.; Matsui, Y.; Uede, T.; et al. Osteopontin plays a critical role in interstitial fibrosis but not glomerular sclerosis in diabetic nephropathy. Nephron Extra 2012, 2, 87–103. [Google Scholar] [CrossRef]
- Yu, Q.; Vazquez, R.; Khojeini, E.V.; Patel, C.; Venkataramani, R.; Larson, D.F. IL-18 induction of osteopontin mediates cardiac fibrosis and diastolic dysfunction in mice. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H76–H85. [Google Scholar] [CrossRef]
- Weber, G.F.; Zawaideh, S.; Hikita, S.; Kumar, V.A.; Cantor, H.; Ashkar, S. Phosphorylation-dependent interaction of osteopontin with its receptors regulates macrophage migration and activation. J. Leukoc. Biol. 2002, 72, 752–761. [Google Scholar] [CrossRef]
- Ashkar, S.; Weber, G.F.; Panoutsakopoulou, V.; Sanchirico, M.E.; Jansson, M.; Zawaideh, S.; Rittling, S.R.; Denhardt, D.T.; Glimcher, M.J.; Cantor, H. Eta-1 (osteopontin): An early component of type-1 (cell-mediated) immunity. Science 2000, 287, 860–864. [Google Scholar] [CrossRef]
- Franzen, A.; Heinegard, D. Isolation and characterization of two sialoproteins present only in bone calcified matrix. Biochem. J. 1985, 232, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Oldberg, A.; Franzen, A.; Heinegard, D. Cloning and sequence analysis of rat bone sialoprotein (osteopontin) cDNA reveals an Arg-Gly-Asp cell-binding sequence. Proc. Natl. Acad. Sci. USA 1986, 83, 8819–8823. [Google Scholar] [CrossRef] [PubMed]
- Martin-Marquez, B.T.; Sandoval-Garcia, F.; Corona-Meraz, F.I.; Martinez-Garcia, E.A.; Sanchez-Hernandez, P.E.; Salazar-Paramo, M.; Fletes-Rayas, A.L.; Gonzalez-Inostroz, D.; Vazquez-Del Mercado, M. Osteopontin: A Bone-Derived Protein Involved in Rheumatoid Arthritis and Osteoarthritis Immunopathology. Biomolecules 2023, 13, 502. [Google Scholar] [CrossRef] [PubMed]
- Kurzbach, D.; Platzer, G.; Schwarz, T.C.; Henen, M.A.; Konrat, R.; Hinderberger, D. Cooperative unfolding of compact conformations of the intrinsically disordered protein osteopontin. Biochemistry 2013, 52, 5167–5175. [Google Scholar] [CrossRef]
- Tagliabracci, V.S.; Pinna, L.A.; Dixon, J.E. Secreted protein kinases. Trends Biochem. Sci. 2013, 38, 121–130. [Google Scholar] [CrossRef]
- Mateos, B.; Holzinger, J.; Conrad-Billroth, C.; Platzer, G.; Zerko, S.; Sealey-Cardona, M.; Anrather, D.; Kozminski, W.; Konrat, R. Hyperphosphorylation of Human Osteopontin and Its Impact on Structural Dynamics and Molecular Recognition. Biochemistry 2021, 60, 1347–1355. [Google Scholar] [CrossRef]
- Tagliabracci, V.S.; Engel, J.L.; Wen, J.; Wiley, S.E.; Worby, C.A.; Kinch, L.N.; Xiao, J.; Grishin, N.V.; Dixon, J.E. Secreted kinase phosphorylates extracellular proteins that regulate biomineralization. Science 2012, 336, 1150–1153. [Google Scholar] [CrossRef]
- Boskey, A.L.; Christensen, B.; Taleb, H.; Sorensen, E.S. Post-translational modification of osteopontin: Effects on in vitro hydroxyapatite formation and growth. Biochem. Biophys. Res. Commun. 2012, 419, 333–338. [Google Scholar] [CrossRef]
- Forsprecher, J.; Wang, Z.; Goldberg, H.A.; Kaartinen, M.T. Transglutaminase-mediated oligomerization promotes osteoblast adhesive properties of osteopontin and bone sialoprotein. Cell Adh. Migr. 2011, 5, 65–72. [Google Scholar] [CrossRef]
- Shao, Z.; Morser, J.; Leung, L.L.K. Thrombin cleavage of osteopontin disrupts a pro-chemotactic sequence for dendritic cells, which is compensated by the release of its pro-chemotactic C-terminal fragment. J. Biol. Chem. 2014, 289, 27146–27158. [Google Scholar] [CrossRef]
- Hu, D.D.; Lin, E.C.; Kovach, N.L.; Hoyer, J.R.; Smith, J.W. A biochemical characterization of the binding of osteopontin to integrins alpha v beta 1 and alpha v beta 5. J. Biol. Chem. 1995, 270, 26232–26238. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, H.J.; Jun, J.I.; Oh, Y.; Choi, S.G.; Kim, H.; Chung, C.W.; Kim, I.K.; Park, I.S.; Chae, H.J.; et al. Intracellular cleavage of osteopontin by caspase-8 modulates hypoxia/reoxygenation cell death through p53. Proc. Natl. Acad. Sci. USA 2009, 106, 15326–15331. [Google Scholar] [CrossRef]
- Christensen, B.; Schack, L.; Klaning, E.; Sorensen, E.S. Osteopontin is cleaved at multiple sites close to its integrin-binding motifs in milk and is a novel substrate for plasmin and cathepsin D. J. Biol. Chem. 2010, 285, 7929–7937. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, M.L.; Zouein, F.A.; Tian, Y.; Padmanabhan Iyer, R.; de Castro Bras, L.E. Osteopontin is proteolytically processed by matrix metalloproteinase 9. Can. J. Physiol. Pharmacol. 2015, 93, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Gimba, E.R.P.; Brum, M.C.M.; Nestal De Moraes, G. Full-length osteopontin and its splice variants as modulators of chemoresistance and radioresistance (Review). Int. J. Oncol. 2019, 54, 420–430. [Google Scholar] [CrossRef]
- Shinohara, M.L.; Kim, H.J.; Kim, J.H.; Garcia, V.A.; Cantor, H. Alternative translation of osteopontin generates intracellular and secreted isoforms that mediate distinct biological activities in dendritic cells. Proc. Natl. Acad. Sci. USA 2008, 105, 7235–7239. [Google Scholar] [CrossRef]
- Chou, C.F.; Huang, C.C.; Bin Dabil, N.; Chang, P.L. Assessing SPP1/Osteopontin (OPN) Splice Variants and Their Association to Nonmelanoma Skin Cancer by Absolute Quantification: Identification of OPN-5 Subvariants and Their Protein Coding Potential. Cancer Investig. 2021, 39, 559–570. [Google Scholar] [CrossRef]
- Bastos, A.; Gomes, A.V.P.; Silva, G.R.; Emerenciano, M.; Ferreira, L.B.; Gimba, E.R.P. The Intracellular and Secreted Sides of Osteopontin and Their Putative Physiopathological Roles. Int. J. Mol. Sci. 2023, 24, 2942. [Google Scholar] [CrossRef]
- Lin, J.; Myers, A.L.; Wang, Z.; Nancarrow, D.J.; Ferrer-Torres, D.; Handlogten, A.; Leverenz, K.; Bao, J.; Thomas, D.G.; Wang, T.D.; et al. Osteopontin (OPN/SPP1) isoforms collectively enhance tumor cell invasion and dissemination in esophageal adenocarcinoma. Oncotarget 2015, 6, 22239–22257. [Google Scholar] [CrossRef]
- Inoue, M.; Shinohara, M.L. Intracellular osteopontin (iOPN) and immunity. Immunol. Res. 2011, 49, 160–172. [Google Scholar] [CrossRef]
- Ito, K.; Kon, S.; Nakayama, Y.; Kurotaki, D.; Saito, Y.; Kanayama, M.; Kimura, C.; Diao, H.; Morimoto, J.; Matsui, Y.; et al. The differential amino acid requirement within osteopontin in alpha4 and alpha9 integrin-mediated cell binding and migration. Matrix Biol. 2009, 28, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Nishimichi, N.; Higashikawa, F.; Kinoh, H.H.; Tateishi, Y.; Matsuda, H.; Yokosaki, Y. Polymeric osteopontin employs integrin alpha9beta1 as a receptor and attracts neutrophils by presenting a de novo binding site. J. Biol. Chem. 2009, 284, 14769–14776. [Google Scholar] [CrossRef] [PubMed]
- Weber, G.F.; Ashkar, S.; Cantor, H. Interaction between CD44 and osteopontin as a potential basis for metastasis formation. Proc. Assoc. Am. Physicians 1997, 109, 1–9. [Google Scholar] [PubMed]
- Mi, Z.; Guo, H.; Russell, M.B.; Liu, Y.; Sullenger, B.A.; Kuo, P.C. RNA aptamer blockade of osteopontin inhibits growth and metastasis of MDA-MB231 breast cancer cells. Mol. Ther. 2009, 17, 153–161. [Google Scholar] [CrossRef]
- Meng, Y.; Yu, W.; Liu, Z.; Zhang, M.; Chen, Y.; Li, S.; Fan, Y.; Hu, S.; Zhang, K.; Zhang, Q.; et al. The inflammation patterns of different inflammatory cells in histological structures of hyperplasic prostatic tissues. Transl. Androl. Urol. 2020, 9, 1639–1649. [Google Scholar] [CrossRef] [PubMed]
- Chughtai, B.; Lee, R.; Te, A.; Kaplan, S. Role of inflammation in benign prostatic hyperplasia. Rev. Urol. 2011, 13, 147–150. [Google Scholar] [PubMed]
- Bushman, W.A.; Jerde, T.J. The role of prostate inflammation and fibrosis in lower urinary tract symptoms. Am. J. Physiol. Renal Physiol. 2016, 311, F817–F821. [Google Scholar] [CrossRef]
- Colinot, D.L.; Garbuz, T.; Bosland, M.C.; Wang, L.; Rice, S.E.; Sullivan, W.J., Jr.; Arrizabalaga, G.; Jerde, T.J. The common parasite Toxoplasma gondii induces prostatic inflammation and microglandular hyperplasia in a mouse model. Prostate 2017, 77, 1066–1075. [Google Scholar] [CrossRef]
- Shinohara, D.B.; Vaghasia, A.M.; Yu, S.H.; Mak, T.N.; Bruggemann, H.; Nelson, W.G.; De Marzo, A.M.; Yegnasubramanian, S.; Sfanos, K.S. A mouse model of chronic prostatic inflammation using a human prostate cancer-derived isolate of Propionibacterium acnes. Prostate 2013, 73, 1007–1015. [Google Scholar] [CrossRef]
- Popovics, P.; Jain, A.; Skalitzky, K.O.; Schroeder, E.; Ruetten, H.; Cadena, M.; Uchtmann, K.S.; Vezina, C.M.; Ricke, W.A. Osteopontin Deficiency Ameliorates Prostatic Fibrosis and Inflammation. Int. J. Mol. Sci. 2021, 22, 12461. [Google Scholar] [CrossRef]
- Thalmann, G.N.; Sikes, R.A.; Devoll, R.E.; Kiefer, J.A.; Markwalder, R.; Klima, I.; Farach-Carson, C.M.; Studer, U.E.; Chung, L.W. Osteopontin: Possible role in prostate cancer progression. Clin. Cancer Res. 1999, 5, 2271–2277. [Google Scholar] [PubMed]
- Castellano, G.; Malaponte, G.; Mazzarino, M.C.; Figini, M.; Marchese, F.; Gangemi, P.; Travali, S.; Stivala, F.; Canevari, S.; Libra, M. Activation of the osteopontin/matrix metalloproteinase-9 pathway correlates with prostate cancer progression. Clin. Cancer Res. 2008, 14, 7470–7480. [Google Scholar] [CrossRef] [PubMed]
- Tilli, T.M.; Silva, E.A.; Matos, L.C.; Faget, D.V.; Dias, B.F.; Vasconcelos, J.S.; Yokosaki, Y.; Gimba, E.R. Osteopontin is a tumor autoantigen in prostate cancer patients. Oncol. Lett. 2011, 2, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Popovics, P.; Awadallah, W.N.; Kohrt, S.E.; Case, T.C.; Miller, N.L.; Ricke, E.A.; Huang, W.; Ramirez-Solano, M.; Liu, Q.; Vezina, C.M.; et al. Prostatic osteopontin expression is associated with symptomatic benign prostatic hyperplasia. Prostate 2020, 80, 731–741. [Google Scholar] [CrossRef]
- Macoska, J.A.; Uchtmann, K.S.; Leverson, G.E.; McVary, K.T.; Ricke, W.A. Prostate Transition Zone Fibrosis is Associated with Clinical Progression in the MTOPS Study. J. Urol. 2019, 202, 1240–1247. [Google Scholar] [CrossRef]
- Yokasaki, Y.; Sheppard, D. Mapping of the cryptic integrin-binding site in osteopontin suggests a new mechanism by which thrombin can regulate inflammation and tissue repair. Trends Cardiovasc. Med. 2000, 10, 155–159. [Google Scholar] [CrossRef]
- Cui, G.; Chen, J.; Wu, Z.; Huang, H.; Wang, L.; Liang, Y.; Zeng, P.; Yang, J.; Uede, T.; Diao, H. Thrombin cleavage of osteopontin controls activation of hepatic stellate cells and is essential for liver fibrogenesis. J. Cell Physiol. 2019, 234, 8988–8997. [Google Scholar] [CrossRef]
- Popovics, P.; Cai, R.; Sha, W.; Rick, F.G.; Schally, A.V. Growth hormone-releasing hormone antagonists reduce prostatic enlargement and inflammation in carrageenan-induced chronic prostatitis. Prostate 2018, 78, 970–980. [Google Scholar] [CrossRef]
- Wei, J.; Marisetty, A.; Schrand, B.; Gabrusiewicz, K.; Hashimoto, Y.; Ott, M.; Grami, Z.; Kong, L.Y.; Ling, X.; Caruso, H.; et al. Osteopontin mediates glioblastoma-associated macrophage infiltration and is a potential therapeutic target. J. Clin. Investig. 2019, 129, 137–149. [Google Scholar] [CrossRef]
- O’Regan, A.; Berman, J.S. Osteopontin: A key cytokine in cell-mediated and granulomatous inflammation. Int. J. Exp. Pathol. 2000, 81, 373–390. [Google Scholar] [CrossRef]
- Gao, X.; Jia, G.; Guttman, A.; DePianto, D.J.; Morshead, K.B.; Sun, K.H.; Ramamoorthi, N.; Vander Heiden, J.A.; Modrusan, Z.; Wolters, P.J.; et al. Osteopontin Links Myeloid Activation and Disease Progression in Systemic Sclerosis. Cell Rep. Med. 2020, 1, 100140. [Google Scholar] [CrossRef]
- Joseph, D.B.; Henry, G.H.; Malewska, A.; Reese, J.C.; Mauck, R.J.; Gahan, J.C.; Hutchinson, R.C.; Mohler, J.L.; Roehrborn, C.G.; Strand, D.W. 5-Alpha reductase inhibitors induce a prostate luminal to club cell transition in human benign prostatic hyperplasia. J. Pathol. 2022, 256, 427–441. [Google Scholar] [CrossRef]
- Suzuki, K.; Takezawa, Y.; Suzuki, T.; Honma, S.; Yamanaka, H. Synergistic effects of estrogen with androgen on the prostate--effects of estrogen on the prostate of androgen-administered rats and 5-alpha-reductase activity. Prostate 1994, 25, 169–176. [Google Scholar] [CrossRef]
- Shibata, Y.; Ito, K.; Suzuki, K.; Nakano, K.; Fukabori, Y.; Suzuki, R.; Kawabe, Y.; Honma, S.; Yamanaka, H. Changes in the endocrine environment of the human prostate transition zone with aging: Simultaneous quantitative analysis of prostatic sex steroids and comparison with human prostatic histological composition. Prostate 2000, 42, 45–55. [Google Scholar] [CrossRef]
- Suzuki, K.; Ito, K.; Suzuki, T.; Honma, S.; Yamanaka, H. Synergistic effects of estrogen and androgen on the prostate: Effects of estrogen on androgen- and estrogen-receptors, BrdU uptake, immunohistochemical study of AR, and responses to antiandrogens. Prostate 1995, 26, 151–163. [Google Scholar] [CrossRef]
- Nicholson, T.M.; Ricke, E.A.; Marker, P.C.; Miano, J.M.; Mayer, R.D.; Timms, B.G.; vom Saal, F.S.; Wood, R.W.; Ricke, W.A. Testosterone and 17beta-estradiol induce glandular prostatic growth, bladder outlet obstruction, and voiding dysfunction in male mice. Endocrinology 2012, 153, 5556–5565. [Google Scholar] [CrossRef] [PubMed]
- Popovics, P.; Skalitzky, K.O.; Schroeder, E.; Jain, A.; Silver, S.V.; Van Fritz, F.; Uchtmann, K.S.; Vezina, C.M.; Ricke, W.A. Steroid hormone imbalance drives macrophage infiltration and Spp1/osteopontin(+) foam cell differentiation in the prostate. J. Pathol. 2023, 260, 177–189. [Google Scholar] [CrossRef]
- Grabowska, M.M.; Kelly, S.M.; Reese, A.L.; Cates, J.M.; Case, T.C.; Zhang, J.; DeGraff, D.J.; Strand, D.W.; Miller, N.L.; Clark, P.E.; et al. Nfib Regulates Transcriptional Networks That Control the Development of Prostatic Hyperplasia. Endocrinology 2016, 157, 1094–1109. [Google Scholar] [CrossRef] [PubMed]
- Lund, S.A.; Giachelli, C.M.; Scatena, M. The role of osteopontin in inflammatory processes. J. Cell Commun. Signal 2009, 3, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Nieves, J.A.; Patalano, S.C.; Almanza, D.; Gharaee-Kermani, M.; Macoska, J.A. CXCL12/CXCR4 Axis Activation Mediates Prostate Myofibroblast Phenoconversion through Non-Canonical EGFR/MEK/ERK Signaling. PLoS ONE 2016, 11, e0159490. [Google Scholar] [CrossRef]
- Capucetti, A.; Albano, F.; Bonecchi, R. Multiple Roles for Chemokines in Neutrophil Biology. Front. Immunol. 2020, 11, 1259. [Google Scholar] [CrossRef] [PubMed]
- Dang, T.; Liou, G.Y. Macrophage Cytokines Enhance Cell Proliferation of Normal Prostate Epithelial Cells through Activation of ERK and Akt. Sci. Rep. 2018, 8, 7718. [Google Scholar] [CrossRef]
- Yoo, K.H.; Thornhill, B.A.; Forbes, M.S.; Coleman, C.M.; Marcinko, E.S.; Liaw, L.; Chevalier, R.L. Osteopontin regulates renal apoptosis and interstitial fibrosis in neonatal chronic unilateral ureteral obstruction. Kidney Int. 2006, 70, 1735–1741. [Google Scholar] [CrossRef] [PubMed]
- Gui, Y.; Zheng, H.; Cao, R.Y. Foam Cells in Atherosclerosis: Novel Insights into Its Origins, Consequences, and Molecular Mechanisms. Front. Cardiovasc. Med. 2022, 9, 845942. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Summer, R.; Chen, Z.; Tian, Y.; Ma, J.; Cui, J.; Hao, X.; Guo, L.; Xu, H.; Wang, H.; et al. Lipid Uptake by Alveolar Macrophages Drives Fibrotic Responses to Silica Dust. Sci. Rep. 2019, 9, 399. [Google Scholar] [CrossRef]
- Ganesan, R.; Henkels, K.M.; Wrenshall, L.E.; Kanaho, Y.; Di Paolo, G.; Frohman, M.A.; Gomez-Cambronero, J. Oxidized LDL phagocytosis during foam cell formation in atherosclerotic plaques relies on a PLD2-CD36 functional interdependence. J. Leukoc. Biol. 2018, 103, 867–883. [Google Scholar] [CrossRef]
- Li, L.; Liu, B.; Haversen, L.; Lu, E.; Magnusson, L.U.; Stahlman, M.; Boren, J.; Bergstrom, G.; Levin, M.C.; Hulten, L.M. The importance of GLUT3 for de novo lipogenesis in hypoxia-induced lipid loading of human macrophages. PLoS ONE 2012, 7, e42360. [Google Scholar] [CrossRef]
- Masetti, M.; Carriero, R.; Portale, F.; Marelli, G.; Morina, N.; Pandini, M.; Iovino, M.; Partini, B.; Erreni, M.; Ponzetta, A.; et al. Lipid-loaded tumor-associated macrophages sustain tumor growth and invasiveness in prostate cancer. J. Exp. Med. 2022, 219, e20210564. [Google Scholar] [CrossRef]
- El-Kenawi, A.; Dominguez-Viqueira, W.; Liu, M.; Awasthi, S.; Abraham-Miranda, J.; Keske, A.; Steiner, K.K.; Noel, L.; Serna, A.N.; Dhillon, J.; et al. Macrophage-Derived Cholesterol Contributes to Therapeutic Resistance in Prostate Cancer. Cancer Res. 2021, 81, 5477–5490. [Google Scholar] [CrossRef]
- Qiao, R.; Qiao, H.; Zhang, Y.; Wang, Y.; Chi, C.; Tian, J.; Zhang, L.; Cao, F.; Gao, M. Molecular Imaging of Vulnerable Atherosclerotic Plaques in Vivo with Osteopontin-Specific Upconversion Nanoprobes. ACS Nano 2017, 11, 1816–1825. [Google Scholar] [CrossRef]
- Han, H.; Ge, X.; Komakula, S.S.B.; Desert, R.; Das, S.; Song, Z.; Chen, W.; Athavale, D.; Gaskell, H.; Lantvit, D.; et al. Macrophage-derived Osteopontin (SPP1) Protects from Nonalcoholic Steatohepatitis. Gastroenterology 2023, 165, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.D.; Nishi, H.; Poles, J.; Niu, X.; McCauley, C.; Rahman, K.; Brown, E.J.; Yeung, S.T.; Vozhilla, N.; Weinstock, A.; et al. Single-cell analysis of fate-mapped macrophages reveals heterogeneity, including stem-like properties, during atherosclerosis progression and regression. JCI Insight 2019, 4, e124574. [Google Scholar] [CrossRef]
- Kim, K.; Shim, D.; Lee, J.S.; Zaitsev, K.; Williams, J.W.; Kim, K.W.; Jang, M.Y.; Seok Jang, H.; Yun, T.J.; Lee, S.H.; et al. Transcriptome Analysis Reveals Nonfoamy Rather Than Foamy Plaque Macrophages Are Proinflammatory in Atherosclerotic Murine Models. Circ. Res. 2018, 123, 1127–1142. [Google Scholar] [CrossRef] [PubMed]
- McArdle, S.; Buscher, K.; Ghosheh, Y.; Pramod, A.B.; Miller, J.; Winkels, H.; Wolf, D.; Ley, K. Migratory and Dancing Macrophage Subsets in Atherosclerotic Lesions. Circ. Res. 2019, 125, 1038–1051. [Google Scholar] [CrossRef] [PubMed]
- Willemsen, L.; de Winther, M.P. Macrophage subsets in atherosclerosis as defined by single-cell technologies. J. Pathol. 2020, 250, 705–714. [Google Scholar] [CrossRef]
- Xiong, J.; Li, Z.; Tang, H.; Duan, Y.; Ban, X.; Xu, K.; Guo, Y.; Tu, Y. Bulk and single-cell characterisation of the immune heterogeneity of atherosclerosis identifies novel targets for immunotherapy. BMC Biol. 2023, 21, 46. [Google Scholar] [CrossRef]
- Schwarz, A.; Bonaterra, G.A.; Schwarzbach, H.; Kinscherf, R. Oxidized LDL-induced JAB1 influences NF-kappaB independent inflammatory signaling in human macrophages during foam cell formation. J. Biomed. Sci. 2017, 24, 12. [Google Scholar] [CrossRef]
- Messex, J.K.; Byrd, C.J.; Thomas, M.U.; Liou, G.Y. Macrophages Cytokine Spp1 Increases Growth of Prostate Intraepithelial Neoplasia to Promote Prostate Tumor Progression. Int. J. Mol. Sci. 2022, 23, 4247. [Google Scholar] [CrossRef]
- Zhang, Y.; Du, W.; Chen, Z.; Xiang, C. Upregulation of PD-L1 by SPP1 mediates macrophage polarization and facilitates immune escape in lung adenocarcinoma. Exp. Cell Res. 2017, 359, 449–457. [Google Scholar] [CrossRef]
- Ladwig, A.; Walter, H.L.; Hucklenbroich, J.; Willuweit, A.; Langen, K.J.; Fink, G.R.; Rueger, M.A.; Schroeter, M. Osteopontin Augments M2 Microglia Response and Separates M1- and M2-Polarized Microglial Activation in Permanent Focal Cerebral Ischemia. Mediators Inflamm. 2017, 2017, 7189421. [Google Scholar] [CrossRef]
- Bai, G.; Matsuba, T.; Niki, T.; Hattori, T. Stimulation of THP-1 Macrophages with LPS Increased the Production of Osteopontin-Encapsulating Exosome. Int. J. Mol. Sci. 2020, 21, 8490. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lv, G.; Wang, B.; Kuang, L. XIST/miR-376c-5p/OPN axis modulates the influence of proinflammatory M1 macrophages on osteoarthritis chondrocyte apoptosis. J. Cell Physiol. 2020, 235, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Xi, F.; Deng, X.; Ni, Y.; Pu, C.; Wang, D.; Lou, W.; Zeng, X.; Su, N.; Chen, C.; et al. Osteopontin Promotes Macrophage M1 Polarization by Activation of the JAK1/STAT1/HMGB1 Signaling Pathway in Nonalcoholic Fatty Liver Disease. J. Clin. Transl. Hepatol. 2023, 11, 273–283. [Google Scholar] [CrossRef]
- Capote, J.; Kramerova, I.; Martinez, L.; Vetrone, S.; Barton, E.R.; Sweeney, H.L.; Miceli, M.C.; Spencer, M.J. Osteopontin ablation ameliorates muscular dystrophy by shifting macrophages to a pro-regenerative phenotype. J. Cell Biol. 2016, 213, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Lund, S.A.; Wilson, C.L.; Raines, E.W.; Tang, J.; Giachelli, C.M.; Scatena, M. Osteopontin mediates macrophage chemotaxis via alpha4 and alpha9 integrins and survival via the alpha4 integrin. J. Cell Biochem. 2013, 114, 1194–1202. [Google Scholar] [CrossRef]
- Zhao, K.; Zhang, M.; Zhang, L.; Wang, P.; Song, G.; Liu, B.; Wu, H.; Yin, Z.; Gao, C. Intracellular osteopontin stabilizes TRAF3 to positively regulate innate antiviral response. Sci. Rep. 2016, 6, 23771. [Google Scholar] [CrossRef]
- Inoue, M.; Moriwaki, Y.; Arikawa, T.; Chen, Y.H.; Oh, Y.J.; Oliver, T.; Shinohara, M.L. Cutting edge: Critical role of intracellular osteopontin in antifungal innate immune responses. J. Immunol. 2011, 186, 19–23. [Google Scholar] [CrossRef]
- Ouyang, J.F.; Mishra, K.; Xie, Y.; Park, H.; Huang, K.Y.; Petretto, E.; Behmoaras, J. Systems level identification of a matrisome-associated macrophage polarisation state in multi-organ fibrosis. Elife 2023, 12, e85530. [Google Scholar] [CrossRef]
- Schack, L.; Stapulionis, R.; Christensen, B.; Kofod-Olsen, E.; Skov Sorensen, U.B.; Vorup-Jensen, T.; Sorensen, E.S.; Hollsberg, P. Osteopontin enhances phagocytosis through a novel osteopontin receptor, the alphaXbeta2 integrin. J. Immunol. 2009, 182, 6943–6950. [Google Scholar] [CrossRef]
- Wei, J.; Chen, Z.; Hu, M.; He, Z.; Jiang, D.; Long, J.; Du, H. Characterizing Intercellular Communication of Pan-Cancer Reveals SPP1+ Tumor-Associated Macrophage Expanded in Hypoxia and Promoting Cancer Malignancy Through Single-Cell RNA-Seq Data. Front. Cell Dev. Biol. 2021, 9, 749210. [Google Scholar] [CrossRef]
- Theyer, G.; Kramer, G.; Assmann, I.; Sherwood, E.; Preinfalk, W.; Marberger, M.; Zechner, O.; Steiner, G.E. Phenotypic characterization of infiltrating leukocytes in benign prostatic hyperplasia. Lab. Investig. 1992, 66, 96–107. [Google Scholar]
- O’Regan, A.W.; Chupp, G.L.; Lowry, J.A.; Goetschkes, M.; Mulligan, N.; Berman, J.S. Osteopontin is associated with T cells in sarcoid granulomas and has T cell adhesive and cytokine-like properties in vitro. J. Immunol. 1999, 162, 1024–1031. [Google Scholar] [CrossRef]
- Hur, E.M.; Youssef, S.; Haws, M.E.; Zhang, S.Y.; Sobel, R.A.; Steinman, L. Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat. Immunol. 2007, 8, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Cantor, H.; Shinohara, M.L. Regulation of T-helper-cell lineage development by osteopontin: The inside story. Nat. Rev. Immunol. 2009, 9, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef] [PubMed]
- Tuong, Z.K.; Loudon, K.W.; Berry, B.; Richoz, N.; Jones, J.; Tan, X.; Nguyen, Q.; George, A.; Hori, S.; Field, S.; et al. Resolving the immune landscape of human prostate at a single-cell level in health and cancer. Cell Rep. 2021, 37, 110132. [Google Scholar] [CrossRef] [PubMed]
- Joseph, D.B.; Henry, G.H.; Malewska, A.; Reese, J.C.; Mauck, R.J.; Gahan, J.C.; Hutchinson, R.C.; Malladi, V.S.; Roehrborn, C.G.; Vezina, C.M.; et al. Single-cell analysis of mouse and human prostate reveals novel fibroblasts with specialized distribution and microenvironment interactions. J. Pathol. 2021, 255, 141–154. [Google Scholar] [CrossRef]
- Aalamian, M.; Pirtskhalaishvili, G.; Nunez, A.; Esche, C.; Shurin, G.V.; Huland, E.; Huland, H.; Shurin, M.R. Human prostate cancer regulates generation and maturation of monocyte-derived dendritic cells. Prostate 2001, 46, 68–75. [Google Scholar] [CrossRef]
- Hirz, T.; Mei, S.; Sarkar, H.; Kfoury, Y.; Wu, S.; Verhoeven, B.M.; Subtelny, A.O.; Zlatev, D.V.; Wszolek, M.W.; Salari, K.; et al. Dissecting the immune suppressive human prostate tumor microenvironment via integrated single-cell and spatial transcriptomic analyses. Nat. Commun. 2023, 14, 663. [Google Scholar] [CrossRef]
- Hawlina, S.; Zorec, R.; Chowdhury, H.H. Potential of Personalized Dendritic Cell-Based Immunohybridoma Vaccines to Treat Prostate Cancer. Life 2023, 13, 1498. [Google Scholar] [CrossRef]
- Shinohara, M.L.; Lu, L.; Bu, J.; Werneck, M.B.; Kobayashi, K.S.; Glimcher, L.H.; Cantor, H. Osteopontin expression is essential for interferon-alpha production by plasmacytoid dendritic cells. Nat. Immunol. 2006, 7, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, M.L.; Kim, J.H.; Garcia, V.A.; Cantor, H. Engagement of the type I interferon receptor on dendritic cells inhibits T helper 17 cell development: Role of intracellular osteopontin. Immunity 2008, 29, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Broekelmann, T.J.; Bodmer, N.K.; Mecham, R.P. Identification of the growth factor-binding sequence in the extracellular matrix protein MAGP-1. J. Biol. Chem. 2020, 295, 2687–2697. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Pascal, L.E.; Stolz, D.B.; Wang, K.; Zhou, Y.; Chen, W.; Xu, Y.; Chen, Y.; Dhir, R.; Parwani, A.V.; et al. E-cadherin is downregulated in benign prostatic hyperplasia and required for tight junction formation and permeability barrier in the prostatic epithelial cell monolayer. Prostate 2019, 79, 1226–1237. [Google Scholar] [CrossRef]
- Romero, F.; Shah, D.; Duong, M.; Penn, R.B.; Fessler, M.B.; Madenspacher, J.; Stafstrom, W.; Kavuru, M.; Lu, B.; Kallen, C.B.; et al. A pneumocyte-macrophage paracrine lipid axis drives the lung toward fibrosis. Am. J. Respir. Cell Mol. Biol. 2015, 53, 74–86. [Google Scholar] [CrossRef]
- Brawer, M.K. Prostatic intraepithelial neoplasia: An overview. Rev. Urol. 2005, 7 (Suppl. S3), S11–S18. [Google Scholar]
- Gleason, D.F. Histologic grading of prostate cancer: A perspective. Hum. Pathol. 1992, 23, 273–279. [Google Scholar] [CrossRef]
- Hugosson, J.; Mansson, M.; Wallstrom, J.; Axcrona, U.; Carlsson, S.V.; Egevad, L.; Geterud, K.; Khatami, A.; Kohestani, K.; Pihl, C.G.; et al. Prostate Cancer Screening with PSA and MRI Followed by Targeted Biopsy Only. N. Engl. J. Med. 2022, 387, 2126–2137. [Google Scholar] [CrossRef]
- Beltran, H.; Rickman, D.S.; Park, K.; Chae, S.S.; Sboner, A.; MacDonald, T.Y.; Wang, Y.; Sheikh, K.L.; Terry, S.; Tagawa, S.T.; et al. Molecular characterization of neuroendocrine prostate cancer and identification of new drug targets. Cancer Discov. 2011, 1, 487–495. [Google Scholar] [CrossRef]
- Lundberg, A.; Zhang, M.; Aggarwal, R.; Li, H.; Zhang, L.; Foye, A.; Sjostrom, M.; Chou, J.; Chang, K.; Moreno-Rodriguez, T.; et al. The Genomic and Epigenomic Landscape of Double-Negative Metastatic Prostate Cancer. Cancer Res. 2023, 83, 2763–2774. [Google Scholar] [CrossRef]
- Feng, Q.; He, B. Androgen Receptor Signaling in the Development of Castration-Resistant Prostate Cancer. Front. Oncol. 2019, 9, 858. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Abdollah, F.; Schiffmann, J.; Trudeau, V.; Shariat, S.F.; Kim, S.P.; Perrotte, P.; Montorsi, F.; Briganti, A.; Trinh, Q.D.; et al. Distribution of metastatic sites in patients with prostate cancer: A population-based analysis. Prostate 2014, 74, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ye, G.; Dong, B.; Huang, L.; Zhang, C.; Sheng, Y.; Wu, B.; Han, L.; Wu, C.; Qi, Y. A pan-cancer analysis of the oncogenic role of secreted phosphoprotein 1 (SPP1) in human cancers. Ann. Transl. Med. 2022, 10, 279. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Chen, Q.; Alam, A.; Cui, J.; Suen, K.C.; Soo, A.P.; Eguchi, S.; Gu, J.; Ma, D. The role of osteopontin in the progression of solid organ tumour. Cell Death Dis. 2018, 9, 356. [Google Scholar] [CrossRef]
- Bramwell, V.H.; Doig, G.S.; Tuck, A.B.; Wilson, S.M.; Tonkin, K.S.; Tomiak, A.; Perera, F.; Vandenberg, T.A.; Chambers, A.F. Serial plasma osteopontin levels have prognostic value in metastatic breast cancer. Clin. Cancer Res. 2006, 12 Pt 1, 3337–3343. [Google Scholar] [CrossRef]
- Zduniak, K.; Ziolkowski, P.; Ahlin, C.; Agrawal, A.; Agrawal, S.; Blomqvist, C.; Fjallskog, M.L.; Weber, G.F. Nuclear osteopontin-c is a prognostic breast cancer marker. Br. J. Cancer 2015, 112, 729–738. [Google Scholar] [CrossRef]
- Kidess, E.; Giesecke, Y.; Eichhorn, I.; Mohr, R.; Jann, H.; Fischer, C.; Wiedenmann, B.; Roderburg, C.; Tacke, F.; Sigal, M. Osteopontin is a prognostic circulating biomarker in patients with neuroendocrine neoplasms. J. Cancer Res. Clin. Oncol. 2023, 149, 10925–10933. [Google Scholar] [CrossRef]
- Rangel, J.; Nosrati, M.; Torabian, S.; Shaikh, L.; Leong, S.P.; Haqq, C.; Miller, J.R., 3rd; Sagebiel, R.W.; Kashani-Sabet, M. Osteopontin as a molecular prognostic marker for melanoma. Cancer 2008, 112, 144–150. [Google Scholar] [CrossRef]
- Forootan, S.S.; Foster, C.S.; Aachi, V.R.; Adamson, J.; Smith, P.H.; Lin, K.; Ke, Y. Prognostic significance of osteopontin expression in human prostate cancer. Int. J. Cancer 2006, 118, 2255–2261. [Google Scholar] [CrossRef]
- Hotte, S.J.; Winquist, E.W.; Stitt, L.; Wilson, S.M.; Chambers, A.F. Plasma osteopontin: Associations with survival and metastasis to bone in men with hormone-refractory prostate carcinoma. Cancer 2002, 95, 506–512. [Google Scholar] [CrossRef]
- Tilli, T.M.; Thuler, L.C.; Matos, A.R.; Coutinho-Camillo, C.M.; Soares, F.A.; da Silva, E.A.; Neves, A.F.; Goulart, L.R.; Gimba, E.R. Expression analysis of osteopontin mRNA splice variants in prostate cancer and benign prostatic hyperplasia. Exp. Mol. Pathol. 2012, 92, 13–19. [Google Scholar] [CrossRef]
- Thoms, J.W.; Dal Pra, A.; Anborgh, P.H.; Christensen, E.; Fleshner, N.; Menard, C.; Chadwick, K.; Milosevic, M.; Catton, C.; Pintilie, M.; et al. Plasma osteopontin as a biomarker of prostate cancer aggression: Relationship to risk category and treatment response. Br. J. Cancer 2012, 107, 840–846. [Google Scholar] [CrossRef]
- Hsieh, I.S.; Huang, W.H.; Liou, H.C.; Chuang, W.J.; Yang, R.S.; Fu, W.M. Upregulation of drug transporter expression by osteopontin in prostate cancer cells. Mol. Pharmacol. 2013, 83, 968–977. [Google Scholar] [CrossRef]
- Khodavirdi, A.C.; Song, Z.; Yang, S.; Zhong, C.; Wang, S.; Wu, H.; Pritchard, C.; Nelson, P.S.; Roy-Burman, P. Increased expression of osteopontin contributes to the progression of prostate cancer. Cancer Res. 2006, 66, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Mauri, G.; Jachetti, E.; Comuzzi, B.; Dugo, M.; Arioli, I.; Miotti, S.; Sangaletti, S.; Di Carlo, E.; Tripodo, C.; Colombo, M.P. Genetic deletion of osteopontin in TRAMP mice skews prostate carcinogenesis from adenocarcinoma to aggressive human-like neuroendocrine cancers. Oncotarget 2016, 7, 3905–3920. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Chakraborty, G.; Kundu, G.C. The crucial role of cyclooxygenase-2 in osteopontin-induced protein kinase C alpha/c-Src/IkappaB kinase alpha/beta-dependent prostate tumor progression and angiogenesis. Cancer Res. 2006, 66, 6638–6648. [Google Scholar] [CrossRef]
- Desai, B.; Rogers, M.J.; Chellaiah, M.A. Mechanisms of osteopontin and CD44 as metastatic principles in prostate cancer cells. Mol. Cancer 2007, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Desai, B.; Ma, T.; Chellaiah, M.A. Invadopodia and matrix degradation, a new property of prostate cancer cells during migration and invasion. J. Biol. Chem. 2008, 283, 13856–13866. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Zhou, C.Q.; Chellaiah, M.A. Osteopontin and MMP9: Associations with VEGF Expression/Secretion and Angiogenesis in PC3 Prostate Cancer Cells. Cancers 2013, 5, 617. [Google Scholar] [CrossRef]
- Robertson, B.W.; Bonsal, L.; Chellaiah, M.A. Regulation of Erk1/2 activation by osteopontin in PC3 human prostate cancer cells. Mol. Cancer 2010, 9, 260. [Google Scholar] [CrossRef]
- Angelucci, A.; Festuccia, C.; D’Andrea, G.; Teti, A.; Bologna, M. Osteopontin modulates prostate carcinoma invasive capacity through RGD-dependent upregulation of plasminogen activators. Biol. Chem. 2002, 383, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Saliganan, A.D.; Meng, H.; Nabha, S.M.; Sabbota, A.L.; Sheng, S.; Bonfil, R.D.; Cher, M.L. Prostate cancer cell-derived urokinase-type plasminogen activator contributes to intraosseous tumor growth and bone turnover. Neoplasia 2008, 10, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Gong, K.; Zhang, X.; Wu, S.; Cui, Y.; Qian, B.Z. Osteopontin as a multifaceted driver of bone metastasis and drug resistance. Pharmacol. Res. 2019, 144, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.Q.; Woodard, A.S.; Tallini, G.; Languino, L.R. Substrate specificity of αvβ3 integrin-mediated cell migration and phosphatidylinositol 3-kinase/AKT pathway activation. J. Biol. Chem. 2000, 275, 24565–24574. [Google Scholar] [CrossRef]
- Moro, L.; Venturino, M.; Bozzo, C.; Silengo, L.; Altruda, F.; Beguinot, L.; Tarone, G.; Defilippi, P. Integrins induce activation of EGF receptor: Role in MAP kinase induction and adhesion-dependent cell survival. EMBO J. 1998, 17, 6622–6632. [Google Scholar] [CrossRef]
- Angelucci, A.; Festuccia, C.; Gravina, G.L.; Muzi, P.; Bonghi, L.; Vicentini, C.; Bologna, M. Osteopontin enhances the cell proliferation induced by the epidermal growth factor in human prostate cancer cells. Prostate 2004, 59, 157–166. [Google Scholar] [CrossRef]
- Vaupel, P.; Kelleher, D.K.; Hockel, M. Oxygen status of malignant tumors: Pathogenesis of hypoxia and significance for tumor therapy. Semin. Oncol. 2001, 28 (Suppl. S8), 29–35. [Google Scholar] [CrossRef]
- Nakamura, K.D.; Tilli, T.M.; Wanderley, J.L.; Palumbo, A., Jr.; Mattos, R.M.; Ferreira, A.C.; Klumb, C.E.; Nasciutti, L.E.; Gimba, E.R. Osteopontin splice variants expression is involved on docetaxel resistance in PC3 prostate cancer cells. Tumour Biol. 2016, 37, 2655–2663. [Google Scholar] [CrossRef]
- Brina, D.; Ponzoni, A.; Troiani, M.; Cali, B.; Pasquini, E.; Attanasio, G.; Mosole, S.; Mirenda, M.; D’Ambrosio, M.; Colucci, M.; et al. The Akt/mTOR and MNK/eIF4E pathways rewire the prostate cancer translatome to secrete HGF, SPP1 and BGN and recruit suppressive myeloid cells. Nat. Cancer 2023, 4, 1102–1121. [Google Scholar] [CrossRef]

| Prostate Disease | Major Finding | Reference | Citation |
|---|---|---|---|
| Benign prostate disease | OPN expression is similar in prostate cancer and BPH. | Thalmann et al., 1999 | [52] |
| The serum level of OPN is elevated in BPH patients compared to age-matched healthy males. | Castellano et al., 2008 | [53] | |
| The presence of OPN-reactive antibodies is increased in men with BPH vs. healthy men. | Tilli et al., 2011 | [54] | |
| OPN tissue expression is increased with BPH progression (incidental BPH vs. surgical BPH), and OPN induces cytokine expression in prostate stromal cells. | Popovics et al., 2020 | [55] | |
| OPN expression is upregulated in the carrageenan-induced prostatic inflammation mouse model. | Popovics et al., 2018 | [59] | |
| Loss of OPN ameliorates prostatic inflammation and fibrosis in E. coli-induced mouse model of prostatitis. | Popovics et al., 2021 | [51] | |
| OPN is highly expressed in prostatic luminal foam cells, and the loss of OPN leads to decreased macrophage numbers and improves urinary function. | Popovics et al., 2023 | [68] | |
| Spp1 expression is elevated in the Nfib-knockout prostate hyperplasia model. | Grabowska et al., 2016 | [69] | |
| Prostate cancer | OPN expression correlates with prostate cancer progression, Gleason score, and reduced survival. | Forootan et al., 2006 | [130] |
| OPN level correlates with prostate cancer progression and MMP9 expression. | Castellano et al., 2008 | [53] | |
| Plasma osteopontin level is associated with worse survival and metastasis in men with castration-resistant prostate cancer. | Hotte et al., 2002 | [131] | |
| OPN splice variant expression is associated with PCa Gleason grade. | Tilli et al., 2012 | [132] | |
| Osteopontin plasma level predicts treatment outcomes in metastatic PCa post radiotherapy. | Thoms et al., 2012 | [133] | |
| Chemotherapy is associated with increased osteopontin levels in prostate tumors and increased drug transporter expression. | Hsieh et al., 2013 | [134] | |
| OPN level is increased predominantly in epithelial cells, as well as in stromal immune cells, in genetic models. | Khodavirdi et al., 2006 | [135] | |
| OPN-targeting antibody inhibited the growth-stimulatory action of endogenous OPN in Pca cells. | Thalmann et al., 1999 | [52] | |
| TRAMP mice develop more aggressive prostate cancer with a neuroendocrine phenotype when crossed with OPN-KO vs. wild-type mice | Mauri et al., 2016 | [136] | |
| OPN stimulates the phosphorylation of Akt and Jun kinase in PIN cells. | Messex et al., 2022 | [89] | |
| OPN stimulates the c-SRC/p50&p65/COX-2 pathway leading to the production of prostaglandin and the stimulation of angiogenesis. | Jain et al., 2006 | [137] | |
| OPN stimulates the secretion and activity of MMP9 and its interaction with the CD44 receptor, thereby promoting cell migration. | Desai et al., 2007 | [138] | |
| OPN overexpressing prostate cancer cells have increased gelatinase activity and more invadopodia. | Desai et al., 2008 | [139] | |
| OPN and MMP9 cooperate to increase the expression and secretion of VEGF, ERK phosphorylation, and angiogenesis. | Gupta et al., 2013 | [140] | |
| OPN stimulates the phosphorylation of MEK, ERK1/2, and Akt in PCa cells. | Robertson et al., 2010 | [141] | |
| OPN stimulates plasminogen activators in PCa cells. | Angelucci et al., 2002 | [142] | |
| OPN increases the urokinase-type plasminogen activator in PCa cells to promote bone metastasis. | Dong et al., 2008 | [143] | |
| OPN enhances migration in PCa cells via PI3-kinase activity/Akt pathway. | Zheng et al., 2000 | [145] | |
| Integrin receptor–OPN interaction co-stimulate EGF receptor signaling. | Angelucci et al., 2004 | [147] | |
| Hypoxia induces OPN expression in PCa cells. | Hsieh et al., 2013 | [134] | |
| Osteopontin splice variant overexpression enhances resistance to docetaxel. | Nakamura et al., 2016 | [149] | |
| PCa-secreted OPN promotes myeloid suppressor cells. | Brina et al., 2023 | [150] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silver, S.V.; Popovics, P. The Multifaceted Role of Osteopontin in Prostate Pathologies. Biomedicines 2023, 11, 2895. https://doi.org/10.3390/biomedicines11112895
Silver SV, Popovics P. The Multifaceted Role of Osteopontin in Prostate Pathologies. Biomedicines. 2023; 11(11):2895. https://doi.org/10.3390/biomedicines11112895
Chicago/Turabian StyleSilver, Samara V., and Petra Popovics. 2023. "The Multifaceted Role of Osteopontin in Prostate Pathologies" Biomedicines 11, no. 11: 2895. https://doi.org/10.3390/biomedicines11112895
APA StyleSilver, S. V., & Popovics, P. (2023). The Multifaceted Role of Osteopontin in Prostate Pathologies. Biomedicines, 11(11), 2895. https://doi.org/10.3390/biomedicines11112895





